Abstract
BACKGROUND:
Detecting volatile organic compounds (VOCs) could provide a rapid, noninvasive, and inexpensive screening tool for detecting cancer.
OBJECTIVE:
In this systematic review, we identified specific exhaled breath VOCs correlated with lung, colorectal, and breast cancer.
METHODS:
We identified relevant studies published in 2015 and 2016 by searching Pubmed and Web of Science. The protocol for this systematic review was registered in PROSPERO and the PRISMA guidelines were used in reporting. VOCs and performance data were extracted.
RESULTS:
Three hundred and thirty three records were identified and 43 papers were included in the review, of which 20 were review articles themselves. We identified 17 studies that listed the VOCs with at least a subset of statistics on detection cutoff levels, sensitivity, specificity, area under the receiver operating characteristic curve (AUC), and gradient.
CONCLUSIONS:
Breath analysis for cancer screening and early detection shows promise, because samples can be collected easily, safely, and frequently. While gas chromatography-mass spectrometry is considered the gold standard for identifying specific VOCs, breath analysis has moved into analyzing patterns of VOCs using a variety of different multiple sensor techniques, such as eNoses and nanomaterials. Further development of VOCs for early cancer detection requires clinical trials with standardized breath sampling methods.
Keywords
Introduction
Hippocrates, considered to be the father of medicine, taught his students to smell the breath of their patients as an indicator of disease in about 400 years BC [1]. In 1971, Linus Pauling and co-workers reported that normal human breath contains a complex mixture of several hundred volatile organic compounds (VOCs) [2] and recently scientists from the Max Planck Institute for Chemistry have observed differing VOC levels in movie audiences depending upon the emotional content of the film [3]. VOCs are a diverse group of chemicals emitted from the human body that often reflect the metabolic condition of the person [4]. Concentrations of specific organic compounds in the exhaled breath of cancer patients are thought to be the result of oxidative stress, abnormal metabolic processes, or an imbalance between the systematic manifestations of reactive oxygen species and a biological system’s ability to detoxify or repair damage [4, 5, 6]. Some VOCs are thought to originate from cancer cells as a result of abnormal metabolism that does not exist in healthy people [5, 7].
Lung, breast, and colorectal cancer are among the top five cancers in terms of both incidence and mortality. Lung cancer has the highest mortality rate of these; in 2016, it is estimated that 224,000 Americans will be diagnosed with lung cancer, and 158,000 will die of it. Breast cancer incidence is estimated at 249,000, with 41,000 deaths. And colorectal cancer is estimated to strike 134,000 Americans and to kill 49,000. Improved screening may reduce mortality for all of these cancers [8] by detecting cancer when it is small and more easily treated. Although there are a variety of techniques available to detect some cancers at an early stage, many are considered by patients to be uncomfortable and inconvenient and are often underutilized by large segments of the population for a variety of reasons, such as access and patient burden [9].
While cancer screening biomarkers have been developed using biological samples such as blood and urine, detecting specific levels of VOCs as cancer biomarkers holds the potential to provide a low patient burden, rapid, noninvasive, and inexpensive cancer screening tool [10, 11, 12]. Breath samples have elicited interest because the samples are easy to collect and analyze, and the literature in this area continues to grow [4, 5, 6, 7, 9, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55]. Testing exhaled breath is painless and noninvasive, and therefore suitable even for children and critically ill patients. Because exhaled breath tests can be performed frequently, they could also be helpful in monitoring cancer to catch recurrence or signs of progression early [9].
Van de Kant et al. provided a systematic review of VOCs in pulmonary diseases in 2012 [56]. In 2015, Krilaviciute et al. completed an extensive, detailed systematic review of cancer detection through exhaled breath [29], including 73 studies. This study found that analyzing exhaled breath is promising, but standardization of breath collection, sample storage and data handling are required in larger studies.
The science of detecting disease through exhaled breath has grown exponentially in the past few years with many technology teams developing new sensor techniques and devices. The aim of the following systematic review was to identify specific exhaled breath VOCs correlated with lung, colorectal, and breast cancer, along with reported detection levels, sensitivity, specificity, area under the receiver operating characteristic curve (AUC), and the concentration gradient in cancer patients. We focused on studies published after the Krilaviciute 2015 review [29].
Methods
Protocol and registration
The protocol for this systematic review was registered in the PROSPERO [57] database (CRD420160 45513) and the PRISMA guidelines were used in reporting the review [58]. Funding was received from Nano Engineered Applications (NEA) [59].
Eligibility criteria
Inclusion criteria were for studies of human breath VOCs related to breast, colorectal, or lung cancer, as discussed in either original articles or review papers. Duplicate articles, articles not directly relevant to the topic, articles without abstracts, and articles not focused on testing in humans were excluded. Two reviewers (Davis and Oakley-Girvan) independently screened the studies for eligibility. Disagreements were resolved by discussion.
Information sources and search process
On June 10, 2016, we searched both the PubMed and Web of Science databases of scientific articles using the following search string:
((cancer OR carcinoma OR adenocarcinoma OR tumor OR malignancy OR malignant disease) AND ((volatile AND (compound OR compounds OR marker OR markers OR biomarker OR biomarkers)) OR (VOC OR VOCs OR breathprint OR breath-print OR breath print) AND (breath OR exhaled OR air))) Filters: LANGUAGE: (English) Publication Years: 2015 AND 2016.
Flow diagram for search strategy methods, reported according to the PRISMA statement. *outdoor/indoor air pollutants (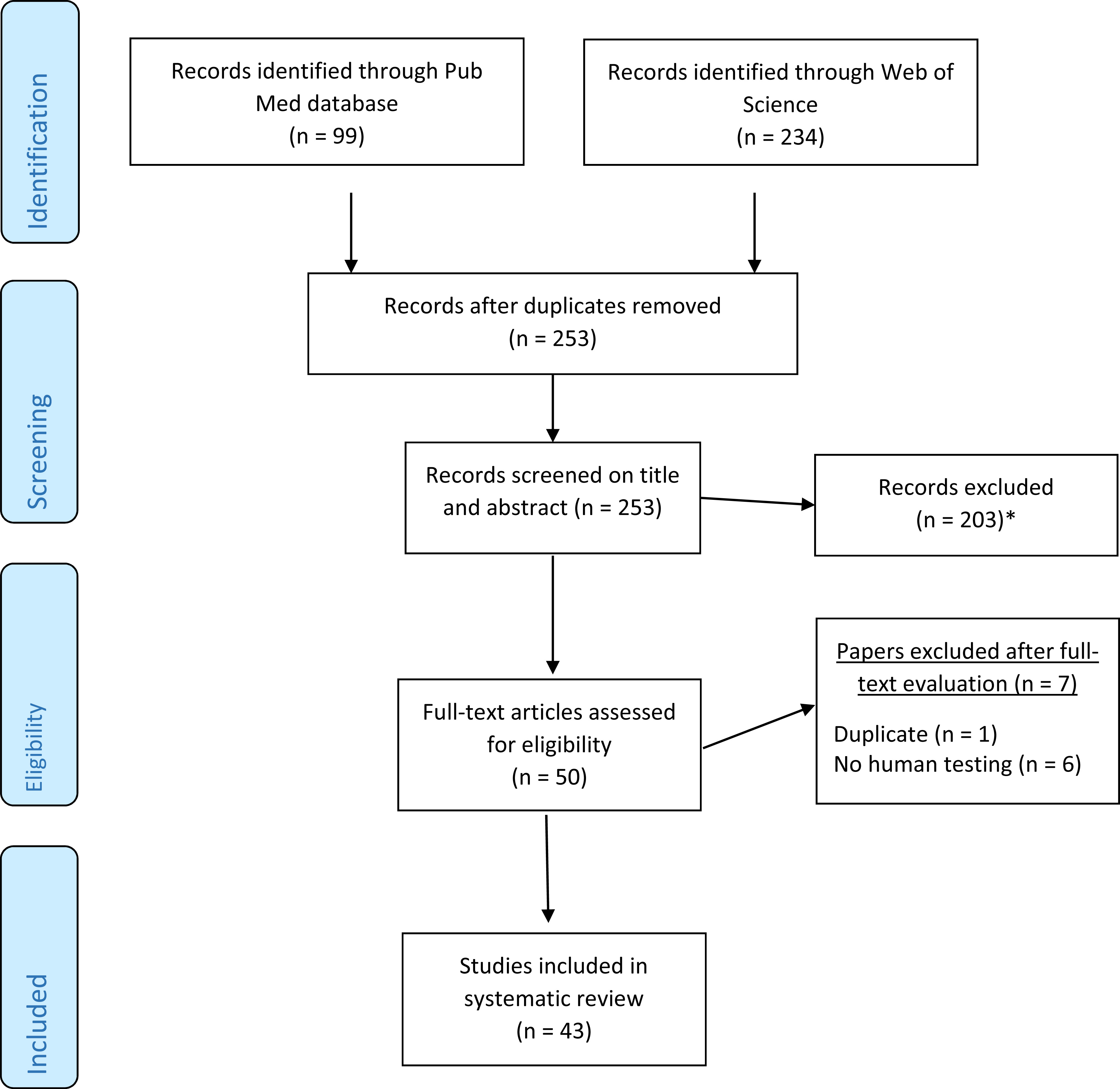
In total, 333 records were identified, 99 fromPubMed, and 234 from Web of Science, of which 80 were duplicates. We reviewed the remaining 253 titles and abstracts to identify studies relevant to the topic. Articles excluded from the review were categorized as shown in Fig. 1. We reviewed 50 full-text papers for inclusion; seven papers were excluded after full-text review. A total of 43 papers were included in the systematic review, of which 20 were review articles themselves, and 23 were studies of VOCs in humans related to lung (19), colorectal (2), or breast cancer (2).
Data collection process
The relevant data were extracted from the 43 selected papers by two reviewers (Davis and Oakley-Girvan). We extracted the following information using a predesigned data extraction form: first author, publication year, number of cases, number of controls, names of volatile compounds tested, cut-off level, sensitivity, specificity, area under the receiver operating characteristic curve (AUC), and gradient. For those papers that did not include this information, we collected the first author, publication year, number of cases, a number of controls, type of study, and the accuracy statistics provided by the study comparing cancer cases with healthy controls where available. Review papers were searched for the same data elements.
Lung cancer compounds, performance, and the concentration gradient in cancer patients compared with controls
Lung cancer compounds, performance, and the concentration gradient in cancer patients compared with controls
Breast cancer compounds, performance, and the concentration gradient in cancer patients compared with controls
Summary
Most of the studies that identified individual VOCs used gas chromatography-mass spectrometry (GC-MS), which is considered the gold standard because it allows metabolite identification that is transferable between instruments with different manufacturers [5]. The GC-MS technique is not only easily reproducible, it has high sensitivity and robustness for individual VOCs [60].
Proton transfer reaction mass spectrometry (PTR-MS) is another method for measuring volatile signatures [61, 62]. Fienberg et al. looked at the volatile signature in lung cancer before and after oral glucose tolerance testing using this method and found differences between lung cancer and control patients [25], but is not included in the table because his findings are not focused on detecting lung cancer and he did not provide the requisite performance statistics.
Unfortunately, only the minority of original studies that we reviewed listed individual VOCs and provided specific information about their performance (N
Of the studies that did test specific VOCs, all were conducted in a case-control approach, in which diagnosed patients were compared with healthy controls, or controls with noncancerous conditions. Most studies enrolled small numbers of participants and several included multiple comparison groups. Overall, moderate to very good diagnostic performance was reported for distinguishing healthy controls from patients [5, 29]. However, reports to date indicate there is no single VOC that would clearly determine the presence of cancer with high performance. Instead, the occurrence of several VOCs in combination was used to increase both overall sensitivity and specificity of cancer diagnosis through breath testing [6, 29, 31, 42].
Lung cancer studies
Of the 19 lung cancer studies our literature review identified, only five included the names of specific VOCs identified in cancer patients, along with most of the statistics required to complete Table 1. In the study conducted by Corradi et al., subjects with suspected lung cancer were enrolled and VOCs were combined and compared for those with a subsequent diagnosis against those with no diagnosis [21]; Schumer and colleagues [46] and Li et al. [30] compared combined VOCs from lung cancer patients with those with benign disease and with healthy controls. Ligor et al. compared combined VOCs in lung cancer cases and controls [31]. Fernandes et al. provided a list of VOCs in his review article along with the cutoff levels, but without further details on individual articles [26]. The number of patients enrolled in the studies ranged from 12 [64] to 156 [46].
Colorectal cancer compounds, performance, and the concentration gradient in cancer patients
Colorectal cancer compounds, performance, and the concentration gradient in cancer patients
Not all studies included the cut-off levels at which the VOC was considered to be abnormal, and not all included sensitivity, specificity, or AUC. A few did not indicate whether the VOC was higher or lower in cancer patients compared with healthy controls (gradient). The five studies prior to 2015 shown in Table 1 are from Krilaviciute et al. [29]. For some studies, the statistics on sensitivity, specificity and AUC was presented for all compounds combined, rather than each compound individually.
The studies not shown in Table 1 either did not provide a list of the VOCs identified, or did not provide any of the statistics required for this table. Four of these studies did list the VOCs [19, 38, 39, 65], but used different statistics to evaluate the accuracy of differences between patients and controls. Capuano et al. provided the percentage of identification for each of the VOCs [19], while Fu et al. utilized the P-value for the difference between lung cancer patients, controls, and benign nodules [65], Peralbo-Molina et al. used the P-value for the difference between lung cancer patients, smokers, and healthy controls [39], and Nardi-Agmon et al. provided the percentage of success in monitoring response to anticancer treatments, contrasting stable disease with partial response [38].
In our review, along with the studies identified by Krilaviciute et al. [29], we identified an additional study evaluating individual VOCs for breast cancer as shown in Table 2. This study in 2015 by Barash [16] recruited 23 healthy women, 13 with benign breast conditions, 10 with ductal carcinoma in situ (DCIS), and 80 with malignant disease across five different subtypes. We report the VOC statistics for the healthy versus the malignant patients in Table 3. Study sizes for the breast cancer compounds ranged from 10 [66] to 80 cases [16].
One of the two studies we identified that reported on human breath compounds used to identify breast cancer did not list the VOCs, although they reported high sensitivity and specificity in their comparison of breast cancer patients with healthy controls [33].
Colorectal cancer studies
Both of the colorectal cancer studies identified in this review listed individual VOCs and provided statistics on performance (see Table 3). Krilaviciute et al.’s 2015 review [29] only identified the 2013 study from Altomare [67], which was also listed in a review by Markar [32]. The final study listed in Table 3 by Peng [68], was found in Markar et al.’s review [32]. All of the colorectal cancer compound studies contrasted patients diagnosed with colorectal cancer compared with healthy controls, except for Altomare et al., 2015 [14], which compared 32 colorectal cancer patients who were disease-free at follow-up with 55 healthy controls. Case size for the studies ranges from 26 [68] to 65 [15].
Review articles
Our review identified 20 systematic review or over- view articles, almost half of the 43 articles we identified through our search strategy. We read the full text of each review article, and identified any prior publications in the review article that included the VOCs for lung, colorectal or breast cancer. While several of the review articles provided VOCs found in the studies that they reviewed, only Krilaviciute et al. [29] and Markar et al. [32] named the VOCs as well as provided statistics on sensitivity, specificity, cutoffs, AUC, and gradient. The topics of the review articles are shown in Table 4.
Discussion
A challenge we encountered in conducting this review was that the majority of studies did not include full statistical details on the VOCs that were evaluated. We identified 19 studies through our search criteria that focused on lung cancer, but only five provided the VOCs with statistics on detection cutoff levels, sensitivity, specificity, AUC, and gradient, and some provided only a subset of these performance statistics. Only one of the two breast cancer studies provided these statistics, but both of the colorectal cancer studies listed the desired information.
Moreover, we encountered a variety of different methods for detecting and reporting on VOCs. Studies that utilized electronic noses (eNoses) to detect VOCs did not provide a list of the VOCs. The eNose experiments were based on the canine olfactory system, which seeks a pattern of organic chemicals, sometimes known as “breath prints”, rather than individual chemicals. ENoses are an assembly of nonspecific sensors which respond to different odors by producing a complex signal [6]. They have the advantage of being less expensive, easier to use, providing more rapid results, lower operating costs, and greater portability along with flexibility in the sensor array, which can be adjusted for specialized applications [54]. However, eNoses cannot identify individual compounds in complex mixtures [6, 69]. ENose studies also utilized a variety of statistics to verify the accuracy of their results, including the percentage correctly identified (e.g., [19]) or cross-validated values (e.g., [23]). Although GC-MS is considered the gold standard for identifying individual VOCs, this technique is also slow, cumbersome and expensive [6].
Topics covered in review articles
Topics covered in review articles
Several studies identified in our review focused on testing newly developed technology to distinguish the VOC pattern found in individuals with specific diseases compared with healthy people. For example, there are several reports that nanotechnology can be used to develop a simple, portable and effective screening device for lung cancer. Chemiresistors are one such example and they leverage monolayered gold nanoparticles for their biocompatibility and their ability to detect VOCs at very low concentrations [26]. Colorimetric sensors are another and use materials that can change color in response to their chemical environment. The sensors can be easily fabricated, and the colors can be read by taking a picture with a smart phone and using simple software. However, many VOCs could only be detected in the range of parts per million [9, 70]. Sun et al. and Adiguzel et al. provide excellent reviews of a variety of types of sensors [9, 13] that is beyond the scope of this review.
There are also several technical hurdles involved in taking, handling, storing and analyzing breath samples for VOC analysis [5, 6]. The method used for exhaling the breath can strongly affect the VOC mixtures [5] and the ambient air which the subject has been breathing may affect what chemicals are identified in their breath [5]. Patients diagnosed with lung cancer are frequently smokers. Therefore, tobacco smoke constituents are identified in breath [31]. There is currently no standard method for collecting breath, with some studies analyzing the ambient air and comparing it with the chemicals from the breath sample, and others using “lung washout” before breath sampling [29] which usually involves participant burden and clinical assistance. In addition, some breath analysis techniques require storing the sample for a short time prior to analysis. Using Tedlar bags, one of the most common storage techniques, can lead to some contaminants leaching into the breath sample if it is stored too long. If sample storage is required, thermal desorption tubes can help avoid confounding compounds [29].
Data analysis also varied widely between studies [6, 71, 72], making replication of results between studies difficult. In future work, internal validation by random sample split or leave-one-out cross-validation is recommended to get closer to estimates that are comparable [29].
Although we used a comprehensive search string in two independent databases, it is possible that we missed relevant studies. The wide variation in breath collection, storage, analysis and reporting in the studies makes summarization of data challenging. We included only studies that evaluated the exhaled breath of cancer patients compared with controls, excluding in vitro studies and studies with canines. We also focused our presentation and discussion on those studies that reported VOCs along with performance statistics, which was a subset of the studies reviewed.
Breath analysis for cancer screening and early detection shows promise, because technology measurement continues to advance and it allows breath samples to be collected easily, safely, and frequently [6, 9, 29]. This review focused on specific VOCs evaluated in human studies with respect to lung, breast, or colorectal cancer published in in the past five years. We noted specific VOCs correlated with these cancers, along with the cutoff levels at which they were detected, the sensitivity, specificity and AUC.
Gas chromatography-mass spectrometry (GC-MS) is considered the gold standard for identifying specific VOCs. However, the science of breath analysis has recently moved into analyzing patterns of VOCs using a variety of different multiple sensor techniques, such as eNoses and nanomaterials. Additional development of breath analysis for early cancer detection will require standardized breath sampling methods and innovative techniques for VOC detection and analysis. As technology advances and clinical studies are conducted breath analysis holds promise not only for detecting cancer early, but also for monitoring the response to cancer treatment and potentially early detection of recurrence when secondary treatment efficacy is at its highest. Although several authors felt that breath analysis requires further development and testing before integration into clinical practice, Brodrick et al. report on a protocol which was successfully used for quickly and accurately collecting breath samples from 323 respiratory patients recruited in 10 different secondary health care clinics [17]. Given the growing body of work and the compelling reasons to detect cancer and its recurrence early, further clinical work validating novel technologies and approaches is warranted.
Footnotes
Acknowledgments
This research was supported by Nano Engineered Applications (NEA).
