Abstract
Background
Urinary volatile organic compounds can accurately reflect individual exposure levels. This study investigated the association between urinary volatile organic compounds and cancer from both single- and combined-exposure perspectives and identified the volatile organic compounds most strongly associated with cancer.
Methods
This cross-sectional study analyzed data from 6056 US adults from the National Health and Nutrition Examination Survey 2011–2018 cycles, including 546 individuals with cancer. Logistic regression and restricted cubic spline models were used to evaluate associations for individual volatile organic compound exposures. Subgroup and interaction analyses were conducted to assess differences among populations. Least absolute shrinkage and selection operator, weighted quantile sum, and Bayesian kernel machine regression models were applied to evaluate mixture associations and to identify volatile organic compounds most strongly associated with cancer.
Results
Nine urinary volatile organic compound metabolites were significantly associated with cancer. Among them, N-acetyl-S-(3,4-dihydroxybutyl)-L-cysteine, a metabolite of 1,3-butadiene, and 2-aminothiazoline-4-carboxylic acid, a metabolite of cyanide, exhibited nonlinear dose–response relationships. Subgroup and interaction analyses indicated that age and body mass index were important effect modifiers of these associations. Mixture exposure analyses revealed that higher overall volatile organic compounds exposure levels were associated with a higher prevalence of cancer, with N-acetyl-S-(3,4-dihydroxybutyl)-L-cysteine contributing most strongly to the observed association.
Conclusion
This study provides epidemiological evidence of an association between urinary volatile organic compounds and cancer, with N-acetyl-S-(3,4-dihydroxybutyl)-L-cysteine consistently showing the strongest association. These findings highlight the potential public health relevance of exposure to specific volatile organic compounds.
Introduction
Cancer poses a serious threat to human health. In 2022, an estimated 20 million new cancer cases and 9.7 million cancer-related deaths were reported worldwide. 1 Among the numerous factors associated with the occurrence of cancer, the role of hazardous air pollutants (HAPs) has gained increasing attention. 2 Volatile organic compounds (VOCs) are a major component of HAPs and are ubiquitous in daily life, arising from natural products, food processing and cooking emissions, personal care and cleaning products, and everyday items such as furniture, pharmaceuticals, and plastics. 3 Several studies have confirmed an association between specific VOCs and cancer. Chlorinated compounds, such as vinyl chloride, are associated with digestive and urinary system malignancies. 4 Aldehydes present in cooking fumes, including acrolein and crotonaldehyde, have been linked to lung cancer in women. 5 Benzene is a well-established risk factor for hematologic malignancies, including leukemia, lymphoma, and multiple myeloma. 6
Inhalation is the primary route of human exposure to VOCs. VOCs can readily diffuse through the alveoli and enter systemic circulation. 7 In addition, dermal contact has been recognized as a potential exposure route for certain VOCs. Most VOCs are catalyzed in the liver by cytochrome P450 enzymes to form reactive intermediates, which mediate their toxic effects. 8 The carcinogenic mechanisms of VOCs may involve the synergistic action of multiple biological pathways, including DNA damage,9,10 oxidative stress,11,12 and pro-inflammatory responses.13,14 To protect the body from such reactive intermediates, glutathione conjugates with these intermediates to form water-soluble metabolites, which are subsequently excreted in urine. Therefore, measuring stable VOC metabolites (mVOCs) in urine has become a reliable approach for assessing individual exposure levels.5,15
Previous epidemiological studies have primarily estimated individual exposure based on ambient concentrations of VOCs. 16 However, ambient concentrations reflect only the overall level of environmental pollution and do not account for individual metabolic variability, lifestyle factors, or behavioral patterns that may substantially influence actual exposure doses. 17 Most existing studies have focused on investigating the association between individual VOCs and cancer, whereas potential synergistic, antagonistic, or additive effects of mixed VOC exposures have been largely overlooked. 18 Epidemiological evidence on the association between mixed VOC exposures and cancer remains limited and warrants further investigation. The present study used data from four cycles (2011–2018) of the US National Health and Nutrition Examination Survey (NHANES). Urinary concentrations of mVOC were measured to evaluate the association between VOC exposure and cancer prevalence and to identify the components most strongly associated with cancer in the American population.
Methods
Study design and participants
This study was a cross-sectional analysis based on data from four cycles (2011–2018) of NHANES. We performed a secondary analysis using publicly available NHANES data. A total of 39,156 participants from the 2011–2018 cycles were initially considered. Participants were excluded sequentially as follows: first, individuals younger than 20 years (n = 16,539); second, those with missing information on cancer diagnosis (n = 1); third, those without urinary creatinine measurements (n = 1476); and finally, those lacking urinary mVOC data (n = 15,084). After applying these exclusion criteria, the remaining 6056 participants were included in the final analysis (Figure 1). Cancer diagnosis data were obtained from the medical conditions questionnaire (MCQ), and urinary mVOC measurements were obtained from NHANES laboratory files. These datasets were linked using the unique participant identifier (SEQN). The study protocol was approved by the National Center for Health Statistics Ethics Review Board and was conducted in accordance with the Declaration of Helsinki 1975, as revised in 2024. All participants provided written informed consent. All data were fully deidentified, and no individual could be identified. The reporting of this study conforms to the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 19

Flowchart of participant inclusion and exclusion in the 2011–2018 NHANES study population.
Exposure variables
VOCs were selected as the primary exposure indicators in this study. All VOC measurements were obtained from the publicly available NHANES laboratory datasets. Detailed analytical procedures and quality control protocols are available on the NHANES website and have been described in previous studies. 20 To ensure the validity of the analysis, only VOCs with detection rates greater than 80% were included. Ultimately, 15 VOCs were included in this study, and their chemical names, precursor compounds, and detection rates are presented in Table S1. For concentrations below the limit of detection (LOD), values were imputed according to the NHANES recommendations, whereby measurements below LOD were replaced with LOD divided by the square root of two (LOD/√2). Data were processed prior to the analysis. First, metabolite concentrations were standardized according to the urinary creatinine level to eliminate the influence of urine dilution on the results. Subsequently, these standardized values were log-transformed to reduce skewness, stabilize variance, and meet regression model assumptions.
Cancer assessment
Cancer status was determined based on participants’ responses to MCQ. Participants were classified as having cancer if they answered “yes” to MCQ220 (Have you ever been told by a doctor or other health professional that you had cancer or a malignancy?). For those who reported a history of cancer, additional information on cancer type was obtained from MCQ230a–MCQ230d, which recorded up to four self-reported cancer diagnoses.
Covariates
A range of potential covariates was included in the analyses to control for confounding bias: (a) sociodemographic variables, including sex, age, race, marital status, and education level; and (b) health-related variables, including body mass index (BMI), smoking status (never/former/current), drinking status (never/low-to-moderate/heavy), hypertension (yes/no), and diabetes (yes/no). Smoking status was categorized as never (<100 cigarettes in a lifetime), former (>100 cigarettes in a lifetime and not currently smoking), and current (>100 cigarettes in a lifetime and currently smoking every day or occasionally). Participants were categorized as never drinkers, low-to-moderate drinkers (≤2 drinks/day for men and ≤1 drink/day for women), and heavy drinkers (>2 drinks/day for men and >1 drink/day for women) based on the average number of drinks consumed on drinking days during the past year. Hypertension was defined as a mean systolic blood pressure >140 mmHg, a mean diastolic blood pressure >90 mmHg, a self-reported physician diagnosis of hypertension, or current use of antihypertensive medication. Diabetes was defined as a fasting plasma glucose ≥7.0 mmol/L, glycated hemoglobin (HbA1c) ≥6.5%, a self-reported physician diagnosis of diabetes, or current use of insulin or oral hypoglycemic agents.
Statistical analysis
Given the complex sampling design of NHANES, which included stratification, clustering, and unequal probabilities of selection, sampling weights, strata, and primary sampling units were incorporated into the analyses to generate nationally representative estimates.
Participants were categorized into the cancer and the non-cancer groups according to cancer status. Categorical variables were presented as weighted frequencies and percentages, and continuous variables were reported as weighted means with SDs. Differences in baseline characteristics between groups were evaluated using survey-weighted chi-square tests for categorical variables and survey-weighted t-tests for continuous variables. To reduce potential selection bias and loss of statistical efficiency associated with complete-case analysis, missing covariate data were handled using multiple imputation. Pearson correlation analysis was performed to assess potential multicollinearity among urinary mVOCs.
Two weighted logistic regression models were constructed to evaluate the association between each urinary VOC and cancer. Model 1 was unadjusted. Model 2 adjusted for covariates including sex, age, race, marital status, education level, BMI, smoking status, drinking status, hypertension, and diabetes. To account for comparisons arising from the evaluation of multiple urinary mVOCs, p-values were adjusted using the false discovery rate (FDR) method based on the Benjamini–Hochberg procedure. Restricted cubic spline (RCS) models with three knots were used to examine potential nonlinear associations, and nonlinearity was assessed using Wald tests. To further evaluate a potential threshold effect, a segmented logistic regression model was applied. The inflection point was estimated using an iterative algorithm implemented in the segmented package, which identifies the breakpoint by maximizing model fit. Subgroup analyses were conducted to explore potential effect modification by demographic and clinical characteristics.
Least Absolute Shrinkage and Selection Operator (LASSO) regression was used to identify the most significant exposure variables. 21 The optimal penalty parameter (λ) was determined using 10-fold cross-validation. The final model was selected based on the one-standard-error rule based on the minimum mean squared error (MSE). 22 Subsequently, weighted quantile sum (WQS) regression was applied to assess mixture exposure and to determine the contribution of each component. 23 Finally, to further explore the complex interactions, the Bayesian kernel machine regression (BKMR) was applied. This model can not only evaluate the overall mixed effects but also simultaneously analyze the effects of individual exposures and the interaction relationships among them. 24 All analyses were performed using R software (version 4.3.2). Two-sided p-values <0.05 were considered statistically significant.
Results
Study population characteristics
Table 1 presents the baseline characteristics of the 6056 participants included in the study from 2011 to 2018. Participants were categorized into the cancer and non-cancer groups, with 546 individuals reporting a cancer diagnosis. Statistically significant differences between the two groups were observed for age, race, marital status, smoking status, drinking status, diabetes, and hypertension.
Baseline characteristics of study participants.
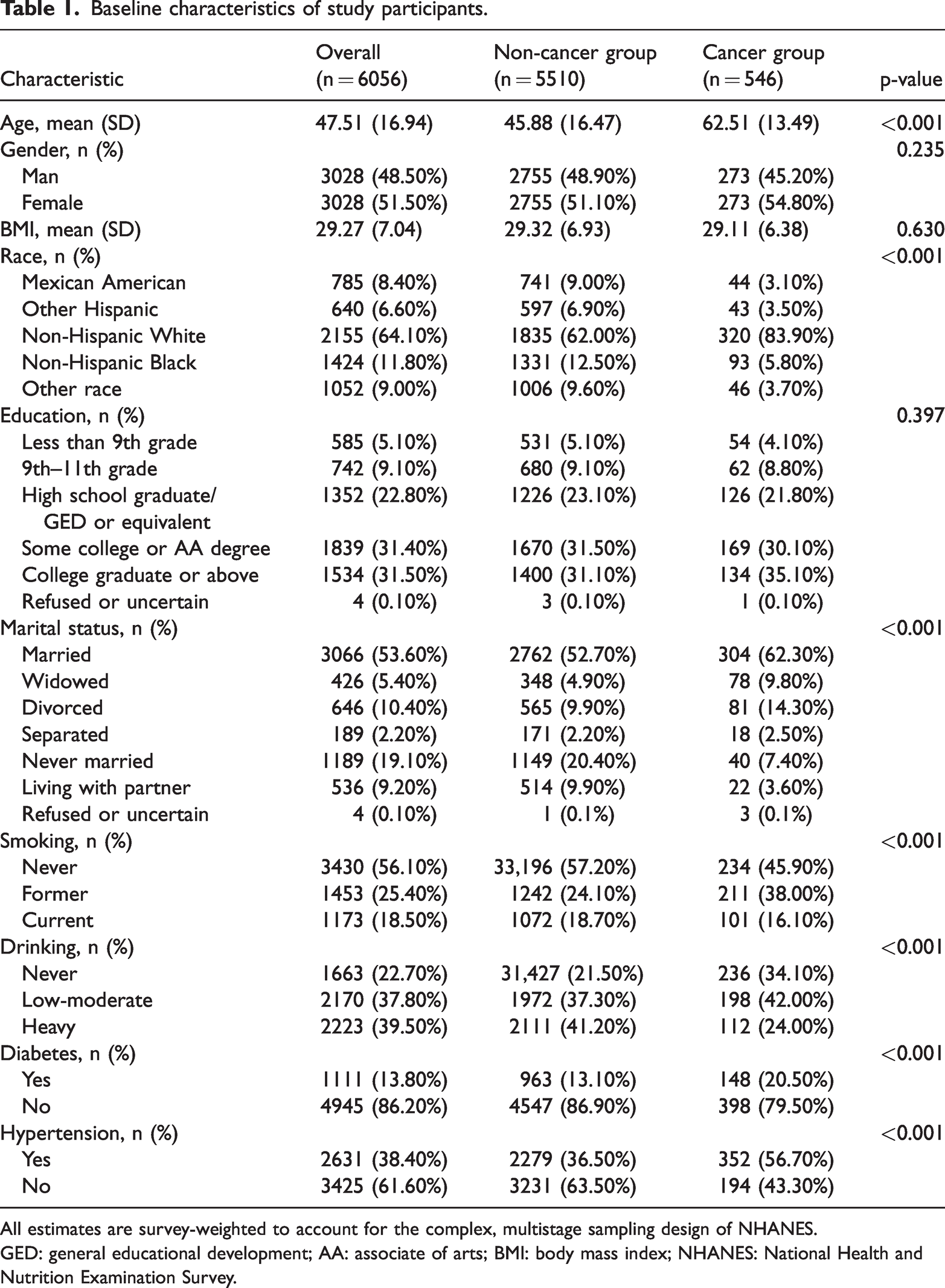
All estimates are survey-weighted to account for the complex, multistage sampling design of NHANES.
GED: general educational development; AA: associate of arts; BMI: body mass index; NHANES: National Health and Nutrition Examination Survey.
Distribution characteristics and interrelationships of urinary VOCs
Tables S2 and S3 show that three metabolites with the highest urinary concentrations were N-acetyl-S-(3,4-dihydroxybutyl)-L-cysteine (DHBMA), a metabolite of 1,3-butadiene; N-acetyl-S-(3-hydroxypropyl)-L-cysteine (3HPMA), a metabolite of acrolein; and N-acetyl-S-(3-hydroxypropyl-1-methyl)-L-cysteine (HMPMA), a metabolite of crotonaldehyde. Table S4 and Figure S1 further demonstrate that several metabolites, including AMCC (a metabolite of N,N-dimethylformamide), SBMA (a metabolite of toluene), CEMA (a metabolite of acrolein), DHBMA (a metabolite of 1,3-butadiene), MA and PGA (metabolites of ethylbenzene and styrene), MHBMA3 (a metabolite of 1,3-butadiene), and HMPMA (a metabolite of crotonaldehyde), were significantly higher in participants with cancer (p < 0.05). Pearson correlation analysis indicated strong positive inter-metabolite relationships (Figure S2), particularly between 2MHA and 3,4MHA (r = 0.83); MHBMA3 and HMPMA (r = 0.76); and 3HPMA and HMPMA (r = 0.70).
Associations of individual VOC exposure with cancer
Figure 2 summarizes the associations between 15 urinary mVOC and cancer based on logistic regression models. In the unadjusted model (Model 1), 10 metabolites, including AAMA, AMCC, CEMA, DHBMA, 3HPMA, MA, MHBMA3, PGA, and HMPMA, were positively and significantly associated with cancer. After adjusting for multiple confounding factors in Model 2, the association between AMCC and cancer was no longer statistically significant, whereas CYMA showed a significant positive association. To account for multiple comparisons, FDR correction was applied. After FDR adjustment, associations for several metabolites, including 3HPMA, CEMA, AAMA, MA, HMPMA, and CYMA (Table S5), remained statistically significant, whereas the associations for other metabolites were attenuated. RCS analysis (Figure 3) demonstrated nonlinear relationships between 2-aminothiazoline-4-carboxylic acid (ATCA) and cancer and between DHBMA and cancer (p for nonlinearity < 0.05). When ATCA levels exceeded 0.49, a significant positive association with cancer was observed (odds ratio (OR) = 1.43, 95% confidence interval (CI): 1.03–1.97, p < 0.001; Table S6). In contrast, a significantly higher prevalence of cancer was observed among participants with DHBMA levels below 0.81 (OR = 7.04, 95% CI: 1.97–36.09, p = 0.008; Table S6). Subgroup analyses (Tables S7–S13) indicated that age (<65 vs. ≥65 years), BMI (<25 vs. ≥25), smoking status (never/former/current), and drinking status (never/low-to-moderate/heavy) significantly modified the associations between urinary VOCs and cancer (p for interaction < 0.05). In contrast, the associations were consistent across subgroups defined by sex, diabetes, and hypertension.

Weighted logistic regression models showing associations between VOCs and cancer. All metabolites were creatinine-adjusted and log-transformed. Model 1 was unadjusted, whereas Model 2 was further adjusted for sex, age, race, marital status, education level, body mass index (BMI), smoking status, drinking status, hypertension, and diabetes. VOCs: volatile organic compounds.

Restricted cubic spline curves depicting nonlinear associations between VOCs and cancer. All metabolites were creatinine-adjusted and log-transformed. All regression models were adjusted for potential confounders, including sex, age, race, marital status, education level, body mass index (BMI), smoking status, drinking status, hypertension, and diabetes. VOCs: volatile organic compounds.
Association of VOC mixture exposure with cancer
Pearson correlation analysis suggested multicollinearity among several urinary VOCs. To address this issue, LASSO regression was applied (Figure 4), and DHBMA was identified as the metabolite most strongly associated with cancer. WQS regression (Table 2) showed a positive association between the VOC mixture and cancer (OR = 1.61, 95% CI: 1.42–1.83, p < 0.001). This association remained significant after adjustment for confounders (OR = 1.26, 95% CI: 1.03–1.56, p = 0.033). Figure 5 presents the contribution weights of each metabolite, with DHBMA showing the largest contribution, accounting for 57.1% in Model 1 and 20.60% in Model 2. The BKMR model was used to further analyze the association between the 15-mVOC mixture and cancer prevalence. Higher overall mixture concentrations were associated with progressively higher prevalence of cancer (Figure 6). DHBMA was identified as the key contributor based on its high posterior inclusion probability (PIP) value (Table S14). Its significant effect was further highlighted in univariate analyses, in which other VOCs were fixed at different percentiles (Figure S3). Additionally, the dose–response curve indicated a nonlinear association for DHBMA (Figure S4), and no significant interactions were observed among the VOCs (Figure S5).

LASSO regression for identifying key VOCs associated with cancer. (a) Coefficient profiles of 15 urinary VOCs across a sequence of penalty parameters (log λ). As the penalty increases, less relevant variables shrink toward zero. (b) Ten-fold cross-validation used to select the optimal λ. The left dotted line indicates the value with minimum binomial deviance, and the right dotted line represents the one-standard-error (1-SE) criterion. LASSO: least absolute shrinkage and selection operator; VOCs: volatile organic compounds.
WQS regression of the association between VOC mixture exposure and cancer.

The concentrations of urinary mVOCs were corrected for urinary creatinine and log-transformed (log (ng/mg Cr)). Model 1 was unadjusted, whereas Model 2 was further adjusted for sex, age, race, marital status, education level, body mass index (BMI), smoking status, drinking status, hypertension, and diabetes.
CI: confidence interval; OR: odds ratio; VOCs: volatile organic compounds; mVOCs: volatile organic compound metabolites; WQS: weighted quantile sum.

Weighted quantile sum (WQS) regression showing the relative contribution of individual VOCs to cancer. All metabolites were creatinine-adjusted and log-transformed. (a) Contribution weights of 15 urinary VOCs estimated without adjustment for covariates. (b) Contribution weights estimated after adjusting for sex, age, race, marital status, education level, body mass index (BMI), smoking status, drinking status, hypertension, and diabetes. VOCs: volatile organic compounds.

Overall effect of VOCs mixture exposure on cancer estimated using the BKMR model. All metabolites were creatinine-adjusted and log-transformed. Models were adjusted for sex, age, race, marital status, education level, body mass index (BMI), smoking status, drinking status, hypertension, and diabetes. BKMR: Bayesian kernel machine regression; VOCs: volatile organic compounds.
Discussion
This study used a nationally representative sample of 6056 adults from the 2011 to 2018 cycles of the NHANES to systematically evaluate the association between VOC exposure and cancer. The results demonstrated that multiple individual mVOC were significantly associated with higher prevalence of cancer. Additionally, the analyses of VOCs mixture exposure revealed a dose–response relationship, with higher overall VOC exposure corresponding to greater odds of cancer. Notably, DHBMA consistently showed the strongest association with cancer prevalence among the metabolites examined.
This study found that elevated urinary levels of AAMA, SBMA, CEMA, DHBMA, 3HPMA, MA, MHBMA3, PGA, and HMPMA were significantly associated with increased prevalence of cancer. AMCC demonstrated statistical significance only in the univariate regression analysis, whereas CYMA showed a significant association exclusively in the multivariable regression model. AMCC is a major metabolite of dimethylformamide (DMF). Studies have reported increased mortality from liver and lung cancers among workers with long-term exposure to high concentrations of DMF. 25 Animal studies have also demonstrated its hepatocarcinogenic potential. 26 Although the association between AMCC and cancer was not confirmed in the multivariate model, its potential carcinogenicity cannot be excluded because of possible residual confounding. In 2024, the International Agency for Research on Cancer (IARC) classified acrylonitrile as a Group 1 human carcinogen based on sufficient epidemiological evidence linking it to lung cancer and clear carcinogenic effects in animal studies. 27 The multivariable regression model in our analysis showed significant positive association between CYMA (a metabolite of acrylonitrile) and cancer, suggesting that its potential carcinogenic effect may become apparent only after adjustment for confounding factors. This finding is consistent with the latest risk assessment by IARC. Based on consistent findings from LASSO, BKMR, and WQS models, DHBMA was identified as the mVOC most strongly associated with cancer, which is likely closely related to the high levels of the parent compound 1,3-butadiene. Notably, 1,3-butadiene is classified as an IARC Group 1 carcinogen, and ethylene oxide is also recognized as a Group 1 carcinogen, underscoring the public health importance of cumulative exposure to carcinogenic VOCs. Major sources of 1,3-butadiene include automobile exhaust, biomass combustion, and tobacco smoke. 28 Its toxicity in humans mainly results from the highly reactive intermediate metabolites formed during hepatic metabolism. 29 Under the action of cytochrome P450 enzymes, 1,3-butadiene can metabolizes into genotoxic and mutagenic metabolites such as 3,4-epoxy-1-butene (EB), 1,2,3,4-diepoxybutane (DEB), 3-butene-1,2-diol (BDD), and 3,4-epoxybutane-1,2-diol (EBD),28,30 among which DEB exhibits the highest toxicity.31,32 MHBMA3 is another terminal metabolite in the 1,3-butadiene metabolic pathway, and its urinary concentration was substantially lower than that of DHBMA in our study, suggesting that DHBMA may better reflect the overall internal exposure. Animal studies have shown that even low-level exposure to 1,3-butadiene can induce tumors in multiple organs in mice. 33 A large cohort study of rubber industry workers also reported a significant positive exposure–response relationship between cumulative 1,3-butadiene exposure and leukemia risk. 34
In the real-world settings, individuals are simultaneously exposed to multiple VOCs. This study applied the WQS and BKMR models, which more accurately reflect mixed exposures. Both approaches consistently showed a significant positive dose–response relationship between VOC mixtures and cancer. The WQS model is particularly useful for assessing the overall effect of highly correlated exposures, 35 whereas BKMR is more suitable for identifying nonlinear relationships and interactions. 36 In the BKMR model, the univariate exposure–response curve for DHBMA exhibited a pronounced nonlinear pattern, consistent with the findings from RCS analysis, further supporting the pivotal role of DHBMA. The link between VOCs and cancer is supported by multiple biological mechanisms. DNA damage is considered one of the most direct pathogenic pathways. VOCs can form DNA–protein crosslinks that disrupt DNA replication and repair, thereby increasing the risk of mutations. 37 Oxidative stress is another critical mechanism for VOC-induced DNA damage. VOC exposure has been shown to correlate in a dose-dependent manner with urinary biomarkers of oxidative damage, including 8-oxo-guanosine, 8-oxo-2′-deoxyguanosine, and 8-oxoguanine. 38 Another study further demonstrated that urinary levels of CEMA, HMPMA, CMEMA, and AAMA were significantly and positively associated with markers of oxidative DNA damage, including 8-hydroxy-2'-deoxyguanosine and 8-hydroxy-guanosine. 39 In addition to directly damaging DNA, VOCs may also promote tumor occurrence through epigenetic modifications. High-level exposure can increase the methylation of key gene promoters, including DNA topoisomerase IIα (TOP2A), superoxide dismutase 1 (SOD1), and tumor necrosis factor-α (TNF-α). 40 Collectively, these findings suggest that VOCs disrupt cellular homeostasis through multiple synergistic pathways, including direct genotoxicity, oxidative stress, chronic inflammation, and epigenetic alterations, all of which play crucial roles in cancer initiation and progression. 41
Subgroup analyses revealed a stronger association between VOC exposure and cancer among individuals younger than 65 years. This age-related difference may be attributed to higher metabolic activity and more frequent exposure-related behaviors in younger individuals. Moreover, the presence of multiple competing health conditions in older adults may partially obscure independent associations between VOC exposure and cancer prevalence. With respect to BMI, this association appeared to be more pronounced among individuals with BMI <25 kg/m2. This observation is consistent with previous studies reporting positive associations between VOC exposure and both visceral adiposity index (VAI) and lipid accumulation product (LAP), with stronger effects observed in non-obese individuals. 42 This may indicate greater susceptibility to lipid metabolic disruption and environmental toxicant effects in normal-weight populations. Although no significant interaction was observed for most VOCs, statistically significant interactions between smoking status and several VOCs (including AMCC, CEMA, CYMA, and MA) were identified with respect to cancer prevalence (p for interaction < 0.05). Overall, the associations between these VOC exposures and cancer prevalence were stronger among current smokers compared with never-smokers.
Despite using nationally representative data from NHANES and applying multiple statistical models to systematically evaluate the associations between urinary VOCs and cancer, this study has several limitations. First, the cross-sectional design limits causal inference. Second, although the NHANES MCQ provides information on cancer type (including melanoma and other site-specific malignancies) and timing of diagnosis, the relatively small number of cancer cases and the substantial heterogeneity across cancer types necessitated focusing on overall cancer as the primary outcome. Notably, previous NHANES-based studies have reported associations between VOC exposure and skin cancer, including melanoma, highlighting the need for future studies with larger sample sizes to explore cancer-specific associations. 43 Third, mVOCs were measured from a single spot urine sample that reflects only recent exposure. Most mVOCs have relatively short biological half-lives, with the majority eliminated within 24 h or less, limiting the ability of a single measurement to capture long-term or cumulative exposure. Additionally, this measurement may be influenced by individual hydration status, metabolic variability, and behavioral differences. Finally, the 15 metabolites included in the analysis were selected based on detection rates, which may have excluded other potentially toxic VOCs, possibly resulting in an underestimation of overall exposure.
Conclusion
This study demonstrated that multiple VOCs were positively associated with cancer prevalence, with DHBMA consistently showing the strongest association across analytical models. The use of urinary VOCs rather than environmental concentrations as exposure indicators enables more accurate quantification of individual intake, directly reflecting the total absorbed internal dose. By integrating multiple statistical approaches, this study provides exploratory evidence of potential mixture effects and nonlinear exposure–response relationships of VOCs. Future prospective studies incorporating mechanistic validation are warranted to strengthen the scientific validity of causal inference.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605261427143 - Supplemental material for Associations of volatile organic compounds with cancer: A cross-sectional analysis of single and mixture exposures
Supplemental material, sj-pdf-1-imr-10.1177_03000605261427143 for Associations of volatile organic compounds with cancer: A cross-sectional analysis of single and mixture exposures by Chenhui Lv, Jianxiang Ge, Hao Chen and Qijia Xuan in Journal of International Medical Research
Footnotes
Acknowledgments
Not applicable.
CRediT authorship contribution statement
Chenhui Lv: Conceptualization, Methodology, Investigation, and Writing – Original Draft Preparation. Jianxiang Ge: Visualization, Software, and Methodology. Hao Chen: Formal analysis. Qijia Xuan: Writing – Review & Editing, Visualization, and Validation.
Consent for publication
All authors have read and approved the published version of the paper.
Declaration of competing interest
The authors declare that they have no conflicts of interest.
Ethics approval and consent to participate
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
