Abstract
Introduction and aims
Atopic dermatitis (AD) is a chronic inflammatory skin disease characterized by relapse, persistent pruritus, and eczema-like lesions. Although AD typically occurs at an early age, adults can also be affected.1,2 Currently, AD affects 20% of children and 5%–10% of adults. 3 AD pathophysiology is complex and includes skin barrier dysfunction, environmental and immunological factors, and gene interactions.2,4 Multiple cytokines and cells are involved in the pathogenesis of AD, including interleukin (IL)-4, IL-5, IL-9, IL-13, eosinophils, basophils, and mast cells.5–7
Thymic stromal lymphopoietin (TSLP) is an IL-7-like four-helical bundle cytokine generated by keratinocytes.8,9 TSLP mainly targets dendritic cells (DCs), and upregulates the expression of CD80, OX40L, and CD86. 10 Additionally, TSLP promotes IL-4, IL-5, IL-9, and IL-13 expression through its action on mast cells, epithelial cells, macrophages, and innate lymphoid cells. 10 The human TSLP gene is located on chromosome 5q22.1, which is adjacent to 5q31, a gene located in the cytokine cluster of atopic dermatitis. 11 TSLP is involved in the pathogenesis of various diseases including asthma, AD, inflammatory arthritis, and cancer.11–13 The three main TSLP polymorphisms under investigation are rs1898671, rs1837253, and rs11466749.14–18 Variants of the TSLP gene can alter TSLP protein activity, leading to inflammatory responses. For example, in patients with AD, the rs1898671 polymorphism is associated with disease persistence and risk of eczema herpeticum.18,19
Gene polymorphisms and TSLP levels are associated with the risk of AD. However, these studies have limitations. For example, the sample sizes were relatively small, and divergent or even opposite results were observed. The statistical power was limited for individual studies. Therefore, we performed a meta-analysis and trial sequential analysis (TSA) to more comprehensively evaluate the association between TSLP and AD.
Materials and methods
Literature search
Six databases, including PubMed, Web of Science, EMBASE, Ovid, Sinomed, and China National Knowledge Infrastructure (CNKI), were searched from their establishment date to 2022.04.30. The search strategies varied for different databases and are described in the Supplemental Materials.
Inclusion and exclusion criteria
The inclusion criteria were 1 : published studies on the relationships between TSLP and AD, including TSLP gene rs1898671, rs1837253, and rs11466749 polymorphisms and blood or serum protein levels 2 ; case-control studies or cohort studies 3 AD diagnosis through clinical diagnostic criteria 4 ; controls were healthy individuals or individuals without related diseases 5 ; sufficient information of TSLP genotype frequencies or levels was provided 6 ; for studies with gene polymorphisms, p-values, odds ratios (ORs), and 95% confidence intervals (95% CIs) were provided or could be calculated from the study data 7 ; for studies with levels, the form of mean ± standard deviation (SD) was provided or could be calculated.
The exclusion criteria were 1 : conference publications, case reports, reviews, commentary, or animal studies 2 ; duplicate data 3 ; studies that failed to provide adequate information to calculate levels or genotype frequencies; and 4 for studies with levels, the original data in the study required transformations to the form of mean ± SD.
Two investigators (Zhili Xu and Boyang Zhou) conducted the selection process. In cases of disagreement, another investigator (Tianhang Yu) checked the procedure.
Data extraction
The following information was extracted from the included studies 1 : first author’s name and year of publication 2 ; country (or region) where the study was performed 3 ; ethnicity and gender of the case and control subjects for studies with gene polymorphisms 4 ; sample size of both groups 5 ; TSLP genotype frequencies or levels of both groups 6 ; units and methods of detection for studies with levels 7 ; results of Hardy Weinberg Equilibrium (HWE) for studies with polymorphisms. Two investigators (Zhili Xu and Suhua Wu) performed the extraction. Another investigator (Linfeng Li) reviewed the information and corrected any errors.
Methodological quality assessment
The Newcastle–Ottawa scale (NOS) 20 was used to evaluate the quality of the included studies. The NOS includes scores for selection, comparability, and exposure. The maximum score is 9; when a study attained a score of ≥6, it was considered to be of high quality. Two investigators (Zhili Xu and Boyang Zhou) completed the assessments. If any controversy was encountered, another investigator (Linfeng Li) checked the evaluation procedure and made a final decision.
Statistical analysis
For studies with TSLP levels, the standardized mean difference (SMD) was applied to eliminate the impact of different units and measurements of detection. If the data was not in the form of mean ± SD in the publication, transformation was conducted based on the methods of Luo et al. and Wan et al.21,22 For studies with gene polymorphisms, Hardy-Weinberg Equilibrium (HWE) in the control group was calculated using STATA 16. 23 Evaluation was performed using ORs and 95% confidence intervals (CIs). The Hartung–Knapp–Sidik–Jonkman (HKSJ) method was employed as an estimator in the meta-analysis because of its greater reliability, particularly in studies with small sample sizes. 24 For each polymorphism in the TSLP gene, three exposure groups (DD, Dd, and dd) were defined based on two alleles (D and d). Five genetic models were applied in our study, including allele contrast (D vs d), homozygous contrast (DD vs dd), heterozygous contrast (Dd vs dd), dominant contrast (DD + Dd vs dd), and recessive contrast (DD vs Dd + dd). For each comparison, a p-value <0.05 was defined as statistically significant.
Based on the between-study heterogeneity, a fixed- or random-effects model was chosen. Q-statistics and I2 tests were used to assess heterogeneity. For a p-value >0.10 and I2 <50%, the heterogeneity was not significant, and the fixed-effects model was selected; otherwise, the random model was used. Sensitivity analysis was used to identify the impact of an individual study by omitting the studies in that order. Funnel plots were generated to estimate the publication bias for comparisons with five or more studies. The analysis was performed using RevMan 5.3 software and R 4.3.0 software.20,24
Trial sequential analysis
A TSA was performed to determine whether the number of cases was sufficient for comparison. In our study, the type I error (false positive) was set at 5%, and the type II error (false negative) was set at 30%. The analysis was performed using a Java-based TSA application.25,26
Results
Selection of the studies
A total of 4,150 publications were obtained from these six databases. Subsequently, 2,041 duplicate records were removed and 2,109 were screened for titles and abstracts. A total of 1,989 publications were excluded and the remaining 120 were screened for the whole content. On the basis of the selection criteria, 107 patients who did not meet the inclusion criteria were excluded. Thirteen studies were initially included; after the entire content and data were checked, all 13 were included. The selection flowchart is shown in Figure 1. Flowchart of selection.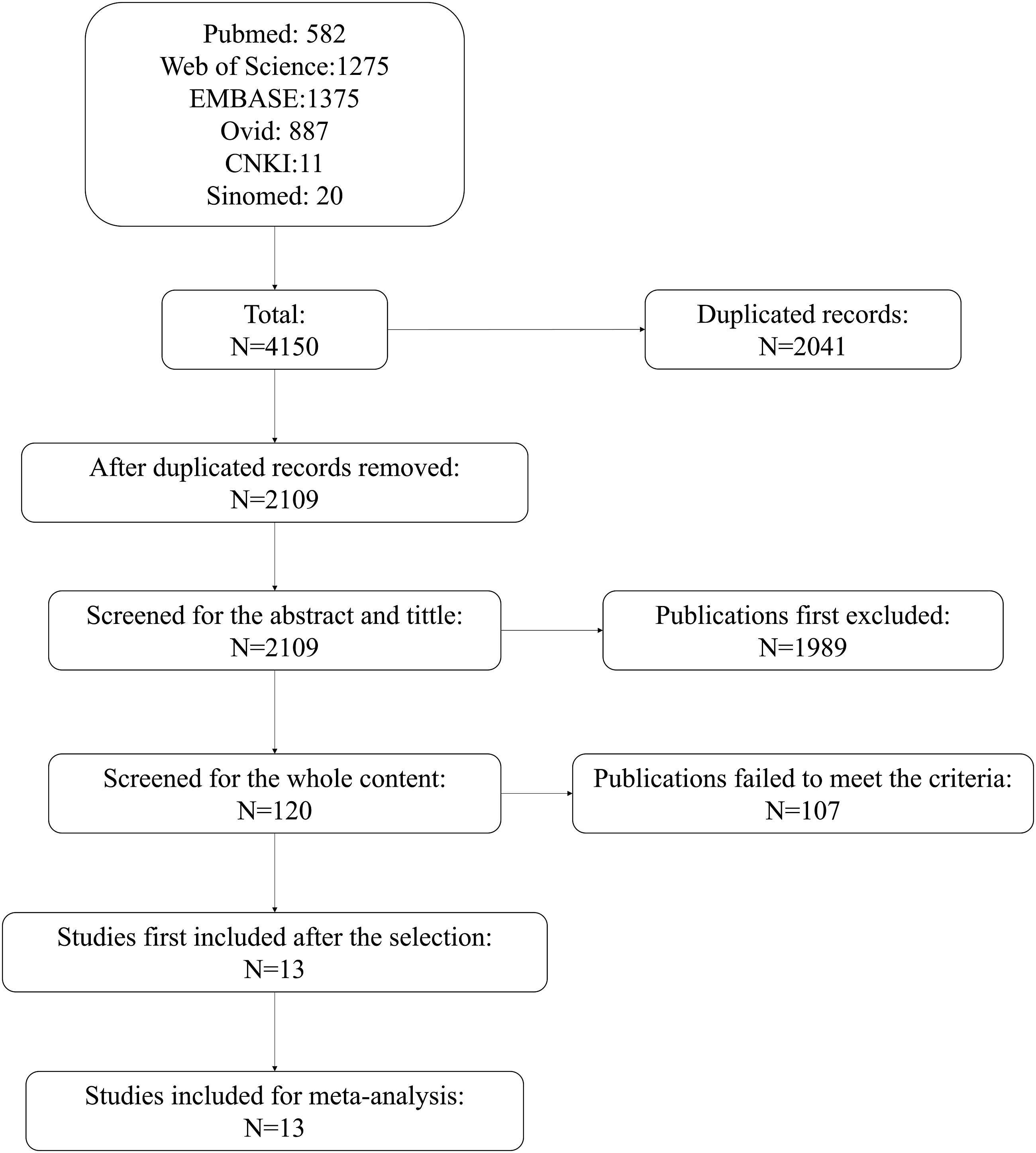
Characteristics of included studies
Thirteen studies were included.27–39 Five studies reported genetic polymorphisms.27–31 Three gene polymorphisms, namely rs1898671, rs1837253, and rs11466749, were investigated.
For rs1898671 polymorphisms, two studies were included, which individually investigated cases from Poland and Taiwan of China.29,31 The mean ages were 25.5 years and 5.94 ± 2.73, respectively. The diagnosis of AD was based on the International Study of Asthma and Allergies in Childhood (ISAAC) questionnaire and Hanifin and Rajka criteria. The sample sizes were 375 and 232 in the case group, and 1145 and 157 in the control group.
For rs1837253 polymorphisms, two studies were included, which individually investigated cases from Japan and Mainland China.28,30 The mean ages were 30.7 ± 4.6 and 5.51 ± 7.93 years respectively. AD diagnosis was based on the ISAAC questionnaire and the Hanifin and Rajka criteria. The sample sizes were 188 and 2,954 in the case group and 565 and 5,347 in the control group.
For rs11466749 polymorphisms, two studies were included, which individually investigated cases from Taiwan of China and Ukraine.27,31 The ages were 5.94 ± 2.73 years and a median of 8 years, respectively. AD diagnosis was based on the ISAAC questionnaire and multiple criteria. The sample sizes were 375 and 47 in the case group and 1,145 and 80 in the control group, respectively.
Both the alleles and detailed case numbers in the exposure groups was reported in the included studies; therefore, all five genetic models could be applied. Among the six study groups, five were compliant with the HWE, whereas one group with the rs1898671 polymorphism was not.
Eight studies reported on TSLP levels and AD (32-39). Among them, three were conducted in Korea,33,35,39 Japan, 36 China, 32 Denmark, 37 Turkey, 38 and Poland. 34 Three studies investigated the levels in children,33,35,38 whereas the other five covered more age groups.32,34,36,37,39 Seven studies adopted the Hanifin and Rajka criteria to diagnose AD (33-39), whereas only one study applied the Williams criteria. 32 All detection methods were ELISA, and the units were pg/mL in seven33–39 and ng/L in one. 32 The sample sizes varied from 25 to 132 in the case group and from 10 to 87 in the control group.
Basic characteristics of studies with TSLP gene polymorphisms and AD.

Basic characteristics of studies with TSLP level and AD.

Results of study quality assessment through Newcastle-Ottawa scale.

Associations between TSLP gene polymorphisms and AD
For rs1898671 polymorphism, there were significant relationships with AD under homozygous (DD vs dd), and dominant contrasts (DD + Dd vs dd), with OR = 0.47 (95% CI: 0.24, 0.92) and 0.46 (95% CI: 0.24, 0.88), respectively (p < 0.05). Under allele (D vs d), heterozygous (Dd vs dd) and recessive contrasts (DD vs Dd + dd), no significant relationship was found, with OR = 0.95 (95% CI: 0.79, 1.15), 0.35 (95% CI: 0.00, 281.55) and 1.14 (95% CI: 0.84, 1.54), respectively (Figure 2). Forest plots of rs1898671 polymorphisms and AD.
For rs1837253 polymorphism, no significant relationship was found under each contrast (p > 0.05), with OR = 0.96 (95% CI: 0.13, 7.24) for allele, 0.98 (95% CI: 0.03, 31.80) for homozygous, 1.08 (95% CI: 0.95, 1.22) for heterozygous, 1.11 (95% CI: 0.99, 1.26) for dominant, and 0.93 (95% CI: 0.05, 17.96) for recessive contrasts, respectively (Figure 3). Forest plots of rs1837253 polymorphisms and AD.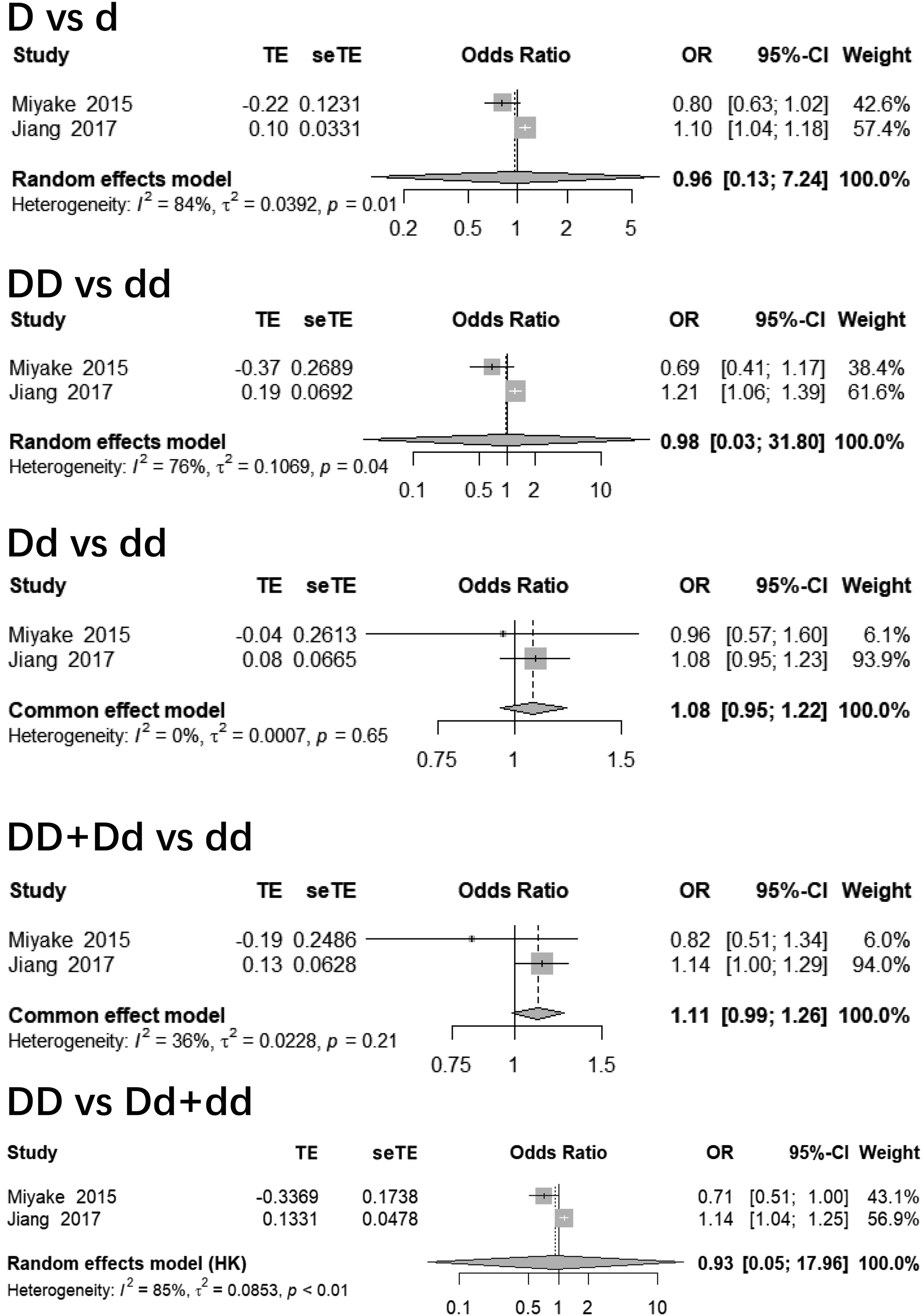
For rs11466749 polymorphism, again no significant relationship was found under each contrast (p > 0.05), with OR = 1.03 (95% CI: 0.78, 1.34) for allele, 0.61 (95% CI: 0.24, 1.59) for homozygous, 0.55 (95% CI: 0.21, 1.47) for heterozygous, 0.61 (95% CI: 0.24, 1.57) for dominant, and 1.08 (95% CI: 0.80, 1.45) for recessive contrasts, respectively (Figure 4). Forest plots of rs11466749 polymorphisms and AD.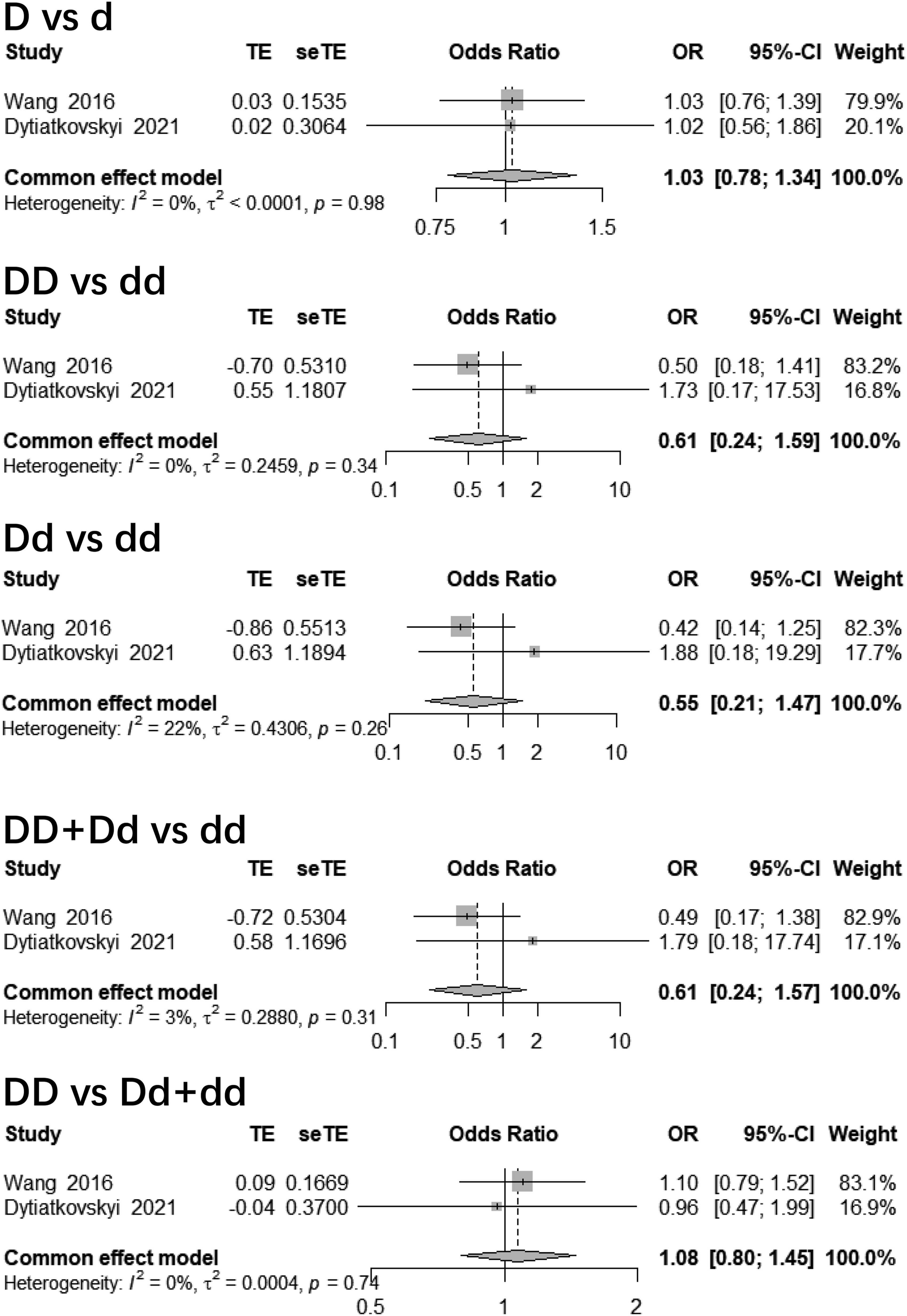
Association between TSLP levels and AD
Overall, 447 cases and 251 control subjects from eight studies were included. The pooled data suggested that serum TSLP levels were significantly higher in patients with AD compared with control subjects, with the SMD = 1.45 (95% CI: 0.65–2.25) (p < 0.05) (Figure 5). Forest plots of TSLP levels and AD.
Sensitivity analysis and publication bias
For comparisons with polymorphisms, the significance changed when a given study was omitted under some contrast in the rs1898671 and rs1837253 polymorphisms. This indicates that the results of these gene polymorphisms were not perfectly robust. In comparisons of the rs11466749 polymorphism, the significance did not change when a given study was omitted. This indicated that the results for the rs11466749 polymorphism were robust (sTable 2 in the Supplemental Material).
For comparison with TSLP levels, the significance did not change when each study was omitted individually. Therefore, the association between TSLP levels and AD was robust.
Eight studies were included for the comparison of TSLP levels. Funnel plots were used to evaluate publication bias. The plot was asymmetric, indicating a publication bias (Supplemental Figure 1).
Subgroup analysis
In each polymorphism comparison, it was unnecessary to perform a subgroup analysis because only two study groups were included.
In the comparison of TSLP levels, eight studies were included, and the heterogeneity was significant. Subgroup analysis was performed according to location and age. In the subgroup analysis by location, significant relationships were observed in both Asia and Europe. However, heterogeneity remained significant in each subgroup. In the subgroup analysis by age group, there were significant relationships both in children and in patients with a mean or median age of >18 years. Heterogeneity was not significant in the subgroup of children, whereas it was significant in older patients (Supplemental Figure 2).
Trial sequential analysis
For rs1898671, the Z-curve failed to reach the required information size (RIS) line under heterozygous contrast (Dd vs dd), indicating an inadequate sample size. The Z-curves crossed the monitoring boundary under homozygous (DD vs dd), heterozygous (Dd vs dd), and dominant contrast (DD + Dd vs dd) conditions, indicating the significance of the meta-analysis results.
For the rs1837253 and rs11466749 polymorphisms, the Z-curves failed to reach the RIS line and the monitoring boundary under all contrasts. The sample sizes were inadequate, and there was no significant difference in the meta-analysis results.
For comparison with the TSLP levels, the Z-curve crossed the RIS line and monitoring boundary. The sample sizes were adequate and the meta-analysis results were significant. The TSA results are presented in Figures 3–6 in Supplemental Material.
Discussion
TSLP is primarily secreted by the epithelial cells, airway smooth muscle cells, keratinocytes, stromal cells, fibroblasts, mast cells, macrophages, monocytes, granulocytes, and DCs. TSLP plays various pathophysiological roles in immune-related diseases. 40 In this study, we analyzed the relationship between TSLP and AD, which is more comprehensive and specific compared with previous studies. This relationship has been diverse in previous studies. Nakamura et al. 36 reported that TSLP levels were not elevated in patients with AD, whereas Uysal et al. 38 found that serum TSLP levels could be used as a biomarker in AD to predict severity. Therefore, we conducted a meta-analysis to comprehensively evaluate this relationship. With further research on AD pathogenesis, novel topical and systemic treatments, including dupilumab, tralokinumab, nemolizumab, crisaborole, and small-molecule Janus kinase (JAK) inhibitors, are being rapidly developed.2,41–43
Tezepelumab (AMG157/MEDI9929), an anti-TSLP monoclonal antibody, has been widely investigated for asthma treatment. Multiple studies have confirmed its efficacy in asthma.44–47 The FDA has approved the use of TSLP-targeted antibodies for severe asthma. 48 In 2019, Simpson et al. reported an optimistic response to tezepelumab in AD patients. 49 However, only a few studies have focused on the clinical application of tezepelumab in AD. We analyzed the association between TSLP gene polymorphisms, and its level, and the risk for AD. Our study lays the foundation for future research and provides a reference for future studies evaluating anti-TSLP mAbs in patients with AD.
Three gene polymorphisms, namely rs1898671, rs1837253, and rs11466749, were investigated in the present study. The rs1898671 polymorphism is an intron variant of the TSLP gene located on chr5:111072304. This polymorphism is mainly associated with allergic diseases such as AD (50), allergic rhinitis, 51 and eosinophilic esophagitis. 52 For the rs1898671 polymorphism, we found significant relationships with AD in homozygous (DD vs dd) and dominant (DD + Dd vs dd) contrasts in the present study. From our results, we conclude that the TSLP rs1898671 polymorphism is associated with AD. However, the sensitivity analysis showed that the results under these contrasts were not robust. Furthermore, in trial sequential analyses, inadequate sample sizes were included only under heterozygous contrast (Dd vs dd). Considering the overall quality of these results, further studies with a larger number of cases are required. The rs1837253 polymorphism is located on chr5:111066174, whereas the rs11466749 polymorphism is a non-coding transcript variant located on chr5:111076887. Both polymorphisms are associated with allergic diseases.14,15,17,51–54 In the present study, no significant relationship was found under each contrast for either polymorphism, and the TSA results indicated that the sample sizes were inadequate. Therefore, further studies with larger sample sizes should be conducted to investigate these relationships more thoroughly. Moreover, considering the diverse results of current studies, more research on both polymorphisms, focusing on their functions, is required.
Single nucleotide polymorphisms (SNPs) in different genomic regions affect gene function through various mechanisms. 55 More than 50% of SNPs are found in non-coding regions, where they primarily influence gene function by affecting the regulatory elements involved in gene expression or by altering splice-site activity, thus affecting translation. In coding regions, synonymous coding SNPs, while not altering the protein sequence, may still affect processes, such as protein folding, whereas non-synonymous SNPs directly change the amino acid composition of the encoded protein, with a functional impact depending on whether the altered amino acid plays a crucial role in the structure or function of the protein, rs1898671 is located in an intronic region, rs1837253 is located upstream of the transcription start site of TSLP, and rs11466749 is a noncoding transcript variant.14,15,17,50 However, no studies have systematically investigated how rs1898671, rs1837253, and rs11466749 polymorphisms specifically affect TSLP protein levels, structure, or function. Further research is required to address this gap.
We also analyzed the relationship between serum TSLP levels and AD. TSLP is primarily generated by the epithelial and stromal cells of the skin, lungs, and gastrointestinal tract. In tissues and blood, TSLP acts as an alarmin. After being released from cells, TSLP participates in inflammatory and immune pathways. 56 The main targets of TSLP are DCs. Moreover, TSLP regulates neutrophils, basophils, eosinophils, group 2 innate lymphoid cells, natural killer T cells, mast cells, and smooth muscle cells.57–59 TSLP plays an important role in the development of AD in humans. For example, Soumelis et al. found that TSLP was overexpressed in the skin lesions of patients with AD, 60 and in patients with asthma, TSLP levels were related to Th2 chemokine levels. 61 In the present study, we included eight studies comprising 447 cases and 251 control subjects and found that TSLP levels were elevated in patients with AD. Previous studies have expressed different views regarding the relationship between TSLP levels and AD. For example, Nakamura et al. 36 suggested that serum TSLP levels are not elevated in patients with AD, whereas Jaworek et al. 34 suggested that serum TSLP levels are higher in patients with AD, and may play a vital role in the mechanism of this disease. Considering the limitations of the individual studies, we performed a meta-analysis. After selection based on the inclusion and exclusion criteria, our analysis included both previous and current studies of TSLP levels and AD. Moreover, adequate cases were enrolled from the TSA, indicating that the meta-analysis results were reliable. Additionally, sensitivity analysis showed that the relationship between TSLP levels and AD was robust. Therefore, it can be concluded that TSLP levels are elevated in patients with AD.
In comparison with TSLP levels, subgroup analysis was subsequently conducted based on location and age range. In each subgroup, the effect remained significant, indicating that TSLP levels remained elevated in patients with AD. However, heterogeneity remained significant, except in the subgroup of children. The genetic backgrounds of patients from different locations may differ, but the mechanism of TSLP in AD may be similar. Therefore, it could be that other factors contributed to the heterogeneity. We further analyzed the age range. AD affects children the most (20%), whereas only 5–10% of adults are affected. 3 In younger patients, the immune function is still developing, which is different from that in adults; thus, the role of TSLP may differ in AD. Age may play a more important role than the region or genetic background. Consequently, the results of the studies included in the child subgroup showed high consistency and low heterogeneity. In the subgroup of patients with a mean or median age >18 years, patients with different age ranges were enrolled, which could result in significant heterogeneity. Since only two studies were included in each comparison of individual polymorphisms, subgroup analysis was unnecessary.
TSLP plays a critical role in the pathogenesis of AD and is a promising therapeutic target. A fully human IgG2λ monoclonal antibody, Tezepelumab, has been developed. Tezepelumab binds to a key part of the TSLP-binding site, preventing its interaction with TSLP receptor, thereby inhibiting various downstream inflammatory pathways. 62 Tezepelumab has also been investigated as a treatment option for AD (49). Phase I studies have demonstrated that tezepelumab exhibits predictable linear pharmacokinetics, acceptable safety, and tolerability. Phase IIa trials have shown a higher rate of clinical improvement in the tezepelumab treatment group than in the placebo group, although this difference was not statistically significant. 49 Other drugs targeting TSLP have also been developed and are currently undergoing clinical trials (e.g. BSI-045B, CM326). 63 TSLP-targeted therapy shows great promise and could significantly improve AD treatment in the future.
Our study had some limitations. First, in the comparison of levels, publication bias may exist in the evaluation of the funnel plot. Second, in our initial research, other TSLP gene polymorphisms were identified; however, for these polymorphisms, only one study was included per polymorphism; thus, we did not perform any further analysis. Additionally, in the selection procedure, we set the inclusion and exclusion criteria, and although some publications tended to meet these criteria, they were still excluded. For example, in the study by Mócsai et al., specific data were only shown in figures, but the bars of the histogram were not clearly defined as standard error (SE) or SD. 64 In a study performed by Wang et al., the mean and SE of different age groups were obtained from the figures, but more steps were needed to merge the data; therefore, errors were not readily noticeable during the procedure. 65 Due to the absence of access to the original data, we excluded such studies. Finally, previous studies confirmed that TSLP is highly expressed in human AD lesions. 66 However, we did not analyze the TSLP levels in these lesions. For different patients with AD, the manifestations of the lesions may differ; consequently, the cytokine expression profile may vary. However, when samples are collected from patients with different manifestations and severities, the results may be inconsistent. Therefore, future studies with well-designed protocols focusing on TSLP levels in the skin lesions of patients with AD are required.
Conclusion
This meta-analysis indicated that TSLP gene rs1898671 polymorphism is associated with AD and that serum TSLP levels are elevated in patients with AD. TSLP is strongly associated with the susceptibility to AD.
Supplemental Material
Supplemental Material - Thymic stromal lymphopoietin and atopic dermatitis: A systematic review and meta-analysis with trial sequential analysis
Supplemental Material for Thymic stromal lymphopoietin and atopic dermatitis: A systematic review and meta-analysis with trial sequential analysis by Zhili Xu, Boyang Zhou, Suhua Wu, Tianhang Yu and Linfeng Li in European Journal of Inflammation.
Footnotes
Author contributions
Zhili Xu: The study concept and design; review of the literature; data collection, and interpretation of data; statistical analysis; writing of the manuscript. Boyang Zhou: Review of the literature; data collection, and interpretation of data; statistical analysis; Suhua Wu: Data collection, interpretation of data, and statistical analysis; Tianhang Yu: Data collection, interpretation of data, and statistical analysis; Linfeng Li: The study concept and design, statistical analysis; data collection; effective participation in the research guidance; final approval of the final version of the manuscript.
Declaration of conflicting interest
The authors declare no potential conflicts of interest with respect to research, authorship and/or publication of this article
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
