Abstract
Prolonged exposure to different occupational or environmental toxicants triggered oxidative stress and inflammatory reactions mediated lung damage. This study was designed to explore the influence and protective impact of flavone on lung injury in rats intoxicated with nicotine (NIC) and exposed to radiation (IR). Forty rats were divided into four groups; group I control, group II flavone; rats were administered with flavone (25 mg/kg/day), group III NIC + IR; rats were injected intraperitoneally with NIC (1 mg/kg/day) and exposed to γ-IR (3.5 Gy once/week for 2 weeks) while group IV NIC + IR + flavone; rats were injected with NIC, exposed to IR and administered with flavone. Redox status parameters and histopathological changes in lung tissue were evaluated. Nuclear factor-kappa B (NF-κB), forkhead box O-class1 (FoxO1) and nucleotide-binding domain- (NOD-) like receptor pyrin domain-containing-3 (NLRP3) gene expression were measured in lung tissues. Moreover, nuclear factor (erythroid-derived 2)-like 2 (Nrf2) and phosphatidylinositol three kinase (PI3K) were measured using ELISA kits. Our data demonstrates, for the first time, that flavone protects the lung from NIC/IR-associated cytotoxicity, by attenuating the disrupted redox status and aggravating the antioxidant defence mechanism via activation of the PI3K/Nrf2. Moreover, flavone alleviates pulmonary inflammation by inhibiting the inflammatory signaling pathway FOXO1/NF-κB/NLRP3- Inflammasome. Collectively, the obtained results exhibited a notable efficiency of flavone in alleviating lung injury induced by NIC and IR via modulating PI3K/Nrf2 and FoxO1/NLRP3 Inflammasome.
Introduction
Prolonged exposure to different occupational or environmental toxicants either from chemical (asbestos, formaldehyde and polycyclic aromatic hydrocarbons) or physical (ionizing and non-ionizing radiations) as well as cigarette smoke and air pollutants negatively affect lung functions promoting lung diseases and eventually lung cancer. 1 These toxicants elicit oxidative stress and inflammatory reactions which deteriorate the lung architecture via the destruction and fibrosis of the lung parenchyma impairing the repair mechanism and consequently increasing morbidity and mortality globally due to pulmonary dysfunction and diseases. 2
Nicotine (NIC) is one of the widely spread and known environmental biohazards. It is the most abundant alkaloid of tobacco, which is responsible for the addiction and toxicity of cigarettes on lung health and immunity. 3 Regarding the excessive nicotine receptors, solubility and transfer across cellular membranes are rapidly absorbed into the blood circulation and deposited in the lung tissue due to the large surface area of the alveoli. 4 Additionally, its metabolism to nitrosamine compounds results in the production of free radical species like reactive oxygen species (ROS) that promote toxicity and oxidative damage of the lung epithelial cells as well as inflammatory response, pulmonary dysfunction and other diseases. 5
Exposure of humans to radiation regarding its source natural environment (background, radon or ultraviolet (UV) medical (diagnostic X-rays or γ-rays radiotherapy), occupational or accidental seriously implies the life quality owing to its adverse effects such as toxicity, organ dysfunction and injury besides body immunosuppression. 1 Collectively, radiation boosts lung injury and toxicity owing to their high radiosensitivity leading to radiation-induced lung injury (RILI).6,7 The detrimental and toxic effect of radiation was attributed to its interaction with biological organs and systems triggering the excessive generation of ROS and oxidative stress that eventually augments cellular oxidative damage and impairs diverse signaling pathways. 8
The excessive production of ROS from both nicotine and γ-IR markedly promotes acute lung injury (ALI) by sustaining oxidative stress and aggravating pulmonary inflammation by activation of many inflammatory pathways such as nuclear factor-kappa B (NF-κB), Forkhead box protein O1 (FoxO1)- nucleotide-binding domain- (NOD-) like receptor pyrin domain-containing-3 (NLRP3) and other inflammatory cytokines. 9 Moreover, the inflammatory signaling pathway FOXO1/NF-κB/NLRP3 is negatively regulated by the PI3k/Akt signaling pathway 10 which regulates cell proliferation, survival, apoptosis and cytoprotective defence via activation of its downstream Nuclear factor-erythroid 2-related factor 2 (Nrf2) signaling pathway. 11 Nrf2 is a cytoprotective transcription factor that maintains the cellular antioxidant defence mechanism, interacts with several signaling pathways, affects the activity of many oxidases associated with inflammation, and has an antioxidant. 12 The activated Nrf2 attenuates the oxidative stress and inflammation induced by FoxO1-NLRP3 beside suppressing the NF-κB which eventually alleviates lung injury. 13
Owing to their potential efficacy and absence of side effects, together with little or no toxicity, medicinal plants are preferable to conventional drugs in treating various diseases that are considered difficult to cure. From these medicinal plants, flavonoids are natural phytochemical compounds that have been used in herbal complementary medicines for a long time now.14,15 Flavonoids are considered derivatives of the parent compound flavone and are classified into flavones, flavonols, isoflavones, and anthocyanidins based on their oxidation state and the degree of conjugation of the central heterocyclic ring.16,17 Flavones (from Latin flavus, “yellow”) are a vital subclass of the flavonoid’s family, which are natural polyphenolic products with the backbone of 2-phenylchromen-4-one (2-phenyl-1-benzopyran-4-one) (as shown in Figure 1). Flavones are usually found in large amounts in a glycosylated form in various common foods such as yellow or orange fruits, celery, parsley, red peppers, chamomile, mint, as well as spices. This class commonly includes flavone, apigenin, luteolin, and chrysin.
18
Based on their role in supporting health, flavones have a valuable effect in many applicable fields (pharmacy, nutrition cosmetic, and therapy) owing to their low toxicity, anti-inflammatory, antimicrobial, anticancer activities, antioxidant, antiallergic, neuroprotective, antidiabetic, cardioprotective, and metal chelating properties, in addition to their immune-regulatory activities in various diseases either prophylaxis or therapeutic.
19
Flavone backbone structure adopted from Catarino et al. study.
20

Therefore, the current study was conducted to examine the efficiency of flavone in mitigating lung injury induced by Nicotine and γ -IR and its modulatory effects on disrupted redox status as well as FoxO1-NLRP3 Inflammasome and PI3K/Nrf2 signaling pathway.
Materials and methods
Materials
Nicotine, synthesis grade Product Code: NI00200100, CAS No. 54-11-5 was obtained from Scharlau, Scharlab S.L Barcelona, Spain. Flavone was purchased from Sigma–Aldrich® (St Louis, Missouri, United States).
Irradiation process
Rats were subjected to whole-body γ-irradiation at the National Centre for Radiation Research and Technology (NCRRT), Egyptian Atomic Energy Authority, using Canadian gamma cell-40 (137Cesium) at a dose of 3.5 Gy once/week for 2 weeks (cumulative dose 7 Gy), at a dose rate of 0.333 Gy•min-1.
Animals
The handling of the experimental animals involved in this study followed the approved ethical guidelines of the Institutional Animal Care and Use Committee Research Ethic Board of Benha University, Faculty of Veterinary Medicine (BUFVTM12-11-22). Male rats weighing between 100 and 120 g were procured from the Nile Company for Pharmaceuticals & Chemical Industries S.A.E. (Egypt) and were housed in hygienic cages at a temperature of 22 ± 2°C, with a consistent 12-h light/dark cycle, and unrestricted access to a commercial pellet diet and potable water.
Experimental design
Experimental rats were divided into the following groups (10 rats each). (1) Control Group: Normal rats served as control. (2) Flavone Group: where rats were treated orally with flavone (25 mg/kg/day) for 2 weeks. (3) Nicotine + IR Group: its rats were daily injected with Nicotine intraperitoneally at a dose of 1 mg/kg body weight with modification
3
for 3 weeks and exposed to whole body γ-IR at a dose of 3.5 Gy once/week for 2 weeks (total dose 7 Gy). (4) Nicotine + IR + Flavone Group: Its rats were daily injected with Nicotine and exposed to IR and at the beginning of the second week, rats were treated with flavone (25 mg/kg/day) for 2 weeks.
At the end of the experimental period, the animals were fasted overnight and sacrificed under anaesthesia using urethane. Lung tissues were dissected out and washed with saline, dried on filter paper, and then divided into two parts. The first part from each lung was stored at −80°C for the biochemical analysis. Moreover, the other part of the lung was immediately preserved in a 10% buffered formalin-saline solution for histopathological examination.
Bioinformatic analysis
Data source and processing
The corresponding Gene Expression Omnibus (GEO) datasets contain mRNA expression profiles of rats exposed to cigarette smoking (CS) and control rats were selected. The target microarray dataset, including GSE178513, was extracted from the GEO database (https://www.ncbi.nlm.nih.gov/geo/). The GSE178513 dataset based on the GPL26961 platform (Agilent-085,985 Arraystar Rat lncRNA V3 microarray) contained 3 CS rats and three control rats.
Screening of differentially expressed genes (DEGs)
The GEO2R web tool was applied to identify the DEGs of GSE178513 with the criteria of adjusted p-value (Adj.p) < 0.05 and | log2 fold-change (FC) | > 2.0.
Functional enrichment analysis of DEGs
KOBAS server 21 was used to perform functional annotation analyses including Kyoto Encyclopedia of Genes and Genomes pathways (KEGG) and Gene ontology (GO terms). This program can provide a functional interpretation of gene lists derived from genomic studies. A p-value <0.05 was applied to get a significant pathway. However, the bar charts for GO terms and KEGG pathways were performed via the R package statistical software. 22
Biochemical measurements
Evaluation of oxidative stress parameters
The levels of Malondialdehyde (MDA: Cat. No. MD 25 29) as an indicator for lipid peroxidation, as well as the antioxidant parameters glutathione (GSH: Cat. No. GR 25 11) and glutathione peroxidase (GPx: Cat. No. GP 25 24) activity were measured by commercial kits obtained from Bio-Diagnostic Company in Cairo, Egypt.
ELISA estimations
The concentrations of tumor necrosis factor-α (TNF-α: Cat #MBS924824), nuclear factor (erythroid-derived 2)-like 2 (Nrf2: Cat # MBS012148), phosphatidylinositol three kinase (PI3K: Cat # MBS26.381)and Caspase-1 (CASP1: Cat# MBS2019421)were measured in the homogenates of lung tissues by commercially ELISA kits from My BioSource, San Diego, USA according to the manufacturer’s guidelines.
Quantitative real-time PCR (RT-PCR) analysis
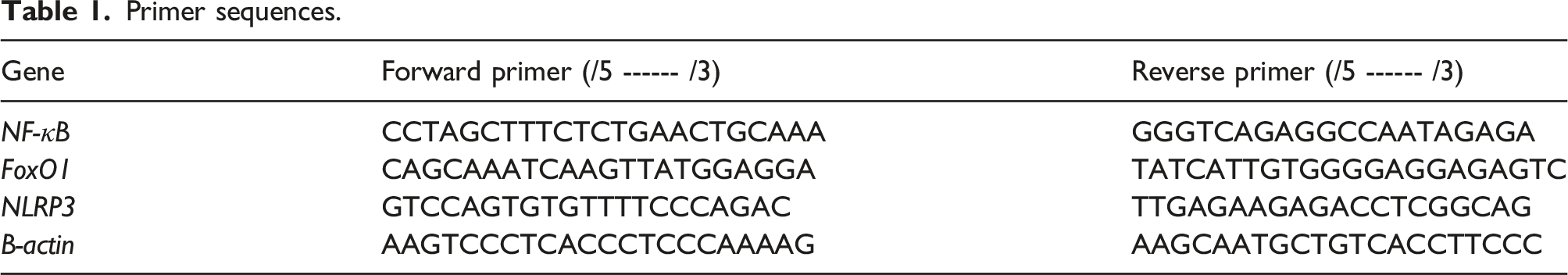
Primer sequences.
Histopathological examination of lung
Lung tissues were initially preserved in a solution of 10% neutral buffered formalin solution, subsequently excised, cleansed, and dehydrated using a series of ascending alcohol concentrations. After that, the dehydrated specimens were embedded in paraffin blocks and sliced into 4-6 µm sections. Finally, the resulting tissue sections were stained with hematoxylin and eosin (H&E) for evaluating the histopathological changes in the lung by electric light microscope. 24 Each lung section was scored from 0 to 4 depending on the area exposed to interstitial inflammation, alveolar wall thickening, peribronchial inflammation and interstitial edema (0 ≤ 10%, 1 = up to 30%, 2 = up to 50%, 3 = up to 70%, 4 ≥ 70%). 25
Statistical analysis
The Statistical Program for Social Science (SPSS 20, SPSS Inc, USA), and a one-way ANOVA test were used to analyze the results. For the comparisons between groups, Duncan’s test at p ≤ 0.05 was used. The results were presented as means ± standard error of the mean (SEM). Moreover, GraphPad Prism 8 Software (GraphPad Software, Inc., San Diego, CA, United States) was used to display the charts.
Results
Differential expression analysis between rats exposed to cigarette smoking and control rats
The transcriptomic analyses of gene expression data of GSE178513 to identify DEGs between rats exposed to cigarette smoking and control rats (Figure 2); the results showed 354 genes were differentially expressed with Adj. p < 0.05 and | log2 fold-change (FC) | > 2.0 (Figure 3). Box plots show the mean expression level for each sample in the dataset. Box plots generated from normalized microarray measurements. X-axis: individual samples grouped into green color (CS) and violet color (control); Y-axis: the expression intensity values. Differentially Expressed Genes. (A) The Venn diagram shows the number of DEGs. B) Volcano plot shows the magnitude of differential expression between CS and control samples, each dot represents one gene that had detectable expression in both groups. The blue dots represent down-regulated genes with Fold change <-2.0 and p value <0.05 while the reds represent up-regulated ones with Fold change >2.0 and p value <0.05.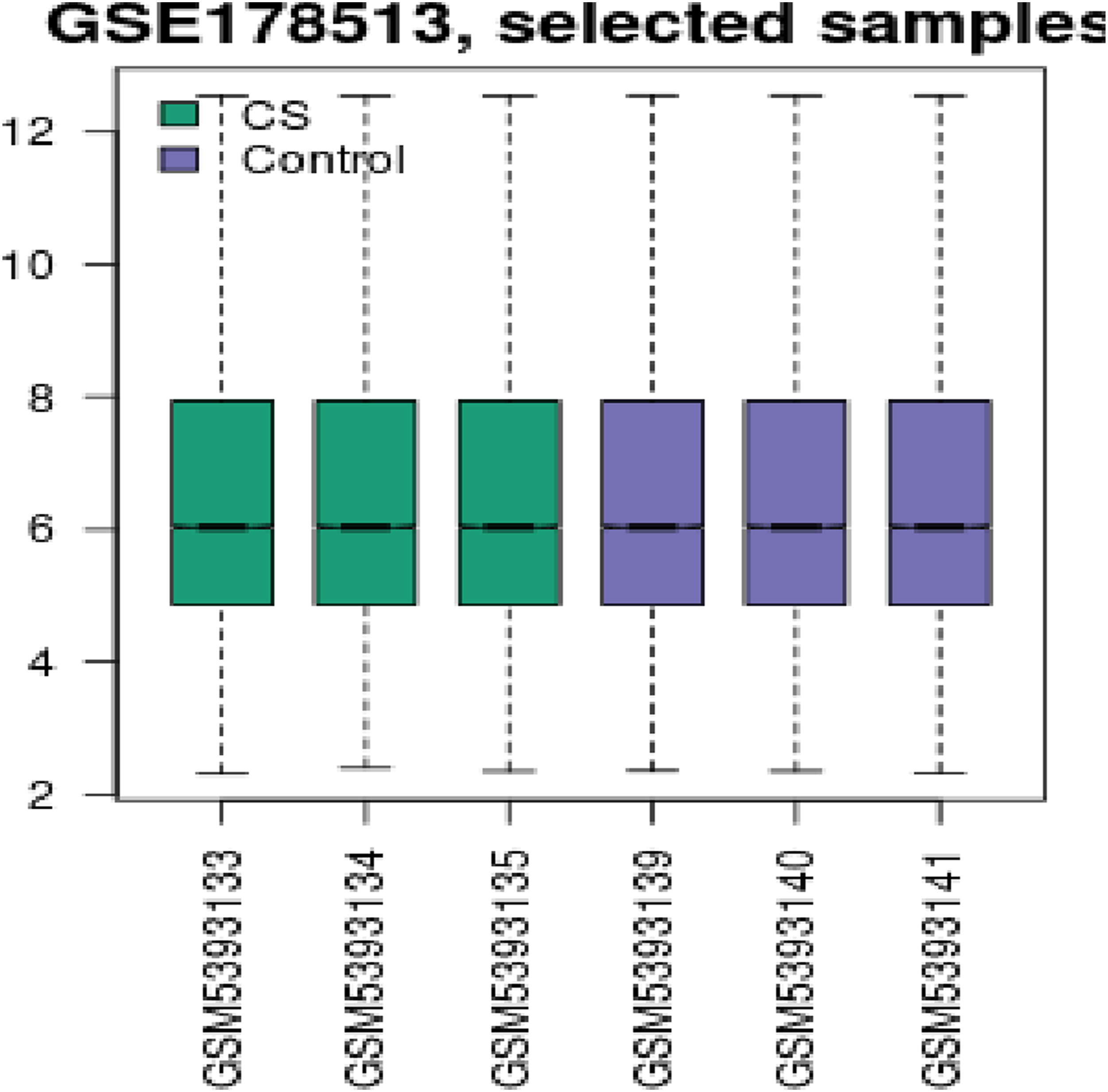

Functional enrichment analysis of DEGs
The Differentially expressed genes analysis exhibited that there were 540 DEGs between CS and control. Additionally, functional enrichment analyses were applied for DEGs to explore the molecular functions and signaling regulated by smoking. GO analysis via KOBAS servers showed that DEGs were markedly enriched in molecular functions related to the oxidation-reduction process, inflammatory response, and immune response. Similarly, KEGG pathways were enriched in Inflammatory mediator regulation of TRP channels, NF-kappa B signaling pathway, TNF signaling pathway, Cytokine-cytokine receptor interaction, IL-17 signaling pathway, Toll-like receptor signaling pathway, and Nicotine addiction via KOBAS servers (Figure 4). Functional enrichment analyses. (A) Bar charts show the GO terms for DEGs in this study. (B) Bar charts show the KEGG pathways for DEGs.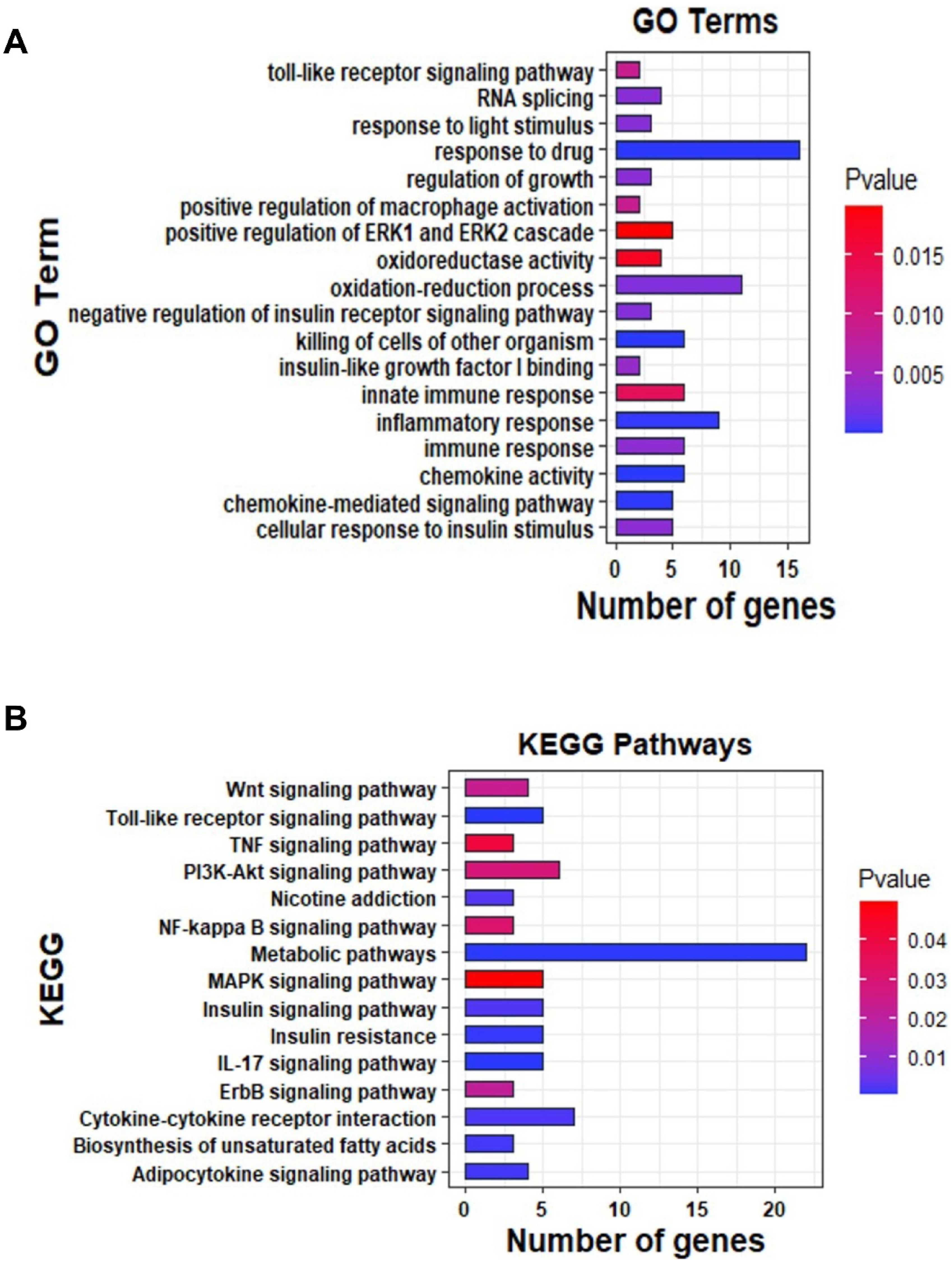
Influence of flavone on pulmonary oxidative stress parameters
The effect of nicotine and IR on oxidative stress is shown in Figure 5. Both nicotine and gamma irradiation significantly elevated ROS levels in the lung leading to impaired redox status. A notable reduction in the pulmonary levels of GSH and GPx associated with marked elevation of MDA level was demonstrated in rats intoxicated with nicotine and exposed to IR as compared to the control group. This indicates pulmonary toxicity and abolished antioxidant capacity. Conversely, there was a significant improvement in the oxidative stress parameters with flavone treatment relative to the Nicotine + IR group as indicated by the significant increase in antioxidant parameters (GSH and GPx) along with the reduction of MDA level. Effect of flavone on oxidative stress status in lung tissues. (A) Lipid peroxidation (MDA), (B) glutathione (GSH), and (C) glutathione peroxidase (GPx). Data is presented as Means ± SE. Columns with different letters are significant (p < 0.05) while those with similar letters are non-significant.
Influence of flavone on the levels of Nrf2, and PI3K in lung tissue
Interestingly, the PI3K/Akt signaling pathway promotes antioxidant, cytoprotective and cell survival mechanisms by activating the Nrf2 signaling pathway against oxidative stress-induced injury. Collectively, in response to the impaired redox balance and blunted antioxidant capacity, the current results revealed a significant decrease in the Nrf2 and its upstream PI3K in the lung tissue of rats injected with nicotine and exposed to IR relative to their control (Figure 6). Whereas, rats that received flavone exhibited a significant upregulation in the pulmonary levels of Nrf2 and PI3K. Thus, suggesting the antioxidant and cytoprotective effect of Flavone by the upregulation of the PI3K/Nrf2 signaling pathway against nicotine and IR-induced lung toxicity. Effect of flavone on the lung levels of PI3K and Nrf2. Data is presented as Means ± SE. (A): PI3K and (B): Nrf2. Columns with different letters are significant (p < 0.05) while those with similar letters are non-significant.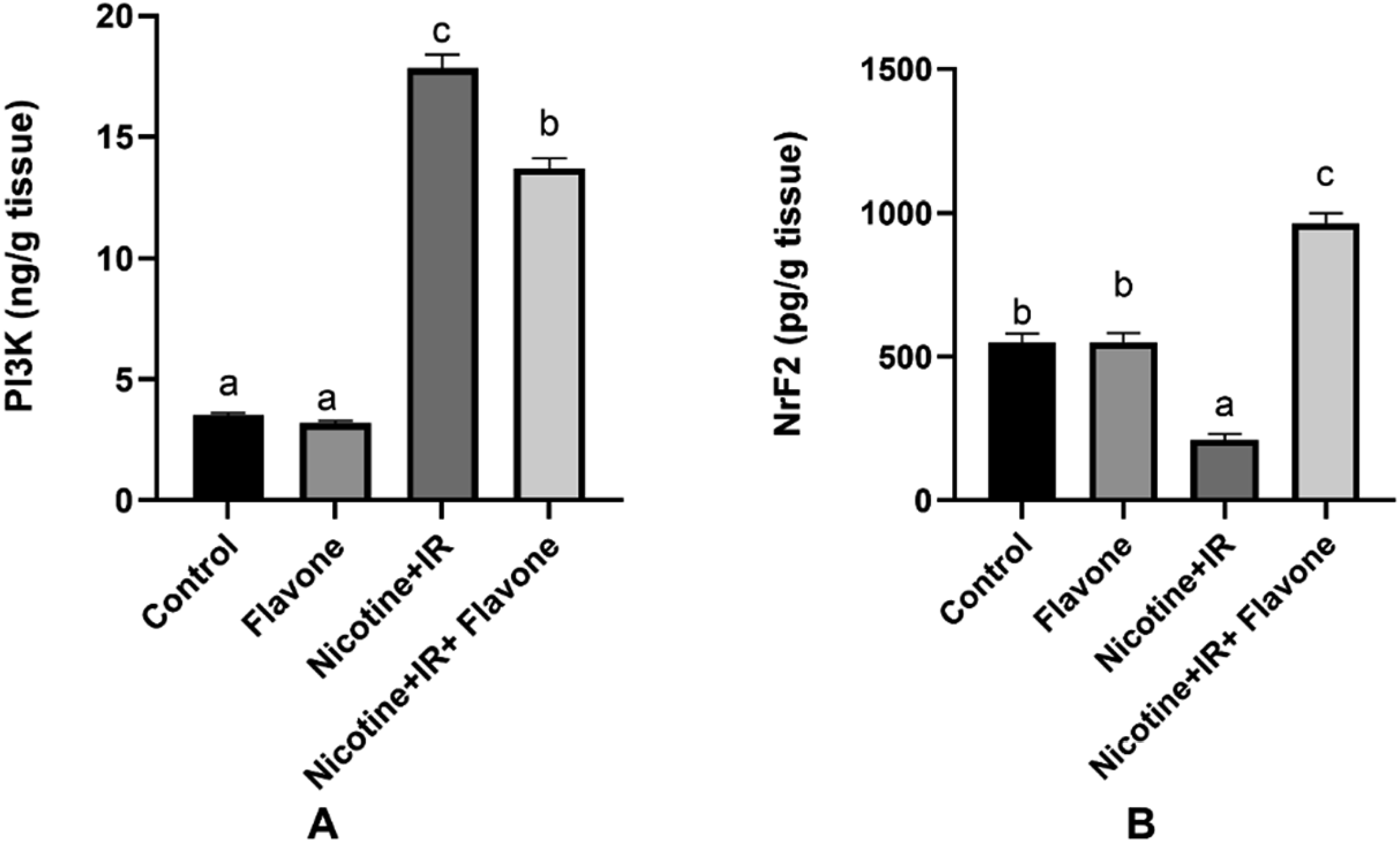
Impact of flavone on the inflammatory mediators (TNF-α and NF-κB) in pulmonary tissues
Regarding the crosstalk of Nrf2 with NF-kB signaling pathway, the interaction between them regulates cellular redox homeostasis and inflammatory response. The marked elevation in the ROS production derived from nicotine and IR coupled with suppression of the PI3K/Nrf2 signaling pathway is associated with inflammatory response and subsequently release of pro-inflammatory mediators as well as further generation of ROS in lung tissues. Herein, this was evident by the remarkable upregulated mRNA expression of the NF κB which in turn increased the levels of the TNF-α in the lung tissues of the nicotine + IR group. Conversely, treatment with flavone hindered this inflammatory response by suppressing the NF κB mRNA expression and consequently reducing the levels of the TNF-α as well (Figure 7). Effect of flavone on inflammatory markers in lung tissues. (A): NF-κB and (B): TNF-α. Columns with different letters are significant (p < 0.05) while those with similar letters are non-significant.
Impact of flavone on FoxO1, caspase-1 and NLRP3 expression in lung tissue
Additionally, the PI3K/AKT signal pathway is the regulator of FoxO1 which has a key role in the inflammatory reaction. The interaction between the activated NF-κB and FOXO1 provoked the production of the pro-inflammatory mediators such as NLRP3, which in turn stimulate caspase-1 activation and eventually mediate pulmonary injury. As shown in Figure 8, our data exhibited that intoxication with nicotine and exposure to IR remarkably upregulated the gene expression of FoxO1, Caspase-1 and NLRP3 in the lung tissue as compared to the control group. However, rats supplemented with flavone showed a significant suppression in the expression of these genes. Effect of flavone on the mRNA expression of FoxO1, Caspase-1 and NLRP3 in the lung tissue. (A): FoxO1, (B): Caspase-1 and (C): NLRP3. Columns with different letters are significant (p < 0.05) while those with similar letters are non-significant.
Histopathological changes
The photomicrographs of the lung tissues of the control group and Flavone group showed normal lung architecture, the airspaces were separated by fine delicate inter-alveolar septa and clear alveolar sacs with regular air sacs, normal vasculature with scant perivascular connective tissue, folded columnar epithelial cells of bronchi and bronchiole, and normal fibrous tissues distribution score 0 (Figure 9(a) and (b)). Conversely, rats intoxicated with nicotine and exposed to IR showed disruption of the normal lung architecture manifested by multiple focal areas of collapsed lung alveoli with marked thickening of the inter-alveolar septa and hyperplasia of the bronchial epithelial lining. Numerous numbers of mononuclear and inflammatory cell infiltration of the alveolar wall and peribronchial areas were seen score 4 (Figure 9(c)). On the other hand, lung sections of rats treated with nicotine + IR + Flavone showed marked improvement in comparison with the previous group evidenced by mild damage of lung tissues, few inflammatory cell infiltrations of the alveolar wall along with reducing thickening of the alveolar wall score1 (Figure 9(d)). Photomicrograph of lung tissue section (A and B) showing normal lung architecture with regular air sacs and airspaces, thin inter-alveolar septa and clear alveolar sacs, normal vasculature with scant perivascular peribronchiolar cells of the lung representing the control and Flavone group respectively. In the Nicotine + IR group (C): a marked disruption of the normal lung architecture, collapsed lung alveoli, thickening of the inter-alveolar septa, hyperplasia of bronchial epithelial lining and inflammatory cells infiltration of the alveolar wall and peribronchial were observed. (D): rats treated with nicotine + IR + Flavone showed marked improvement of the lung tissues with mild thickening of the alveolar wall and lower numbers of inflammatory cells.
Discussion
Oxidative stress associated with excessive environmental pollutants alters many signaling pathways which is involved in the pathomechanisms of various disorders. 17 Herein the Gene Expression Omnibus (GEO) datasets contain mRNA expression profiles of rats exposed to cigarette smoking (CS) in addition to DEGs analysis were applied to explore the molecular functions and signaling regulated by smoking. GO analysis via KOBAS servers 21 showed that DEGs were markedly enriched in molecular functions related to the oxidation-reduction process, inflammatory response, and immune response. Accordingly, the novelty of our study is to elucidate the influence and protective effect of flavone on PI3K/Nrf2 and FoxO1/NF-κB/NLRP3 signaling pathway-mediated inflammation and injury in the lung of rats intoxicated with nicotine and exposed to radiation.
Herein, nicotine coupled with exposure to γ-IR markedly triggered oxidative stress in the lung leading to impaired redox status, injury and disrupted signaling pathways. Hanania et al. 26 indicated that the cytotoxic effect of radiation on lung tissue was attributed to their high radiosensitivity as well as the generation of ROS triggering oxidative stress, vascular damage, and inflammation which consequently results in alveolar walls edema, vascular permeability and thickening of interalveolar septa. 27 Moreover, the histopathological examination showed that ROS derived from nicotine and IR was associated with disruption of the normal lung architecture manifested by multiple collapsed lung alveoli with marked thickening of the inter-alveolar septa and hyperplasia of bronchial epithelial lining besides fibrosis of the lung parenchyma and inflammatory cells infiltration of the alveolar wall.28,29 In line with our results, Cha et al. 30 indicated that ROS generated from nicotine modified the cellular membrane permeability besides disrupting the antioxidant defence system which was evidenced by the elevation of lipid peroxidation (MDA) accompanied with the depletion of the antioxidant components (GSH content and GPx activity) reflecting lung tissues damage.
Based on the aforementioned results, it was found that the sustained oxidative stress was associated with the depletion of the antioxidant defence system and diminished response to excessive ROS leading to suppression of Nrf2 which is a major redox cellular homeostasis transcription factor maintaining cellular antioxidant defence mechanism, 31 detoxifying xenobiotics and pollutants 32 and protecting many organs such as the lungs against impaired redox status and oxidative damage. 33 our results agree with many previous results which showed that lung injury was associated with inhibition of the Nrf2.34,35
In addition to its role in cell proliferation, differentiation and apoptosis, PI3K/Akt signaling pathway promotes antioxidant, cytoprotective and cell survival mechanisms via activation of the Nrf2 signaling pathway against oxidative stress-induced injury. 36 Considerably, in response to the impaired redox balance and blunted antioxidant capacity, a significant decrease in the protein levels of the PI3K alongside that of its downstream Nrf2 was observed. The current results coincide with many studies indicating the suppressive potential of cigarette smoke and nicotine on Nrf2 or its upstream regulator PI3K in lung tissues.37–39
Previous data indicated that the PI3K/Akt and Nrf2 signaling pathways regulate cellular homeostasis and defence system versus inflammatory and oxidative damage 40 due to the crosstalk with NF-kB signaling pathway. 41 Cha et al. 30 indicated that the deactivation of Nrf2 with respect to impaired redox status and an insufficient response to oxidative stress promote redox-sensitive proinflammatory signaling in lung tissues. This was attributed to the competition between both transcription factors for the binding DNA site of cAMP- response-element-binding-protein (CREB)-binding protein (CBP), leading to the upregulation of NF-κB. 35
Collectively, the excessive ROS resulting from exposure to both toxin nicotine and γ-IR act synergistically as inflammatory signaling molecules in the lung to stimulate alveolar macrophages and neutrophils42,43 as well as many pro-inflammatory transcription factors (NF-κB due to the proteasomal degradation of IκBα 35 and protein (NLRP3) leading to upregulation of the pro-inflammatory cytokine TNF-α coupled with infiltration of many inflammatory cells and edema which ultimately induced lung tissue damage and injury.9,44 Accordingly, our results are in line with previous data indicating that nicotine activated NF-κB which aggravates the release of the pro-inflammatory cytokines TNF-α in lung tissues.45,46 Additionally, exposure to radiation (X-ray) promoted pulmonary inflammation via the excessive production of ROS causing the recruitment of inflammatory cells, activation of the NF-kB pathway and eventually the release of pro-inflammatory cytokines such as TNF-α.47,48 Lu et al. 40 and Wang et al. 49 exhibited the involvement of NLRP3 inflammasome in the pathogenesis and progression of the inflammatory diseases of the lung whereas it was upregulated subsequent to the activation of the NF-κB. The activation of the NLRP3 was associated with another inflammasome component caspase-1 which in turn aggravates the inflammatory response mediating diverse lung damage.50,51
The existing inflammation boosted the activity of FoxO1 which is a transcription factor involved in the regulation of the inflammatory response via transcriptional regulation, and signal transduction besides its role in maintaining tissue homeostasis and regulation of many cellular processes (proliferation, differentiation apoptosis and oxidative stress.52,53 The increased activity of FOXO1 along with the activated NF-κB synergistically enhances the expression of pro-inflammatory mediators NLRP3 and IL-1β. Meanwhile, the inflammatory signaling pathway FOXO1/NF-κB/NLRP3 is negatively regulated by the PI3k/Akt signaling pathway. 10 Moreover, both FOXO and Nrf2, downstream factors of the PI3K/Akt signaling pathway, 54 are two major conflicting pathways against cellular oxidative stress. 55
Conversely, Alsemeh and Abdullah 56 indicated that the activation of the PI3K/Akt signaling pathway has a crucial role in alleviating lung tissue toxicity and injury. This was attributed to boosting the function of alveolar sodium channels and Na+-K+-ATP, consequently eliminating excess edema fluid in the alveoli. 40 Furthermore, the activation of the Nrf2 signaling subsequent to the activation of the PI3K/Akt signaling pathway activation resulted in stimulation of Nrf2-mediated antioxidant/phase-II detoxification enzymes thus, restoring the cellular homeostasis by the attenuation of the impaired redox status and enhancement of the antioxidants system.57,58 Notably, Li et al. 59 and Zhao et al. 60 reported that Nrf2 activation clearly suppressed the inflammatory response and hampered the release of the pro-inflammatory mediators by suppressing many inflammatory signaling pathways such as NF-κB suggesting the anti-inflammatory effect of Nrf2. Additionally, the inhibition of the NF-κB transcriptional activity hindered the inflammasome components (NLRP3, caspase-1), therefore alleviating lung injury. 61 Moreover, Fu et al. 34 and Cui & Zhang 62 indicated that activation of the PI3K/Akt/Nrf2 signaling pathway alleviated the pulmonary oxidative damage and inflammation via the downregulation and inhibition of the FoxO1 as well as NLRP3-Caspase1 inflammasome in LPS induced lung injury.
Consistent with the aforementioned results, our study revealed that treating rats intoxicated with nicotine and γ-IR with the natural product flavone significantly attenuated the impaired redox status by minimizing the lipid peroxidation contents and aggravating the antioxidant defence system (Nrf2, GSH and GPx). The powerful antioxidant effect of the flavone as one of the flavones was appointed to its chemical structure whereas the presence of Chromone (1-benzopyran-4-one) backbone (2,3-double bond in conjugation with 4-keto functional group provides electron delocalization from the ring B and the electron-donating groups on the ring B reduce the O–H bond dissociation energy) provoking scavenging of the various free radicals either reactive oxidative or nitrosative species owing to the rapid hydrogen transfer, hindering lipid peroxidation 63 alongside with chelating metals ions and impeding various redox reactions. 64 Moreover, it modulates the Nrf2 pathway which in turn alleviates oxidative stress 65 together with modulating various oxidative stress-related processes by hampering the activity of central free radical-producing enzymes as well as enhancing the intracellular levels of antioxidant enzymes thus preventing the subsequent damage to the cellular biomolecules such as lipids, proteins, and DNA. 66 Interestingly, the hydrophilic and lipophilic fragments in flavone potentiated its antioxidant activity and maintained the cellular membrane’s structure and functions along with abolishing the effects of the harmful molecules through its partitioning in the hydrophobic core of the cellular membrane lipid bilayers and formation of hydrogen bonds between the polar groups of the lipids and the hydrophilic fragments of flavones. 67 Another prominent role of flavone is radioprotection whereas Singh et al. 68 showed that flavones have a photo-protective activity by which they can protect normal living cells from the deleterious effects of radiation through quenching the free radicals and absorption of UV radiation.
Several studies have indicated that the anti-inflammatory effect of flavone as one of the flavonoids was attributed to the activation of the Keap1-Nrf2 signaling pathway, repression of several inflammatory enzymes activities (cyclooxygenase and lipoxygenase activities) 69 in addition to blockage of various signaling pathways such as Toll-like receptor (TLR)/NF κB axis and NLRP3, consequently blunting the excessive release of the pro-inflammatory mediators (TNFα and IL-1β) and inflammatory metabolites. Thus, diminishing the cell damage and mitigating the inflammatory response.19,70 Additionally, Geraets et al. 71 reported that flavone alleviated NF-κB- derived inflammatory response in a Chronic Obstructive Pulmonary Disease (COPD) model. This was confirmed by Rudrapal et al.72,73 who exhibited the protective roles of dietary polyphenols against CS-induced inflammation-mediated chronic disorders such as COPD and other lung diseases indirectly by activating endogenous defense systems and modulating cellular signaling pathways, such as NF-κB activation, glutathione biosynthesis, the PI3-kinase/Akt pathway, and Nrf2 translocation into the nucleus.
Furthermore, Tianzhu et al. 74 and Lim et al. 75 exhibited the suppression of the NLRP3 inflammasome and its components (caspase-1 and inhibition of IL-1β) by flavonoid (apigenin) which belong to the flavone class therefore alleviating inflammatory diseases related to the NLRP3 inflammasomes and the supplementation with flavones minimized the treatment period in inflammatory diseases. 76 This was attributed to their potential for free radical scavenging, signaling pathways modulation, and inhibiting the inflammatory enzymes. 77
Our study has some limitations. First, the sample size was not calculated. Second, BALF samples were not aspirated for further analysis in addition to the lack of using a siRNA or an inhibitor of PI3K/Nrf2. Therefore, further studies are required as an extensive in-depth analysis regarding the molecular proteins PI3K/Nrf2 and FoxO1/NLRP3 inflammasome to elucidate the mechanism of protective effect of flavone in the future along with this study.
Conclusion
The current results manifested that flavone mitigates lung injury by attenuating the disrupted redox balance and aggravating the antioxidant content via activation of the PI3K/Nrf2. Moreover, flavone alleviates lung inflammation by inhibiting the inflammatory signaling pathway FOXO1/NF-κB/NLRP3. Consequently, flavone may be used to alleviate oxidative stress and inflammation-mediated lung injury in rats.
Footnotes
Acknowledgements
The authors are grateful to Prof. Dr Ahmed Osman (Prof. Of Pathology, Faculty of Veterinary Medicine, Cairo University) for his help in histopathology. We also thank Dr Ahmed Hammad (Radiation Biology Research Department, National Centre for Radiation Research and Technology, Egyptian Atomic Energy Authority) for his help in the bioinformatic analysis.
Authors’ contributions
The authors participated in the study’s design, conception, and implementation as well as the experimental component, investigations and findings analysis, and manuscript writing. All authors read and approved the manuscript for publication.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical statement
Data availability statement
All data obtained from this study are included in the current manuscript.
