Abstract
Introduction
The incidence of diabetes shows a rising trend in the developing countries during the past 30 years 1 It estimates that the number of diabetics will have increased to almost 600 million until 2035 around the world. 2 Diabetic foot is one of the feared complications of diabetes, which can cause diabetic disability and even death. Diabetic foot ulcers (DFUs) are one of the leading causes of hospitalization for people with diabetes.3,4 It was estimated that 15%–25% of people with diabetes may develop a DFU in their lifetime. 5 Data from a multi-center survey shows that the proportion of DFUs complicated by infection accounts for about 70% in China. 6
Diabetic foot ulcer infection (DFI) is a frequent complication of diabetes, which translates to a significant burden on healthcare systems. 7 Leading factors related to DFI are neurological, ischemic and mixed lesions, including severe clinical manifestations such as ulcers, osteomyelitis, infection and gangrene. 8 In fact, the concerning infections’ aetiology is usually complicated. The main management of DFI involves empirical antibiotic. Two additional factors leading to treatment failure are non-accurate diagnosis of the infecting pathogen relying on swab cultures instead of bone biopsy cultures and inappropriate initial surgical management.9,10 However, the increasingly prevalence of antibiotic-resistant pathogens is one of the greatest challenges, representing limited efficacy antimicrobial agents to treat DFI. 11 Moreover, the overuse of existing antimicrobial agents, long use of multiple broad-spectrum agents was responsible for the prevalence and spread of multidrug-resistant (MDR) bacteria. 12 DFI due to Gram-negative rods (GNRs) MDR, especially Klebsiella pneumoniae, Escherichia coli, Pseudomonas aeruginosa, and Acinetobacter baumannii are associated with increased morbidity and mortality and prolonged hospitalization. 13 In particular, MDR strains of Gram-positive cocci (GPCs) , Staphylococcus aureu, have emerged as particularly serious concerns. 14 All these pose a serious threat to global public health in treating DFI owing to the limited treatment options available.15,16
Increasing evidence indicates that the varying microbiological profiles of DFI were associated with the type and severity of the infection. Early identification of the type and drug resistance of pathogenic bacteria in DFI is helpful for clinical selection of highly sensitive antibiotics for treatment, timely control of infection, improvement of the prognosis, and effective reduction of amputation rate and mortality of patients.17,18 Moreover, there is a paucity of data with regard to the MDR pathogens in this hospital. Hence, this study analyzed the distribution of pathogenic bacteria in DFI and the characteristics of their resistance to antibiotics, in order to provide a reference for the rational selection of antibiotics in clinical practice and better guide treatment.
Materials and methods
General data
We prospectively collected all patients with DFU admitted to our hospital from January 2018 to January 2020. The inclusion criteria were as follows: (1) diagnostic criteria for DFI are based on the standard stipulated by International Working Group on the Diabetic Foot (IWGDF). DFI was based on the 2011 IWGDF Guideline for the treatment of DFIs 19 ; (2) patients who had clear and comprehensive clinical medical records; (3) patients who agreed to take part in the research project. The exclusion criteria were patients with previous amputation. Ethical approval for this study was obtained from the ethical committees of Beihua University (formal ethical approval number: 2018-01-01), and written informed consent was obtained from all subjects before the study.
Diagnostic criteria
MDR bacterial infection was defined based on pathogen resistance to three or more different types of antibiotics. 20 DFI also termed as lower extremity infection, or referring to infection in the area below the ankle (excluding the ankle). 21 Diabetic patients with DFU and DFI were graded from 0 – five according to the reference. 22 The patients with osteomyelitis were diagnosed based on the combination of the probe-bone, plain X-ray and magnetic resonance image results with inflammatory markers in serum. Besides, the patient’s severity of any DFU and DFI is sub-classified into the categories of mild, moderate and severe infection group according to the Infectious Diseases Society of America guidelines. 23
Data collection
The main data were collected from the department of endocrinology and metabolism computerized database in our hospital. All information included patients gender, age, type and duration of diabetes, prior history of ulceration (duration of ulcer, ulcer size), Wagner classification, with or without osteomyelitis, antimicrobial agents usage in 6 months prior to hospitalization. Besides, the physical examination and laboratory tests information included glycosylated hemoglobin, insulin use, fasting blood glucose, hyperlipidemia, hypertension. Other major chronic diseases including microvascular disease and macrovascular disease were recorded.
Collection of specimens and identification of etiological pathogens
Patients with suspected DFIs were obtained cultures before prescribing of empirical antibiotics on the first day of admission. All the specimens were collected from deep tissues with biopsy or curettage after cleaning and debridement of ulcera. Besides, the pus at the bottom of the ulcer was taken with a syringe for anaerobic culture. The obtained specimens were immediately sent to the laboratory of avoiding any delay. Culture in patients with DFI usually taken three times during one treatment course. No repetitive isolates from a single patient were included. Culturing, isolating and identification of etiological pathogens were performed by using the Vitek two Compact System with GN card and ASTGN13 card (bioMérieux, Marcy l’Etoile, France).
Antimicrobial susceptibility
Antimicrobial susceptibility to a panel of antimicrobial agents was determined by using the broth microdilution method according to Clinical and Laboratory Standards Institute recommendations (CLSI, 2012). 24 The MRSA and ESBL confirmatory tests were performed according the reference described previously.25,26 The tested antibiotics (AB Biodisk, Solna, Sweden) were included: ampicillin, oxacillin, cefazolin, cefotetan, cefuroxime, ceftazidime, ceftriaxone, cefepime, ciprofloxacin, levofloxacin, amikacin, gentamicin, vancomycin, tobramycin, polymyxin B, rifampicin, clindamycin, tetracycline, tigecycline, erythromycin, linozelid, imipenem, meropenem, aztreonam, ampicillin/azobactam, piperacillin/tazobactam, compound sulfamethoxazole. Reference strains in all tests were Escherichia coli ATCC 25922, Klebsiella pneumoniae ATCC 700603, Staphylococcus aureus ATCC 25923 and Pseudomonas aeruginosa ATCC 27853.
Statistical analysis
Statistical analysis was performed using SPSS22.0 statistical software with a threshold of p<0.05 that was considered to be statistically significant. The numerical data were analyzed using t-test, while categorical data was analyzed using chi square.
Logistic regression analysis was performed to investigate the risk factors of the patients with DFI.
Results
Clinical features of patients with DFIs
Clinical features of patients with DFIs in the study.
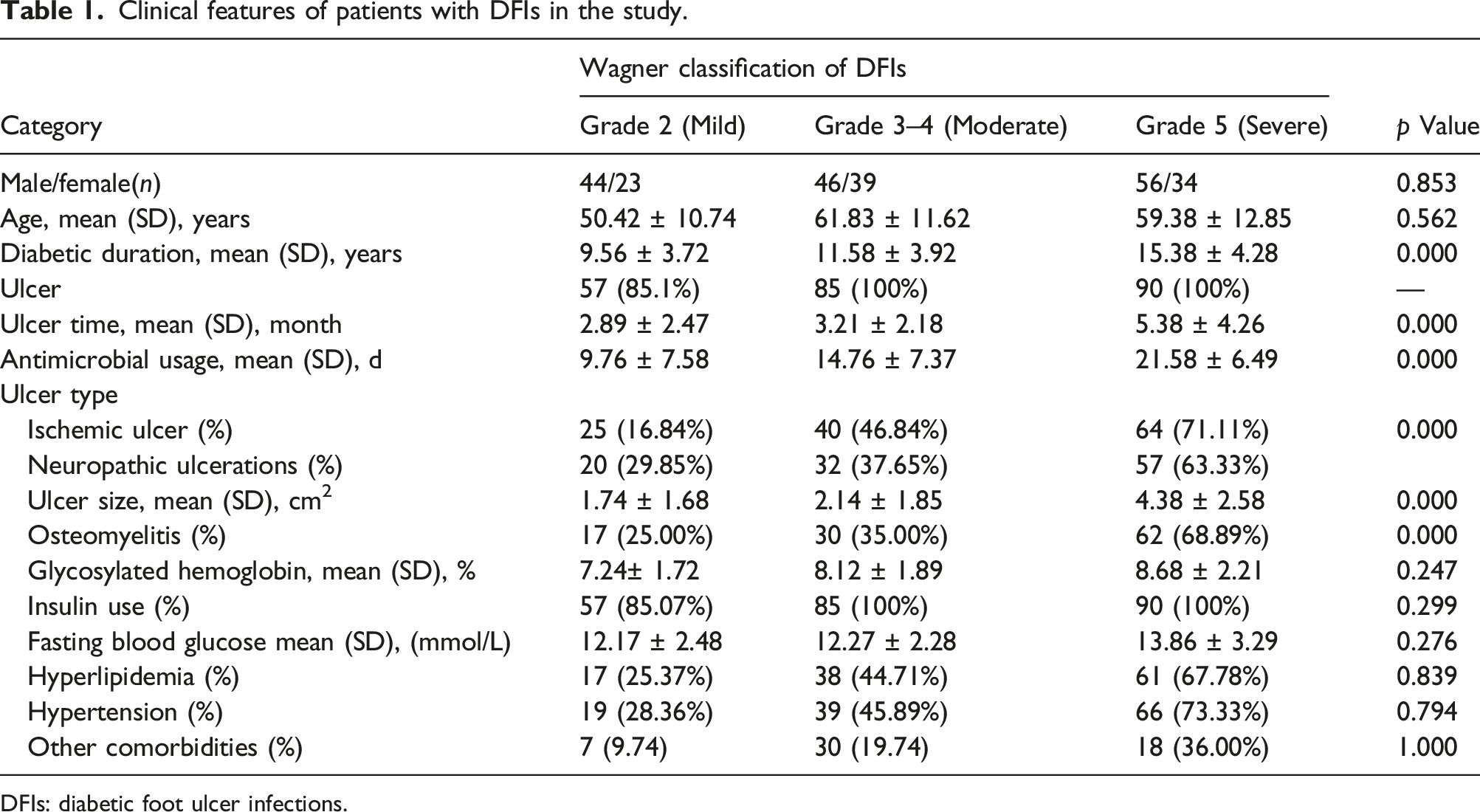
DFIs: diabetic foot ulcer infections.
Profiles of pathogens and antimicrobial susceptibility patterns
A total of 251 isolates were isolated from 242 patients samples, the total percentage of Gram-negative rods (GNRs).and Gram-positive cocci (GPCs) were 138 (54.92%) and 113 (45.02%), respectively. Among them, 138 strains of GNRs included Pseudomonas aeruginosa (56), Escherichia coli (28), Proteus mirabilis (16), Klebsiella pneumonia (16), Acinetobacter baumannii (10), Enterobacter aerogenes (7) and Enterobacter cloacae (5) ; whereas, 113 strains of GPCs included Staphylococcus aureus (50), Staphylococcus epidermidis (23), Staphylococcus haemolyticus (15), Enterococcus faecalis (12), Streptococcus agalaciate (8) and Streptococcus equismilis (5). The most common isolates were Pseudomonas aeruginosa and Staphylococcus aureus, accounting for 22.37% and 19.74%, respectively. Notably, no fungi isolates were identified from all patients.
Generally, the resistance rates of GNRs and GPCs showed an increasing tendency along with the DFIs severity among three groups. In the mild infection group, the resistance rates of GNRs to cefuroxime (10.7%), cefotetan (11.8%), ceftriaxone (12.3%), cefepime (12.3%), and ceftazidime (15.7%) increased to cefuroxime (30.7%), cefotetan (31.8%), ceftriaxone (37.8% ), cefepime (12.3%) and ceftazidime (32.4%) in the severe infection groups (Figure 1). Similarly, the resistance rates of Staphylococcus aureus to ceftriaxone (16.8%), ceftazidime (22.3%) and cefuroxime (17.8%) increased to ceftriaxone (36.7%), ceftazidime (39.3) and cefuroxime (37.8). The antibiotic sensitivity patterns indicated that tigecycline for GNRs and vancomycin for GNCs except for MRSA isolates had good sensitivity, respectively (Figure 2). Comparison with the resistance rates of GNRs showing an increasing tendency among three groups. Comparison with the resistance rates of GPCs showing an increasing tendency among three groups.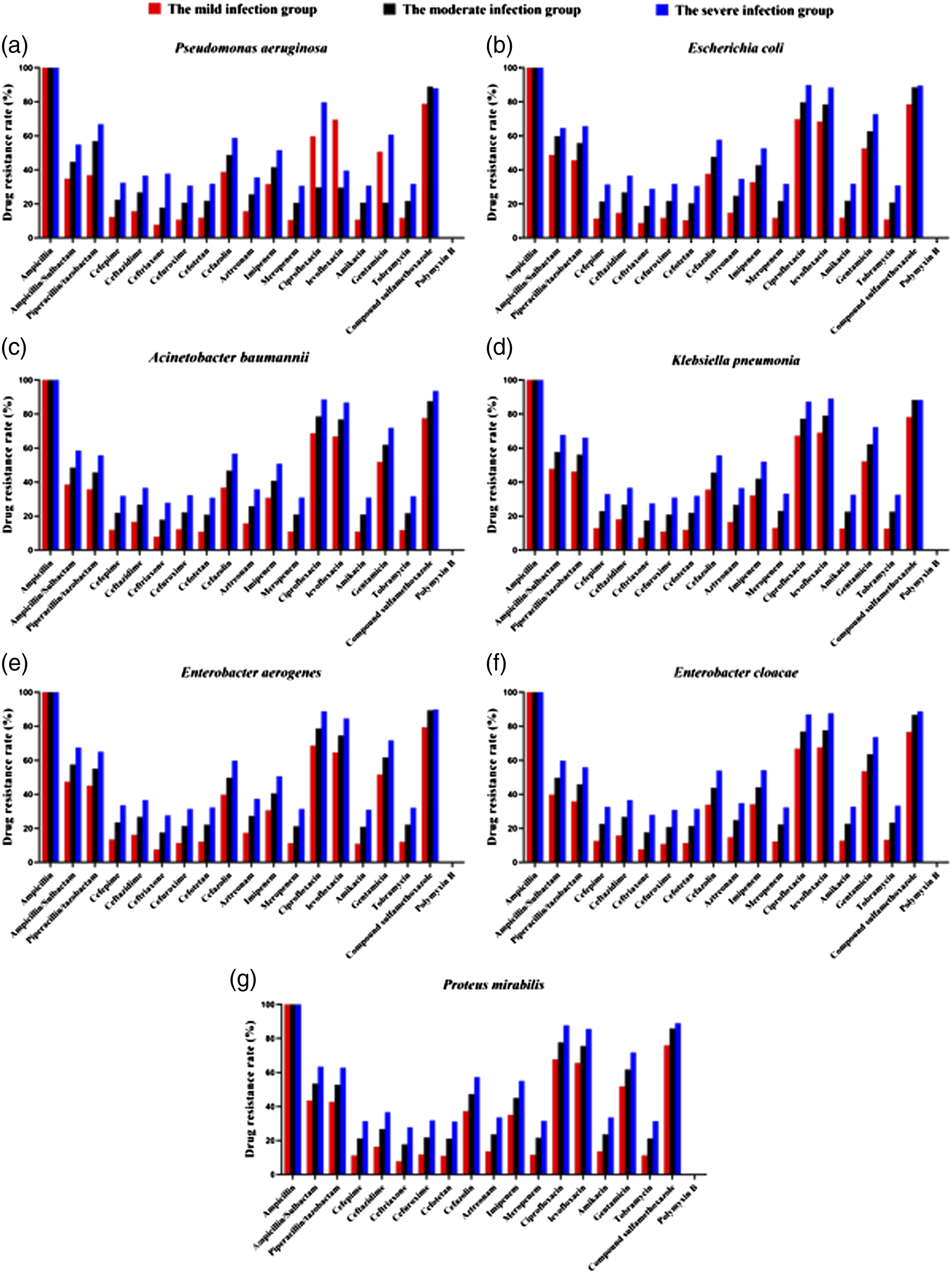

Out of all isolates, 152 (60.56%, 152/251) isolates were confirmed to be MDR from 11 patients in the mild infection group, 29 patients in the moderate infection group and 50 in the severe infection groups. Among them, 64 strains were GPCs and 88 strains were GNRs (Figure 3). Distribution of MDR isolates among three groups.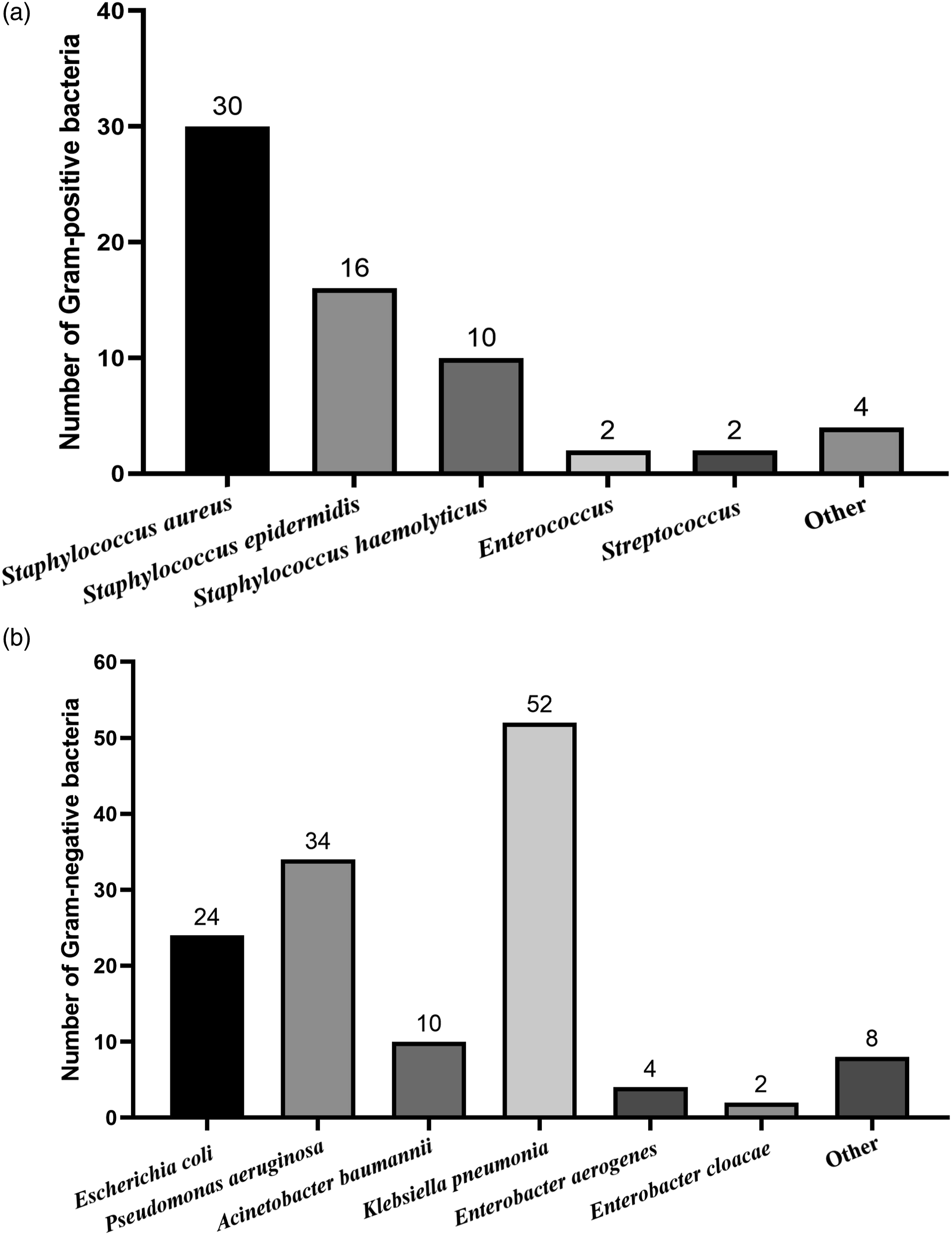
In the study, 47 (34.1%) isolates of GNRs were ESBLs producers and 15 (30%) isolates of Staphylococcus aureus were MRSA producers. And even one isolates of MRSA was confirmed to be vancomycin-resistant Staphylococcus aureus (VRSA). Additionally, no polymycin-resistant GNRs and vancomycin-resistant enterococci (VRE) strains were found, however, 15 of vancomycin-resistant S. aureus isolates were found.
Risk factors for MDR in patients with DFIs
Logistic regression analysis of DFIs association with risk.
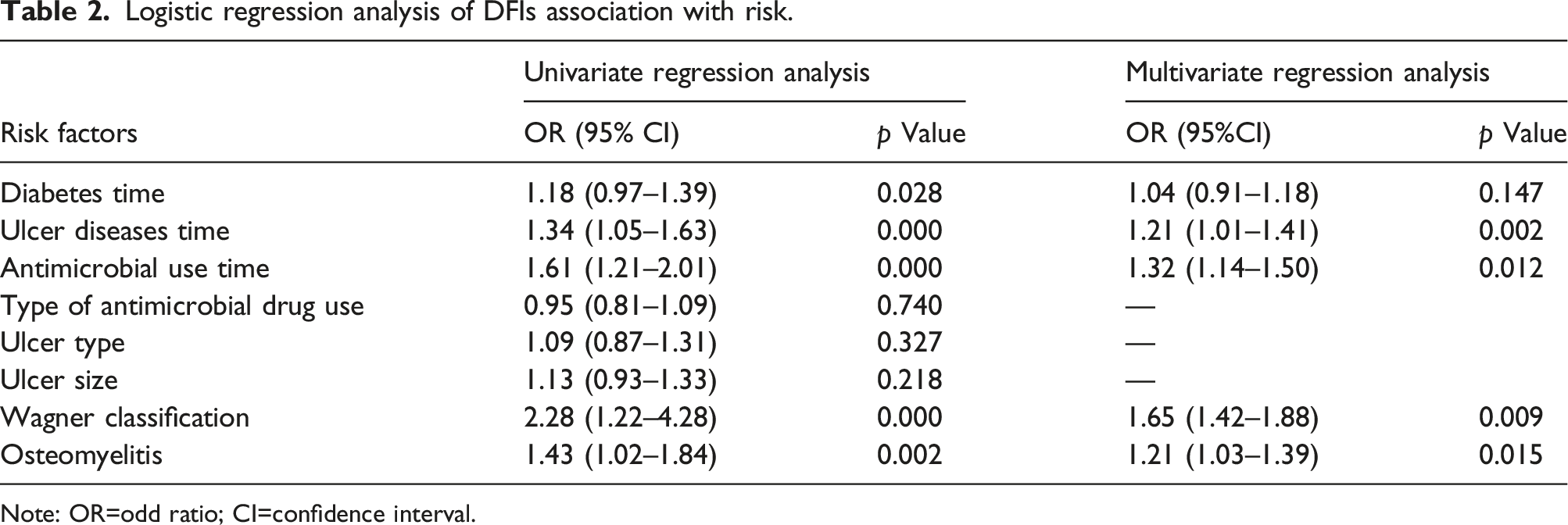
Note: OR=odd ratio; CI=confidence interval.
Discussion
Our study was a cross-sectional and retrospective study. All participants had diabetic complications in addition to long diabetes duration. Compared with mild infection group, the patients were apt to suffer from malnutrition in going diabetes duration. This was reported in a previous study by Wu et al. and Jiang et al.27,28 This suggests that it is essential to rectify the patients with malnourished state in a timely manner.
In this study, the most common GNR was Pseudomonas aeruginosa, followed by Escherichia coli, Acinetobacter baumannii, Klebsiella pneumonia, Enterobacter aerogenes. The most common GPC was Staphylococcus aureus, followed by Staphylococcus epidermidis, Staphylococcus haemolyticus and Enterococcus. This finding was similar to that of a reporter from South India, which explored bacterial etiology of DFIs. 29 A numbers of studies have reported that GNRs were the predominant microbes isolated from DFIs. However, some studies showed the contrary results along with different regions in China. Feng et al. 13 reported that in 559 pathogens from 429 ulcer specimens, 59.0% were Gram-positive and 37.7% were Gram-negative. Whereas, Wu et al. explored the distribution of microbes and drug susceptibility in patients with diabetic foot infections in southwest China, demonstrating that GNRs were the predominant bacteria (51.0%), and GPCs accounted for 36.9%, and 12.1% of fungi were observed. 30 Our results were in agreement with existing studies.1,6
Recent studies revealed that the etiology variation of pathogens distribution in patients with DFIs. 31 It was noteworthy that MRSA/Staphylococcus aureus and ESBLs-producing Enterobacteriaceae were more and more commonly isolated from patients with DFIs. 32 In this study, the presence of MRSA was also reported in 15 (30%) isolates of GPCs and ESBLs producers in 47 isolates of GNR. Moreover, one VRSA strain was also found. The prevalenc of VRSA is extremely warring. Fortunately, the isolation and proper hygiene methods were adopted to block the dissemination into other settings in the hospital.
Factors associated with the major changes included: long-time or repeated hospitalization, cross-infection, the inappropriate usage of antibiotics, advanced age, osteomyelitis, severity of the ulcer, and so on. MRSA and ESBLs-producing bacteria infections prolong ulcer healing times and hospitalization times, resulting in treatment failure and even increasing the emergence of MDR. 33 A studies of Mendes et al. showed that 24.5% of patients with DFI developed methicillin-resistant Staphylococcus aureus. Mu et al. found that the infection rate of patients with diabetic foot disease increased to 85.50%. A total of 754 strains of bacteria were cultured from 444 patients with DFD, of which 112 were MDR bacterial infections in China. 14 Our findings showed that the 60.56% isolates were confirmed to be MDR in the region, thus requiring more attention. They had the same MDR spectrum to at least three antibiotics.
Clinically, broad-spectrum antibacterial drugs are often long-term used which increases the risk of multi-drug resistant bacterial infections in patients with DFIs.32,23 Our results demonstrated that the resistance rates of GNRs and GPCs showed an increasing tread along with the DFIs severity among three groups, and GNRs were the most common MDR in DFIs. Clinicians should pay particular attention to strengthening the microbial culture when using antibiotics to treat MDR in DFIs. Appropriate antibacterial drugs should be utilized according to the results of drug susceptibility. Besides that, we systematically analyzed the clinical features, risk factors. In addition, we found that a long duration of diabetes, long duration of ulcer, long-term use of antibacterial drugs, ischemic ulcers, large ulcer wounds, increased Wagner grade and osteomyelitis were associated with MDR infections in patients with DFI. Logistic regression analysis indicated that duration of ulcer, duration of antibiotic use, Wagner grade and osteomyelitis were risk factors for MDR bacterial infection in patients with DFIs. Worthly to note, osteomyelitis is a common and serious complication in the process of DFIs. Once the patients have osteomyelitis, they are prone to difficulty of cureation, and even threatening the patients of death. For such patients, debridement and drainage through surgery should be performed to reduce drug resistance and to improve the clinical prognosis. 34
There are some limitations in the study. Firstly, the study was a cross-sectional in a single-center, and the sample size was small. Secondly, genotypic or molecular data of all MDR isolates were not documented and the survey on molecular epidemiology was not performed. Further studies are needed to conduct a multicenter study and follow-up study of the participants. Besides, we need confirm the genetic types of all MDR isolates and the mechanism of transmission.
Conclusion
The predominate pathogens of patients with DFI were GNRs, followed by GPCs in the region. They exhibited the different antibiotic resistance phenotypes. As the patient’s severity of DFI along with Wagner classification increases, the MDR infections tend to be more seriously. The data provided advice on selection of highly sensitive antibiotics in the management of DFIs.
Footnotes
Acknowledgement
We thank anonymous reviewers for excellent criticism of the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China project (81402979), the Jilin Science and Technology Development Program (No. 20190905006SF; JJKH20220076KJJ).
