Abstract
The Gleason grading system is among the most important prognostic factors in patients with prostate cancer. From the 2005 to the 2014 consensus conferences, organized by the International Society of Urological Pathology, the morphologic criteria for the identification of the Gleason patterns were redefined, thus resulting in the shrinkage of the Gleason pattern 3. This led to the expansion of the Gleason pattern 4. The newly proposed grade group system reduces the Gleason scores of prostate cancer to the lowest number, each associated with a unique behavior from the prognostic point of view. The advantage is that the simplified system with five groups allows for a more accurate stratification of the patients in comparison with the Gleason system. Cribriform, fused, ill-defined and glomeruloid glands are part of the histologic spectrum of the Gleason pattern 4. Cribriform morphology has a prognosis that is worse in comparison with the other non-cribriform Gleason 4 patterns. One of the major implications of the cribriform growth is that it precludes a patient from choosing active surveillance.
Keywords
The Gleason grading system is among the most important prognostic factors in patients with prostate cancer (PCa). From the 2005 1 to the 2014 2 consensus conferences, organized by the International Society of Urological Pathology (ISUP), the morphologic criteria for the identification of the Gleason patterns (GPs) were redefined, thus resulting in the shrinkage of the GP 3. This led to the expansion of the GP 4.
From the Gleason system to the Grade Groups
The Gleason grading systems modified both in 2005 1 and 2014 2 are suboptimal for several reasons:
Patients with Gleason score (GS) 3+3=6 think that their PCa belongs to the intermediate risk, even though the GS 6 is the lowest score used by pathologists when reporting prostate biopsies.
GS 7 includes PCas with 3+4=7 and 4+3=7. Studies have shown a better clinical outcome for patients with GS 7 with primary pattern 3 versus 4.
PCas with GS 9 to 10 have a prognosis that is worse than that seen in tumors with GS 4+4=8.
Different grouping systems and approaches have been adopted to combine PCas with different GSs for treatment purposes and prognostic scopes.
Based on a 2013 investigation by Epstein and his team 3 , a new grading system approach was adopted in 2014 at the time of the Chicago Conference held by the ISUP to address such limitations; that is, grade groups (GGs) from 1 to 5. The clinical value of the GGs has been demonstrated in several clinical studies4,5. The new GG system is used in parallel with the 2014 revised Gleason system.
This simplified system with five groups allows for a more precise stratification of the patients than current revised Gleason systems, with the potential of reducing overtreatment in men with PCa. From the psychological point of view, the shift from a lowest value of “6” to “1” has a positive consequence on patients’ awareness of the disease. This facilitates patient choice of treatment options, including radical prostatectomy, cryoablation, radiotherapy, hormonal therapy, or active surveillance (AS). GS 6 (GG 1) cancers are characterized by indolent behavior with an excellent prognosis. However, it should still be considered as a cancer due to the fact that usually it is organ-confined, so as not to mislead a patient seeking a definitive cure 6 . The international community has considered it as a “new grading system,” whereas most uropathologists consider it as a “novel grouping” of the 2014 modified Gleason grading system.
Correlation of GGs with tissue genetic changes
A genomic study was conducted by Rubin et al. 7 based on whole exome and whole genome sequencing data from 426 localized PCa treated by radical prostatectomy (RP). The results give support to the clinical importance of the GGs. In particular, the investigation showed an increased frequency of both genomic amplifications and deletions together with an increasing risk strata and of non-synonymous point mutations. In particular, GG 1 was haploid, whereas GGs 2 to 5 showed an increasing frequency of nuclear polyploidy. Distinct genomic profiles between the 5 GGs were seen by using the principal component analysis. This gives further support to GG 1 through GG 3 as distinct categories. However, genomic similarity was observed between GG 4 and GG 5.
2018 implementations of the 2014 revised Gleason system and of GGs
The current approach in the implementation of the 2014 revised Gleason system (also known as the 2016 World Health Organization grading system) and of the GGs includes reporting the percentage pattern 4 in GS 7 on biopsy and RP; reporting minor high-grade (i.e., tertiary) patterns in biopsies and RP specimens; grading score versus jar; and grading separate tumor nodules in an RP specimen. 8
Reporting percentage pattern 4 in GS 7 on biopsy and RP
There are several advantages in reporting the percentage of pattern 4 in the GS 7 tumors: 3+4 (GG 2) or 4+3 (GG 3), such as:
Consistency between needle biopsy core and RP grading
Consistency among uropathologists in the grading of limited pattern 4
GS 3+3=6 (GG 1) is accepted for men to undergo AS. However, selected patients—depending on their age and comorbidity, the magnetic resonance imaging (MRI) findings, the extent of cancer, and patient choice—could be candidates for AS with a GS of 3+4=7 (GG 2), provided that the pattern 4 is ⩽10% and a cribriform morphology is absent. 8
Reporting minor high-grade (tertiary) patterns in biopsies and RP specimens
On needle biopsy, the GS is based on the predominant pattern + the highest grade pattern; that is, the tertiary pattern is not included. For example, in a biopsy core with 60% GP 3, 35% pattern 4, and 5% pattern 5, the cancer would be considered as GS 3+5=8 (GG 4).
For the percentage of pattern 5 to be considered as a tertiary pattern it should represent less than 5% of the tumor lesion in RP. A tumor with a GS 3+4 =7 with less than 5% of pattern 5 is 3+4 = 7 with tertiary 5 (GG 2 with minor high-grade pattern), whereas a GS 3+4 = 7 with more than 5% of pattern 5 becomes 3+5=8 (GG 4). A similar approach is adopted for a GS 4+3= 7 with tertiary 5 (GG 3 with a minor high-grade pattern) versus 4+5 = 9 (GG 5) 8 .
Grading core versus jar
The uropathologist assigns individual GSs to separate biopsy cores as long as the cores are sent to the pathology laboratory in separate containers, or the cores are in the same container; however, they need to be identified by the clinician as to their location with different color inks. In clinical practice the highest GS is used for prognosis and treatment. The various nomograms and tables proven to be prognostically useful utilize the highest GS in those cases where there are multiple positive cores of different GSs 8 . The GSs of multiple cores in the same jar from a certain location (e.g., based on multiparametric MRI (mpMRI)) are averaged together. This is due to the fact that they are from the same location of the prostate. In those cases with fragmented multiple cores in the same jar, the agreement is to give a global GS for that jar 8 . The uropathologist has the option, in addition to giving separate cores individual GSs, to give an overall score at the end of the pathology report.
Grading separate tumor nodules in an RP specimen
A uropathologist could have, for example, two separate cancer nodules. One could be high grade (i.e., GS 4+4=8, GG 4) and there could be a larger nodule of GS 3+3=6 (GG 1). If one were to average both tumor nodules together, the grade would be GS 3+4=7 (GG 2). This is misleading as the prognosis of the tumor in the overall RP would be expected to be based on the higher grade nodule, which would not be diluted by a separate lower grade focus. The uropathologist should list the separate tumor nodules as tumor nodule #1, tumor nodule #2, etc., and give the separate grade and stage for each nodule 9 .
Further definition of GP 4: cribriform morphology
Cribriform, fused, ill-defined, and glomeruloid glands are part of the morphologic spectrum of current GP 4. Cribriform, derived from the Latin word cribrum (i.e., sieve), was introduced by Gleason to describe “glands composed of solid sheet with perforations or lumina” (Figure 1). Cribriform morphology has a worse prognosis in comparison with the other, non-cribriform, morphologies. Following the 2014 ISUP conference, a rapidly growing body of evidence has demonstrated a poor prognosis in association with cribriform morphology 10 .
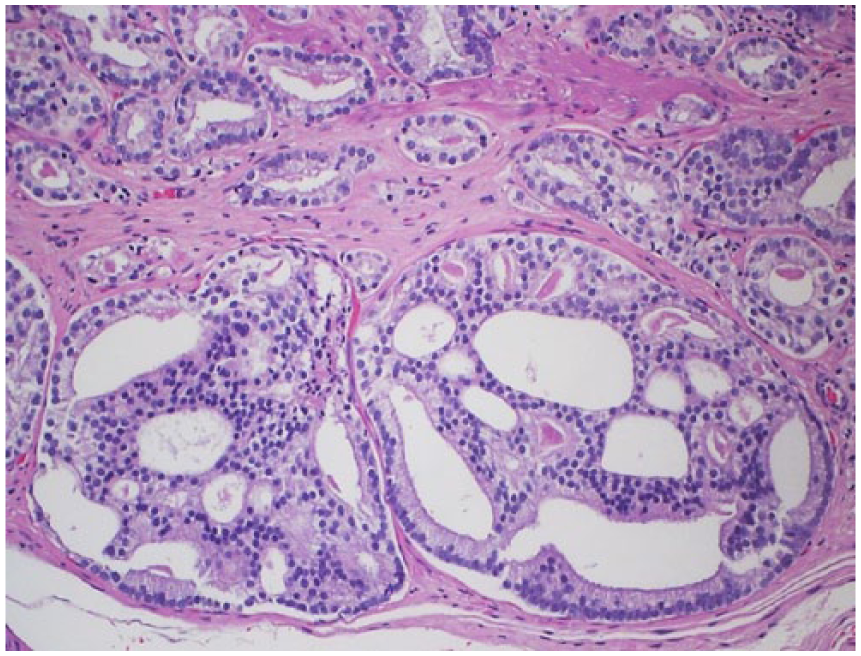
Prostate cancer with cribriform morphology.
Cribriform morphology observed on prostate biopsy is linked to increased upgrading, upstaging, and positive surgical margins in the subsequent RP. Flood et al. 11 showed that perineural invasion on prostate biopsy—considered a strong predictor of adverse pathological features—was inferior to the cribriform pattern in the prediction of non-organ confined PCa in the RP. Kweldam et al. 12 showed that the cancer-specific survival of GS 3+4 (GG 2) PCa without cribriform glands in prostate biopsy was similar to that with GS 3+3 (GG 1) PCas.
Dong et al. 13 demonstrated that after a follow-up time of 10 years, 13% of the men with cribriform morphology in the RP developed metastasis in comparison with 2.6% with GP 4 without the cribriform pattern. Other investigations supported the information that the presence of any cribriform morphology was associated with higher biochemical recurrence. Kryvenko et al. 14 showed that men with lymph node metastases had higher volumes of cribriform morphology in the RP. Within GS 8 (GG 4) PCa, cribriform was also predictive of cancer-specific survival.
mpMRI and GP 4 morphologies
In a study of men who underwent RP with pre-operative mpMRI, cribriform predominant cancers were less visible than non-cribriform predominant tumors 15 . In a further investigation, cribriform tumors in their pure form on RPs were poorly visible on mpMRI (only 17% of foci were seen). An increase in visibility was seen when it was surrounded by non-cribriform morphology 16 . Although the mechanism of reduced visibility of cribriform tumors is still poorly understood, the less compact structure of cribriform glands could theoretically reduce their visibility on diffusion-weighted imaging.
Voided urine, GP 4 morphologies, and biological markers
A variety of molecular changes have been reported in cribriform tumors, including the loss of phosphatase and tensin homolog; a few investigations have evaluated whether epigenetic changes are present in cribriform tumors. Since the prostate glands drain in the urethra, genetic material from PCa with the cribriform pattern that has spread within preexisting ducts and acini could be detected in voided urine more easily than from invasive tumor glands 17 .
Conclusions
The newly proposed GG system reduces the GSs of PCa to the lowest number—each of which is associated with a unique behavior from the prognostic point of view. The advantage is that the simplified system with five groups allows for a more accurate stratification of the patients in comparison with the Gleason systems. Cribriform, fused, ill-defined, and glomeruloid glands are part of the histologic spectrum of GP 4. Cribriform morphology has a prognosis that is worse in comparison with the other non-cribriform GP 4 patterns. One of the major implications of cribriform growth is that it precludes a patient from choosing AS (Table 1).
Pathologic items that exclude a patient from choosing AS.

Footnotes
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
