Abstract
Objective:
Breast cancer is the most widespread cancer in women, with a high mortality rate. This study aims to assess the association between single nucleotide polymorphisms (SNPs) of LOC643714 (rs12922061) and TOX3 (rs3803662) and breast cancer, as well as the clinical characteristics of tumors.
Materials and methods:
In total, 104 breast cancer patients and 118 healthy controls were recruited to our study. The genotyping was performed by the SNP scan method. General characteristics, the clinical characteristics of tumors and reproductive factors were included in the analysis. Statistical tests included the Student t-test, the Chi-square test (X2) or Fisher’s exact test, and unconditional logistic regression analysis. The receiver operating characteristic curves were used to evaluate the predictive role of rs12922061 in breast cancer.
Results:
The LOC643714 polymorphism was a risk factor for breast cancer under a dominant model (TT+TC vs. CC: OR 1.801; 95% CI 1.048, 3.095; statistical power=60%), recessive model (TT vs. TC + CC: OR 4.297; 95% CI 1.164, 15.867; statistical power=64%) and log-additive (TT vs. CC: OR 5.163; 95% CI 1.368, 19.485; statistical power= 73%). Furthermore, the rs12922061 polymorphism was associated with menopause status in patients (P=0.005). No statistically significant association was found between the rs3803662 polymorphism and breast cancer in patients or healthy controls.
Conclusions:
Our study found that rs12922061 of LOC643714 was related to breast cancer risk. With a limited sample size and statistical power, further multi-center studies are needed to confirm the influence of the LOC643714 polymorphisms on breast cancer based on larger populations.
Introduction
Breast cancer (BC) is one of the most frequent types of malignant neoplasms in women. It is the second most common cause of mortality and is the leading cause of cancer morbidity among females.1-3 In 2016, it is estimated that there were 249,260 new cases of BC and a total of 40,890 deaths in American women. 4 Furthermore, the incidence of BC also shows an increasing tendency in China. 1 As a multi-factorial disease, BC is a complicated disease affected by genetic and environmental factors. 5 Genetic factors act as an important factor in the pathogenesis of BC, with FGFR2, CHEK2, BRIP1, PALB2, TP53, BRCA2, and BRCA16-12 being implicated, and several polymorphisms in many different genes have been found to be associated with the risk of BC. 8
The TOX3 and LOC643714 genes are located on chromosome 16q12. TOX3 is a nuclear protein including a nuclear localization signal and a high mobility group (HMG)-box that can modify chromatin structure. 13 The researchers found that the expression of TOX3 is associated with BC,13-14 and the high expression of TOX3 may influence BC progression and prognosis. 13 The LOC643714 gene was shown by several studies to be associated with BC,15-17 but at the time of writing, the gene of LOC643714 has no known function.
The polymorphisms rs12922061 and rs3803662 are located in LOC643714 and TOX3, and previous studies have shown that rs3803662 can increase the risk of BC in Caucasian women. 18 Furthermore, Japanese and Chinese Genome-wide Association Study (GWAS) found that the polymorphisms rs12922061 and rs3803662 were significantly related to BC risk.17,19 However, a recent meta-analysis suggested that there was no association between rs3803662 and BC in Asian women. 18 Therefore, the relationship between rs3803662 and BC risk is not clear. 20 In this study, we investigated and assessed the association between single nucleotide polymorphisms (SNPs) (rs12922061 and rs3803662) and BC risk as well as clinical variables in the southern Chinese population.
Material and Methods
Patients
The case-control study included 104 BC patients evaluated by immunohistochemical staining and 118 healthy controls who were selected from the First Affiliated Hospital of Guangxi Medical University from 2013 to 2015. The patients and healthy controls were independent without familiarity, while patients with secondary tumors, other malignant neoplasms, or severe infectious diseases were excluded from the study. The demographic and clinical data of the patients and healthy controls were collected from physical examinations, structured questionnaires, and medical records. The information collected included age, height, weight, body mass index (BMI), ethnicity, menarche age, menopause status and tumor characteristics. The BMI was classified into two groups: non-obese <23.9 kg/m2, and obese >24 kg/m2. Furthermore, the ethnicity was classified into Han, Zhuang, and other groups. The tumor characteristics involved metastasis distance, clinical stage, molecular classification, pathology type, menopausal status, obesity status. The metastasis distance and clinical stage (I, II, III, and IV) was according to the tumor-node-metastasis (TNM) classification of BC. The pathological result was used to assess the type of cancer. The molecular type of the patient was performed by the 2011 St. Gallen Consensus 21 : Luminal A subtypes (ER and/or PR positive, 22 HER-2 negative, 23 ki-67 <14%), Luminal B1 (ER and/or PR positive, HER-2 negative, ki-67 >14%), Luminal B2 (ER and/or PR positive, HER-2 positive, ki-67 any level), HER-2 overexpressing (ER and PR negative, HER-2 positive), and triple-negative (ER negative, PR negative, HER-2 negative). Before recruitment, each participant signed a written informed consent, and the study was approved by the Institutional Ethics Review Board of the Guangxi Medical University.
DNA extraction and genotyping
The DNA was extracted from the blood samples from cases and controls using a DNA isolation kit (Sangon Biotech, Shanghai, China). The genotyping of rs12922061 and rs3803662 was performed by SNPscan system (Genesky Biotechnologies Inc., Shanghai, China). The details were presented in other studies (http://biotech.geneskies.com/en/index.php/Index/fuwuer/id/29). SNPscan technology is a multi-gene mutation screening technology that uses the specificity of the ligase-linked reaction to identify the SNP allele; it then introduces different length non-specific sequences at the end of the probe; polymerase chain reaction amplification was performed by using fluorescently-labeled universal primers; and the amplified products were separated by capillary electrophoresis. Finally, Gene Mapper software was used to analyze the genotype of each SNP locus. The sequences of specific ligase probes were as follows: TOX3, GGTCAGTCCACAGTTTTATTCTTCGCTATGG and GGTCAGTCCACAGTTTTATTCTTCGCTACGA; and LOC643714, CCTGTGAGAGAGATAGAAGAGAATGTGATAAAGG, and GACTATCTCATGTGCTGAGATGTGAGTAGA.
Statistical analysis
Data were recorded as a Microsoft Excel file and analyzed by Statistical Package for Social Sciences program, version 22.0 (IBM, São Paulo, Brazil). The Chi-square test (X2) or Fisher’s exact test were used to calculate the qualitative data, depending on the genotypes or groups. The Student t-test was used for quantitative data (mean ± SD) to compare the variables of the patients and control groups. The association between BC risk and genotype distribution or population characteristics was calculated by simple unconditional logistic regression and multiple logistic regression analysis with an odds ratio (OR) and 95% confidence interval (CI) applied in the assessments. In this study, four genotype models (dominant model: TT+TC vs. CC/GG+GA vs. AA; recessive model: TT vs. TC+CC/GG vs. GA+AA; over-dominant model: TT+CC vs. TC/GG+AA vs. GA; log-additive: TT vs. TC/GG vs. GA, TT vs. CC/GG vs. AA) were applied. 24 The statistical power was evaluated by Power and Precision V4 software (http://www.power-analysis.com/). SHEsis (http://analysis.bio-x.cn/myAnalysis.php) was applied to perform the haplotype analysis and Hardy–Weinberg equilibrium (HWE).25,26 Receiver operating characteristic (ROC) curves were tested to evaluate the predictive role of significant loci in BC risk. In the whole analysis, P values of less than 0.05 were considered statistically significant. All of the P values were two tailed.
Result
Characteristics of the study population
No significant difference was observed in the distribution of age, race, BMI, or age at menarche between the case and control groups (P>0.05), which suggested that the cases and controls of this study were matched adequately for general characteristics (Table 1). In addition, the stratified analyses of obesity status, age, ethnicity, and menarche age were performed in the case and control groups, which showed a significant association between menarche age and BC. Menarche age of 15 to 17.9 years old is protective against BC when compared to an age of 12 to 14.9 years old (OR=0.451; 95% CI 0.253, 0.805) (Table 2).
The characteristics of the study population.
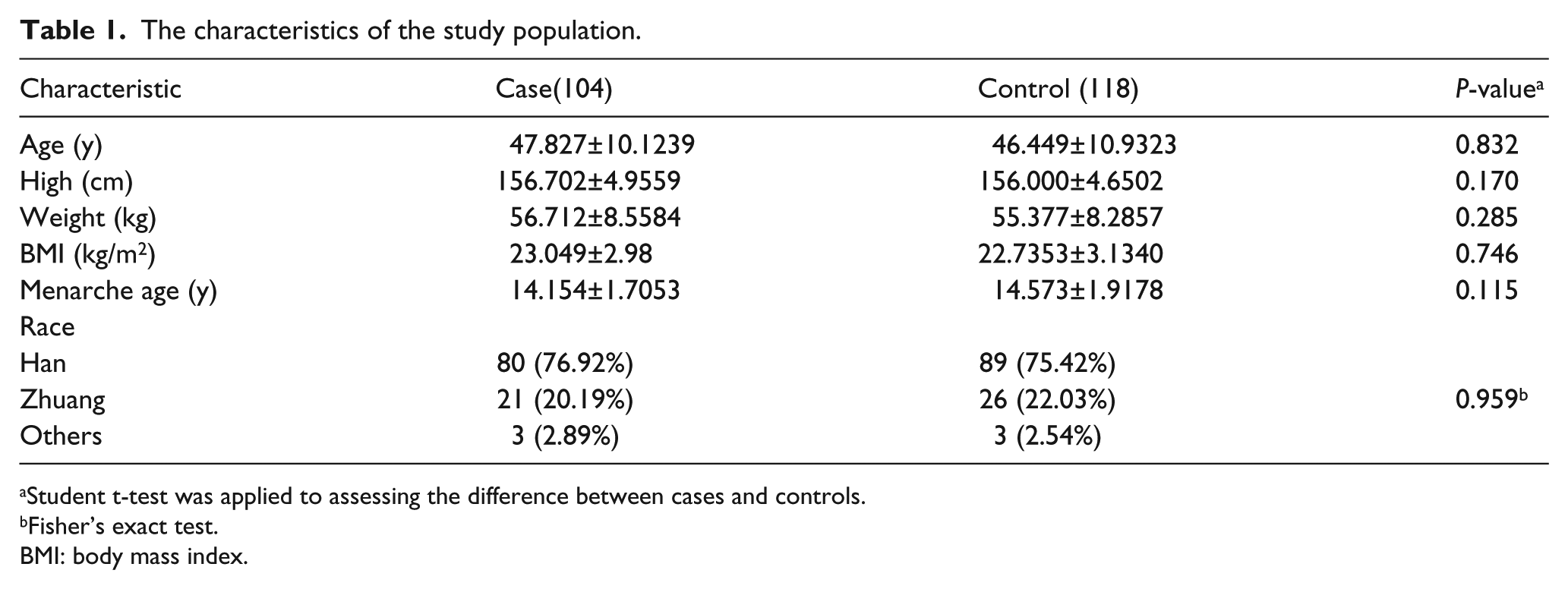
Student t-test was applied to assessing the difference between cases and controls.
Fisher’s exact test.
BMI: body mass index.
Stratified analysis of BMI, age, ethnicity, menarche age in case and control.
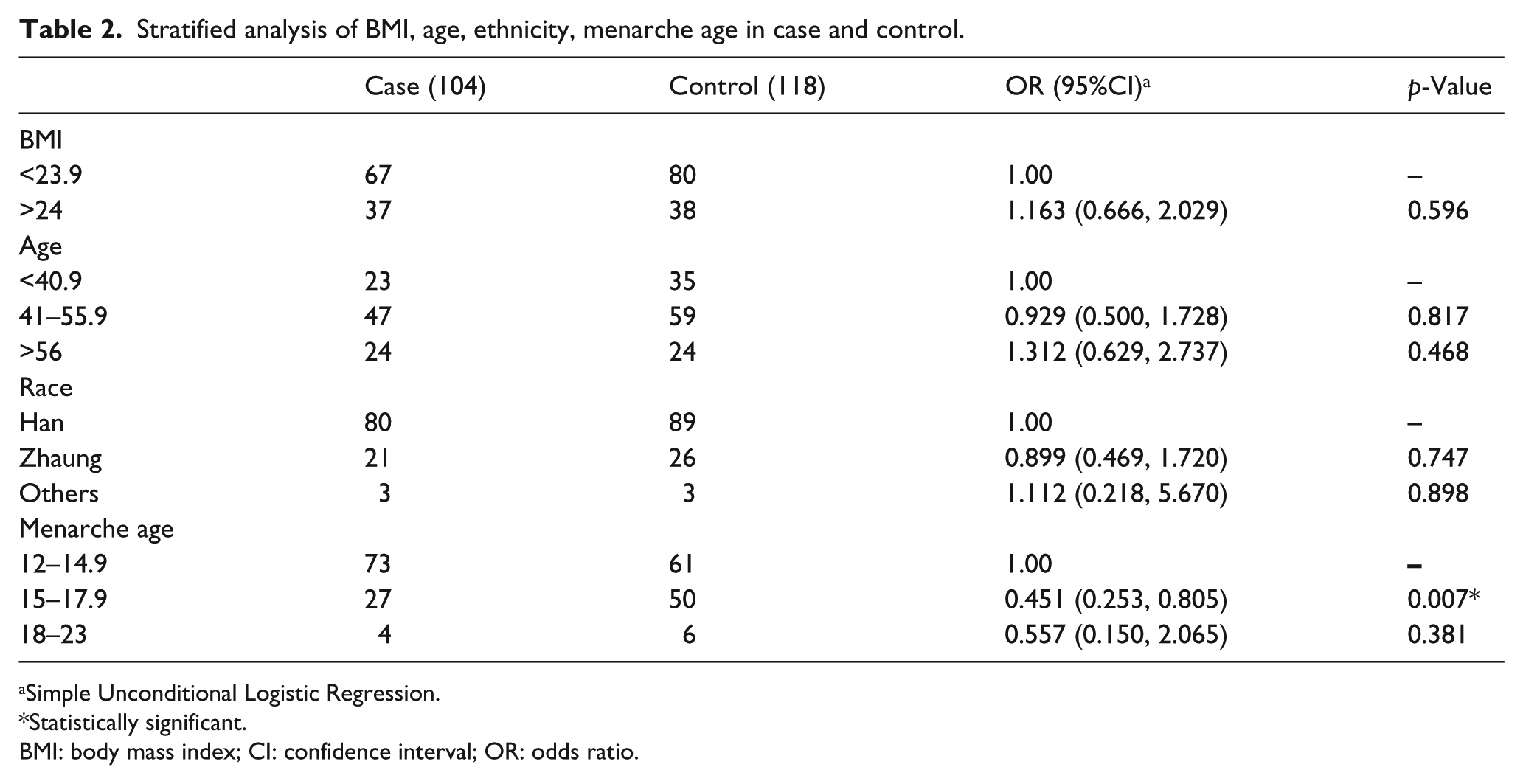
Simple Unconditional Logistic Regression.
Statistically significant.
BMI: body mass index; CI: confidence interval; OR: odds ratio.
The polymorphisms rs12922061 and rs3803662 are associated with the risk of BC
We chose two SNPs to test the association between LOC643714 and TOX3 gene polymorphisms and BC risk. The genotype distribution of these two SNPs is shown in Table 3. Based on the data, SNP rs12922061 was associated with an increased risk of BC under a dominant model (TT+TC vs. CC: OR=1.801; 95% CI 1.048, 3.095; statistical power=60%), recessive model (TT vs. TC + CC: OR=4.297; 95% CI 1.164, 15.867; statistical power=64%), and log-additive model (TT vs. CC: OR=5.163; 95% CI 1.368, 19.485; statistical power=73%). The over-dominant model (TT+CC vs. TC: OR=1.573; 95% CI 0.919, 2.694; statistical power 15%) and the log-additive model (TT vs. TC: OR=3.333; 95% CI 0.867, 12.813; statistical power=46%) were not statistically significant. Furthermore, compared with the C allele, the T allele of 12922061 was associated with a significantly increased risk of BC (Table 3). After adjusting for age, SNP rs12922061 was significantly associated with BC under a dominant model (TT+TC vs. CC: OR=1.753; 95% CI 1.016, 3.025), a recessive model (TT vs. TC+CC: OR=4.318; 95% CI 1.167, 15.985) and a log-additive model (TT vs. CC: OR= 5.109; 95% CI 1.352, 19.316). The results indicated that rs12922061 might have a significant association with BC risk. However, in the current study, there was no significant association between rs3803662 and BC risk.
The genotype distribution of rs12922061 and rs3803662 polymorphisms in cases and controls.
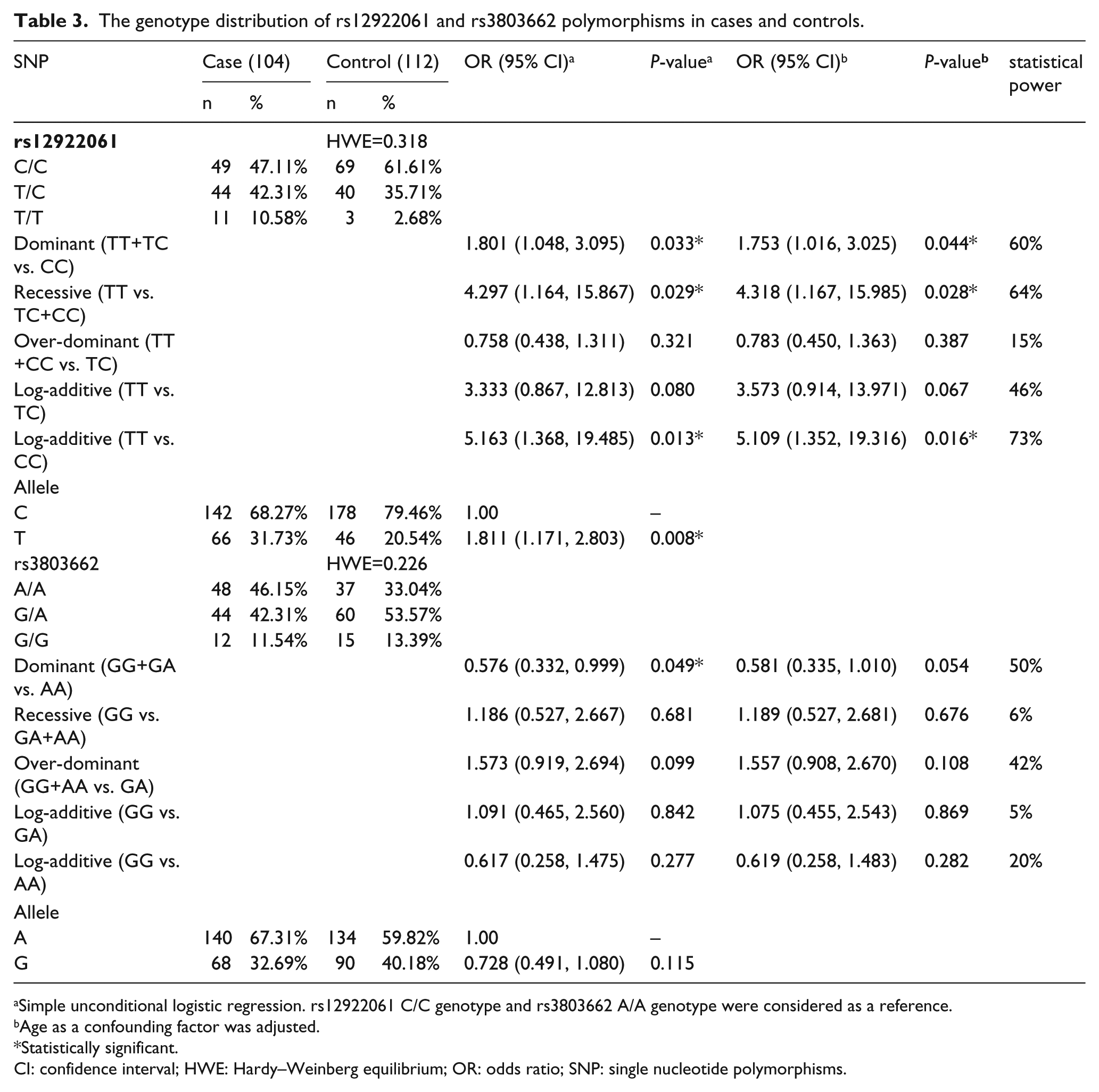
Simple unconditional logistic regression. rs12922061 C/C genotype and rs3803662 A/A genotype were considered as a reference.
Age as a confounding factor was adjusted.
Statistically significant.
CI: confidence interval; HWE: Hardy–Weinberg equilibrium; OR: odds ratio; SNP: single nucleotide polymorphisms.
SHEsis software was used to conduct haplotype analysis. The results suggested that the TA haplotype was significant (OR=1.893; 95%; CI 1.213, 2.956, P=0.005) (supplementary Table 1). In addition, the ROC curves suggested that the area under the curve of the dominant model for predicting BC is the largest (AUC=0.572; 95% CI 0.496, 0.649) compared with the recessive model (AUC=0.539; 95% CI 0.462, 0.617), the over-dominant model (AUC=0.533; 95% CI 0.456, 0.6101), the log-additive model (TT vs. TC) (AUC=0.565; 95% CI 0.452, 0.679), and the log-additive model (TT vs. CC) (AUC=0.571; 95% CI 0.472, 0.670) (supplementary Figure 1).
Meanwhile, we used statistical analysis to assess the association of rs12922061 genotype distribution and clinical characteristics in the patient group. A significant difference in postmenopausal patients and premenopausal patients was seen under the dominant model (OR=3.158; 95% CI 1.410, 7.073; statistical power=82%) and the over-dominant model (OR= 2.629; 95% CI 1.170, 5.907; statistical power=76%) (Table 4, Table 5). However, there were no significant differences between the genotype distribution of rs12922061 and other clinicopathological features of BC patients (Table 4).
The relationship between rs12922061 polymorphism and tumor clinic characteristic.
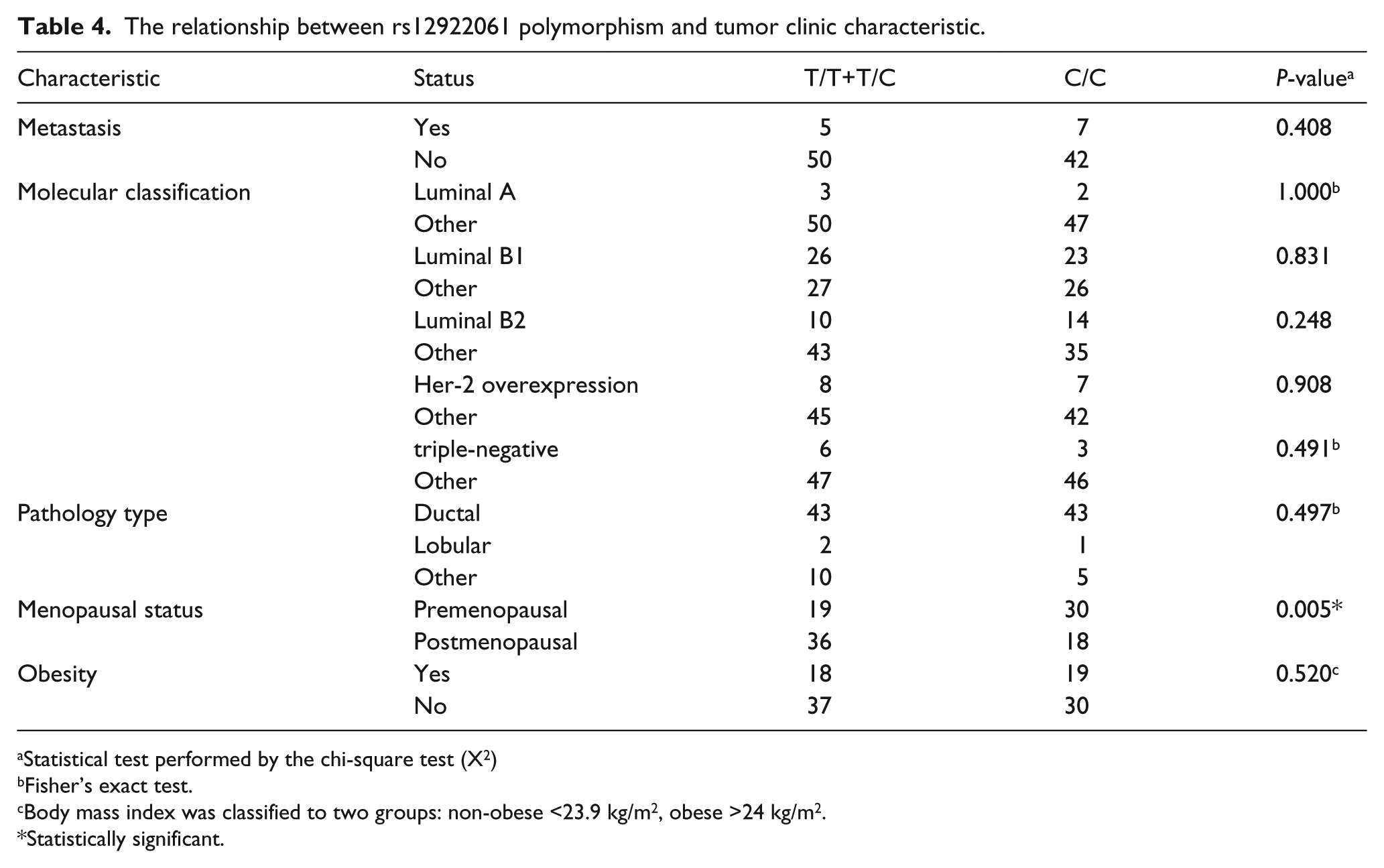
Statistical test performed by the chi-square test (X2)
Fisher’s exact test.
Body mass index was classified to two groups: non-obese <23.9 kg/m2, obese >24 kg/m2.
Statistically significant.
The association between rs12922061 polymorphism and menopausal statue.
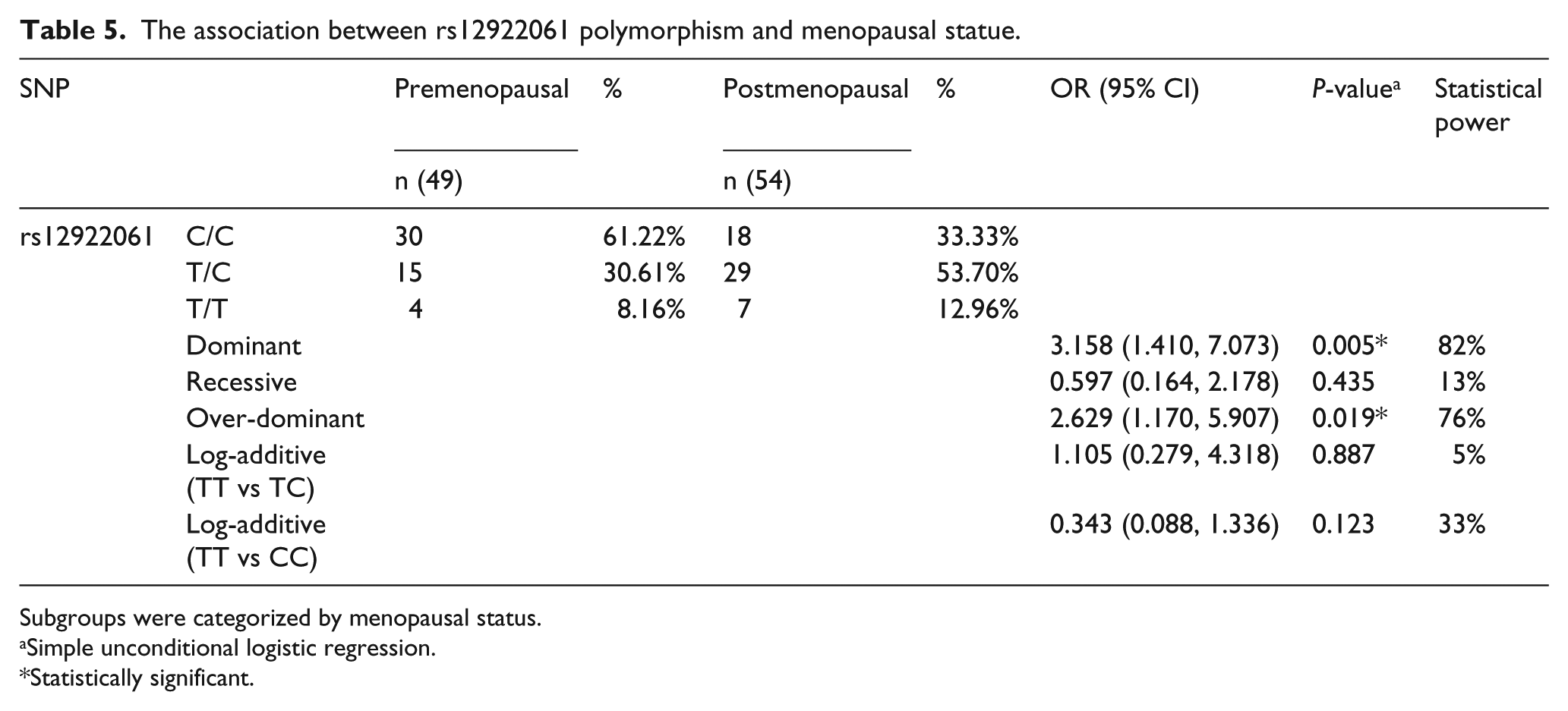
Subgroups were categorized by menopausal status.
Simple unconditional logistic regression.
Statistically significant.
Discussion
It is well known that BC is a public health problem with a complex etiology, different clinical appearances, and tumor aggressiveness.27,28 Genetics plays an important role in the occurrence and evolution of BC. In this study, we aimed to evaluate the association between two SNPs of TOX3 and LOC643714, and BC risk as well as the clinical characteristics of the tumor in a total of 104 BC cases and 112 control individuals. We found that the SNP of rs12922061 was a risk factor for BC and associated with the menopausal status in BC patients. Previous studies reported that there was a relationship between SNPs and BC. Al Zoubi et al. 29 found that polymorphisms and mutations in RAD51 and XRCC3 genes increased the risk of BC; Krivokuca et al. 30 reported an association between XRCC1 polymorphism and BC; and the results of meta-analysis showed that the MDM4 SNP34091 A>C polymorphism may function as a protective factor against cancer risk. 31 Our study found that rs12922061 of LOC643714 was a risk factor for BC.
The SNP rs12922061 is located in the first intron of LOC643714. A previous study reported that the LOC643714 gene codes for a small mRNA would be translated into a protein of 55 amino acids and suggested that causal variant(s) are not located in the TOX3 gene but rather at the LOC643714 locus. 15 In the present study, our results demonstrated that rs12922061 polymorphism was a risk factor for BC. We confirmed the previous findings of the GWAS from Japan and China, showing that there was an association between BC risk and a locus in the LOC643714 region.17,19 In our study, we found a significant difference of rs12922061 genotype distribution in postmenopausal and premenopausal patients under the dominant model (OR=3.158; 95% CI 1.410, 7.073, statistical power=82%), and the over-dominant model (OR= 2.629; 95% CI 1.170, 5.907; statistical power=76%). A previous study reported that the level of serum estrogen concentrations in postmenopausal women is significantly lower than premenopausal women. 32 Also, experimental data strongly suggest that estrogen plays a role in the development and growth of BC. 33 The epidemiologic studies reported that low serum estrogen concentrations were associated with a low risk of BC. Meanwhile, high concentrations were associated with a high risk. 34 Therefore, to some extent, the risk effect of rs12922061 for BC might be masked by the high serum estrogen concentrations in premenopausal period. After the menopause, with the decrease in female serum estrogen concentrations, the risk effect of rs12922061 might appear. Further study is needed to explain this relationship. Meanwhile, the statistical significance in the genotype distribution of rs12922061 and metastasis distance, clinical stage, molecular classification, pathological type and obesity status were not observed. The clinical pathological characteristics of tumors may be influenced by other genetic and environmental factors.
On the other hand, rs3803662 is located at 16q12.1. It specifically binds to the BRCA1 promoter to down-regulates its expression by altering the methylation status of the promoter. BRCA1 expression was found to be significantly reduced in a proportion of BC.13,35 The high expression of TOX3 may influence BC progression and prognosis, and Gudmundsdottir et al. 13 reported that rs3803662 participates in the regulation of TOX3 mRNA expression. Our results are consistent with a recent meta-analysis, which indicated no association between rs3803662 polymorphism and BC risk in Asian women. 18 However, a significant association was identified in the study involving 1156 cases and 1256 controls in China. 19 Several other studies also reported the association between rs3803662 and BC.13,14,18,35-37 The discrepancies of the above-mentioned studies may have been caused by different populations and sample sizes. Further studies are needed to confirm the findings of our study.
Limitations
There are some limitations in our study which should be highlighted. First, the selection bias could not be avoided; our patients and controls were selected from only one hospital. Second, the sample size was small; especially in subgroups, statistical power may be limited to finding differences between the groups.
Conclusions
BC is ubiquitous, with a high mortality in patients. In summary, our present results suggest that the BC is associated with polymorphism rs12922061 in LOC643714. Furthermore, we found that the polymorphism rs12922061 was significantly increased in postmenopausal patients compared with premenopausal patients. Further studies are needed to identify the role of LOC643714 SNPs in BC and evaluate the possibility of using them to diagnose BC in the Chinese population.
Supplemental Material
Supplementary_Material_(JBM) – Supplemental material for Polymorphisms in the TOX3/LOC643714 and risk of breast cancer in south China
Supplemental material, Supplementary_Material_(JBM) for Polymorphisms in the TOX3/LOC643714 and risk of breast cancer in south China by Jinling Liao, Yang Chen, Jia Zhu, Qiuyan Wang and Zengnan Mo in The International Journal of Biological Markers
Footnotes
Author contributions
Liao and Chen contributed equally to this paper.
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the National Natural Science Foundation of China (grants number:81370857), Innovation Project of Guangxi Graduate Education (YCBZ2017037).
Supplementary Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
