Abstract
Background:
The purpose of this case report is to describe the potential that metabolomics breath analysis may have in cancer disease monitoring. The advances in mass spectrometry instrumentation allow the accurate real-time analysis of volatile metabolites exhaled in the breath. The application of such non-invasive devices may provide innovative and complementary monitoring of the physio-pathological conditions of cancer patients.
Case presentation:
A 59-year-old Caucasian woman with spindle cell malignant mesenchymal sarcoma of the presacral region started a first-line therapy with non-pegylated liposomal doxorubicin and ifosfamide associated with pelvic radiant treatment. After two cycles of chemotherapy plus radiotherapy, a significant pulmonary disease progression was reported. Thus, a second-line therapy with trabectedin was administered. However, after only two cycles of treatment a re-staging computed tomography scan reported further cancer disease progression of the target pulmonary lesions as well as occurrence of new satellite bilateral nodules. Real-time analysis of breath exhaled volatile organic compounds, performed by select ion flow tube mass spectrometry (SIFT-MS) during the follow-up of the patient, showed a specific metabolic pattern not observed in the breath of other soft tissue sarcoma patients who achieved clinical benefit from the treatments.
Conclusions:
This case report revealed the importance of the non-invasive real-time volatile organic compounds breath analysis to distinguish individual specific chemo-resistance phenotypes among soft tissue sarcoma patients. Such observation seems to suggest that breath metabolomics may be particularly useful for monitoring cancer disease progression in soft tissue sarcoma patients where only cost-effective diagnostic tools, such as positron emission tomography and computed tomography, are available.
Introduction
The cancer patient’s journey from diagnosis to treatment is characterized by the strict monitoring of the cancer disease, which is commonly performed mainly by tumor serum markers and instrumental monitoring. Most of these investigations are focused on tumor parameters and few regard the overall host health status. However, with the advent of the omics era, new opportunities are emerging to study cancer patients and to better describe the complex interplay between tumor and host. Metabolomics is the most recent of the omics sciences, which is particularly helpful to improve our general understanding of cancer-related processes and to identify new diagnostic and prognostic cancer biomarkers. 1 Metabolomics addresses the simultaneous monitoring of hundreds to thousands of small molecule metabolites (<1 kDa) in bio-fluids and tissues to better identify a specific individual phenotype. Since the metabolomics profile results from the complex interplay between host genetic and environmental factors, it may be more informative than genomics and proteomics profiles in describing the health and disease status of an individual. Therefore, the metabolomics approach is becoming an attractive tool for the development of precision medicine.
The volatile organic compounds (VOCs) present in the exhaled breath constitute a sub-component of the metabolome, which was able to give precise biochemical information on the individual’s physio-pathological status. 2 Moreover, the introduction of real-time breath analysis of VOCs represents an attractive, fast, and non-invasive approach for cancer disease diagnosis and monitoring.3,4 Here, we describe the case of a patient with soft tissue sarcoma (STS) who showed cancer disease progression, despite all the treatments performed, which was characterized by a unique breath VOCs profile.
Case report
In February 2017, a 59-year-old Caucasian woman underwent a surgical visit for evacuation disorders. The pelvic magnetic resonance imaging detected a lesion (Ø 6×5×10 cm) in the presacral region that the biopsy ascertained as a high-grade spindle cell malignant mesenchymal sarcoma. A staging computed tomography (CT) scan revealed two bilateral secondary pulmonary nodular lesions with a diameter section from 5 mm to 14 mm. The first-line therapy was initiated with non-pegylated liposomal doxorubicin and ifosfamide, at the dose of 50 mg/m2 intravenously (IV) for 1 day, and 3000 mg/m2 IV for 3 days, every 3 weeks, together with pelvic radiant treatment (44 Gy in 22 fractions). After two cycles of chemotherapy, at the end of the radiation treatment, a restaging CT scan showed a stable disease of the primary pelvic lesion, but revealed significant pulmonary disease progression. Then, a second-line therapy with trabectedin at the dose of 1.5 mg/m2 IV every 3 weeks was started. After two cycles of treatment, the patient was included in an investigation aimed to evaluate the feasibility of real-time breath VOCs analysis. This involved a group of 5 STS metastatic cancer patients whose VOCs profiles were compared with that of 12 healthy control subjects. The patients and controls were scrutinized for their diet and smoking habits, and only non-smokers were enrolled in this investigation. Concomitantly to the VOCs profile analysis, the CT scan for the case report patient revealed further cancer disease progression of the pulmonary lesions as well as the occurrence of new satellite bilateral nodules with diameters from 4 mm to 22 mm (Figure 1). The multiparametric partial least squares differential analysis (PLS-DA) of the VOCs profiles showed the space data separation between the group of STS patients and the healthy individuals, indicating that there are significant differences in their VOCs profiles. The VOCs that better contributed with variable importance in projection (VIP) >1.5 to such differentiation were: ammonia, carbon disulfide, acetone, and phenol. Intriguingly, among the STS group, an outlier patient who showed a significantly different VOCs profile, which corresponded to the case study patient, was recognized (Figure 2). This breath VOCs profile seemed to be associated with her differential individual behavior to cancer treatments. In fact, the patient was characterized by an extremely intrinsic resistance to all performed treatments (first and second line), which is rarely observed in clinical practice.
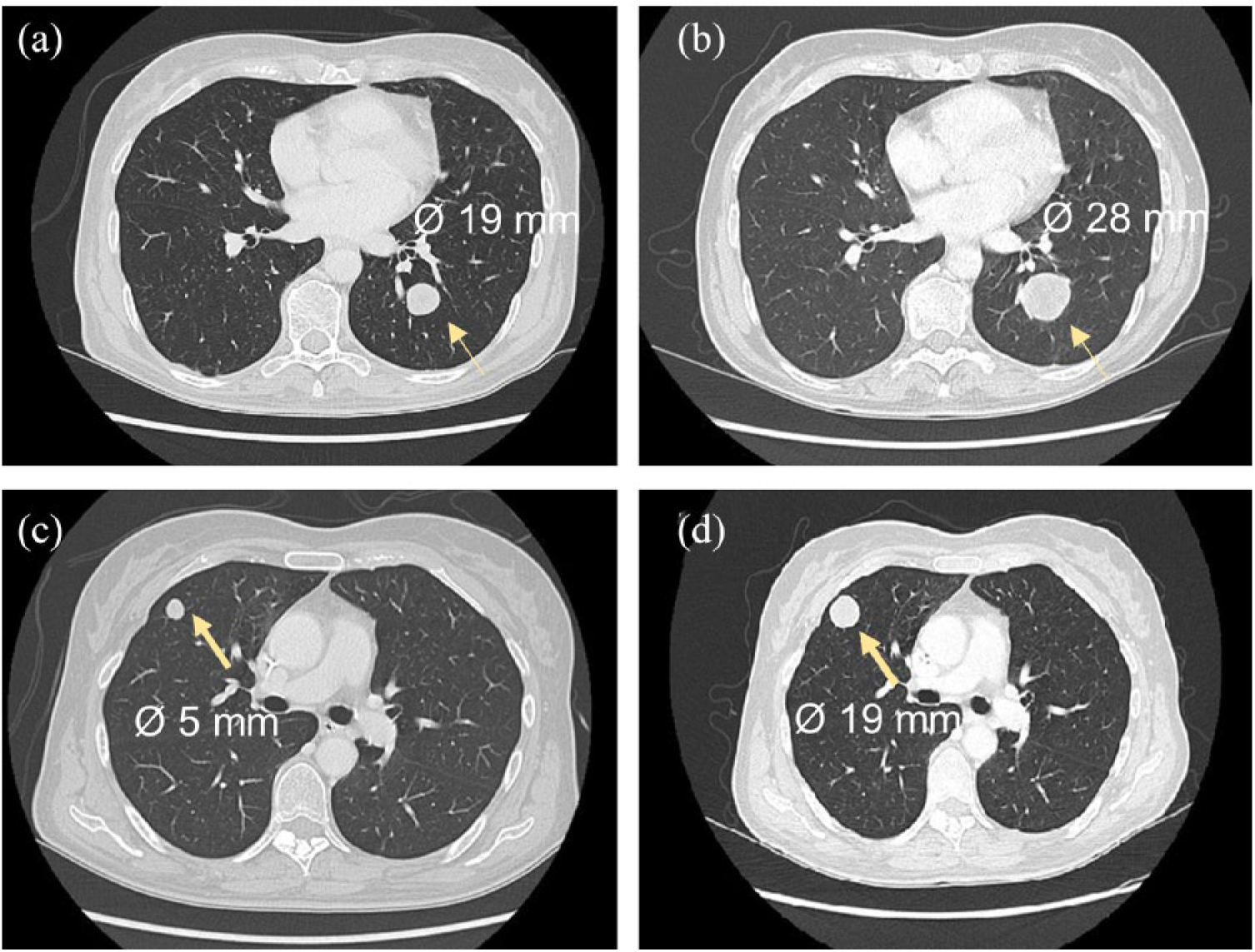
Computed tomography scan of the lung metastases. Both (a) and (c) indicate two nodular pulmonary lesions at the baseline of the second line trabectedin treatment, while (b) and (c) show the same lesions after two cycles of treatment.
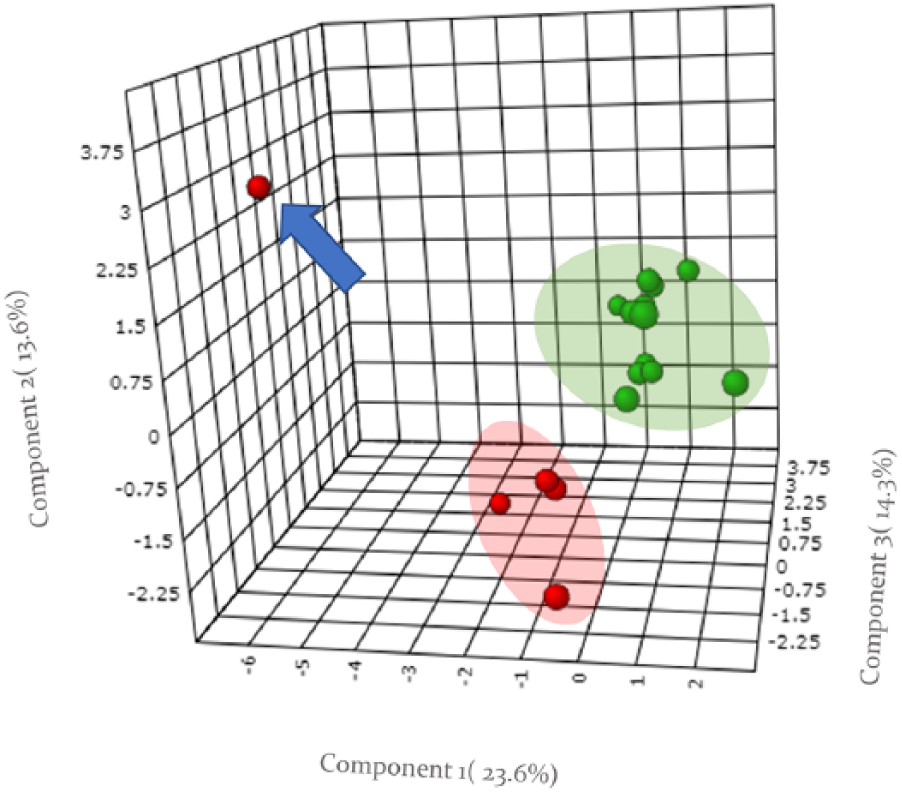
PLS-DA of VOCs profiles data for STS patients (red dots, n=5) and healthy individuals (green dots, n=12). The outlier VOCs profile, indicated by the blue arrow, corresponds to the STS case report patient. The real-time analysis of VOCs exhaled in the breath has been performed with select ion flow tube mass spectrometry (SIFT-MS) Voice 200UltraTM instrument (Syft Technologies Christchurch, New Zealand). The targeted VOCs panel (n=32) included: aliphatic alcohols (n=5, C1–C5), cyanuric acid, low chain aliphatic acids and their corresponding aldehydes (n=12, C1–C6) including aromatic benzaldehyde, sulfur ethers derivatives (n=5), and phenol related compounds (n=3). The low limit of quantification for each VOC investigated was of 1.0 part-per-billion (ppb) (v/v) while the measurement precision was less than 15% on single breath analysis. Patients were taught about “how to breathe” into the breath-inlet and every sampling session consisted of three single exhalations interspersed with no-breathing phases suitable for background sampling. The three background-corrected VOCs signals were averaged, giving the output VOCs concentration expressed as mean ppb. To avoid overestimation of isobaric molecules, specific ion adducts, generated by the H3O+, NO+, and O2+ ions were selected and monitored for each targeted VOC.
Results and discussion
The real-time analysis of the breath VOCs by SIFT-MS is a feasible bio-analytical methodology, which is easy to apply for monitoring cancer patients along their clinical therapeutic program. A targeted VOCs profile can be obtained in less than 5 minutes during the medical examination with high precision and accuracy. The real-time VOCs breath analysis was performed for all patients under fasting conditions between 9 a.m. and 12 p.m., and before the administration of the anticancer treatment. Compared with the other STS patients, the breath of the case report patient was characterized by a high concentration of ethanol, butanol alcohols, formic, acetic, propanoic and butanoic acids, and propionyl and benzyl aldehydes (Figure 3). The occurrence of ethanol in the breath of this patient may reflect an abnormally high glucose fermentation rate associated with the activity of gut microbiome. 5 Analogously, the occurrence of butanol could be ascribed to carbohydrates fermentation of specific bacteria, such as Clostridium acetobutylicum, and/or Streptococcus pneumoniae and Haemophilus influenzae. 6 Interestingly, breath butanol concentration was significantly higher in the breath of lung cancer patients, suggesting a potential role for butanol as a cancer biomarker. 7 It is worth noting that the case report patient’s condition is characterized by the pulmonary metastatic localization of the STS, which may directly influence the VOCs composition of her exhaled breath.
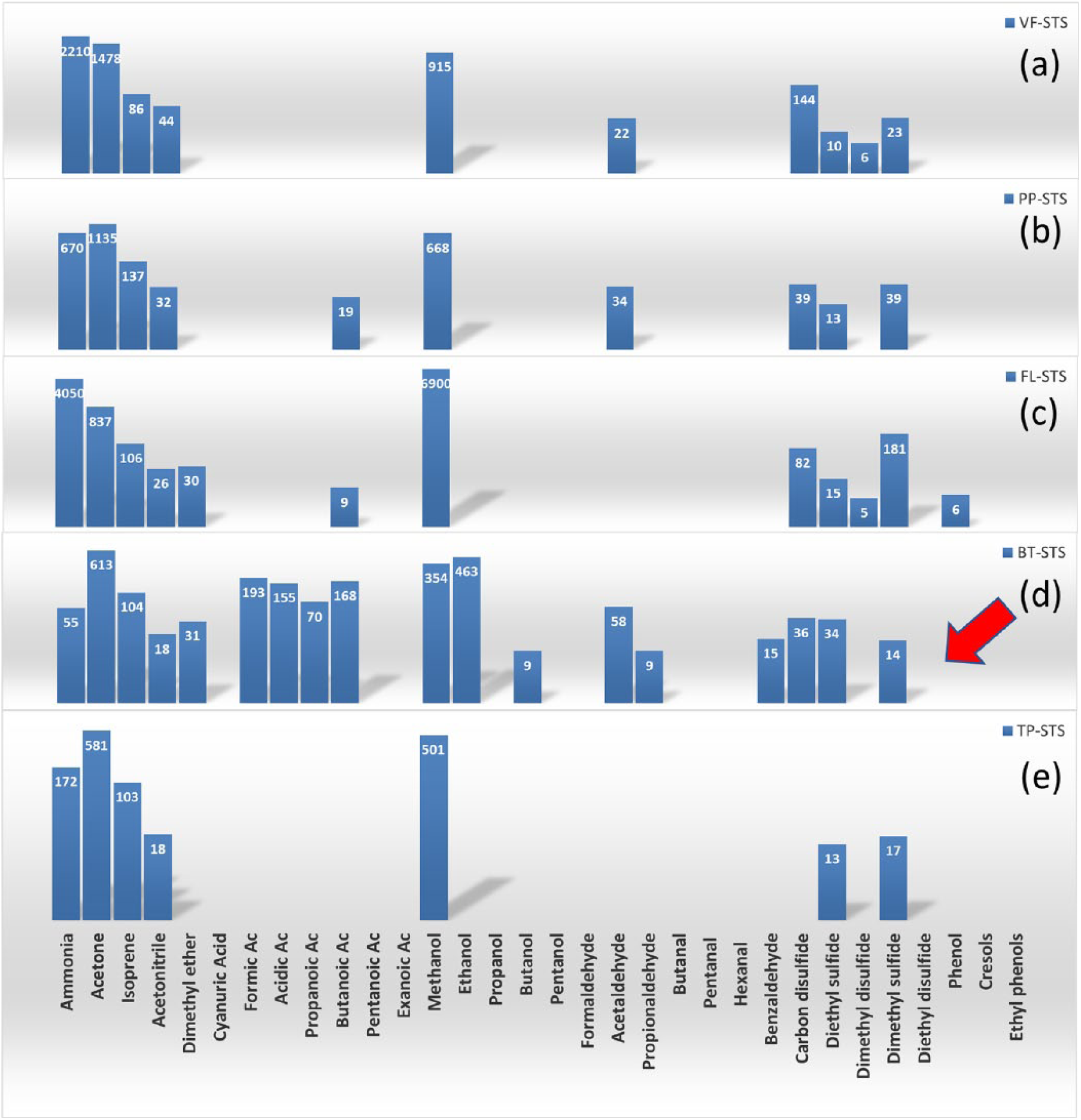
Breath VOCs profile of the case report patient compared with that of the other cancer STS patients. At the time of the breath VOCs analysis, all STS patients had metastatic disease. Two patients (including the case report patient) who had lung and bone lesions, or lung plus abdomen lesions, respectively, were treated with trabectedin as a second-line regimen (a), (d). One patient with bone and skin metastatic lesions received weekly paclitaxel as a first-line treatment (b). Two patients that had metastatic lung lesions, and lung plus abdomen lesions, respectively, received dacarbazine plus gemcitabine as a third-line treatment (c), (e). The red arrow indicates the profile of the case report patient. Inside the box the corresponding VOCs concentration, expressed as ppb(v/v), is shown. The log scale is applied to the y-axis.
The short-chain fatty acids (SCFAs), such as acetate, propionate, and butyrate, which were all observed in the VOCs profile of this case report patient, also represent metabolites derived from gut microbiome; changes in their levels have been shown to markedly influence the physiology and pathology of the host. Some SCFAs, such as butyrate and propionate, were found to inhibit the activity of histone deacetylases in colonocytes and immune cells, promoting the hyperacetylation of histones that indirectly influence the immune system as well as the metabolism of glucose and lipids. 8 Thus, the presence of these metabolites in the breath of this patient may reflect a differential microbiome activity that may harmfully influence both the immune system and the systemic host metabolism. Besides microbiome activity, the SCFAs occurrence in the breath also may derive from elevated aldehyde dehydrogenase activity, which is responsible for the oxidation of the cytotoxic aldehydes to carboxylic acids. In this context, the occurrence of propionyl and benzyl aldehydes, which are not detectable in the breath of the other patients and controls, constitutes a further specific metabolic feature of this case report patient. In general, the aldehydes are produced through the alcohol pathway metabolism and by the cytochrome p450 activity during the detoxification pathway of hydroperoxide, or generated, as secondary bioproducts, from lipid peroxidation. Tumor cells themselves can constitute a source of aldehydes since their high rate of cell proliferation produces high intracellular oxidative stress, which may increase the concentration of aldehydes in blood and, of course, in exhaled breath. Different studies seem to support this evidence by reporting high levels of breath aldehydes, such as propionyl and benzyl derivatives, in the breath of oral and lung cancer patients. For these aldehydes, this suggests a possible role as biomarkers of cancer disease.9,10 All of these specific qualitative and quantitative VOCs’ metabolic features attracted our attention since they appear representative of a particular and distinctive individual pathological and pharmacological phenotype. The specific VOCs profile observed in this patient was not linked to intestinal malfunction symptoms or to dietary aspects. Indeed, the clinical history of this patient revealed an intrinsic resistance to all previously performed cancer treatments, and experienced marked cancer disease progression in contrast to others STS patients, who achieved a partial response or cancer disease stability. Such treatment failures could be associated with both tumor and/or individual characteristics. The snapshot features of the VOCs’ measurement, as performed in this study, did not enable us to establish if the observed specific VOCs profile was present at the baseline of the treatments or was due to the cancer disease progression. However, in spite of this limitation, the observations generated by this case report indicate that the real-time analysis of the exhaled VOCs by SIFT-MS may be a promising feasible and non-invasive approach to distinguish patients with specific phenotypes along their clinical journey from cancer diagnosis to treatment. The further establishment of the effective role of VOCs as signatures in cancer disease monitoring may be particularly useful for STS where only cost-effective diagnostic tools, such as positron emission tomography and CT, are available.
Footnotes
Acknowledgements
The authors would like to thank all cancer patients and the individuals who participated in this study, and Leslie Sonnenschein for the English editing of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by 5 x mille CRO intramural Grant.
Ethical approval
The investigations obtained ethical approval from the Medical Research Ethics Committee of the Centro di Riferimento Oncologico, IRCCS, Aviano, Italy. All the participants in the study were asked to accept and sign the written informed consent.
