Abstract
The purpose of this study was to investigate the expression of pyroptosis-related factors (NLRP3, IL-18, NF-κB, HMGB-1, and GSDMD) in patients who died of COVID-19. The expression levels of NLRP3, IL-18, NF-κB, HMGB-1, and GSDMD in lung and spleen tissues of the COVID-19 group and the control group were detected by tissue immunofluorescence. The control group includes lung tissues and spleen tissues of two patients who died unexpectedly without SARS-CoV-2 infection, and the COVID-19 group includes the lung and spleen tissues of three patients who died of SARS-CoV-2 virus infection. The positive rates of NF-κB, NLRP3, IL-18, and GSDMD in the lung tissues from the control group and COVID-19 group were 9.8% vs 73.4% (p = 0.000), 5.5% vs 63.6% (p = 0.000), 24.4% vs 76.2% (p = 0.000), and 17.5% and 46.8% (p = 0.000) respectively. The positive rates of NF-κB, NLRP3, IL-18, HMGB-1, and GSDMD in the spleen tissues from the control group and COVID-19 group were 20.6% vs 71.2% (p = 0.000), 18.9% vs 72.0% (p = 0.000), 15.2% vs 64.8% (p = 0.000), 27.6% vs 69.2% (p = 0.000), and 23% and 48.8% (p = 0.000), respectively. The positive rates of SARS-CoV-2 spike protein in the CD68 positive cells of the lung and spleen in the control group and COVID-19 group were 2.5% vs 56.8% (p = 0.000); 3.0% vs 64.9% (p = 0.000) respectively. The rates of NF-κB positive nuclei in the control group and COVID-19 group were 13.4% vs 51.4% (p = 0.000) in the lung and 38.2% vs 59.3% (p = 0.000) in the spleen. The rates of HMGB-1 positive cytoplasm in the control and the COVID-19 group were 19.7% vs 50.3% (p = 0.000) in the lung and 12.3% vs 45.2% (p = 0.000) in the spleen. The targets of SARS-CoV-2 are the lung and spleen, where increased macrophages could be involved in the up-regulation of pyroptosis-related inflammatory factors such as NF-κB, HMGB-1, NLRP3, IL-18, and GSDMD.
Introduction
The novel coronavirus pneumonia (Corona Virus Disease 2019, COVID-19) has been in a global pandemic since its outbreak in December 2019. To date, the death toll has reached 5,068,233(https://coronavirus.jhu.edu/map.html), with a tremendous impact on human life, health, and the global economy. The pathogen of COVID19 was recognized as the SARS-CoV-2 virus, a β-coronavirus. This virus has about 50% genome sequence homology with Mers-CoV and about 80% genome sequence homology with SARS-CoV-1.1,2 Studies have demonstrated that the excessive inflammatory response mechanism caused by Mers-CoV and SARS-CoV-1 is associated with pyroptosis.3,4 The levels of GSDMD in the lung tissues of 2019 coronavirus disease patients were significantly higher than that in the control donor tissues, which suggests elevating the pyroptosis in the lung tissues of patients with COVID-19. 5 In patients with 2019 coronavirus disease, SARS-CoV-2 induces the up-regulation of pyroptosis-related cytokines in lung and spleen tissues of COVID-19 patients, but there is no pathological evidence yet.
Once the SARS-CoV-2 virus enters the human upper respiratory tract, it combines with respiratory epithelial cells and enters into the cells to replicate, which induces a strong immune response.6,7 The main pathological manifestation of the lung is diffuse alveolar injury with cellular fibrous mucus-like exudation, cellular exfoliation, hyaline membrane formation, and interstitial mononuclear inflammatory infiltration. 8 The spleen is an important site where the immune response was involved. Human infection with SARS-CoV-2 is characterized by a decrease in lymphocytes and an increase in the ratio of red to white marrow. 9 This suggests that the lung and spleen play an important role in the immune response. In existing studies, it has been suggested that the excessive inflammation caused by SARS-CoV-2 is the main cause of death in COVID-19 patients. 10 The main finding of this study is a significant increase of macrophages in the lung and spleen in patients with COVID-19 deaths and more than half of the cells are positive for SARS-CoV-2 spike protein. In addition, there is a significant increase in the expression of pyroptosis-related inflammatory factors, which probably could be related to the translocation activation of NF-κB and HMGB-1.
Materials and methods
Processing of tissues
Tissues: The lung tissues and spleen tissues of the control group are obtained from two patients who die unexpectedly without SARS-CoV-2 infection, and the lung and spleen tissues of the experimental group are taken from three patients who died of SARS-CoV-2 virus infection. The clinical diagnosis of subjects was confirmed by qRT-PCR for SARS-COV-2 in throat samples and lung CT. All specimens are provided by the Department of Forensic Medicine, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China. (Ethics number: KY-2020–15.01).
Specimen processing: The lung and spleen tissues are fixed in 4% paraformaldehyde at room temperature for 24 h. Then they are washed with phosphate-buffered saline (PBS), dehydrated by ethanol, transparently treated with xylene, soaked in wax, embedded in paraffin, and sectioned with a thickness of 4 μm.
Experimental methods
Reagent
paraffin wax (Sinopharm Group, 69,019,361), absolute ethanol (Sinopharm Group, 10,009,218), xylene (Sinopharm Group, 10,023,418), goat serum (Boster, AR1009), anti-fluorescence quenching mounting tablets (Southernbiotech, 0100–01). Primary antibody: SARS-CoV-2 spike protein antibody (Abcam, 1:500), CD68 antibody (Absin, 1:500) NF-κB antibody (Abcam, 1:500), HMGB-1 antibody (Abcam, ab18256, 1:1000), IL-18 antibody (Abcam, ab1376664, 1:100), NLRP3 antibody (Abgent, AP8564A-EV, 1:100), anti-GSDMD against N-terminus antibody (Proteintech, Cat No. 66,387-1-Ig 1:50) recognizes both active and total GSDMD. Secondary antibody: fluorescence-labeled goat anti-rabbit IgG (Boster, BA1032, 1:800), fluorescence-labeled the goat anti-mouse IgG (Abcam, ab150116, ab150113, 1:800).
Immunofluorescence
Paraffin sections receive xylene, absolute ethanol, gradient alcohol, EDTA antigen retrieval, serum blocking for 1 hour, primary antibody incubated overnight at 4°C, fluorescent secondary antibody incubated for 1 h in the dark, and then mounted in sequence.
Quantitation of immunofluorescence
The immunostaining of the lung and spleen tissues is observed and counted using an inverted fluorescent microscope (model: OlympusX73). Cells stained in the slice are considered positive, while cells lacking fluorescent staining are considered negative cells. Three slices from each person’s lung and spleen tissue and three fields of view from each slide were taken for photographing and counting.
Statistical analysis
Spss20 software is used for data analysis, and values of p < 0.05 are statistically significant.
Result
Increased expression of inflammatory factors such as NF-κB, NLRP3, IL-18, and GSDMD in the lung of patients with COVID-19
Three patients with COVID-19 and two uninfected patients were tested for the lung tissue with anti-NF-κB antibody (Figure 1(a)), anti-NLRP3 antibody (Figure 1(b)), anti-IL-18 antibody (Figure 1(c)), and anti-HMGB-1 antibody (Figure 1(d)), and anti-GSDMD antibody (Supplementary Figure 1(a)) respectively. We found that NF-κB, NLRP3, and IL-18 were up-regulated in lung tissues of the COVID-19 group. In uninfected lung tissues, NF-κB was mainly expressed in the cytoplasm; while HMGB1 was expressed in the nucleus. NLRP3 and IL-18 were expressed both in the nucleus and cytoplasm. NF-κB, NLRP3, IL-18, and HMGB1 were expressed in the nucleus and cytoplasm in the lung tissues of COVID-19 patients in the lung tissues from patients with COVID-19. The positive rates of NF-κB (Figure 1(e)) in the lung tissues of the control group and COVID-19 group were 9.8% and 73.4%, respectively (p = 0.000); the positive rates of NLRP3 (Figure 1(e)) were 5.5% and 63.6%, respectively (p = 0.000); the positive rates of IL-18 (Figure 1(e)) were 24.4% and 76.2% (p = 0.000), the positive rates of GSDMD (Supplementary Figure 1(c)) were 17.5% and 46.8% (p = 0.000), while there was no difference between the positive rate of HMGB-1 (Figure 1(e)) in lung tissue in the control group (91.1%) and those in the COVID-19 group (93.7%) (p = 0.198). Pyroptosis-related inflammatory factors in lungs of COVID-19 patients were upregulated. Anti-NF-κB antibody, anti-NLRP3 antibody, anti-IL-18 antibody, and anti-HMGB-1 antibody were used to detect lungtissues in the control group and the COVID-19 group respectively. The blue on the left is DAPI nuclear stain, the right picture is the superposition of the first two. (a) (x400): the red in the middle is NF-κB; (b) (x400): the red in the middle is NLRP3; (c) (x400): the red in the middle is IL-18; (d) (x400): the red in the middle is HMGB1; E: the statistical graphs of the positive rate of NF-κB, NLRP3, IL-18, HMGB-1 in lung tissues.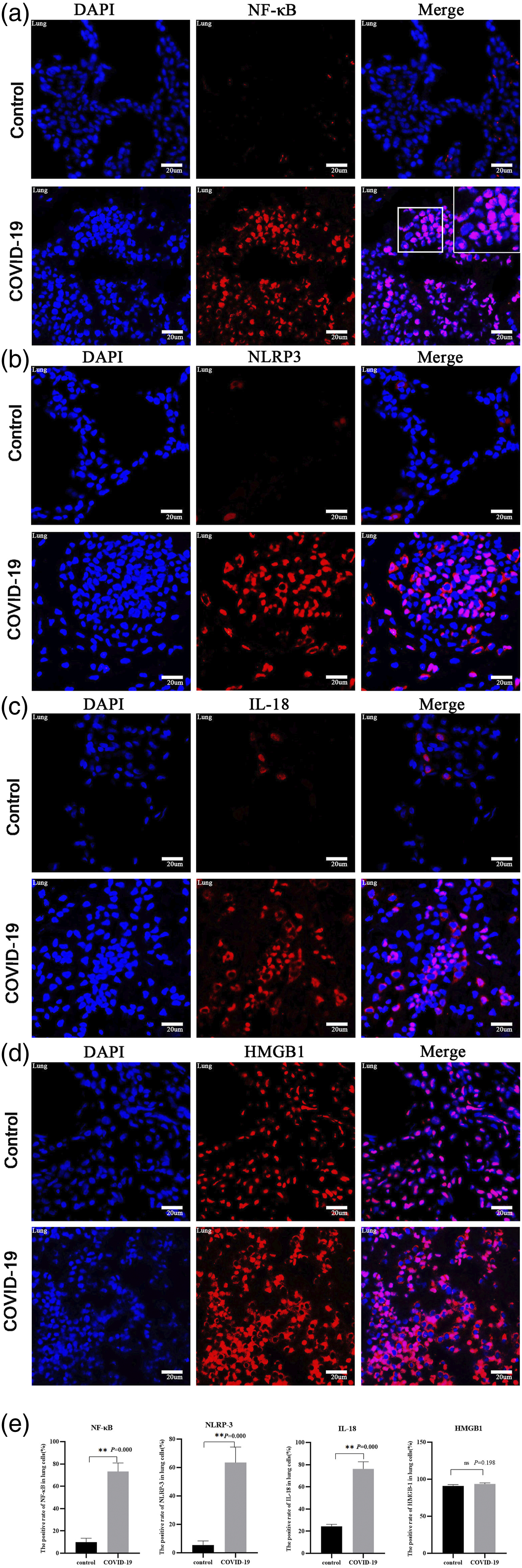
2. Increased expression of inflammatory factors such as NF-κB, NLRP3, IL-18, HMGB-1, and GSDMD in the spleen of patients with COVID-19
Immunofluorescence assays were performed with anti-NF-κB antibody (Figure 2(a)), anti-NLRP3 antibody (Figure 2(b)), anti-IL-18 antibody (Figure 2(c)), anti-HMGB-1 antibody (Figure 2(d)), and anti-GSDMD antibody (Supplementary Figure 1(b)) in the spleen tissues of three COVID-19 patients and two uninfected patients, respectively. We found that NF-κB, NLRP3, IL-18, and HMGB-1 were up-regulated in spleens of the COVID-19 patients. In spleen tissues of uninfected people, NF-κB was mainly expressed in the cytoplasm; HMGB1 was mainly expressed in the nucleus, while NLRP3 and IL-18 were both expressed in the nucleus and cytoplasm. In the spleen tissues of COVID-19 patients, NF-κB, NLRP3, IL-18, and HMGB1 were both expressed in the nucleus and cytoplasm. In the control group and COVID-19 spleen, the NF-κB positive rates (Figure 2(e)) were 20.6% and 71.2%, respectively (p = 0.000); the positive rate of NLRP3 (Figure 2(e)) were 18.9% and 72.0%, respectively (p = 0.000); the positive rates of IL-18 (Figure 2(e)) were 15.24% and 64.8% respectively (p = 0.000); the positive rates of HMGB-1 (Figure 2(e)) were 27.6% and 69.2% (p = 0.000); the positive rates of GSDMD (Supplementary Figure 1(c)) were 23% and 48.8% (p = 0.000). Pyroptosis-related inflammatory factors in spleens of COVID-19 patients were upregulated. Anti-NF-κB antibody, anti-NLRP3 antibody, anti-IL-18 antibody, and anti-HMGB-1 antibody were used to detect the spleen in the control group and the COVID-19 group respectively. The blue on the left is DAPI nuclear stain, the picture on the right is the superposition of the first two. (a) (x200): the red one in the middle is NF-κB; (b) (x200): the red one in the middle is NLRP3; (c) (x200): the red one in the middle is IL-18; (d) (x100): the red one in the middle is HMGB1; (e): the statistical graphs of the positive rate of NF-κB, NLRP3, IL-18, HMGB-1 in spleens.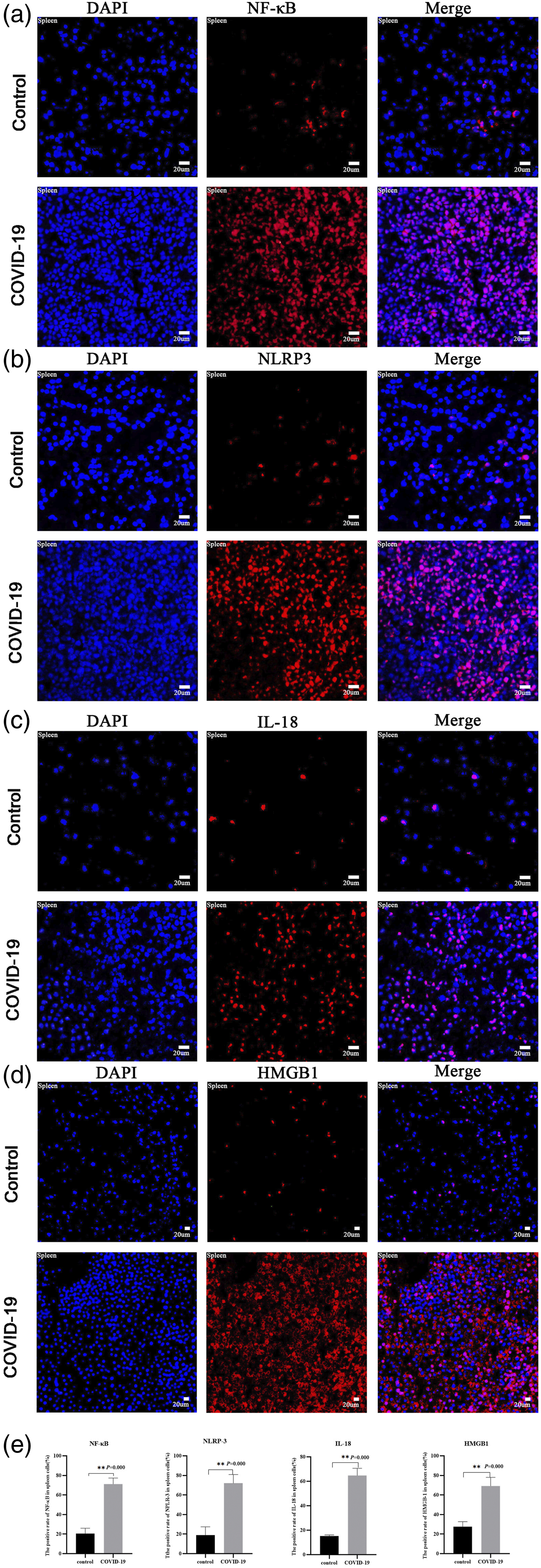
SARS-CoV-2 spike protein was expressed in the lungs and spleens of COVID-19 patients
The lung and spleen tissues of three COVID-19 and two uninfected patients were examined with anti-SARS-CoV-2 spike protein antibody (Figure 3(a), Figure 3(b)). The SARS-CoV-2 spike protein was mainly expressed in the cytoplasm. The positive rates of SARS-CoV-2 spike protein in lung tissues (Figure 3(c)) of the control group and the COVID-19 group were 2.5% and 47.2% (p = 0.000), respectively. The positive rates of SARS-CoV-2 spike protein in spleen tissues (Figure 3(c)) of the control group and the COVID-19 group were 2.7% and 55.5% (p = 0.000), respectively. The expression of SARS-CoV-2 spike protein in lungs and spleens of COVID-19 group. Anti-SARSCoV-2 spike protein antibody was used to detect lungs and spleens of the control group and COVID-19 group respectively. The blue on the left is DAPI nuclear stain, the red in the middle is the SARS-CoV-2 spike protein, and the right one is the superposition of the first two. (a) (x400): lung tissues; (b) (x400): spleentissues; (c): the statistical graphs of the positive rate of SARS-CoV-2 spike protein in lungs and spleens.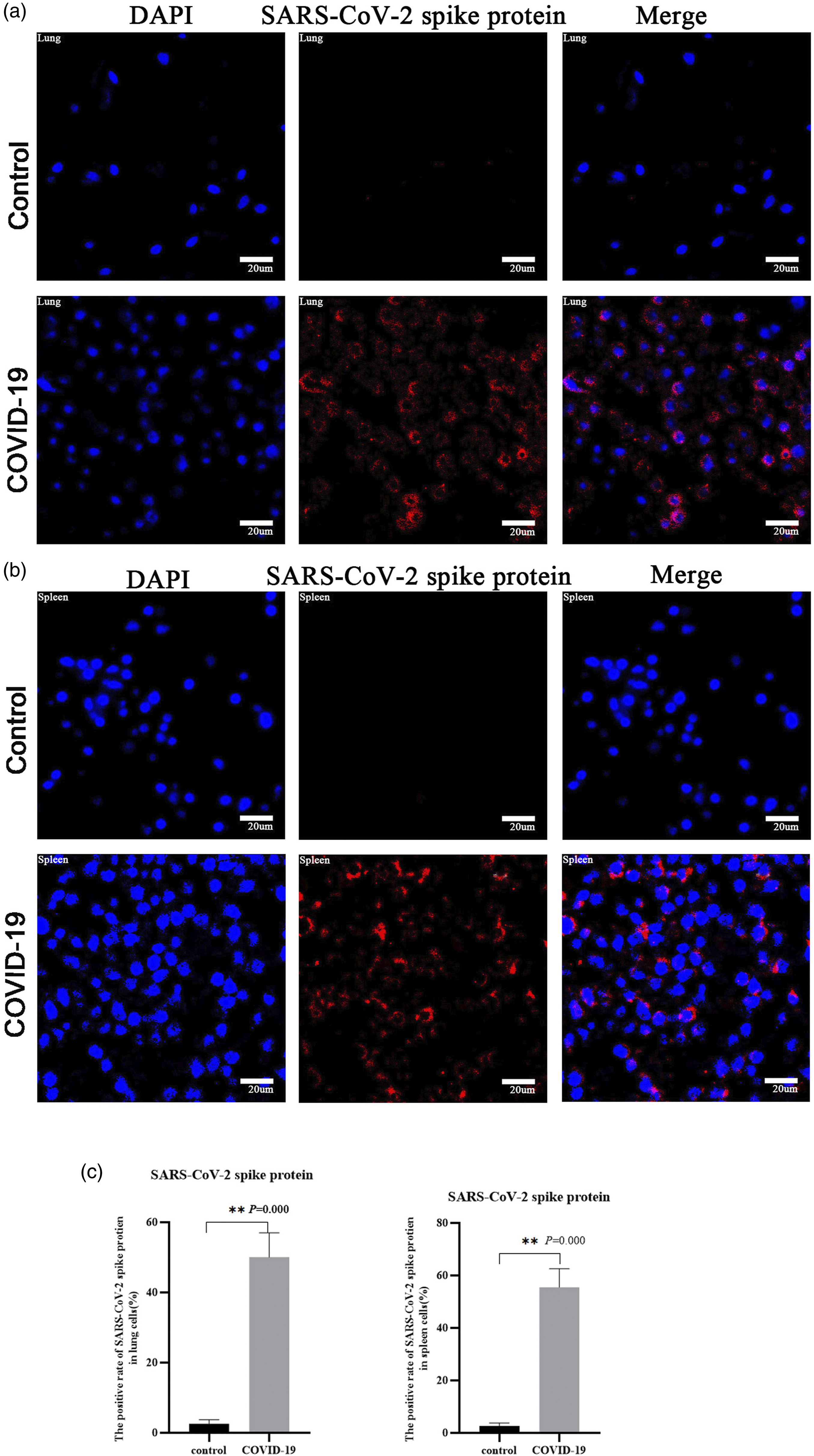
To investigate the virus infection in macrophages, tissues of three COVID-19 dead patients and two uninfected patients were co-stained with anti-SARS-CoV-2 spike protein antibody and anti-CD68 antibody. Both CD68 and SARS-CoV-2 spike protein were expressed in the cytoplasm in lung tissues (Figure 4(a)) and spleen tissues (Figure 4(b)). In the lungs of the control group and COVID-19 group, the positive rates of CD68 (Figure 4(c)) in lung tissues were 16.7% and 63.3%, respectively (p = 0.000), and the positive rates of CD68 (Figure 4(c)) in spleen tissues were 22.1% and 54.0% (p = 0.000), respectively. It was shown that the proportion of macrophages in both the lung and spleen tissues increased in COVID-19 patients. The positive rates of SARS-CoV-2 spike protein in macrophages in lungs (Figure 4(c)) of the control group and COVID-19 group were 2.5% and 56.8% (p = 0.000), respectively; and the positive rates of SARS-CoV-2 spike protein in macrophages in spleens (Figure 4(c)) of the control group and COVID-19 group were 3.0% and 64.9% (p = 0.000). SARS-CoV-2 spike protein was positive in a small number of cells in the control group could be a non-specific background. The macrophages in the lung and spleen of COVID-19 patients express SARS-CoV-2 spike protein. The lungs and spleens of the COVID-19 group and the control group were co-infected with anti-SARS-CoV-2 spike protein antibody and anti-CD68 antibody, respectively. The blue is the cell nucleus, the red is CD68, the green is SARS-CoV-2 spike protein, the right picture is the superposition of the first three pictures. (a) (x400): lung tissues; (b) (x400): spleen tissues; (c): the statistical graphs of CD68 positive rate in lungs and spleens.
The translocation of HMGB-1 and NF-κB in lungs and spleens of COVID-19 patients
To further investigate the signaling pathways that are involved in pathogenesis, the lung and spleen tissues of the COVID-19 group and the control group were co-stained with anti-NF-κB antibody and anti-CD68 antibody. The results revealed that in the lung and spleen tissues of the COVID-19 group, some CD68+ cells are nuclear positive for NF-κB (Figure 5(a), Figure 5(b)). Among these CD68+ cells, the positive rates of NF-κB nuclei in the lung tissues (Figure 5(c)) of the control group and COVID-19 group are 13.4% and 51.4% (p = 0.000), and that in the spleen tissues (Figure 5(c)) of the control group and COVID-19 group were 38.2% and 51.4% (p = 0.000), respectively. NF-κB may be partially translocated from the cytoplasm to the nucleus in the lung and spleen of COVID-19 patients. NF-κB and CD68 were co-stained in lungs and spleens of COVID-19 patients. The lungs and spleens of the COVID-19 group and the control group were co-stained with anti-NF-κB antibody and anti- CD68 antibody. The blue is the cell nucleus, the red is CD68, the green is NF-κB, the right picture is the superposition of the first three pictures. (a) (x400): lung tissue; (b) (x400): spleen tissue; (c): the statistical graphs of the NF-κB nuclear positive rate in lungs and spleens.
Similarly, the lung and spleen of the COVID-19 group and the control group were co-stained with anti-HMGB-1 antibody and anti-CD68 antibody (Figure 6(a), Figure 6(b)). The positive rates of cytoplasmic HMGB-1 in lung tissues (Figure 6(c)) of the control group and the COVID-19 group were 19.7% and 50.3% (p = 0.000), and the positive rate of cytoplasmic HMGB-1 in spleen tissues (Figure 6(c)) of the control group and the COVID-19 group were 12.3% and 45.2% (p = 0.000), respectively. Thus, HMGB-1 could be partially translocated from the nucleus to the cytoplasm in the lung and spleen of COVID-19 patients. HMGB1 and CD68 were co-stained in lungs and spleens of COVID-19 patients. The lungs and spleens of the COVID-19 group and the control group were co-stained with anti-HMGB-1 antibody and anti- CD68 antibody. The blue is the cell nucleus, the red is CD68, the green is HMGB1, the right picture is the superposition of the first three pictures. (a) (x400): lung tissue; (b) (x400): spleen tissue; (c): the statistical graphs of the HMGB1 cytoplasm positive rate in lungs and spleens.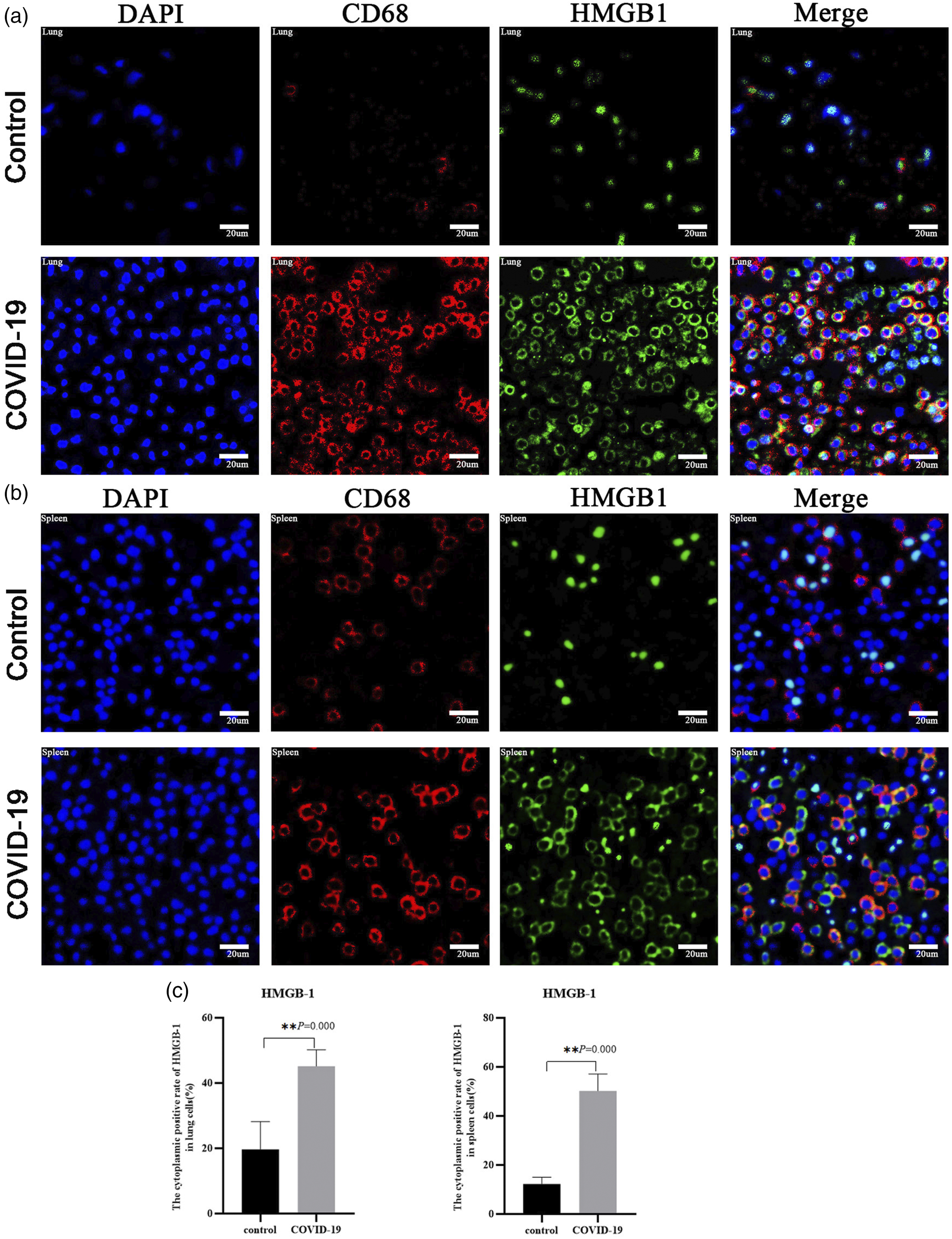
Discussion
SARS-CoV-2 binds to the ACE2 receptors on cytomembranes through the SARS-CoV-2 spike protein and enters into cells. 11 ACE2 receptors are expressed in many organs of the human body. For instance, ACE2 receptors are highly expressed in the heart, intestine, kidney, testis, thyroid, and adipose tissue, while the expression in the lung and spleen is medium or low. 12 In this study, the SARS-CoV-2 spike protein was detected in the lung and spleen of three patients. Meanwhile, there were more macrophages in the lung and spleen of COVID-19 deceased, and the positive rates of SARS-CoV-2 spike protein were as high as 54.8% and 64.9% in the lung and spleen, respectively. Although the single-cell RNA sequencing analysis of normal human tissues shows that the expression of ACE2 on macrophages is low, some scholars believe that when macrophages are activated by inflammatory signals (such as interferon Ⅰ), the expression of the ACE2 receptor on macrophages increases. 13 However, it cannot be ruled out that some macrophages expressing SARS-CoV-2 spike protein were due to phagocytosis.
NF-κB, NLRP3, IL-18, and HMGB-1 have been reported to be associated with pyroptosis.14,15 Pyroptosis is a programmed cell death mediated by GSDMD and characterized by the spontaneous release of cytoplasmic inflammatory factors (IL-1β, IL-18, HMGB1, etc.) into the extracellular space.16,17 The transcription factor NF-κB is a key regulator of the immune response and inflammation. NF-κB is involved in the pathophysiological process of a variety of inflammatory diseases such as chronic inflammation, viral infection, and septic shock syndrome. 18 The activation of the NF-κB is mainly manifested by the translocation from the cytoplasm to the nucleus. 19 Studies have shown that viral proteins of SARS-CoV-1 and Mers-CoV, such as S protein and N protein, can cause the excessive activation of the NF-κB. 20 In our study, the expression of the NF-κB in the COVID-19 group was higher than that in the control group, and the nuclear positive rate of the NF-κB was significantly higher than that of uninfected individuals, indicating the nuclear translocation of the NF-κB. This nuclear translocation probably could be related to the activation of macrophages. In patients with severe COVID-19 reported previously, similar activation of the NF-κB pathway was found in B cells. 21 Therefore, the NF-κB was activated in the lungs and spleens of COVID-19 patients. The activated NF-κB regulates the expression of the downstream NLRP3, pro-IL-1β, and pro-IL-18 through the transcription in the nucleus, which thereby regulates the release of a variety of cytokines, chemokines, and adhesion molecules. 18
As a member of the inflammasome family, NLRP3 is a sensor of pyroptosis and is involved in the inflammatory response to multiple viral infections, such as dengue fever and respiratory syncytial virus. It has been shown of the binding of S protein to ACE2 over-activates NLRP3, which triggers caspase 1 to cleave GSDMD into GSDMD N-terminus and C-terminus. 22 The GSDMD N-terminus forms a pore in the cell membrane, and cytokines such as Il-18 and Il-1β can be released to the outside of the cell through the pores. 23 Thus, cleaved GSDMD is an indicator of pyroptosis. The high expression of GSDMD in the lung and spleen of the COVID-19 group indicated the occurrence of pyroptosis. This study has found that in the lungs and spleens of the COVID-19 group, NLRP3 was highly expressed, which was consistent with the results of the active NLRP3 inflammasome in the lungs of patients who died of COVID-19 reported by Rodrigues TS. 24 NLRP3 was found to induce coagulation and arterial thrombosis through the regulation of platelet integrin αIIbβ3 outside-in signaling. 25 Recent clinical studies have shown that platelets and their activation are associated with higher rates of venous and arterial thrombosis in severe COVID-19 patients. 26 Therefore, the upregulation of NLRP3 may be involved in thrombosis in severe COVID-19 patients. In addition, the high expression of NLRP3 may be one of the reasons for the cytokine storm in COVID-19. On one hand, NLRP3 was found to activate lipoxinB4 to promote the release of pro-inflammatory cytokines by inducing caspase-7-dependent pyroptosis.22,27 On other hand, the active NLRP3 can lead to pyroptosis. The expression of IL-18 in the lungs and spleens of the COVID-19 group was also significantly higher than that of the control group. This suggests that SARS-CoV-2 could activate the downstream pyroptosis pathway of NLRP3, which leads to IL-1β, IL-18, and other inflammatory factors being overproduced and released.28,29 Then the IL-18 and IL-1β further stimulate NK cells and T cells to produce other cytokines, such as tumor necrosis factor-α (TNF-α), granulocyte-macrophage colony-stimulating factor (GM-CSF), thereby starting the cytokine cascade reaction, and causing cytokine storms.30,31
HMGB-1 is found in the nucleus of resting eukaryotic cells. On the one hand, it can be passively released by damaged or dead cells. On the other hand, innate immune cells such as monocytes, macrophages, tissue cells, neutrophils, and dendritic cells can release HMGB-1 after stimulation by inflammatory cytokines 32 Compared to the control group, the intracellular distribution of HMGB-1 in the lungs and spleens of the COVID-19 group changed since the cytoplasmic positive rate is significantly higher. It was reported that HMGB-1 in some macrophages of the lung and spleen of patients who die from COVID-19 is mobilized from the nucleus to the cytoplasm This is the first critical step in the release of HMGB1 from activated immune cells. Chen R et al. also detected the elevated HMGB1 in the serum of patients with severe COVID-19. 33 After infection of SARS-CoV-2, immune cells such as macrophages could actively secrete HMGB1 outside the cell. The excessive extracellular HMGB1 can further lead to the release of pro-inflammatory cytokines, including TNF, IL-10, IL-1β, and IL-6, which form a positive feedback regulation of inflammatory response in patients. 34 In addition, research has shown that platelet-derived HMGB1 mediates platelet aggregation and thrombosis. 35 The activation of HMGB-1 in the COVID-19 group may play an important role in thrombosis in COVID-19 patients. The limitations of this study are the lack of relevant clinical data and the small sample size. Additional studies with larger sample sizes and experiments in vitro will be needed to corroborate these conclusions.
Conclusion
In the lung and spleen of patients who die from COVID-19, macrophages may be over-activated and cause the release and activation of NF-κB, NLRP3, IL-18, HMGB-1, and GSDMD. The results suggested that SARS-CoV-2 could induce the pyroptosis of macrophages. The cell membrane ruptures lead to the release of intracellular inflammatory factors and trigger an intense inflammatory response (Figure 7). This excessive inflammatory response is associated with lung injury, multiple organ failure, acute respiratory distress syndrome (ARDS), systemic inflammatory response syndrome (SIRS) and thrombosis. Our data have provided evidence to support the use of corticosteroids to suppress excessive inflammation in the treatment of patients with severe COVID-19. The SARS-CoV-2 induces the lung and spleen pyroptosis. Once the SARS-CoV-2 virus infects the human body, it attacks the lung and spleen, causing excessive activation of macrophages and pyroptosisrelated inflammatory factors such as NF-κB, HMGB-1, NLRP-3, IL-18, etc. It suggests that SARS-CoV-2 may rupture the cell membrane by inducing macrophage pyroptosis, leading to the release of intracellular inflammatory factors and triggering a cytokine storm.
Supplemental Material
Supplemental Material - Pyroptosis in the lung and spleen of patients died from COVID-19
Supplemental Material for Pyroptosis in the lung and spleen of patients died from COVID-19 by Xin Tong, Haiqin Ping, Xiaoming Gong, Kai Zhang, Zhaojun Chen, Caiyun Cai, Zhiyan Lu, Rongrong Yang, Shicheng Gao, Yunyun Wang, Xinghuan Wang, Liang Liu and Hengning Ke in European Journal of Inflammation
Footnotes
Acknowledgements
I thank the patients who donated organs.
Author’s contribution
TX wrote the manuscript; KHN, LZY, YRR, GSC and WXH supervised the project and revised the manuscript; TX, PHQ, GXM, ZK, CZJ, and CCY did the immunofluorescence; LL and WYY did the tissues collection.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The funding for this research was provided by the National Natural Science Foundation of China (No.31760263), the Non-profit Central Research Institute Fund of the Chinese Academy of Medical Sciences (2020-PT320-004) and Huazhong University of Science and Technology (2020kfyXGYJ098).
Ethics approval
We have obtained written consent from the subjects before study initiation (No.1.0/2020–02-09). Ethical approval for this study was obtained from the Medical Ethics Committee of Wuhan Infectious Diseases Hospital (No.KY-2020–15.01).
Supplemental Material
Supplementary material for this article is available on the online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
