Abstract
In order to investigate whether polymorphisms of genes encoding some factors of innate and adaptive immunity play a role in the development of, or protection against atopic dermatitis (AD) and condition its severity, we genotyped 33 candidate genes and 47 single nucleotide polymorphisms (SNPs) using Custom TaqMan Array Microfluidic Cards and an ABI 7900HT analyser (Applied Biosystems, Foster City, CA, USA). The study involved 104 children with AD (29 with mild-to-moderate and 75 with severe disease; 42 girls; mean age ± SD, 5.8 ± 3.3 years) and 119 healthy controls (49 girls; mean age, 4.8 ± 3.0 years). IL10-rs1800872T, TG and MBL2-rs500737AG were all significantly more frequent among the children with AD (P = 0.015, P = 0.004 and P = 0.030), whereas IL10-rs1800896C and TC were more frequent in those without AD (P = 0.028 and P = 0.032). The VEGFA-rs2146326A and CTLA4-rs3087243AG SNPs were significantly more frequent in the children with mild/moderate AD than in those with severe AD (P = 0.048 andP = 0.036). IL10-rs1800872T and TG were significantly more frequent in the children with AD and other allergic diseases than in the controls (P = 0.014 and P = 0.007), whereas IL10-rs1800896TC and C were more frequent in the controls than in the children with AD and other allergic diseases (P = 0.0055 and P = 0.0034). These findings show that some of the polymorphisms involved in the immune response are also involved in some aspects of the development and course of AD and, although not conclusive, support the immunological hypothesis of the origin of the inflammatory lesions.
Introduction
Atopic dermatitis (AD), the most common chronic inflammatory skin disease, is clinically characterised by acute flares of eczematous, pruritic lesions on dry skin that typically affect the flexural folds. 1 Although the skin lesions are quite homogenous, various clinical phenotypes of AD have been described on the basis of age at onset, severity, racial modifiers, reactions to triggers such as infections, allergens, stress and irritant thresholds, and therapeutic responses. Genetic variations are the main reasons for the skin lesions, for which main hypotheses have been proposed. The first is a defective skin barrier: gene dysfunction and loss-of-function mutations in the genes encoding filaggrin or other structural skin proteins have been found in more than 50% of AD patients. 2 The second is an imbalance in the adaptive immune system: variants of genes controlling innate and adaptive immune responses, high levels of activation of the Th2 cytokine pathway and significant inflammatory responses have been reported in some patients with AD,3,4 as it has been found in patients with allergic rhinitis and asthma. 5
The different AD phenotypes may be explained by the fact that all of these variations may complement each other. Subjects with homozygous filaggrin-null mutations or compound heterozygotes experience an early onset of skin disease and more persistently severe eczema, 6 but their disease may be even more clinically relevant if they also have systemic immune activation. 6 Immune studies suggest that the different AD phenotypes are associated with distinct patterns of activation or suppression of the polar immune axes and the corresponding tissue responses. 6 This highlights the importance of precisely defining the relationships between genetic defects, the development of AD and the different phenotypes not only in order to improve our understanding of disease pathophysiology, but also to assist in the development of a more rational and effective individual approach to its treatment.
The aim of this study was to evaluate whether polymorphisms of the genes encoding some factors of innate and adaptive immunity play a role in the development of, or protection against AD, and condition its severity.
Materials and methods
Study population and recruitment
The study was carried out between 1 December 2014 and 31 January 2015, and involved Caucasian children with AD who are regularly followed up by the Unit of Allergy and Immunology of the Pediatric Highly Intensity Care Pediatric Unit, Department of Pathophysiology and Transplantation, University of Milan, Italy. AD was diagnosed on the basis of the diagnostic criteria defined by the American Academy of Dermatology 7 and its severity at the time of enrolment was graded using the SCORAD index. 8 Upon enrolment, the demographic characteristics and medical history of the children were systematically recorded using standardised written questionnaires, and a 3 mL sample of whole blood was obtained for genetic studies.
Blood samples were taken from a control group of otherwise healthy children of similar age and gender with a negative personal and family history of AD or allergic diseases, a negative family history of asthma and no previous episodes of wheezing.
The study protocol was approved by the Ethics Committee of the Fondazione IRCCS Ca’ Granda, Ospedale Maggiore Policlinico, Milan, Italy, and written informed consent was obtained before enrolment from the parents or legal guardian of each child, and the children aged ⩾7 years.
Genetic studies
Table 1 shows the 33 candidate genes and 47 single nucleotide polymorphisms (SNPs) selected for analysis, which included genes involved in immune regulation, and the pathogenesis of inflammation and allergic diseases. The selected genes encode transcription (CHUK, IKIKB, JUN, NFKB1A, NFKB1B) and transduction factors (MAP3K1), the regulator of T cell activity (CTLA4), pattern recognition receptors (IL1RL1,TLR1, TLR3, TLR4, TLR5, TNFRSF1B), pro-inflammatory cytokines and related receptors (IL13, IL18, IL18RAP), anti-inflammatory cytokines (IL10, IL19, IL20), the cytokine regulating lymphoid cell survival (IL7), chemokines (CCL5, IL8, CXCL9), the regulator of IgE production (IL4R), collectins (MBL2), the protein responsible for the acute response to bacterial lipopolysaccharides (LBP), growth factors (NGF, VEGFA), the protein involved in the structural maintenance of chromosomes (RAD50), the spermatogenesis-associated serine-rich 2-like SPATS2L, the thromboxane A2 receptor (TBXA2R), and the adrenergic beta 2 surface receptor (ADRB2), and are all located on autosomes. Most of the SNPs were functional variants or tagging SNPs characterised by the International HapMap Project that are involved in causing or determining the severity or outcome of wheezing in experimental animals or humans, or have previously been found to be associated with an increased risk of developing asthma or an abnormal immune response.9–39
Gene and single nucleotide polymorphisms (SNPs).
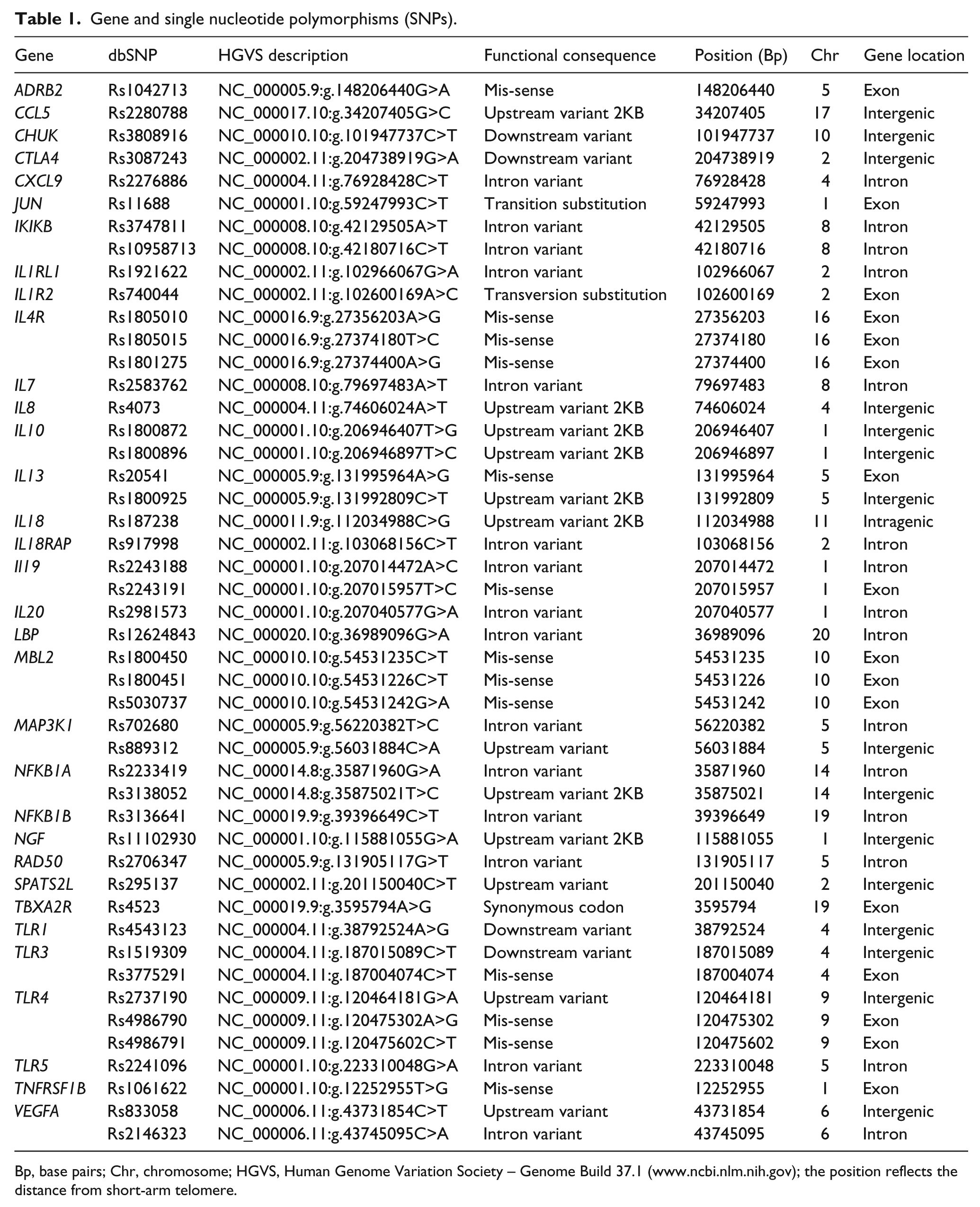
Bp, base pairs; Chr, chromosome; HGVS, Human Genome Variation Society – Genome Build 37.1 (www.ncbi.nlm.nih.gov); the position reflects the distance from short-arm telomere.
DNA was extracted and quantified as previously described. 40 Following nucleic acid purification, the samples were stored at −20°C until use. The 47 SNPs in the 33 genes were genotyped using the Custom TaqMan Array Microfluidic Cards genotyping system on an ABI 7900HT analyser (Applied Biosystems, Foster City, CA, USA). After PCR amplification, the alleles were detected by means of endpoint analysis using SDS and TaqMan Genotyper software (Applied Biosystems, Foster City, CA, USA). The data were entered into a Progeny database (Progeny Software, LLC, South Bend, IN, USA) in order to generate datasets for analysis.
Statistical analysis
It was calculated that a sample size of 104 subjects with AD and 119 in the control group would have 80% power (with alpha = 0.05) to detect an unadjusted odds ratio (OR) of 2.2, assuming that the proportion of children in the control group with a given polymorphic allele for a given gene was 0.30. The test statistic used was the two-sided Mantel-Haenszel test.
The categorical data in the two groups were compared using contingency table analysis and the χ2 or Fisher’s exact test, as appropriate, and the continuous data were analysed using a two-sided Student’s t test (if they were normally distributed on the basis of the Shapiro-Wilk statistic) or a two-sided Wilcoxon rank-sum test. Genotype frequencies were determined by means of direct counting. In order to investigate their Hardy-Weinberg equilibrium (HWE), the expected number of each genotype was compared with the observed number, and potential deviations were assessed using the χ2 or likelihood ratio test, as appropriate. Age-adjusted ORs and their 95% confidence intervals (CIs) were calculated in order to measure the associations between the selected SNPs and: (1) susceptibility to AD by comparing all of the children with AD with the controls; (2) susceptibility to severe AD by comparing the children with mild-to-moderate (SCORAD ⩽50) with those with severe AD (SCORAD >50). The data were controlled for multiple testing using the false discovery rate method and the Benjamini-Hochberg procedure.
All of the statistical analyses were made using SAS software, version 9.2 (Cary, NC, USA)
Results
The study involved 104 children with AD (29 with mild-to-moderate and 75 with severe disease; 42 girls; mean age ± SD, 5.8 ± 4.4 years) and 119 healthy controls (49 girls; mean age, 4.8 ± 3.0 years). Table 2 shows the demographic, clinical and familial characteristics of the two groups of AD patients, which were similar in terms of all of the studied variables except for the type of topical treatment used and the presence of skin symptoms in the previous year, which were significantly more frequent in the children with severe AD than in those with mild-to-moderate AD. These significant differences were expected because of the clinical characteristics of the two groups.
Demographic, clinical and familial characteristics of subjects with atopic dermatitis, by disease severity. a
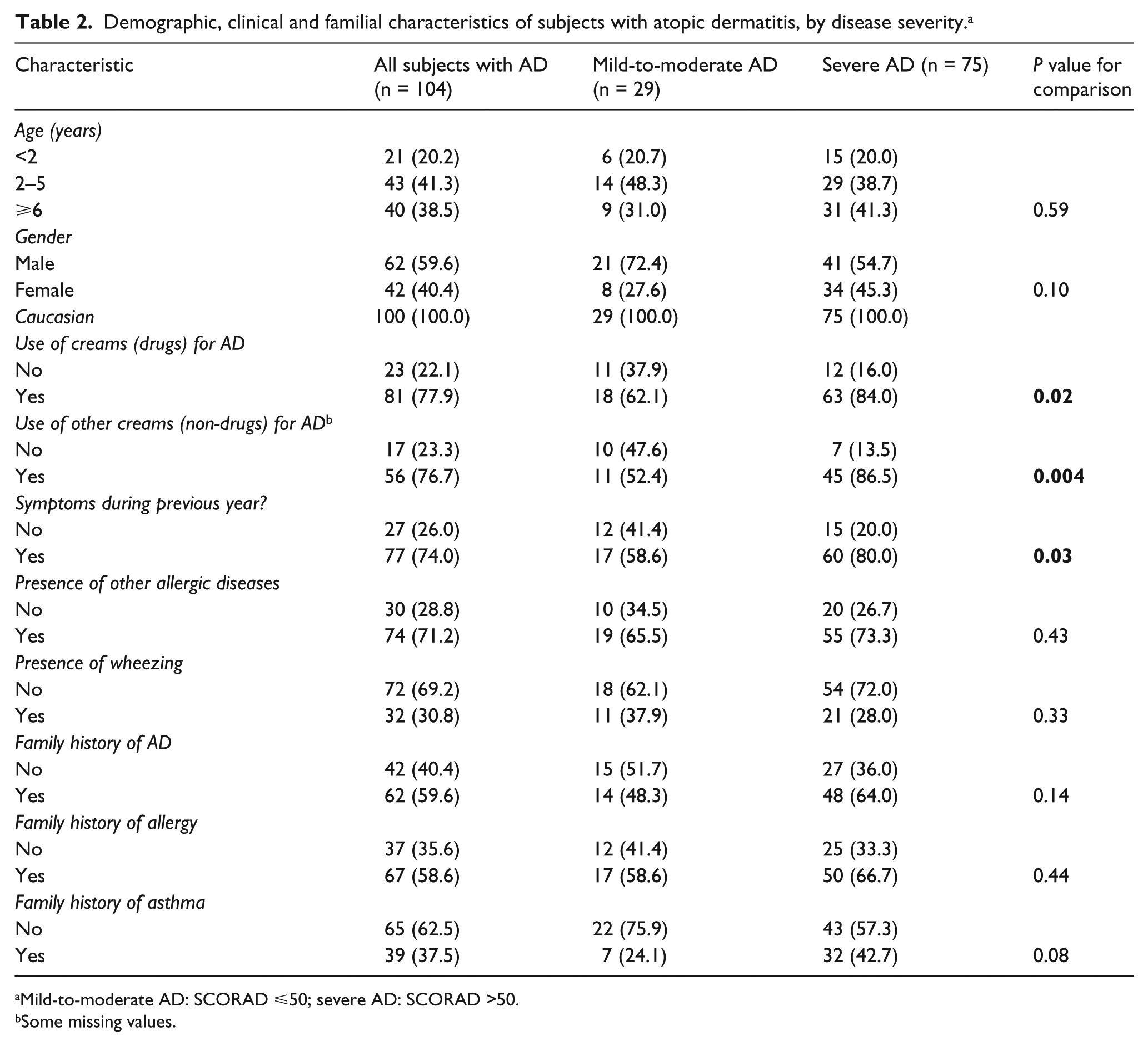
Mild-to-moderate AD: SCORAD ⩽50; severe AD: SCORAD >50.
Some missing values.
Tables 3 and 4, respectively, show the significantly different SNP genotype frequencies, between the children with AD and controls, and between the children with mild-to-moderate or severe AD. IL10-rs1800872T (P = 0.015), TG (P = 0.004) and MBL2-rs500737AG (P = 0.030) were all significantly more frequent in the children with AD than in the controls, whereas IL10-rs1800896C (P = 0.028) and TC (P = 0.032) were significantly more frequent in the controls. These SNPs seemed to favour or reduce the risk of developing AD without conditioning diseases severity because there was no statistical difference in their frequency between the children with mild-to-moderate or severe AD. On the contrary, VEGFA-rs2146326A and CTLA4-rs3087243AG were similarly frequent in the children with and without AD, but their frequency was significantly higher in the children with mild-to-moderate AD than in those with severe AD (P = 0.048 and P = 0.036). There were no other significant differences in genotype frequency between the groups or sub-groups.
Genotype frequencies of selected SNPs in children with atopic dermatitis and controls, and odds ratios (ORs) with 95% confidence intervals (CIs). a

Some missing values.
P values from logistic regression analyses, adjusted for age.
HWE, Hardy-Weinberg equilibrium.
Genotype frequencies of selected SNPs in children with mild-to-moderate and severe atopic dermatitis, and odds ratios (ORs) with 95% confidence intervals (CIs). a
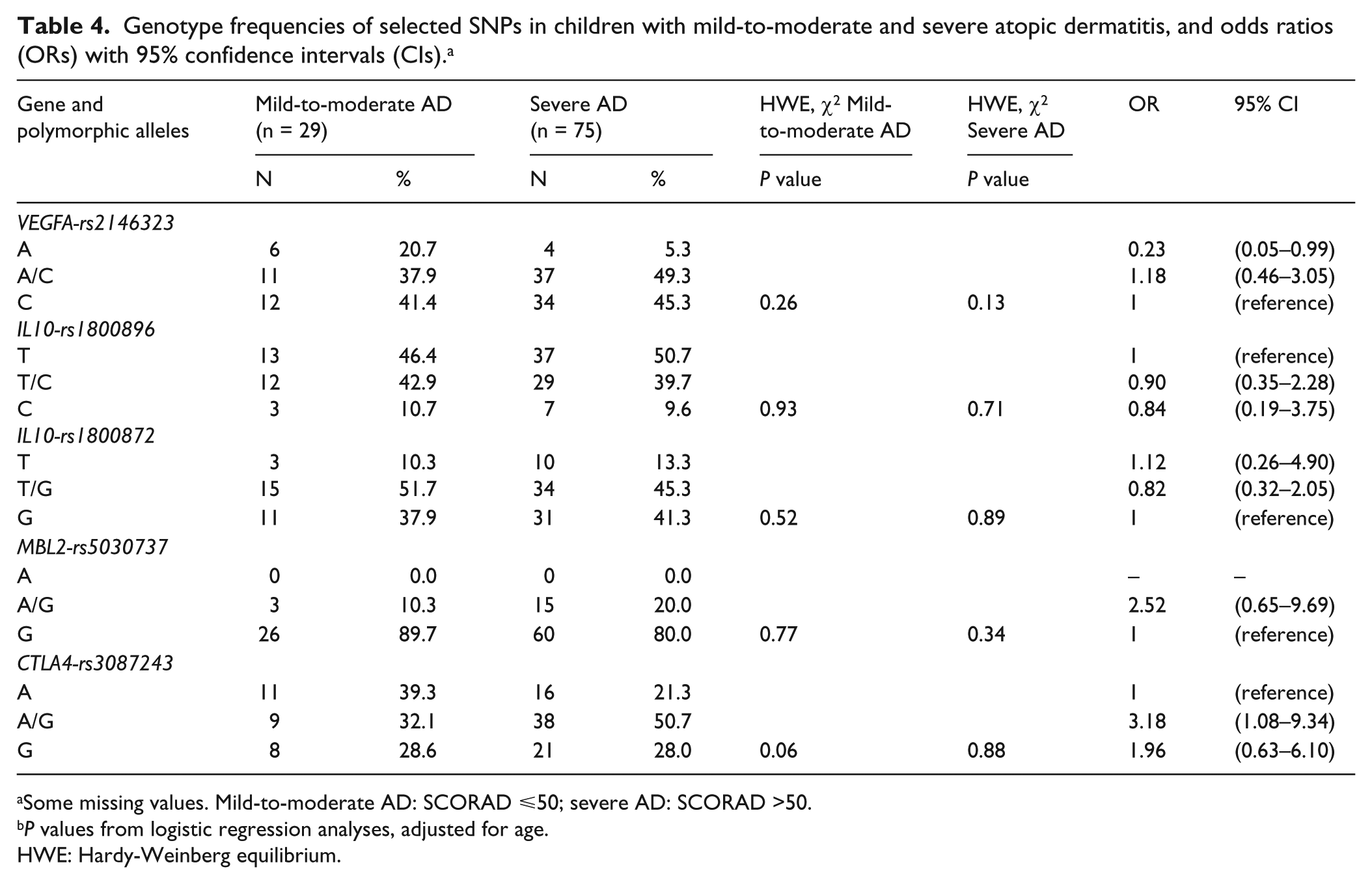
Some missing values. Mild-to-moderate AD: SCORAD ⩽50; severe AD: SCORAD >50.
P values from logistic regression analyses, adjusted for age.
HWE: Hardy-Weinberg equilibrium.
Table 5 shows the genotype frequency of the selected SNPs in the children with AD suffering from wheezing and other allergic diseases and controls. IL10-rs1800872T (P = 0.014) and TG (P = 0.007) were significantly more frequent in the former, whereas IL10-rs1800896TC (P = 0.0055) and C (P = 0.0034) were more frequent in the controls.
Genotype frequencies of selected SNPs in children with atopic dermatitis and other allergic diseases and controls, and odds ratios (OR) with 95% confidence intervals (CI). a

Some missing values.
P values from logistic regression analyses, adjusted for age.
HWE, Hardy-Weinberg equilibrium.
None of the P values was significant after correcting for multiple testing using the false discovery rate method and Benjamini-Hochberg procedure.
Discussion
The results of this study seem to confirm that some genetic polymorphisms may condition the development of AD and its severity. IL10 and MBL2 SNPs seem to play a role in regulating the development of AD, the severity of which seems to depend mainly on VEGFA and CTLA4 SNPs.
The association with IL10 SNPs is not surprising because the protein has important immune regulatory functions and conditions the susceptibility and course of a large number of immune-mediated diseases. Its primary function is to limit inflammatory responses by means of mechanisms that include inhibiting the production of pro-inflammatory cytokines and chemokines, downregulating the expression of major histocompatibility (MHC) class II and co-stimulatory molecules, regulating immunoglobulin class switches in B cells, and attenuating CD4+ T cell responses. 41 However, under particular conditions, IL-10 can have the opposite effect of stimulating immune responses by promoting the proliferation and cytotoxic activity of CD8+ T and natural killer cells, and the survival and antibody secretion of B cells. 42 It has been shown that SNPs in the IL10 gene are associated with allergy and asthma,43 –45 systemic lupus erythematosus, 46 rheumatoid arthritis, 47 psoriasis 48 and infectious diseases. 49 A direct relationship between IL10 genetic polymorphisms and AD alone or associated with other allergic diseases is also indicated by our finding that IL10-rs1800872T and TG were significantly more frequent among children with AD and other allergic diseases than in control subjects.
However, this does not answer the question insofar as the two studied SNPs of IL10 (IL10-rs 1800896 and IL10-rs 1800872) were associated with opposite conditions: the first with a reduced risk of AD and the second with more frequent disease.
There are many conflicting reports concerning the role of IL10 in AD. Chen and Zhao carried out a meta-analysis of seven studies of the SNP that we found seemed to protect children against AD that considered a total of more than 800 AD subjects and about 1200 controls, 50 and found no direct association between IL10-rs 1800896 and susceptibility to, or the severity of AD, whereas Sohn et al. 51 and Lesiak et al. 52 have found close relationships between some IL10 promoter haplotypes and susceptibility to AD, and Lesiak et al. have also found increased serum concentrations of IL10 in AD subjects with this SNP. 53 Moreover, Raedler et al. found that carriers of three IL10 SNP blocks and distal promoter SNPs (including IL10-rs1800872) showed reduced levels of the expression of T reg cell markers, reduced IL-5, TNF-α and GM-CSF levels, and partially increased IFN-γ levels, and that the same SNPs were associated with the development of AD in 3-year-old children, 54 whereas Kijohara et al. did not find any relationship between IL10 genetic variations and AD. 55 These conflicting findings may have been due to various factors, including differences in the ethnicity and general characteristics of the study populations, differences in the time of analysis and/or the assessment of clinical phenotypes, a failure to model gene/environment interactions adequately, 56 or the dual functional nature of IL10 itself.
We found that the severity of AD was associated with VEGFA-rs2146323A and CTLA4-rs3087243. VEGFA is a major regulator of angiogenesis and enhancer of vascular permeability, 57 and seems to induce airway inflammation and airway remodelling in patients with asthma. 17 It has been suggested that there is an association between VEGFA SNPs and AD on the grounds that lesioned epidermis produces approximately 25 times more VEGF than normal stratum corneum, 56 and Zablotna et al. have found a statistical association between the presence of SNPs in a promoter region of the VEGF gene and a diagnosis of AD. 59 Our own findings confirm an association between variations in this gene and the severity of AD, but not the risk of its development.
CTLA4 plays an essential role in the function of the regulatory T cells that control Th1 and Th2 immune responses. 25 As particularly acute-phase AD is associated with a predominance of Th2 lymphocytes, 1 it has been suggested by Jones et al. 60 that a genetic modification in the characteristics of CTLA4 may lead to the development of the disease or increase its severity, a hypothesis that is partly confirmed by our findings. Polymorphisms of CTLA4 have also been associated with severe asthma 61 and it is well known that the most severe cases of AD are associated with bronchial obstruction, and so it is likely that CTLA4 genetic variations may be markers of severe AD.
In conclusion, our findings show that polymorphisms involved in the immune response are related to some aspects of the development and course of AD and, although not conclusive, support the immunological hypothesis of the origin of AD inflammatory lesions. However, given the conflicting nature of the published results, further larger studies are needed in order to establish the real associations between genetic variations and AD, and identify candidate genes that may allow a more precise definition of all aspects of the disease and the early identification of cases with a more negative prognosis.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by a grant from the Italian Ministry of Health (Bando Giovani Ricercatori 2009).
