Abstract
Background:
T-helper (Th)1/Th2 inflammatory responses are responsible for secretory otitis media (SOM) development. However, the mechanisms underlying these immune responses remain unknown. This study aims to identify the primary cytokines that play essential roles in chronic SOM.
Methods:
Two groups were established for the present study: chronic SOM group (n = 21) and control group (n = 10). The middle ear effusion and serum samples of the expression cytokines (interleukin IL-2, IL-4, IL-5, IL-13, IL-17, IL-25, IL-33, interferon [IFN]-γ, thymic stromal lymphopoietin [TSLP], immunoglobulin IgE, and pepsins) were analyzed by enzyme-linked immunosorbent assay.
Results:
The levels of IL-4, IL-5, IL-13, IL-17, IL-25, IFN-γ, TLSP, pepsins, IL-2, and IL-33 (all, p < 0.001) were higher in middle ear effusion, when compared to those in serum, in chronic SOM group (non-paired sample). However, there was no significant difference in serum expression for those cytokines compared chronic SOM group and control group. The paired sample expression for IL-33 and TLSP (both, p = 0.046) were higher compared the effusion and serum in chronic SOM group.
Conclusions:
IL-33 produces inflammatory cytokines, such as IL-1b, IL-6, TNF-α, IL-10, IL-4, and TGF-β, which through nucleus into cytoplasm causing inflammatory responses. The present study revealed that IL-33 also produce IL-17, IL-4, IL-5, and IL-13 inflammatory factors, triggering an inflammatory response. Study reported that the combined stimulation of TSLP and IL-33 elicits an approximately 10-fold increase in cytokine production, when compared to the stimulation of IL-33 alone. This suggests that IL-33 and TLSP may be the primary cytokines involved in Th1/Th2 inflammatory responses in chronic SOM.
Introduction
Secretory otitis media (SOM) is a common middle-ear inflammatory disease characterized by middle ear effusion, ear congestion, and conductive hearing loss. It is diagnosed on the otoendoscopic evidence of middle ear effusion, or by a type B or C tympanogram. SOM often occurs after acute middle ear infection by a bacterium (Streptococcus pneumoniae, Haemophilus influenza, or Moraxella catarrhalis 1 ) or virus (influenza virus, adenovirus, rhinovirus, or respiratory syncytial virus 2, 3). However, some patients with no previous ear infection develop SOM due to mechanical obstruction and dysfunction of the eustachian tube caused by upper respiratory infection, adenoid enlargement, allergic reaction (e.g. anaphylaxis or allergic rhinitis), the presence of a tumor, or gastroesophageal reflux.4,5 Inflammatory and immune factors, such as interleukin (IL)-2, IL-4, IL-5, IL-13, IL-17a, interferon (IFN)-γ, immunoglobulin (Ig)E, and pepsins, have been reported to be present in middle ear effusion.6-10 However, SOM treatment in clinic remains controversial, and its etiology and pathogenesis remain unresolved.4,5 A middle ear effusion that does not vanish for more than 3 months will progress into chronic otitis media. Chronic SOM affects 5–6% of children in high-income countries in their second year of life, becomes less prevalent in older children, and is rare in adults. 11 For the management of SOM, 3 months of watchful waiting is recommended. When the disorder persists, and the patient presents no improvement after 3 months, surgery (balloon dilation eustachian tuboplasty/tympanic tube insertion) should be considered. Despite such treatment, the recurrence rate remains at approximately 25%. 12 Furthermore, chronic SOM would develop, which can cause continuous chronic inflammation, and even aggravate, ultimately resulting in permanent deafness.4,5
The etiology and pathogenesis of chronic SOM remains incompletely understood. The middle ear is an anatomic extension of the upper airway via the eustachian tube. This has been included in the unified airways concept 13 because it is capable of mounting an inflammatory response similar to those occurring in other areas of the respiratory tract. Chronic SOM has been considered to be caused by inflammation and immune response to rhinopharyngeal infection. Inflammation leads to the production of cytokines and exudates that are rich in proteins and inflammatory mediators. The accompanying dilation of blood vessels leads to increased gas exchange in the middle ear, which reduces endotympanic pressure. 14
SOM is a multifactorial inflammatory disease, in which one risk factor is allergy, which results in edema of the eustachian mucosa, increased pressure, and eustachian tube obstruction. People with allergies are 2.0–4.5 fold more likely to develop SOM, when compared to those without allergies.10,15 Compared to that in non-allergic patients with SOM, the middle ear effusion of allergic patients with this disease contains more IgE, Th2 cytokines, mast cells, and eosinophil-derived immune mediators. 10 In atopic and non-atopic children with SOM, high concentrations of pro-inflammatory cytokines IL-1b, tumor necrosis factor (TNF)-α, IL-6, and IL-8 are locally involved in the zone of inflammation, and IL-1b levels are persistently high in atopic patients. 9
IL-1b, a member of the IL-1 family, has been reported to be upregulated in SOM. 9 IL-33, which is another member of the IL-1 family, is involved in the regulation of chronic inflammatory and immune reactions in the lung, brain and gastrointestinal epithelium.16,17 Thymic stromal lymphopoietin (TSLP) and IL-25 also are involved in chronic inflammatory and immune reactions, such as allergic reactions, asthma, and aseptic necrosis. Tissue damage can increase the quantities of immune cells (including dendritic cells, neutrophils, eosinophils, monocytes, natural killer [NK] cells, and mast cells), and trigger the production of cytokines (e.g., IL-4, IL-5, IL-13, and IL-9) and IgE. 18 The middle ear and lungs are derived from the endoderm. The cell components of the middle ear mucosa are similar to those of the lung mucosa, and are characterized by the high expression of Th1/Th2 cytokines. Thus, it can be hypothesized that Th2 inflammatory factors IL-33, TSLP, and IL-25 may be involved in chronic SOM. In order to verify the presence of these factors in chronic SOM cases, enzyme-linked immunosorbent assay (ELISA) was performed to detect the cytokine expression in the effusion and serum samples (obtained by repeated puncture or via tympanostomy tubes) obtained from patients with a disease course of >3 months.
Patients and Methods
Subjects
The present study is a single-blind non-randomized prospective study. The physician and examiner were not the same person, and the examiner was blinded of the patient’s information. Patients with chronic SOM who attended the outpatient and inpatient departments of Shenzhen People’s Hospital (The Second Clinical Medical College of Jinan University) between July 2019 and January 2020 were enrolled for the present study. Healthy controls were selected with minimized differences in age and gender between groups. Chronic SOM was defined, as follows: (a) disease course >3 months; (b) recurrence after repeated puncture for at least three times, or at least one tympanostomy; (c) stuffy or tinnitus sensation; (d) tympanum integrity, invagination, or effusion; (e) tympanogram type B or C; (f) topical fluticasone propionate nasal spray and mucus stimulating agent eucalyptol limonene pinene enteric soft capsules were continuously administered for 8 weeks, and the symptoms did not obviously improve or aggravated for 3 months; (g) patients with good compliance, and regular return visits. Patients with nasopharyngeal tumors, chronic suppurative otitis media, tympanic membrane perforation and severe systemic diseases, patients who were lost to follow-up, and patients with acute infection, gastroesophageal reflux, and allergic symptoms were excluded from the present study.
The Scientific Research Ethics Committee of Shenzhen People’s Hospital approved the study protocol (Permit No. KY- LL-2020257). A written informed consent was obtained from all subjects or their guardians.
Specimens and Preservation
The middle ear effusion samples were collected by sterile acupuncture using a 1-ml syringe, and stored in sterile Eppendorf (EP) tubes at −80°C. Peripheral venous blood was collected into SST-tubes. Within 2 hours of collection, the blood was centrifuged at 2500 r/min for 5 minutes at room temperature. Then, the supernatant (serum) was collected into sterile EP tubes, and stored at −80°C until analysis.
ELISAs
The IgE, IL-2, IL-4, IL-5, IL-13, IL-17, IL-25, IL-33, IFN-γ, TLSP, and pepsins were detected using ELISA kits (1–10 ng/L sensitivity; Shanghai Langton Biotechnology, Shanghai, China), according to manufacturer’s instructions. The middle ear effusion and serum samples were diluted and evaluated in triplicate. The prepared standards and samples (50 µl) were added to a 96-well Microelisa strip plate. For blank control wells, no solution was added. The plate was incubated for 1 hour at 37°C, and washed for five times with 1× washing solution. Next, biotin-conjugated antibody (10 µl) was added to the wells, and the plate was incubated for 30 min at 37°C, followed by five washes with 1× washing solution.
Afterward, Avidin–horseradish peroxidase (50 µl) was added to the wells, and the plate was incubated for another 30 min at 37°C, followed by five washes with 1× washing solution. Next, Chromogen Solution A (50 µl) was added to the wells, and the plate was incubated for 10 min at 37°C for color development. In order to terminate the color development, 50 µl of Stop Solution was added. A microplate reader was used to measure the optical density of each well at 450 nm wavelength. The concentrations of the cytokines, IgE(IU/ml), and pepsin (ng/ml) were determined using the standard curves generated from the prepared standards.
Statistical Analysis
SPSS 20.0 (IBM Corporation, Armonk, NY, USA) and GraphPad Prism 7 (GraphPad Software, Chicago, IL, USA) were used for the statistical analysis. Normally distributed data were expressed as mean ± standard deviation, while non-normally distributed data were expressed in median (Q25, Q75). The non-parametric stats two independent-samples Mann–Whitney U test was used to analyze the ELISA data obtained from the chronic SOM and control groups, and analyze the ELISA data for the tympanogram types obtained from the chronic SOM. Non-parametric stats two-related samples Wilcoxon test was used to analyze the effusion and serum sample data obtained from the chronic SOM group, and chi-squared test and one-way analysis of variance were used to analyze the general characteristics of the study groups. A p-value of <0.05 was considered statistically significant.
Results
Clinical material
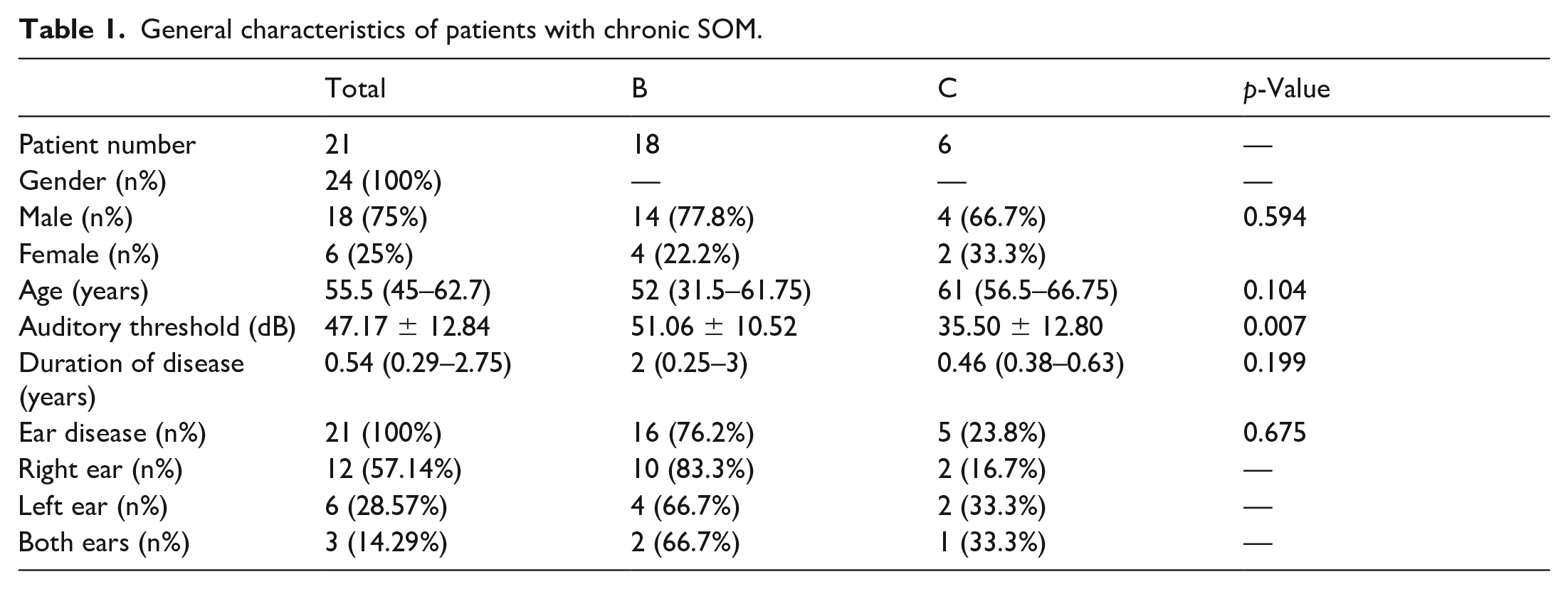
The chronic SOM group comprised of 20 adults and 1 child. The mean duration of disease was 0.54 (range, 0.29–2.75) years. A total of 24 ears were included for the chronic SOM group (the right ears of 12 patients, the left ears of six patients, and both ears of three patients). The auditory threshold was 47.17±12.84 dB. The tympanograms were type B in 18 cases and type C in six cases. Based on the tympanogram types, and in comparing type B with type C, the auditory threshold was statistically significant (Table 1). The number of eosinophils in peripheral blood exhibited no obvious increase (data not shown). The general characteristics of the patients are presented in Table 1.
General characteristics of patients with chronic SOM.
Peripheral blood was collected from 13 patients, and the effusion samples were collected from 14 patients in the chronic SOM group. These patients were obtained from the non-paired samples. Since the middle ear effusion is microscale or thick, this was only collected from some patients. The serum sample was also partially collected, because this was required in the patient’s consent. The blood and effusion paired samples were collected from six patients, which were the same subjects (Table 2).
General characteristics of subjects whose serum was analyzed.

The control group comprised of 10 healthy subjects (one child of 3.25 years old and nine adults [six male and four female]), with a mean age of 40.03 years old (Table 2). Peripheral blood was collected from all healthy subjects.
3.2. Cytokine concentrations in effusion and serum
The levels of IL-4, IL-5, IL-13, IL-17, IL-25, IFN-γ, TLSP, pepsins, IL-2, and IL-33 (all, p<0.001) were higher in the middle ear effusion, when compared to those in serum, in the chronic SOM group (Figure 1). The IgE expression did not significantly differ among these samples. The serum expression of IL-2, IL-4, IL-5, IL-13, IL-17, IL-25, IL-33, IFN-γ, TSLP, and pepsins did not significantly differ between the chronic SOM and control groups. However, the serum expression of IgE significantly differed between the two groups (p=0.03, data not shown). Based on the tympanogram type, the level of cytokines between type B and type C were compared, and the levels of IL-4, IL-5, IL-13, IL-25, IFN-γ, TLSP, pepsins, IL-2, IL-33 and IgE (all, p>0.05) were compared, in terms of effusion level expression or serum levels expression. The results revealed that there was no significant difference between type B and type C patients. Merely the serum levels expression of IL-17 had statistically significant differences (p =0.021), while the effusion level expression for IL-17 had no significant difference (data not shown).

The expression levels of IL-2, IL-4, IL-5, IL-13, IL-17, IL-25, IL-33, IFN-γ, TLSP, pepsins, and IgE in the middle ear effusion in the chronic SOM group (n=14), and serum in the chronic SOM group (non-paired samples) (n=13). ns, not significant. ***p<0.001.
The cytokines and IgE expression profiles were analyzed in the effusion and serum samples collected from six patients (paired samples), and it was revealed that there were significant differences for IL-17, IL-4, IL-33, TLSP (all, p=0.046), IL-13, IFN-γ, IL-2, and pepsins (both, p=0.028). However, no significant difference was observed for IgE, IL-5, and IL-25 (Figure 2).

The expression levels of IL-2, IL-4, IL-13, IL-33, IFN-γ, TLSP, pepsins, IgE, IL-5, IL-17, and IL-25 in the middle ear effusion and serum samples (n=6 each) (paired samples) in the chronic SOM group. ns, not significant. *p<0.05.
Discussion
SOM is common in children. The occurrence of otitis media with effusion in adults can be multifactorial. SOM in adults is a syndrome, rather than a disease, induced by inflammatory reactions and immune responses. The present study revealed the high expression of Th1 cytokines IL-2 and IFN-γ in the middle ear effusion of patients with chronic SOM. The strong Th1 cytokine expression upon infection in patients with SOM suggests that these cytokines are involved in the SOM inflammatory state. 19
In addition, allergy mediators, including Th2 cytokines, play major roles in the pathogenesis of SOM. 20 Th2 cells produce primarily IL-4, IL-5, and IL-13, which play important roles in allergies. 21 Some scholars have reported that high serum IgE expression can upregulate IFN-γ. The IL-13 Arg130Gln genotype affects IFN-γ gene expression via the regulation of the serum IgE level, and plays a role in allergy susceptibility. 22 IFN-γ is secreted mainly by natural dead cells and NK T cells. Upon the establishment of antigen-specific immunity, this is also secreted by CD4 Th1 and CD8 cytotoxic T cells. 23
Th2 cytokines IL-4, Il-5, and IL-13 were detected in the effusion and serum of patients with chronic SOM, with a high expression in the effusion. It was also found that there was greater expression for immune-related protein IgE in serum obtained from the chronic SOM group, when compared to that obtained from the control group. These results indicate that Th2 cytokines IL-4, IL-5, IL-13, and IgE are involved in the expression of chronic otitis media. Kariya et al. 24 reported the detection of IL-2, IL-4, IL-5, IL-10, IL-12, and IFN-γ in 60 (75.0%), 33 (41.3%), 42 (52.5%), 14 (17.5%), 80 (100%), and 66 (82.5%) middle ear effusion samples obtained from patients with SOM. Another study revealed that in a sample of 50 children with otitis media, the levels of IL-2, IL-4, IL-6, and IgE were significantly higher in the middle ear effusion, when compared to those in peripheral blood. 25 Other conclusions revealed that for IL-4, IL-5, IL-13, IFN-γ, and TNF-α, the ELISA detected significantly higher levels of IL-5 in the serum of 35 children with SOM, when compared to those obtained from 28 healthy controls, after adjusting for confounding variables. 26 These findings are consistent with the present detection results. In a prospective study, the serum and effusion IgE levels were positively correlated, while the serum and effusion pepsinogen 1 (PG1) levels were negatively correlated (with an effusion level of 1/10 in serum) in 30 pediatric patients with SOM, suggesting that allergic reaction is a risk factor for SOM development. 27 Similar results were reported by Karyanta et al. 28 They reported that the SOM prevalence ratio was 4.5 times greater for subjects with and without laryngopharyngeal reflux, suggesting that gastroesophageal reflux is a SOM risk factor.
The present study revealed high levels of Th2 cytokines, including IL-33, TLSP, and IL-25, accompanied by IL-17, in middle ear effusion of patients with chronic SOM. The levels of IL-33 and TLSP, but not the levels of IL-25, significantly differed between the effusion and serum samples obtained from these patients, suggesting that IL-33 and TSLP are involved in chronic SOM. IL-33, a member of the IL-1 family, can intracellularly act as a nuclear factor, and extracellularly act as a cytokine. IL-33 and its receptor are expressed in a wide range of immune cells (including mast cells, Th2 cells, activated Th1 cells, regulatory T cells, ILC2 cells, CD8+ T cells, and NK cells). Furthermore, IL-33 plays an important role in innate and adaptive immunity, as well as in anti-inflammatory responses and tissue repair. 29 Moreover, it is involved in the pathogenesis of various inflammatory diseases.16-18 Researchers have identified the crucial role of IL-33 in driving lung remodeling, and verified that anti–IL-33 can restore ciliation on mucus-generating cells, reduce the number of myofibroblasts, and even lead to the improvement of lung function under continuous exposure to allergens. 30 In an in vitro model of asthma exacerbation induced by human airway primary epithelial cell virus culture, the high IL-33 expression persisted in the chronic phase, and aggravated the type 2 inflammatory response. 31 Anti–IL-33 or dexamethasone can inhibit the degree of rhinovirus-induced type 2 inflammation in the acute exacerbation stage, and merely anti–IL-33 can enhance antiviral immunity and reduce virus replication. 31 Anti–IL-33 can also significantly reduce ILC2 cells, CD41 Th2 cells, and mucus hypersecretion. 31 Furthermore, IL-33 induces the production of IL-13 cytokines, and mast cell sensitization enhances the induction of degranulation-specific IgE to release the inflammatory mediators of IL-13, along with a slight increase in expression of IL-33 receptor ST2. This enhances the IgE synthesis from innate immune cells via IL-4, triggering mast cell degranulation in the absence of allergens. 32 The IL-33 receptor ST2 is involved in inflammatory responses after the IgE-mediated response. Mast cells induce IL-33 after the activation of this response. The regional activation of mast cells promotes edema, and subsequently leads to inflammatory cell aggregation. The IL-33/ST2 pathway is critical in IgE-dependent inflammation. 33 IL-33 in activated dendritic cells can induce naive CD4+ T cells to produce large amounts of IL-5 and IL-13, enhancing the atypical Th2 response. 34 In addition, the IL-2 and IL-6 levels in the middle ear effusion are positively correlated with the peripheral blood CD4+/CD8+ ratio in the SOM group. 25
TSLP is highly expressed on epithelial cells, especially keratinocytes, in atopic dermatitis, and is expressed in airway epithelial cells, mast cells, smooth muscle cells, and lung fiber cells.35–37 TSLP can activate the differentiation of dendritic cells ferrite CD4+ T cell into the Th2 cells, which induces the production of many cytokines, such as IL-4, IL-5, IL-13, and TNF-α. It also downregulates IL-10 and IFN-γ, and triggers the production of IgE, leading to eosinophil multiplication and mucus production.35–37 TSLP transgenic mice presented with symptoms of airway inflammation, including major inflammatory cell infiltration, goblet cell proliferation, and airway hyperreactivity, while mice that lack TSLP presented with strong Th1 responses.38,39 These results suggest that TSLP participates in the immune response. Kato et al. 36 reported that glucocorticoids can effectively inhibit IL-4 and IL-13, which mediate inflammatoryresponses and significantly inhibit TSLP.
Taken together, this evidence shows that IL-33 and TLSP mediate Th1/Th2 inflammatory responses, and play key regulatory roles in chronic inflammation. In a mouse model of airway inflammation, Ford et al. 40 detected a mixed Th1 and Th2 immune response associated with IL-13- and IFN-γ-regulated inflammatory disease, in which IFN-γ inhibited the IL-13–induced goblet cell proliferation and airway eosinophil and neutrophil infiltration. D'Andrea et al. 41 reported similar results, showing that the short-term treatment of peripheral blood monocytes with IL-4 or IL-13 can enhance their antigen-presenting function and their support of antigen-dependent IFN-γ production.
The present study revealed the significantly increased expression of IL-2, IL-4, IL-5, IL-13, IL-17, IL-25, IL-33, IFN-γ, TLSP, and pepsins in the effusion and the reduced expression in serum obtained from patients with chronic SOM. The expression of IL-2, IL-4, IL-13, IL-33, INF-γ, TLSP, and pepsins significantly differed between the middle ear effusion and serum samples obtained from the same patient. These respective studies have shown that the imbalance of Th1/Th2 response lead to SOM.24,42,43 Thus, Th1 and Th2 inflammatory factors are highly expressed in patients with chronic SOM, suggesting that this condition is induced by multiple factors, such as inflam-matory and immune response. That is, Th1 and Th2 cells are affected by inflammatory responses. Furthermore, IL-33 and TLSP, which are the key cytokines that regulate inflammation, were highly expressed in the chronic SOM group, suggesting that these serve this function in patients with chronic SOM. Nevertheless, the serum levels of IL-2, IL-4, IL-5, IL-13, IL-17, IL-25, IL-33, IFN-γ, TLSP, and pepsins did not significantly differ between the patient and control groups. Thus, it was hypothesize that chronic SOM is dominated by local inflammation. However, the expression levels of IgE significantly increased, suggesting that allergy is one of the influencing factors of chronic SOM.
In summary, damage-associated molecular patterns (DAMPs) are endogenous molecules that can be released from within the cell into the extracellular space when cells are stressed or damaged. IL-33 is a common DAMP, which binds to the ST2(IL-1RL1) receptor and produces inflammatory cytokines, such as IL-1B, IL-6, TNF-α, IL-10, IL-4, and TGF-β through the NF-kB inflammatory pathway in the nucleus, and inflammatory cytokines are released into the cytoplasm, causing inflammatory responses. 44 Another study revealed that IL-33 binds to Th17 and ILC2 via the ST2 receptor to produce IL-17A, and IL-4, IL-5, IL-9, and IL-13 inflammatory factors, in order to trigger an inflammatory response. 45 The dual function of IL-33 is mainly due to the different types of immune responses on different cells. IL-33 induces Th2 cells, 46 mast cells, 47 and basophils to secrete large amounts of IL-4, IL-5, IL-13, IgE, and IgA, 47 which in turn, induces pathological changes related to Th2 immune response. Toki S et al. reported that the combined stimulation of TSLP and IL-33 elicits an approximately 10-fold increase in cytokine production through ILC2, when compared to the stimulation of IL-33 alone. 48 At low concentrations, DAMPs regulate homeostasis and correct altered physiological states, while at high concentrations, these enhance and spread inflammatory responses. Due to trauma or inflammatory processes, the excessive release of DAMPs can lead to the immune activation of surrounding cells, and the recruitment of distant cells. This further leads tissue damage, which sets up a vicious cycle of damage that can lead to chronic inflammation. 49 IL-33 and TLSP were detected in the effusion of patients with chronic SOM, suggesting the potential roles in the regulation of chronic SOM Th1/Th2 response.
In clinical practice, for adults suffering from chronic SOM, a duration of more than 3 months is rare. Due to the difficulty of obtaining such effusion samples, the sample obtained for the present study was relatively small. Hence, there is a need to continue to expand the sample size, in order to further confirm these findings. In addition, further basic research and studies on chronic SOM at the molecular (cytokine) and cellular (inflammatory cell) level are needed. Further research on cell culture and animal models of chronic SOM should also be performed to reveal the mechanisms of IL-33/TSLP–mediated Th1/Th2 responses in chronic SOM.
Conclusions
IL-33 produces inflammatory cytokines, such as IL-1b, IL-6, TNF-α, IL-10, IL-4, and TGF-β, which through nucleus into cytoplasm causing inflammatory responses. The present study revealed that IL-33 also produce IL-17, IL-4, IL-5, and IL-13 inflammatory factors, triggering an inflammatory response. Study reported that the combined stimulation of TSLP and IL-33 elicits an approximately 10-fold increase in cytokine production, when compared to the stimulation of IL-33 alone. This suggests that IL-33 and TLSP may be the primary cytokines involved in Th1/Th2 inflammatory responses in chronic SOM.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by grants from the Cultivating Fund Project of Shenzhen People’s Hospital (No. SYKYPY201933).
