Abstract
Objective
To investigate whether dexmedetomidine (Dex) can reduce the severity of tourniquet-induced lung injury.
Methods
36 patients undergoing total knee arthroplasty with a tourniquet were randomly assigned to the control (ischemia/reperfusion [I/R]) group and Dex group. Patients in the Dex group received a loading dose of Dex (0.8 μg/kg over 10 min intravenously) followed by continuous infusion of Dex (0.5 μg/kg/h intravenously) until the end of the surgery. The I/R group received an equal amount of 0.9% saline instead of Dex. The serum concentrations of tumor necrosis factor-α (TNF-α), Clara cell protein (CC-16), soluble receptor for advanced glycation end products (sRAGE), and brain-derived neurotrophic factor (BDNF) were measured and arterial blood gas analysis was performed before anesthesia and 30 min, 6 h, and 24 h after tourniquet release.
Results
In the I/R group, compared with baseline, the TNF-α, CC-16, and sRAGE concentrations were higher (p < 0.05) and the BDNF concentration was lower (p < 0.05) at most time points. In the Dex group, the TNF-α, CC-16, and sRAGE concentrations were lower than those in the I/R group (p < 0.05), whereas the concentration of BDNF was higher (p < 0.05). In the arterial blood gas analysis, the Dex group showed a significantly higher partial pressure of oxygen and arterial/alveolar oxygen tension ratio (p < 0.05) and a significantly lower alveolar/arterial oxygen tension difference than the I/R group (p < 0.05).
Conclusion
Dex administration partly inhibits the release of proinflammatory cytokines, affording protection against tourniquet-induced lung injury.
Introduction
Tourniquets are widely used in vascular or orthopedic surgery, and their main purpose is to create a bloodless environment for smooth performance of the operation. 1 However, tourniquets can also cause serious acute limb ischemia/reperfusion (I/R) injury, such as tissue edema, muscle necrosis, and other symptoms.2,3 In some cases, distal organs may also be damaged, and such complications significantly restrict the promotion and application of tourniquets. 4 A means to attenuate multiple-organ injury in surgeries requiring tourniquet application would enhance patients’ recovery.
Dexmedetomidine (Dex) is a new type of highly selective α-2 adrenergic receptor agonist that has been widely used in surgery. As an adjuvant anesthetic, it can provide sedative and analgesic effects. Numerous studies have demonstrated that Dex can play valuable antioxidation and anti-inflammatory roles, and it has a protective effect against I/R injury in multiple organs.5,6 However, no study to date has evaluated the effects of Dex on the lung injury and inflammation responses that develop after tourniquet-induced I/R injury.
Therefore, in this clinical trial, we analyzed the intervention effect and mechanism of Dex on tourniquet-induced lung injury and found that Dex played a protective role mainly by inhibiting the release of inflammatory cytokines.
Methods
Study design and patient groups
The experimental scheme was approved by the ethics committee of the Tianjin Hospital, and the trial was registered at chictr.org.cn (No. ChiCTR2100045801). This trial was conducted in the hospital from June 2021 to January 2022.
Based on a previous study (expected difference in means, 13; expected standard deviation, 9.5), 7 the sample size of the present study was estimated with an α of 0.05 and power set at 80%. The calculation showed that 18 patients in each group would ensure significant differences between the groups. However, because loss to follow-up was inevitable, a 6% loss rate was set. Thus, 40 patients were screened.
Using a random data table, an anesthesiologist who was involved only in preparing the medication in a 100-mL syringe randomly allocated the 40 patients to the control group (I/R group) and the Dex group. An independent researcher who was not involved in the anesthetic procedures or patient enrollment conducted the outcome assessment. All patients provided written informed consent.
The inclusion criteria of the present trial were adult patients (60–75 years of age) with an American Society of Anesthesiologists physical status of I or II who were scheduled for total knee arthroplasty (TKA) and required 60–90 min of tourniquet application during the surgery. The exclusion criteria were patients with basic metabolic diseases, arrhythmia, heart failure, renal dysfunction, hepatic or cardiopulmonary dysfunction, carotid or brain vascular disease, frequent use of antidepressants, unsuitability for or refusal to undergo spinal and epidural anesthesia, and poor compliance. Four patients were excluded according to the above criteria (Figure 1). Flowchart of this study.
The patients underwent basic preoperative preparation, mainly fasting for 8 h and setting up venous access. During the operation, we applied standard monitoring of the patients’ electrocardiogram, heart rate, noninvasive blood pressure, and peripheral capillary oxygen saturation. A total of 2.5 mL of 0.5% ropivacaine was administered at the L3/4 spinal level to anesthetize the patients, and the level of intraoperative control block was T8–10. Ringer’s lactate solution and hydroxyethyl starch injection were infused to maintain hemodynamic stability.
Before inflation of the tourniquet, patients in the Dex group received a loading dose of Dex (0.8 μg/kg over 10 min intravenously) followed by continuous infusion of Dex (0.5 μg/kg/h intravenously) until the end of surgery, while the patients in the control group received injection of 0.9% normal saline at the same dose. Blood samples were obtained from a peripheral vein in the upper extremity, and radial arterial blood gas analysis was performed at the following time points: before anesthesia (T0) and at 30 min (T1), 6 h (T2), and 24 h (T3) after tourniquet release. Venous blood samples were separated by centrifugation at 3000 r/min for 5 min, and the plasma was stored at −80°C.
Enzyme-linked immunosorbent assay was used to detect the serum concentrations of human tumor necrosis factor-α (TNF-α) (Proteintech, Rosemont, IL, USA), Clara cell protein (CC16) (Elabscience, Houston, TX, USA), brain-derived neurotrophic factor (BDNF) (Proteintech), and soluble receptor for advanced glycation end products (sRAGE) (Yifeng Ltd., China). Radial arterial blood gas analysis included measurement of the arterial partial pressure of oxygen (PaO2), arterial partial pressure of carbon dioxide (PaCO2), arterial/alveolar oxygen tension ratio (a/A ratio), alveolar/arterial oxygen tension difference (A-aO2), and hemoglobin.
Statistical analysis
Statistical analysis was processed by SPSS 17.0 software. Data were expressed as mean ± standard deviation (SD). Discrete variables were analyzed with the chi-square test. Continuous variables were compared using t test. For multi-group comparisons, one-way analysis of variance with Bonferroni’s post hoc test was used to determine statistical significance. p < 0.05 was considered as statistically significant.
Results
Patients’ clinical characteristics
Clinical characteristics of the patients.

I/R: ischemia-reperfusion; Dex: dexmedetomidine; BMI: body mass index. Data are expressed as mean ± SD.
Concentrations of TNF-α, CC16, sRAGE, and BDNF
Plasma TNF-α, CC-16, sRAGE and BDNF levels.
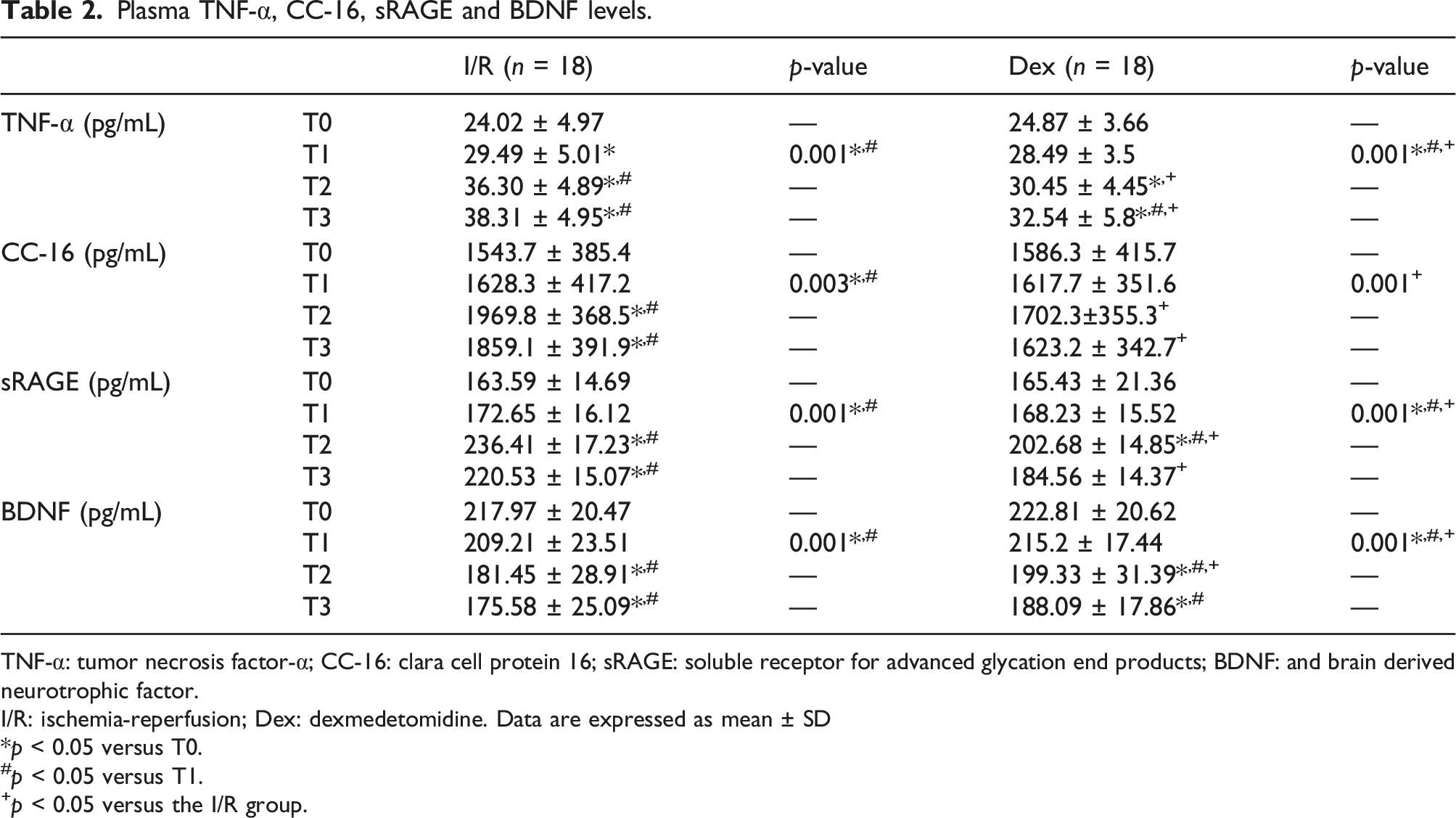
TNF-α: tumor necrosis factor-α; CC-16: clara cell protein 16; sRAGE: soluble receptor for advanced glycation end products; BDNF: and brain derived neurotrophic factor.
I/R: ischemia-reperfusion; Dex: dexmedetomidine. Data are expressed as mean ± SD
p < 0.05 versus T0.
#p < 0.05 versus T1.
+p < 0.05 versus the I/R group.
Blood gas analysis
Blood gas analysis.

PaCO2: arterial partial pressure of carbon dioxide; PaO2: arterial partial pressure of oxygen; a/A ratio: arterial-alveolar oxygen tension ratio; A-aO2: arterial-alveolar oxygen tension difference; Hb: hemoglobin.
I/R: ischemia-reperfusion; Dex: dexmedetomidine. Data are expressed as mean ± SD
p < 0.05 versus T0.
#p < 0.05 versus T1.
+p < 0.05 versus the I/R group.
Discussion
Prolonged use of a tourniquet can result in inflammation in the local ischemic skeletal muscle, and various inflammatory factors can also produce a cascade reaction that damages associated secondary remote organs. 8 Accumulating evidence in many acute inflammatory animal models demonstrates that Dex can significantly inhibit I/R-related organ injury, and the underlying mechanism may be that Dex has certain anti-inflammatory effects. 9 Given that the lung is the organ most vulnerable to I/R-related injury, 10 we hypothesized that Dex would mitigate lung injury through an anti-inflammatory mechanism.
TNF-α plays a key role in the innate immune system, so it is closely related to inflammatory reactions. 11 In this study, we found that during surgery, tourniquet-induced acute limb I/R significantly increased the TNF-α concentration, triggering a systemic inflammatory response.
A previous study indicated that the serum CC16 concentration can increase in several situations associated with a disruption of the air–blood barrier, suggesting that this low-molecular-weight protein may serve as a potential predictor of lung injury.12,13 sRAGE is mainly expressed on alveolar type I cells, and some scholars have found that an elevated sRAGE level is associated with lung injury; thus, sRAGE might serve as an acute bronchiolitis biomarker.14,15 In the present clinical trial, the CC16 and sRAGE concentrations were significantly elevated at T2 and T3 in both groups, but the increment in the Dex group was relatively low. Although not statistically significant, the CC16 and sRAGE concentrations were slightly higher in the Dex group at T1.
The PaO2, a/A ratio, and A-aO2 can be used to evaluate the pulmonary gas exchange function. The results of this study showed that the PaO2 and a/A ratio significantly decreased while A-aO2 significantly increased after tourniquet release. This result suggests that limb I/R will lead to a decline of the lung gas exchange function. Importantly, we found that Dex can play a certain role in restoring this function. Overall, this study suggests that Dex intervention is beneficial to alleviate the lung injury after tourniquet use in patients undergoing TKA.
Related studies have shown that postoperative systemic inflammation can affect cognitive function by acting on the central system and induce related cognitive syndrome. 16 According to the viewpoint of some scholars, neuritis also plays an important role in the pathological changes of cognitive dysfunction and is the main influencing factor. 17 BDNF is a typical neurotrophic factor and has a certain inhibitory effect on neuroinflammation. 18 In this study, the plasma BDNF concentration remained minimally changed in both groups at T1, but it was significantly higher at T2 in the Dex group than in the I/R group. Our study indicates that Dex intervention is beneficial to reduce the risk of cognitive impairment, which is consistent with the research results of Su et al. 19
This study has several limitations. First, the mechanism by which Dex inhibits tourniquet-related injury was not explored in depth. Second, based on our experience and previous research results, 7 we choose 0.8 μg/kg Dex followed by continuous infusion of Dex (0.5 μg/kg/h) throughout surgery; however, different doses of Dex were not tested. Finally, the long-term effects of Dex were not studied. Further investigation of the long-term effects of this treatment is warranted.
Conclusions
The results of this study suggest that patients undergoing TKA requiring tourniquet application can develop tourniquet-induced lung injury, and Dex administration partly inhibits the release of proinflammatory cytokines, affording protection against this effect.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability
The data used and analyzed during this study are available upon request.
