Abstract
High pretreatment levels of plasma fibrinogen have been widely reported to be a potential predictor of prognosis in digestive system tumors; however, the conclusions are not consistent. Therefore, we performed a meta-analysis to comprehensively assess the prognostic roles of high pretreatment plasma fibrinogen levels in digestive system tumors. We searched for eligible studies in the PubMed, Embase, and Web of Science electronic databases for publications from the database inception to 1 September 2017. The endpoints of interest included overall survival, disease-free survival, and recurrence-free survival. We investigated the relationship between fibrinogenemia and overall survival in colorectal cancer (10 studies), gastric cancer (6), pancreatic cancer (6), hepatocellular carcinoma (7), and esophageal squamous cell carcinoma (10); the pooled results indicated that fibrinogenemia was significantly related to a worse overall survival (hazard ratio (HR) 1.73; 95% confidence interval (CI) 1.52, 1.97; P <0.001; HR 1.71; 95% CI 1.28, 2.28; P <0.001; HR 1.57; 95% CI 1.13, 2.17; P = 0.007; HR 1.89; 95% CI 1.57, 2.27; P <0.001, and HR 1.67; 95% CI 1.35, 2.07; P <0.001). Taken together, an increased pretreatment plasma fibrinogen level was related to worse survival in digestive system tumors, indicating that it could be a useful prognostic marker in these types of tumors.
Introduction
The digestive system comprises a number of ducts and glands and is sensitive to various pathological stimuli, which may increase the risks for various diseases, especially malignancies. Digestive system malignancies are a formidable threat to human life and health. 1 Unfortunately, an increasing prevalence of digestive system tumors (DSTs), such as hepatocellular carcinoma, esophagus cancer, pancreatic cancer, and intestinal neoplasm, was reported in 2015, although cancer-associated deaths have been continuously declining over the past two decades. 2 Early diagnosis is one of the major strategies to reduce mortality and improve patient quality of life. Adverse tumor characteristics, such as advanced clinical stage, poor tumor differentiation, lymph node invasion, and distant metastasis, are closely correlated to a poor prognosis, whereas patients with similar clinicopathological features might have diverse clinical outcomes. 3 Thus, there is a need for clinically applicable biomarkers with high sensitivity and specificity that permit early detection and intervention in patients with DSTs and that allow effective therapeutic strategies to improve life quality and clinical outcomes of patients with DSTs.
Plasma fibrinogen is mainly associated with the maintenance of hemostatic function. However, a recent body of evidence indicated that plasma fibrinogen also plays an important role in both inflammation response and tumor progression. 4 In vitro studies suggested that fibrinogen was able to enhance malignant cell invasion, proliferation, angiogenesis, and epithelial-to-mesenchymal transition.5–7 Furthermore, numerous clinical studies indicated that pretreatment plasma fibrinogen (PPF) levels were higher than those in healthy controls and that PPF serum levels were inversely related to the prognosis of patients with various tumors, including DSTs.8–17 In addition, an elevated PPF level was reported to be closely correlated with a more advanced tumor stage and lymph node metastasis of some DSTs, such as esophageal, 18 gastric, 19 and pancreatic 20 cancers. Consequently, increased PPF is associated with tumor progression and a poor prognosis for patients with malignant disease. Considering that the small sample size in a single study might challenge the statistical power, several studies have also reported that PPF was not an independent predictor of unfavorable prognosis in patients who underwent operations for colorectal cancer and ESCC.21–23 To the best of our knowledge, no meta-analysis has investigated the prognostic value of PPF in patients with DSTs. Therefore, to further validate the independent prognostic role of PPF in DSTs, we performed a systematic meta-analysis of the relationship between PPF and the prognosis of patients with DSTs.
Methods
Literature search strategy and selection criteria
This meta-analysis was performed according to Preferred Reporting Items for Systematic Review and Meta-analyses (PRISMA) guidelines. We performed the systematic search by retrieving eligible studies from the PubMed, Embase, and Web of Science electronic databases that assessed the prognostic value of pretreatment plasma fibrinogen levels in DSTs from database inception to 1 September 2017. The combination of the following terms: ((fibrinogen(Title/Abstract)) AND (pancreatic(Title/Abstract)) OR (esophageal(Title/Abstract)) OR (colon(Title/Abstract)) OR (gallbladder(Title/Abstract)) OR (hepatocellular(Title/Abstract)) OR (colorectal(Title/Abstract)) OR (gastric(Title/Abstract)) OR (stomach(Title/Abstract)) OR (liver cancer(Title/Abstract)) OR (cholangiocarcinoma(Title/Abstract)) OR (biliary duct(Title/Abstract)) OR (bile duct(Title/Abstract)) OR (rectal(Title/Abstract))) AND prognosis constituted the search strategy. Limitations regarding the language and region were not applied in this meta-analysis.
The criteria for eligible studies included: (a) perspective or retrospective study; (b) assessment of the association between pretreatment fibrinogen and prognosis in DSTs; (c) articles with full text were published in English; and (d) the hazard ratio (HR) with its 95% confidence interval (CI) or survival curve were extractable. Moreover, the following criteria were used to exclude unqualified studies: (a) case reports, reviews, comments, and systematic reviews; (b) studies on non-DSTs; (c) studies in which no adequate data could be extracted to estimate the HR and 95% CI; and (d) studies performed on animals.
Data extraction and quality assessment
Data were extracted by two independent researchers. A third investigator joined the discussion to reconcile inconsistencies. The following information was obtained: the first author’s name, year of publication, country of research, study design, type of cancer, tumor stage, metastasis, the number of patients, mean age of patients, mean follow-up time, fibrinogen cut-off level, and outcome measures. If the results of both univariate and multivariate analysis were available in the studies, only the multivariate results were extracted due to its control of a larger number of confounding factors. When HRs for overall survival (OS), progression-free survival (PFS), and disease-free survival (DFS) could not be obtained directly, Engauge Digitizer version 4.1 (http://digitizer.sourceforge.net/, freely downloaded software) was used to calculate the survival data from the Kaplan–Meier curves.
A quality assessment of the included studies was performed based on the Newcastle–Ottawa Scale (NOS), which assessed aspects regarding selection, comparability, and outcome. 24 The scores obtained from the NOS varied from 0 to 9; the studies that scored 6 or more were defined as high quality.
Statistical analysis
Statistical analyses in this meta-analysis were performed using RevMan5.3 (The Cochrane Collaboration, The Nordic Cochrane Center, Copenhagen, Denmark). Egger’s and Begg’s tests were performed using STATA version 12.0 (Stata Corporation, College Station, TX, USA). Pooled HRs with 95% CIs were used to estimate the quantitative aggregation of the survival results and the association between PPF and clinicopathological features in DSTs was assessed using HRs with 95% CIs. The heterogeneity across studies was assessed using Cochran’s Q and Higgins I2 statistics. P < 0.05 and I2 > 50% were considered to indicate significant heterogeneity, while I2 < 25 % and 25 % < I2 < 50 % were defined as no and moderate heterogeneity, respectively. A random-effects model was applied when statistical heterogeneity was observed (P < 0.05, I2 > 50 %); otherwise, a fixed effects model was applied. HR > 1 (using low fibrinogen as reference) indicated a higher risk of poor outcomes for high fibrinogen and was considered to be statistically significant if the 95% CI did not include 1 and P <0.05. A sensitivity analysis was performed by sequentially excluding single studies. If the pooled results did not significantly alter when single studies were omitted, they were considered stable. Publication bias was assessed using Begg’s tests and Egger’s tests; P < 0.05 indicated the potential for a statistically significant publication bias.25,26
Results
Study search and study characteristics
The search strategy and selection process are shown in Supplement Figure 1. A total of 717 studies were identified from fully searching the PubMed, Embase, and Web of Science databases. After excluding 167 duplications, 550 studies were evaluated by two researchers who screened the titles and abstracts. A total of 432 studies were excluded as case reports, reviews, irrelevant topics, animal studies, and studies that were not published in English, leaving 118 studies for full-text review. Finally, 43 articles (44 studies) with 17,510 patients were included in the present meta-analysis. 5,6,9,10,14,16,18-21,23,27–58
A total of 25 studies were carried out in China; 12 studies were from Japan; two studies each were performed in Korea, Italy, and Australia; and one study was conducted in the USA. The accrual period of 44 studies ranged from 1995 to 2014 and the numbers of participants ranged from 41 to 1512. A total of 12 studies were on esophageal carcinoma, six were on gastric cancer, six were on pancreatic cancer, 12 were on colorectal cancer, seven were on hepatocellular carcinoma, and one was on biliary tract cancer. The majority of the included studies were retrospective studies and only one was a prospective design. With the exception of one study, the others studies reported fibrinogenemia cut-offs ranging from 2.345 to 4 g/L. Regarding the outcomes of interest, 41 studies investigated the relationship between fibrinogenemia and OS, 12 examined the association between DFS and fibrinogenemia, five studies involved recurrence-free survival (RFS), and three studies referred to PFS. The Newcastle–Ottawa scale scores ranged from 5 to 7, indicating that the quality of the included studies was moderate to high. These study characteristics are shown in Table 1.
Main characteristics of the included studies.
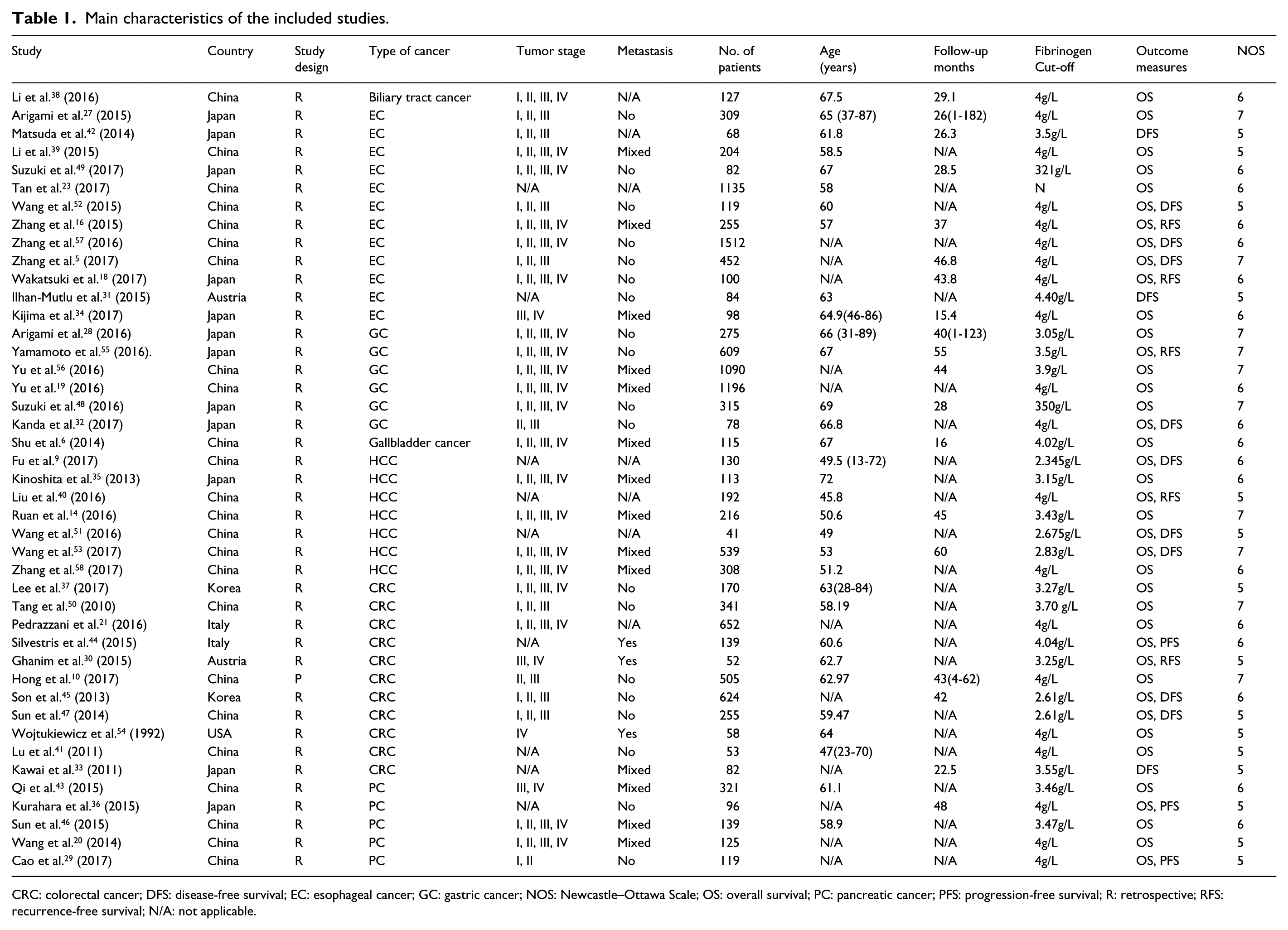
CRC: colorectal cancer; DFS: disease-free survival; EC: esophageal cancer; GC: gastric cancer; NOS: Newcastle–Ottawa Scale; OS: overall survival; PC: pancreatic cancer; PFS: progression-free survival; R: retrospective; RFS: recurrence-free survival; N/A: not applicable.
Prognostic value of fibrinogenemia in colorectal cancer
Fibrinogenemia and OS
A total of 10 studies investigated the relationship between fibrinogenemia and OS in colorectal cancer.10,21,30,37,41,44,45,47,50,54 A random-effects model was conducted to calculate the HR and 95%CI due to the severe heterogeneity (I2 = 60 %; P = 0.007) among those studies. The pooled analysis indicated that fibrinogenemia was significantly related to a worse OS (HR 1.87; 95% CI 1.40, 2.48; P <0.001) (Figure 1(a)).
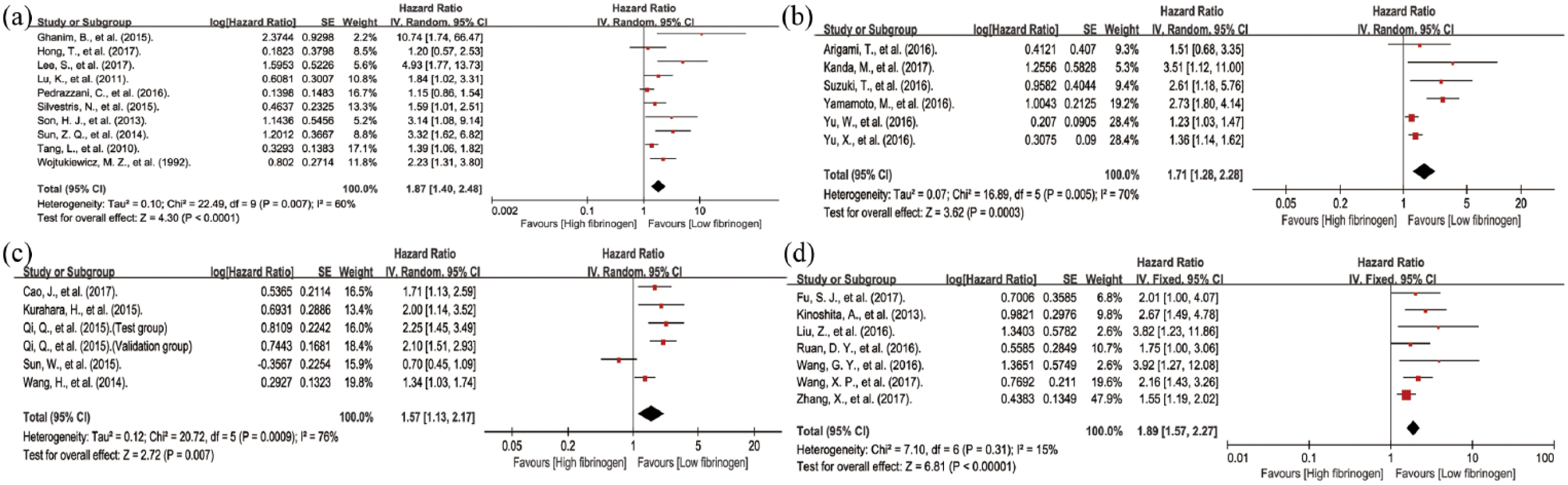
Pooled hazard ratios of overall survival among patients with fibrinogenemia. (a) Pooled hazard ratios of overall survival of fibrinogenemia in patients with colorectal cancer. (b) Pooled hazard ratios of overall survival of fibrinogenemia in patients with gastric cancer. (c) Pooled hazard ratios of overall survival of fibrinogenemia in patients with pancreatic cancer. (d) Pooled hazard ratios of overall survival of fibrinogenemia in patients with hepatocellular carcinoma. (e) Pooled hazard ratios of overall survival of fibrinogenemia in patients with esophageal squamous cell carcinoma.
Due to the substantial heterogeneity, we performed subgroup analyses by ethnicity, metastasis, and cut-off value to explore the potential sources of heterogeneity. The results of subgroup analysis revealed a significant association between high fibrinogen level and poor OS in Asian (HR 1.68; 95% CI 1.36, 2.08; P <0.01) and non-Asian patients (HR 1.44; 95% CI 1.15, 1.79; P <0.01) (Table 2). Additionally, we also identified a significant association between a high fibrinogen level and a more unfavorable OS in the non-metastasis (HR 1.68; 95% CI 1.36, 2.08; P <0.01), and metastasis (HR 1.95; 95% CI 1.39, 2.74; P < 0.01) (Table 2) groups. As for cut-off values, a high fibrinogen level was associated with worse OS in the ≥4 g/L and <4g/L groups (HR 1.42; 95% CI 1.17, 1.74; P <0.01 and HR 1.77; 95% CI 1.39, 2.24, respectively; P <0.01) (Table 2). Although the results of the subgroup analyses indicated that those factors might not be the sources of the heterogeneity, they validated the robustness of the pooled HR for OS.
Subgroup analysis for the influence of preoperative plasma fibrinogen levels on OS of various cancers.
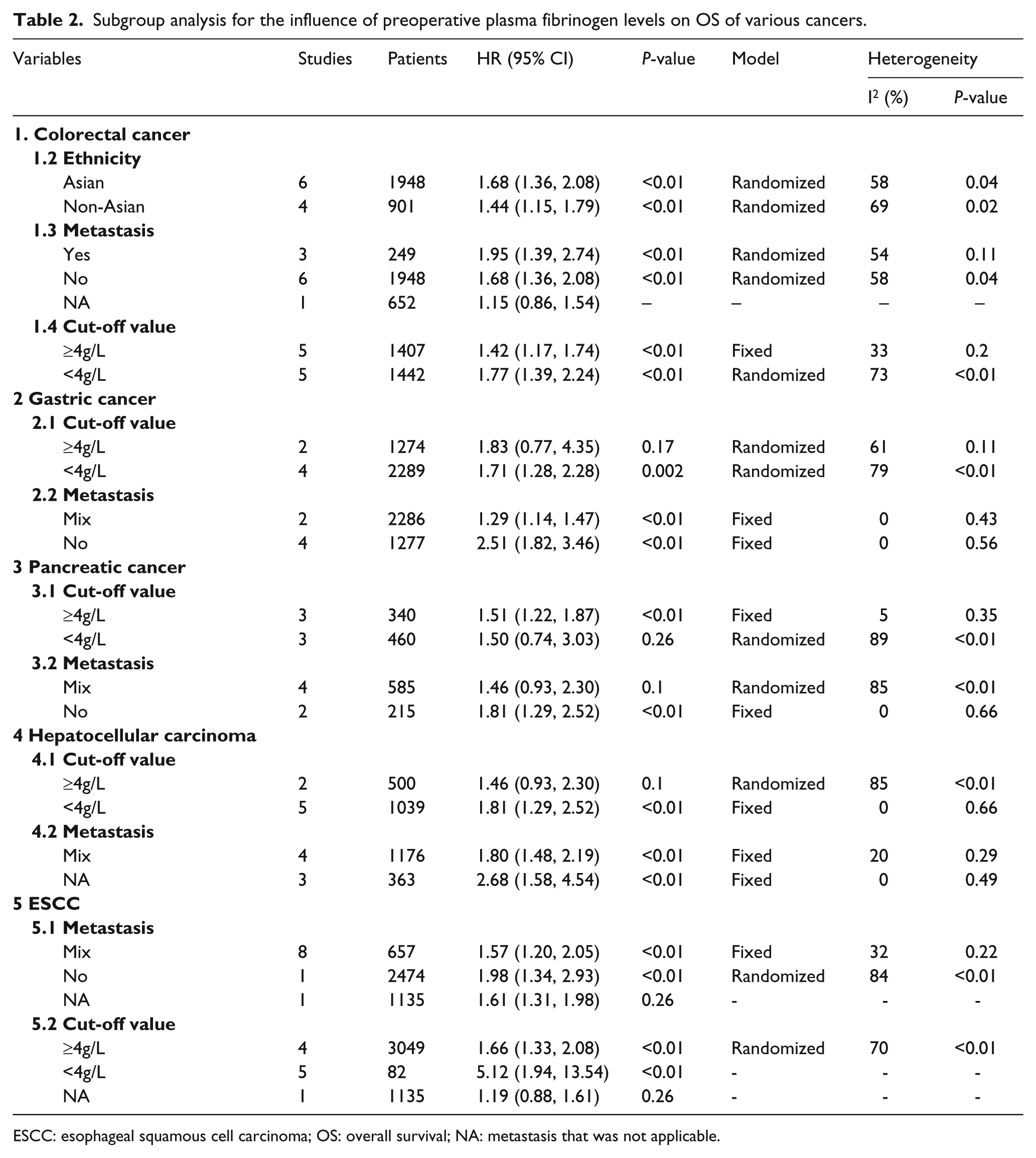
ESCC: esophageal squamous cell carcinoma; OS: overall survival; NA: metastasis that was not applicable.
Fibrinogenemia and DFS/RFS
A total of four studies explored the association between fibrinogenemia and DFS or RFS.30,33,45,46 The pooled results showed an inverse correlation between fibrinogenemia and DFS/RFS (HR 2.69; 95% CI 1.88, 3.86; P <0.0001) (Supplement Figure 2A).
Prognostic value of fibrinogenemia in gastric cancer
Fibrinogenemia and OS
A total of six studies investigated the relationship between fibrinogenemia and OS in gastric cancer.19,28,32,48,55,56 A random-effects model was conducted to calculate the HR and 95% CI due to the severe heterogeneity (I2 = 70 %; P = 0.005) among those studies. The pooled analysis indicated that fibrinogenemia was significantly related to a worse OS (HR 1.71; 95% CI 1.28, 2.28; P <0.001) (Figure 1(b)).
Due to the substantial heterogeneity, we performed subgroup analyses by metastasis and cut-off value to explore the potential sources of heterogeneity. The subgroup analysis revealed a significant association between a high fibrinogen level and a more unfavorable OS in the non-metastasis (HR 2.51; 95%CI 1.82, 3.46; P <0.01) and mixed metastasis (HR 1.29; 95% CI 1.14, 1.47; P < 0.01) groups (Table 2). Regarding cut-offs, a high fibrinogen level was associated with worse OS in the <4 g/L group (HR 1.71; 95% CI 1.28, 2.28; P <0.01) (Table 2).
Prognostic value of fibrinogenemia in pancreatic cancer
Fibrinogenemia and OS
A total of six studies investigated the relationship between fibrinogenemia and OS in pancreatic cancer.20,29,36,43,46 A random-effects model was conducted to calculate the HR and 95% CI due to the severe heterogeneity (I2 = 76%, P <0.001) among those studies. The pooled analysis indicated that fibrinogenemia was significantly related to a worse OS (HR 1.57; 95% CI 1.13, 2.17; P = 0.007) (Figure 1(c)).
Due to the substantial heterogeneity, we performed subgroup analyses by metastasis and cut-off value to explore the potential sources of heterogeneity. The subgroup analysis revealed a significant association between a high fibrinogen level and a more unfavorable OS in the non-metastasis group (HR 1.81; 95% CI 1.29, 2.52; P <0.01) (Table 2). As for cut-off value, a high fibrinogen level was associated with a worse OS in the ≥4 g/L group (HR 1.51; 95% CI 1.22, 1.87; P <0.01) (Table 2).
Prognostic value of fibrinogenemia in hepatocellular carcinoma
Fibrinogenemia and OS
A total of seven studies investigated the relationship between fibrinogenemia and OS in hepatocellular cancer.9,14,35,40,51,53,58 A fixed-effects model was conducted to calculate the HR and 95% CI among those studies. The pooled analysis indicated that fibrinogenemia was significantly related to a worse OS (HR 1.89; 95% CI 1.57, 2.27; P <0.001) (Figure 1(d)).
Due to the substantial heterogeneity, we performed subgroup analyses by metastasis and cut-off value to explore the potential sources of heterogeneity. The subgroup analysis revealed a significant association between high fibrinogen level and poor OS in the mixed metastasis (HR 1.80; 95% CI 1.48, 2.19; P <0.01) and NA metastasis (HR 2.68; 95% CI 1.58, 4.54; P < 0.01) groups (Table 2). As for the cut-off value, high fibrinogen level was associated with a worse OS in the <4 g/L group (HR 1.81; 95% CI 1.29, 2.52) (Table 2).
Fibrinogenemia and DFS/RFS
A total of four studies explored the association between fibrinogenemia and DFS or RFS.9,40,51,53 The pooled results showed that there was an inverse correlation between fibrinogenemia and DFS/RFS (HR 2.17; 95% CI 1.65, 2.85; P <0.0001) (Supplement Figure 2B).
Prognostic value of fibrinogenemia in ESCC
Fibrinogenemia and OS
A total of 10 studies investigated the relationship between high fibrinogenemia and OS in ESCC.5,16,18,23,27,34,39,49,52,57 A random-effects model was used to calculate the HR and 95% CI due to the severe heterogeneity (I2 = 71%, P<0.001) among those studies. The pooled analysis indicated that fibrinogenemia was significantly related to a worse OS (HR 1.67; 95% CI 1.35, 2.07; P <0.001) (Supplement Figure 1).
Due to the substantial heterogeneity, we performed subgroup analyses by metastasis and cut-off value to explore the potential sources of heterogeneity. The subgroup analysis revealed a significant association between a high fibrinogen level and poor OS in the mixed metastasis group (HR 1.57; 95% CI 1.20, 2.05; P <0.01) (Table 2). In the cut-off group, a high fibrinogen level was associated with a worse OS in the ≥4 g/L and <4 g/L groups (HR 1.66; 95% CI 1.33, 2.08; P <0.01 and HR 5.12; 95% CI 1.94, 13.54; P <0.01, respectively) (Table 2). Although the results of the subgroup analyses indicated that those factors might not be responsible for the heterogeneity, they validated the robustness of the pooled HR for OS.
Fibrinogenemia and DFS/RFS
A total of seven studies explored the association between fibrinogenemia and DFS or RFS.5,16,18,31,42,52,57 The pooled results showed an inverse correlation between fibrinogenemia and DFS/RFS (HR 1.64; 95% CI 1.31, 2.05; P <0.0001) (Supplement Figure 2(c)).
Sensitivity analysis
In order to further verify the robustness of the pooled HR for OS and DFS/RFS, sensitivity analyses were conducted to assess the influence of each individual study on the pooled HRs for OS and DFS/RFS by omitting a single study in each step. The results showed that the pooled estimates of the effect of fibrinogenemia on the OS of patients with colorectal cancer, gastric cancer, pancreatic cancer, hepatocellular carcinoma, and ESCC (Figure 2 and Supplement Figure 3(a)) were not substantially altered when any individual study was excluded, which implied that the results of our meta-analysis were robust. Furthermore, the results also suggested that the pooled estimates of the effect of fibrinogenemia on DFS/RFS (Supplement Figure 3(b)) in patients with ESCC were not substantially altered. Sensitivity analysis was not applicable to the correlation of fibrinogenemia with DFS/RFS in patients with colorectal cancer, gastric cancer, pancreatic cancer, and hepatocellular carcinoma because of the limited number of studies exploring the relationship between fibrinogenemia and DFS/RFS.
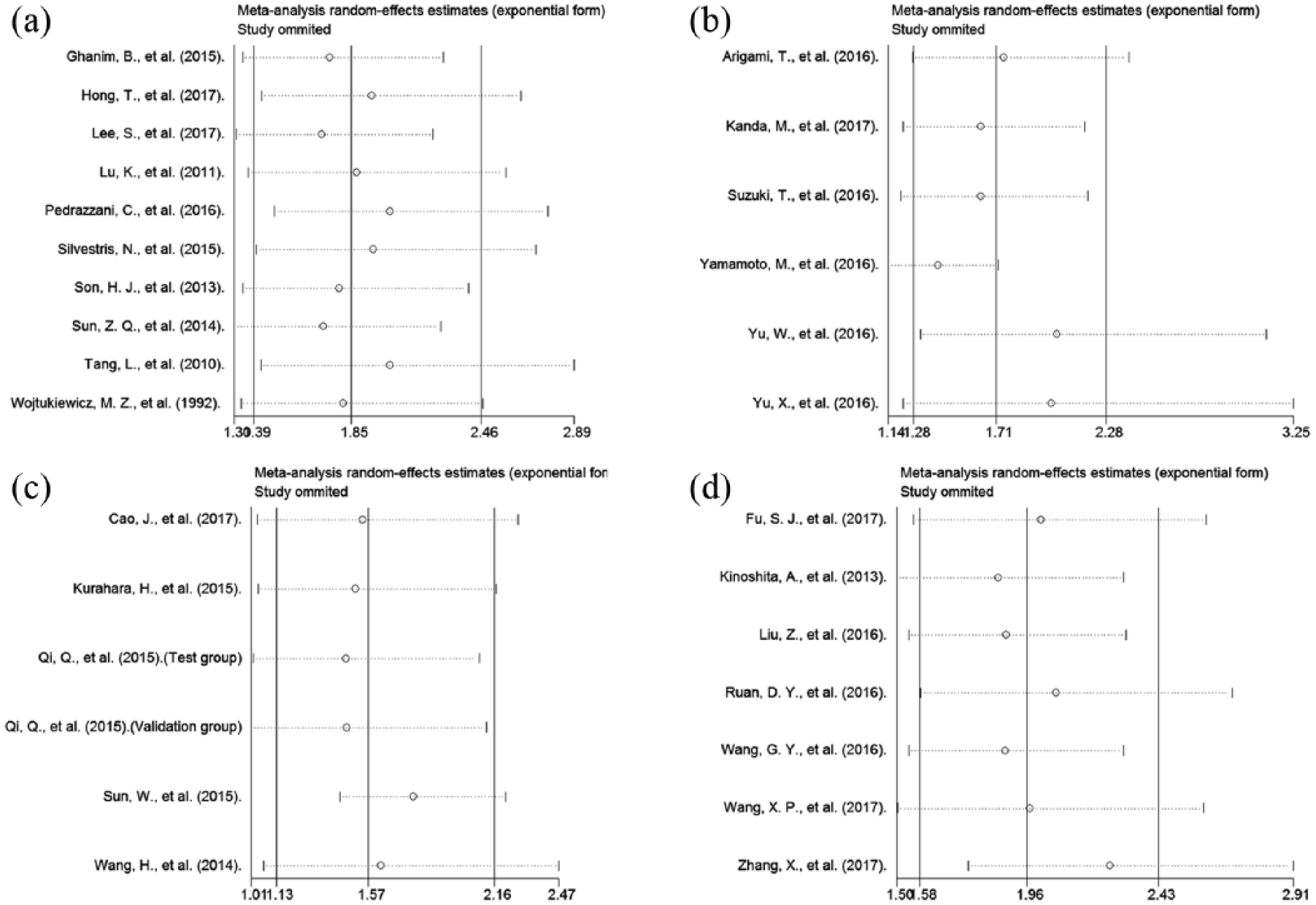
Sensitivity analysis of the pooled effects of overall survival. (a) Sensitivity analysis of the pooled effects of overall survival in patients with colorectal cancer. (b) Sensitivity analysis of the pooled effects of overall survival in patients with gastric cancer. (c) Sensitivity analysis of the pooled effects of overall survival in patients with pancreatic cancer. (d) Sensitivity analysis of the pooled effects of overall survival in patients with hepatocellular carcinoma.
Publication bias
Begg’s and Egger’s tests were used to assess publication bias in our meta-analysis. The results revealed no significant bias in the pooled HRs of OS in patients with gastric cancer and pancreatic cancer (Begg’s test, z = 0.94; P = 0.348; Egger’s test, t-bias = 2.29; P = 0.084 and Begg’s test, z = -0.19; P = 0.851; Egger’s test, t-bias = 0.20; P = 0.848) (Figure 3). However, statistically significant publication bias was observed for the OS in patients with colorectal cancer, hepatocellular carcinoma, and ESCC (Begg’s test: z = 2.06; P = 0.04; Egger’s test: t-bias = 4.33; P = 0.003; Begg’s test: z = 2.25; P = 0.024; Egger’s test: t-bias = 3.73; P = 0.014; and Begg’s test: z = 2.24; P = 0.025; Egger’s test: t-bias = 3.46; P = 0.009) (Supplement Figure 4). Publication bias was not possible when assessing for potential bias in the pooled HRs for the correlation between fibrinogenemia and DFS/RFS owing to the limited number of studies investigating this relationship.
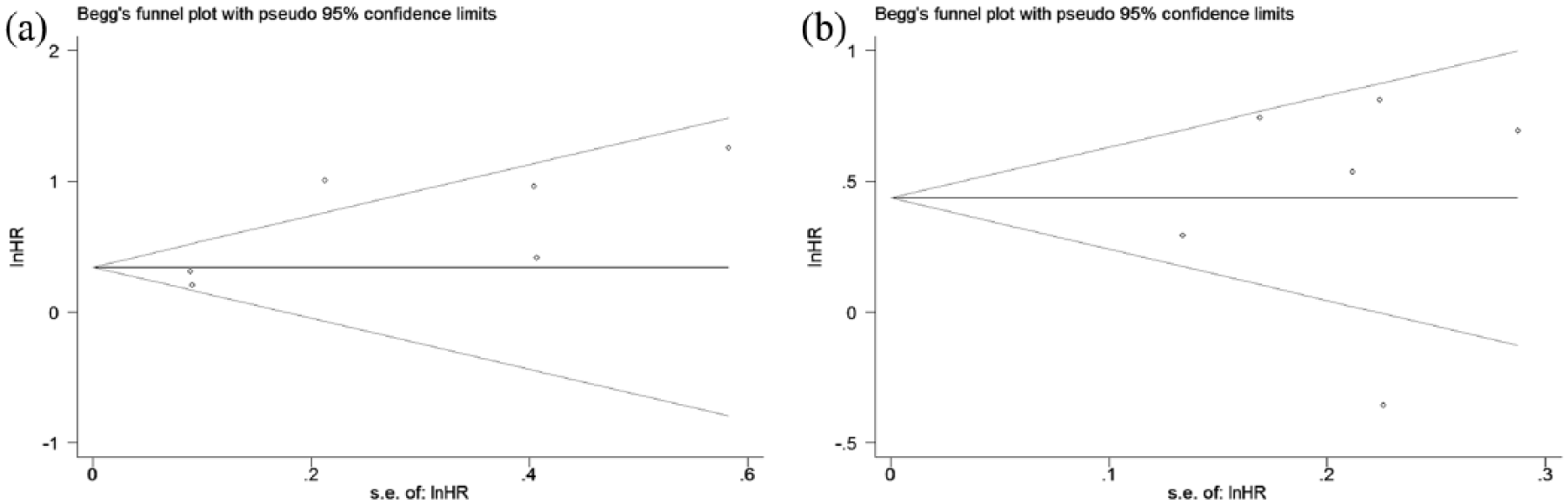
Funnel plots depicting the publication bias among the included studies on overall survival. (a) Funnel plots depicting the publication bias among the included studies on overall survival in patients with gastric cancer. (b) Funnel plots depicting the publication bias among the included studies on overall survival in patients with pancreatic cancer.
To explore whether the publication bias for OS substantially impacted the stability of the pooled HRs in our meta-analysis, we further performed a trim-and-fill analysis. The results showed that the reasonable number of included studies should be 14, including the addition of four missing studies on OS in patients with colorectal cancer; 11 with four missing studies on OS in patients with hepatocellular carcinoma; and 15 with five missing studies on OS in patients with ESCC filled in our pooled analysis; in addition, the updated pooled funnel plots for publication bias on the correlation between SF and OS were relatively symmetrical. More importantly, the updated pooled HRs also did not significantly change (HR 1.458; 95% CI 1.066, 1.993; P = 0.018), (HR 0.502; 95% CI 0.255, 0.749; P <0.001) and (HR 1.284; 95% CI 1.023, 1.611; P = 0.031) (Supplement Figure 5), suggesting that the pooled HR of OS in our meta-analysis was stable, although the publication bias regarding the correlation between SF and OS was significant in our meta-analysis.
Discussion
A series of studies have suggested that a high PPF level is correlated with an adverse prognosis in patients with various cancers. However, the relatively small sample sizes and limited tumor subtypes in these studies largely compromised the power of their statistical analyses and the quality of information. Although it has been further confirmed by meta-analyses of studies exploring the prognostic role of PPF in patients with solid tumor, renal cell carcinoma, and epithelial ovarian cancer,59–61 to the best of our knowledge, no specific meta-analysis has been conducted to assess the prognostic value of PPF in patients with DSTs. Therefore, we combined 43 studies with 17,510 patients to perform a systematic review and meta-analysis to validate the hypothesis that an elevated PPF level predicts poor long-term oncological outcomes in patients with DSTs.
Our meta-analysis included 43 eligible studies investigating the association between PPF and the prognosis of patients with DSTs. The survival data from these studies, including OS, DFS, RFS, and PFS, were pooled in the statistical analyses. Overall, we confirmed that an elevated PPF is a predictor of worse OS, DFS, RFS, and PFS in patients with DSTs. Furthermore, our subgroup and sensitivity analyses verified the robustness of the pooled results for OS and DFS.
PPF is closely correlated with the outcomes of DSTs and other malignancies, but the underlying mechanisms have not been thoroughly clarified. Nevertheless, a large body of evidence from experimental studies has demonstrated that fibrinogen plays an essential role in tumor progression by promoting the invasion and proliferation, angiogenesis, metastasis, and epithelial-to-mesenchymal transition (EMT) of cancer cells.5–7,62 These effects might be partly due to the ability of fibrinogen to bind various growth factors secreted by other elements, such as platelet-derived growth factor, transforming growth factor-b, fibroblast growth factor, and vascular endothelial growth factor. It also functions as a reservoir for these factors, which are involved in the promotion of cell proliferation, metastasis, and angiogenesis, as well as the inhibition of apoptosis.63,64 In addition, fibrinogen could induce the EMT through activation of the p-AKT/p-mTOR pathway, enhancing the motility of esophageal squamous cells. 5 Furthermore, fibrinogen could promote tumor metastasis by binding to platelets and intercellular cell adhesion molecule-1. For instance, Desgrosellier and Cheresh 65 showed that fibrinogen could function as a bridge between platelets and circulating tumor cells (CTCs), which facilitated platelet adherence to CTCs. In particular, thrombin can catalyze the transformation of fibrinogen in the circulatory system into a dense fibrin matrix, which then junctions with platelets to form a favorable shield around CTCs, protecting them from natural killer cell-mediated cytotoxicity.66,67 In addition, fibrinogen could also directly interact with intercellular cell adhesion molecule-1 expressed on endothelial cells and help CTCs to guest on endothelia, which may facilitate the tumor metastasis. 68 Moreover, fibrinogen-dependent inflammatory responses are involved in cancer initiation and progression. Therefore, elevated serum levels of fibrinogen may indicate an active inflammatory tumor microenvironment that favors the progression of inflammation-related cancers. 7 For instance, Qi et al. 43 reported that plasma fibrinogen levels were positively related to neutrophil-lymphocyte, platelets-lymphocyte, and lymphocyte-monocyte ratios, which are widely recognized as markers of systemic inflammatory response.
Although this meta-analysis provided evidence to support the prognostic significance of elevated PPF levels in patients with DSTs, several limitations should be considered. First, the majority of the included studies were retrospective, which might cause bias in patient selection and data analysis. Second, the methods applied to detect PPF, the cut-off values, and the treatment methods differed among the eligible studies, which probably contributed to the heterogeneity. Third, the literature search may not have been comprehensive, since we only included studies published in English, which might have led to a language bias. Fourth, several HRs and 95% CIs were indirectly obtained from the survival curves, which may have introduced some errors. In addition, the HRs and 95% CIs calculated from the survival curves were the statistical results of univariate analyses. Univariate analysis does not take several potential confounding factors into account, which may also cause heterogeneity and bias, and reduce the independent prognostic role of PPF in patients with DSTs. Fifth, most of the enrolled subjects were from Asia, which decreased the generalizability of the results across different ethnicities. Further studies investigating the diagnostic accuracy of PPF among different patient populations in terms of genetics, ethnicity, and geography are warranted. Lastly, only a few of the eligible studies described in their methods that patients with inflammatory conditions or coagulation disorders were excluded. Elevated plasma fibrinogen levels in these patients might be partly attributed to non-tumor-related inflammatory activities or aberrant coagulation. Thus, the pooled analysis of the patients with inconsistent inflammation or coagulation status may affect the actual association between PPF and the prognosis of patients with DSTs. The differences in inflammation or coagulation status might be possible sources of the heterogeneity in our meta-analysis. Therefore, we stress that the results of this meta-analysis should be cautiously interpreted and that more large-scale studies are needed to evaluate the diagnostic accuracy of PPF in patients with DSTs.
In conclusion, the results of this meta-analysis suggest that an elevated PPF level predicts a worse OS and DFS/RFS in patients with DSTs, indicating that it could be a useful prognostic marker and therapeutic target. Considering the aforementioned limitations, prospective multi-center studies are needed to further validate our findings.
Supplemental Material
SupFig-2 – Supplemental material for Prognostic significance of pretreatment plasma fibrinogen level in patients with digestive system tumors: a meta-analysis
Supplemental material, SupFig-2 for Prognostic significance of pretreatment plasma fibrinogen level in patients with digestive system tumors: a meta-analysis by Rui Ji, Qian Ren, Suyang Bai, Yuping Wang and Yongning Zhou in The International Journal of Biological Markers
Supplemental Material
Sup_Fig-1 – Supplemental material for Prognostic significance of pretreatment plasma fibrinogen level in patients with digestive system tumors: a meta-analysis
Supplemental material, Sup_Fig-1 for Prognostic significance of pretreatment plasma fibrinogen level in patients with digestive system tumors: a meta-analysis by Rui Ji, Qian Ren, Suyang Bai, Yuping Wang and Yongning Zhou in The International Journal of Biological Markers
Supplemental Material
Sup_Fig-3 – Supplemental material for Prognostic significance of pretreatment plasma fibrinogen level in patients with digestive system tumors: a meta-analysis
Supplemental material, Sup_Fig-3 for Prognostic significance of pretreatment plasma fibrinogen level in patients with digestive system tumors: a meta-analysis by Rui Ji, Qian Ren, Suyang Bai, Yuping Wang and Yongning Zhou in The International Journal of Biological Markers
Supplemental Material
Sup_Fig-4 – Supplemental material for Prognostic significance of pretreatment plasma fibrinogen level in patients with digestive system tumors: a meta-analysis
Supplemental material, Sup_Fig-4 for Prognostic significance of pretreatment plasma fibrinogen level in patients with digestive system tumors: a meta-analysis by Rui Ji, Qian Ren, Suyang Bai, Yuping Wang and Yongning Zhou in The International Journal of Biological Markers
Supplemental Material
Sup_Fig-5 – Supplemental material for Prognostic significance of pretreatment plasma fibrinogen level in patients with digestive system tumors: a meta-analysis
Supplemental material, Sup_Fig-5 for Prognostic significance of pretreatment plasma fibrinogen level in patients with digestive system tumors: a meta-analysis by Rui Ji, Qian Ren, Suyang Bai, Yuping Wang and Yongning Zhou in The International Journal of Biological Markers
Footnotes
Author contributions
Rui Ji and Qian Ren contributed equally to this work.
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Science and Technology Support Program (2014BAI09B02) and The First Hospital of Lanzhou University Foundation (ldyyynqn201201).
Supplemental Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
