Abstract
Oxymatrine (OMT), a natural quinolizidine alkaloid, has been known to have anti-inflammation, anti-anaphylaxis, and chemopreventive effects on various cancer cells. To clarify the underlying role and molecular mechanisms of OMT in human hemangioma (HA), in the present study, we examined the expression of hypoxia-inducible factor-1a (HIF-1a) and vascular endothelial growth factor (VEGF) in different phases of human HA. After HA derived endothelial cells (HDEC) were pretreated with different concentrations of OMT, cell proliferation, apoptosis, and cycle distribution were evaluated by MTT assay and flow cytometry analysis, respectively. The effects of OMT on expression of HIF-1a signaling were determined by real-time PCR and western blot assays. Our results showed that, the expression of HIF-1a and VEGF was significantly increased in proliferating phase HA, but decreased in involuting phase HA. Moreover, OMT in vitro inhibited proliferative activities and induced cell apoptosis and cycle arrest in G0/G1 phase in HA cells with decreased expression of HIF-1a, VEGF, Bcl-2, and CyclinD1, and increased expression of p53. Taken together, our findings suggest that, the expression of HIF-1a and VEGF is increased in proliferating phase HA, and OMT suppresses cell proliferation and induces cell apoptosis and cycle arrest in proliferative phase HA through inhibition of the HIF-1a signaling pathway, suggesting OMT may provide a novel therapeutic strategy for the treatment of HA.
Introduction
Hemangiomas (HAs), the most common benign tumors, are characterized by an aberrant growth of endothelial cells. The life cycle of HAs differs from that of most tumors in that it progresses from a phase of rapid proliferation followed by spontaneous involution. 1 Most HAs do not require any treatment because they spontaneously regress over the years. However, in a minority of patients, HAs are life-threatening and exert functional impairment or severe systemic complications. 2 Treatment for these aggressive HAs represents an unmet clinical need.
Oxymatrine (OMT), an active component of Chinese traditional medicine, has a great effect on anti-inflammation, anti-arrhythmia, and anti-fibrosis of tissue cells. 3 But current evidence indicates that OMT plays an important role in anti-tumor via inhibiting telomerase activity and inducing G1/G0-phase arrest in colon cancer cells. 4 OMT reduces cell cytotoxicity probably through inhibition of the metabolic activation, and mediates cell division of cancer cells. 5 It triggers oxidative stress, results in the collapse of the mitochondrial transmembrane potential and in turn leads to cytochrome c release and apoptosis through the intrinsic caspase-9/caspase-3 pathway in melanoma cells. 6 Cancer stem cells (CSCs) play a critical role in cancer initiation and relapse as they are resistant to most cytotoxic agents and able to proliferate indefinitely. OMT inhibits CSC growth through inhibition of the Wnt/β-catenin signaling pathway, and represents a novel therapeutic drug for targeting breast CSCs. 7
Hypoxia induces transcription of a range of physiologically important genes including vascular endothelial growth factor (VEGF), which is mediated by hypoxia-inducible factor-1 (HIF-1). Importantly, immunohistochemical analyses demonstrate upregulation of HIF-1a expression in renal clear cell carcinomas, hemangioblastomas, 8 and nerve HAs, 9 and the expression of HIF-1a is associated with angiogenesis, invasion, and progression of brain tumors. 10 However, HIF-1a-mediated effects of OMT on proliferation and apoptosis of HA cells are not understood. In the present study, we investigated the expression of HIF-1a and VEGF in human HA. Through OMT pretreatment of HA cells, we observed the effects of OMT on proliferation and cell apoptosis of HDEC cells.
Materials and methods
Materials
The primary HDEC cells used in the experiments were exacted from human HA tissue cells. The primers of HIF-1a and VEGF were synthesized by ABI (CA, USA). All antibodies were from Santa Cruz Biotechnology (Santa Cruz, CA, USA).
Drugs and reagents
OMT was from Santa Cruz Biotechnology (Santa Cruz, CA, USA); Dulbecco’s Modified Eagle medium and fetal bovine serum were from Thermo Fisher Scientific Inc. (Waltham, MA, USA); TRIzol Reagent and Lipofectamine 2000 were from Invitrogen (Carlsbad, CA, USA); M-MLV Reverse Transcriptase was purchased from Promega (Madison, WI, USA); SYBR Green Master Mixture was from Takara (Otsu, Japan). ECL-PLUS/Kit was from GE Healthcare (Piscataway, NJ, USA). Cell cycle analysis kit, apoptosis kit, RNase A, and Annexin V-FITC were from KeyGEN biology (Nanjing, PR China).
Tissue samples
Human HA samples were collected at the Department of General Surgery, and were classified according to Mulliken criteria. Tissues and clinical information were collected from Xinhua Hospital affiliated to Shanghai Jiao Tong University School of Medicine. There were 10 cases of proliferating phase HA and 10 cases of involuting phase HA. A portion of each tissue sample was fixed with 10% formalin for histopathological examination. All HA tissues were determined by two independent pathologists.
IHC staining
IHC staining for HIF-1a and VEGF using monoclonal antibodies was carried out. Unstained sections were deparaffinized and incubated overnight at 4°C with primary antibodies against the proteins, then with biotinylated secondary antibody (1:200) at room temperature for 1h, followed by incubation with ABC peroxidase and 3,3’ diaminobenzidine. Sections were counterstained with hematoxylin. Digital images were acquired and the integrated optical density (IOD) of semi-quantitative analysis was performed.
Cell culture and pretreatment
The proliferating phase HDEC were cultured in DMEM medium supplemented with 10% heat-inactivated FBS, 100 U/mL of penicillin, and 100 μg/mL of streptomycin, and were placed in a humidified atmosphere containing 5% CO2 at 37°C. Proliferating phase HDEC pretreated with different concentrations of OMT (0, 0.5 mg/mL, 1 mg/mL, and 2 mg/mL) were selected for further study.
Quantitative real-time PCR
To examine the mRNA expression levels of HIF-1a and VEGF in proliferating phase HDEC, real-time PCR was used. Total RNA of each clone was extracted with TRIzol according to the manufacturer’s protocol. Reverse-transcription was carried out using M-MLV and cDNA amplification was carried out using SYBR Green Master Mix kit according to the manufacturer’s protocol. Target genes were amplified using specific oligonucleotide primer and human glyceraldehyde-3-phosphate dehydrogenase (GAPDH) gene was used as an endogenous control. Three separate experiments were performed for each clone.
Western blot assay
The proliferating phase HDEC cells were harvested and extracted using lysis buffer (Tris-HCl, SDS, Mercaptoethanol, Glycerol). Separated protein bands were transferred into polyvinylidene fluoride (PVDF) membranes and the membranes were blocked in 5% skim milk powder. The primary antibodies against HIF-1a, VEGF, Bcl-2, p53, and CyclinD1 were diluted according to the instructions of antibodies and incubated overnight at 4°C. Then, horseradish peroxidase-linked secondary antibodies were added at a dilution ratio of 1:1000, and incubated at room temperature for 2 h. The relative protein level was normalized to β-actin concentration. Three separate experiments were performed for each clone.
Cell proliferation assay
HDEC proliferation was analyzed with the MTT assay. Briefly, HDEC treated with OMT were incubated in 96-well-plates with DEME medium supplemented with 10% FBS. HDEC were treated with 20 μL MTT dye at 48 h and incubated with 150 μL of DMSO for 5 min. The color reaction was measured at 570 nm with enzyme immunoassay analyzer (Bio-Rad, USA). The proliferation activity was calculated for each clone.
Cell apoptosis analysis
Proliferating phase HDEC were trypsinized, washed with cold PBS, and resuspended in binding buffer according to the instruction of the apoptosis kit. FITC-AnnexinV and PI were added to the fixed cells for 20 min at room temperature. Then, Annexin V binding buffer was added to the mixture before the fluorescence was measured on FACsort flow cytometer. Cell apoptosis was analyzed using Cell Quest software (Becton Dickinson, Mountain View, CA, USA). Three separate experiments were performed for each clone.
Cell cycle analysis
Proliferating phase HDEC were trypsinized, washed by PBS and fixed with 80% cold ethanol overnight at −20°C. After PBS washing, fixed cells were stained with PI in the presence of RNase A for 30 min at room temperature. Each sample was filtered through a 50 μm nylon filter to obtain single-cell suspension. The samples were analyzed on FACsort flow cytometer (Becton Dickinson, Mountain View, CA, USA). ModFit3.0 software (Verity Software House, Topsham, ME, USA) was used for cell cycle analysis. Three separate experiments were performed for each clone.
Statistical analysis
SPSS 20.0 was used for the statistical analysis. One-way analysis of variance (ANOVA) was used to analyze the differences between groups. The LSD method of multiple comparisons was used when the probability for ANOVA was statistically significant. Statistical significance was P <0.05.
Results
The expression of HIF-1a and VEGF in human HA
The expression of HIF-1a and VEGF was detected by IHC analysis. As shown in Figure 1a, the expression of HIF-1a and VEGF was increased in proliferating phase HA, but decreased in involuting phase HA. As indicated in Figure 1b, the average IOD of HIF-1a and VEGF in proliferating phase HA was significantly higher than that in involuting phase HA (**P <0.01).

The expression of HIF-1a and VEGF in human HA. (a) The expression of HIF-1a and VEGF was examined by IHC analysis. The expression of HIF-1a and VEGF was increased in proliferating phase HA, but decreased in involuting phase HA. (b) The average IOD of HIF-1a and VEGF was much higher in proliferating phase HA than that in involuting phase HA (**P <0.01).
The effect of OMT on HIF-1a expression
To assess the effect of OMT on HIF-1a expression in proliferating phase HDEC cells, the expression level of HIF-1a was measured by real-time PCR and western blot assays. As shown in Figure 2a, a significant decrease of HIF-1a mRNA expression was investigated in OMT-treated groups compared with the untreated group (**P <0.01). As for the protein expression, indicated by western blot assay, the expression level of HIF-1a was decreased in OMT-treated groups compared with the untreated group (Figure 2b, **P <0.01).
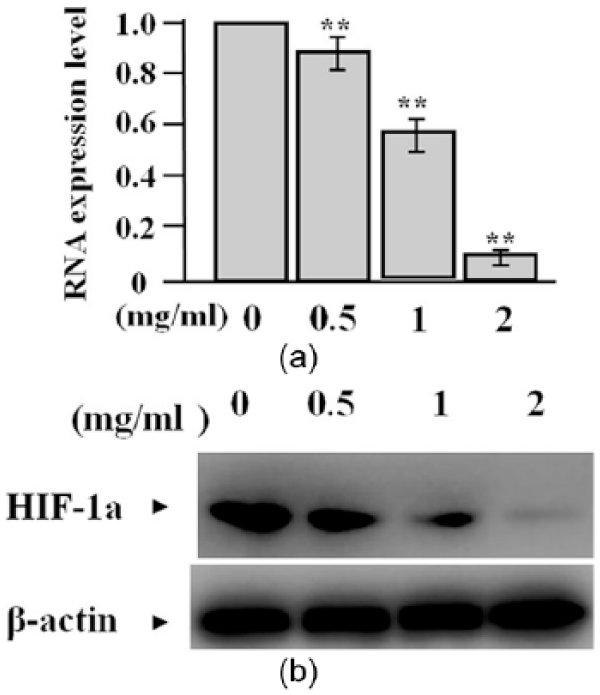
The effect of OMT on HIF-1a expression. (a) The expression level of HIF-1a mRNA, detected by real-time PCR, was significantly lowered in OMT-treated groups compared with the untreated group (**P <0.01). (b) The expression amount of HIF-1a protein, evaluated by western blot assay, was markedly decreased in OMT-treated groups compared with the untreated group.
The effect of OMT on cell proliferation
To confirm the effect of OMT on HDEC cell proliferation, we examined their proliferative activities by MTT assay. We found that OMT significantly reduced the proliferative activities of HDEC cells compared with the untreated group (**P <0.01) (Figure 3a). In addition, the effect of OMT on VEGF expression was evaluated by western blot assay. The amount of VEGF protein was significantly decreased in OMT-treated groups compared with the untreated group (Figure 3b).

The effect of OMT on cell proliferation. (a) MTT assay showed that proliferative activities of HDEC cells were attenuated in OMT-treated groups compared with the untreated group (**P <0.01). (b) The expression amount of VEGF, examined by western blot assay, was significantly reduced in OMT-treated groups compared with the untreated group.
The effect of OMT on cell apoptosis
To determine the effect of OMT on HDEC cell apoptosis, we carried out flow cytometry analysis indicating that apoptotic indexes were markedly elevated in OMT-treated groups compared to the untreated group (**P <0.01) (Figure 4 a, b). To decide the effect of OMT on the expression of p53 and Bcl-2, we further examined the expression levels of p53 and Bcl-2 by western blot assay, indicating that the amount of Bcl-2 protein was decreased but p53 was increased in OMT-treated groups (Figure 4c).
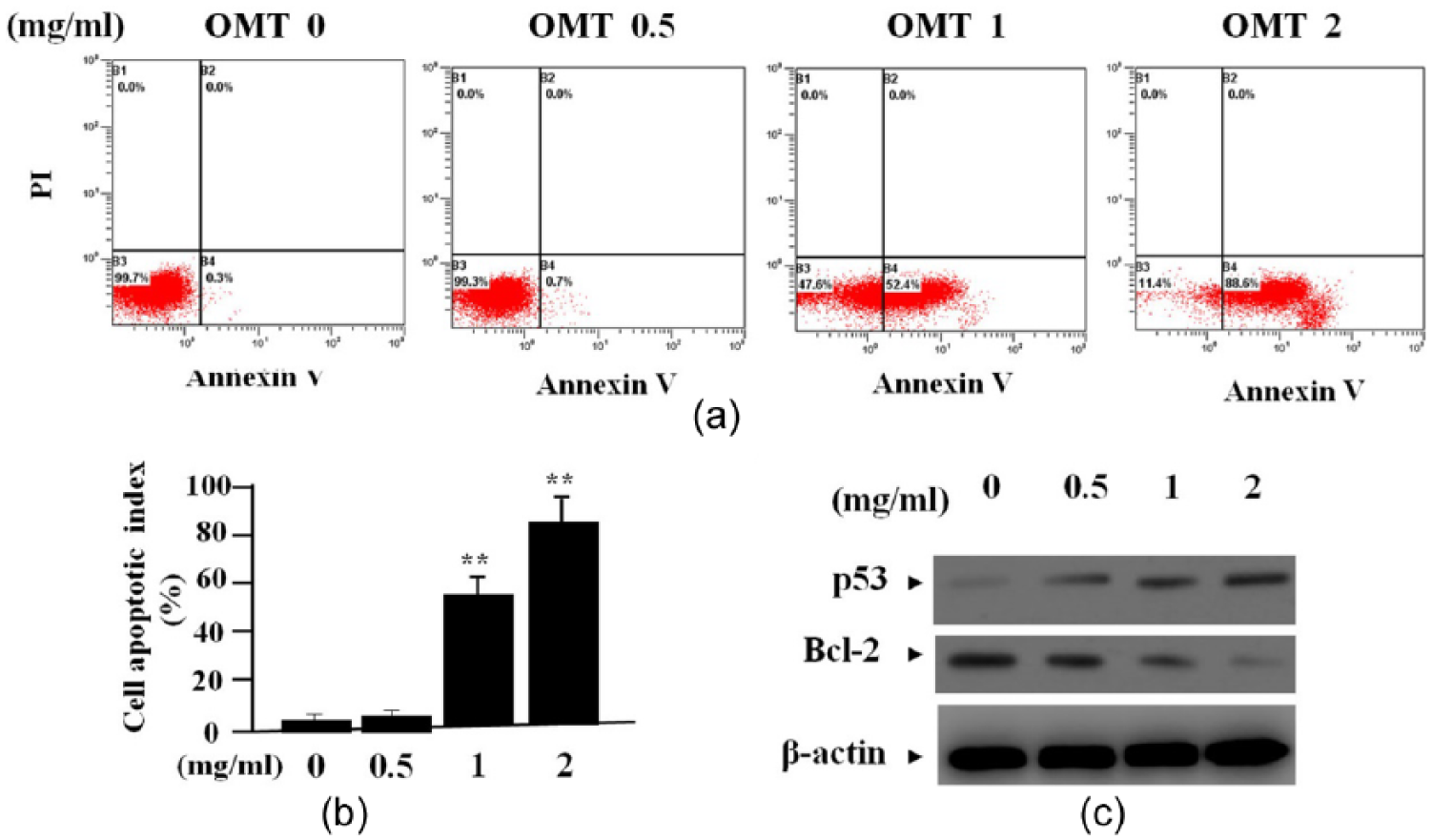
The effect of OMT on cell apoptosis. (a, b) Flow cytometric analysis indicated that apoptotic indexes of HDEC cells were obviously raised in OMT-treated groups compared to the untreated group (**P <0.01). (c) The expression of p53, examined by western blot assay, was upregulated while that of Bcl-2 was downregulated in OMT-treated groups compared to the untreated group.
The effect of OMT on cell cycle distribution
To verify the effect of OMT on cycle distribution of HDEC cells, we performed flow cytometry analysis, indicating that the G0/G1 phase fraction was increased, but S phase fraction was decreased, thus cell cycle was arrested in G0/G1 phase in OMT-treated groups compared with the untreated group (Figure 5 a, b). To determine the effect of OMT on cyclinD1 expression, we examined the expression level of CyclinD1 by western blot assay, indicating that cyclinD1 was significantly decreased in OMT-treated groups compared with the untreated group (Figure 5c).
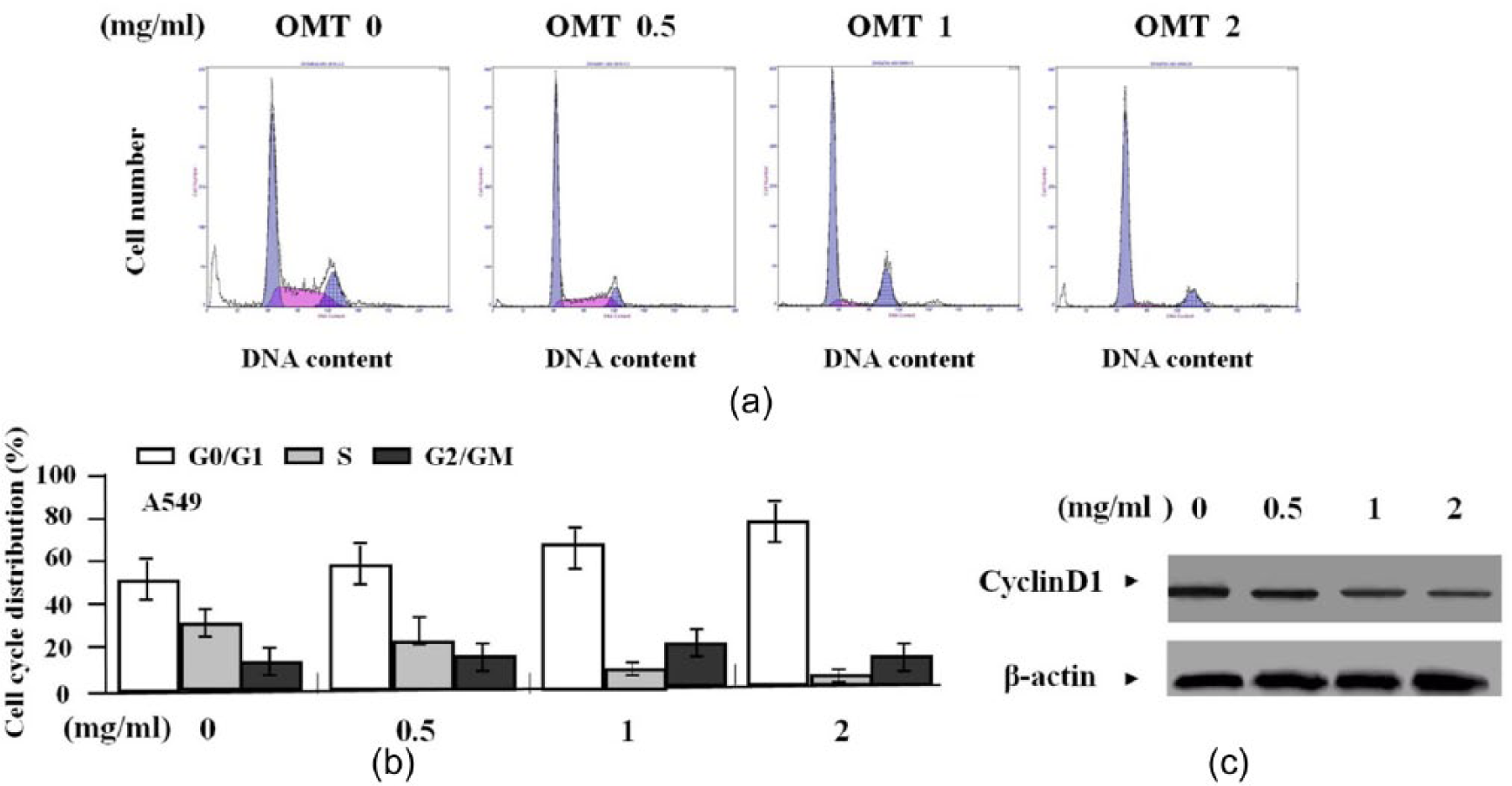
The effect of OMT on cell cycle distribution. (a, b) Flow cytometric analysis demonstrated that G0/G1 phase fraction was increased, but S phase fraction was decreased in OMT-treated groups compared to the untreated group (**P <0.01). (c) The expression level of CyclinD1, detected by western blot assay, was weakened in OMT-treated groups compared to the untreated group.
Discussion
The hypoxic environment leads to an upregulation of HIF-1a responsive chemokines including VEGF, known to promote the recruitment and proliferation of endothelial progenitor cells, and increase of HIF-1 promotes new blood vessel development, ultimately leading to the pathogenesis of infantile HAs.11, 12 Proliferating HAs express known mediators of vasculogenesis such as HIF-1a and VEGF, which are involved in the initiation or progression of Has. 13 Activation of HIF-2 alpha and consequent overexpression of VEGF by the endothelial cells is involved in the pathogenesis of Has. 14 In our study, the expression of HIF-1a and VEGF was increased in proliferating phase HA, but decreased in the involuting phase HA, suggesting that HIF-1a signaling might be implicated in the pathogenesis of HA. 15
OMT, a natural quinolizidine alkaloid, has cytotoxic and chemopreventive effects on various cancer cells, and inhibits the proliferation and induces apoptosis of human hepatoma cells mediated by a cycle arrest in G2/M and S phase through downregulation of bcl-2 and upregulation of p53. 16 OMT induces the apoptosis of esophageal carcinoma cells related to the upregulation of p21 and downregulation of p-ERK1/2 and CyclinD1, and contributes to apoptotic cell death of pancreatic cancer via regulation of Bcl-2 and IAP families, release of mitochondrial cytochrome c and activation of caspase-3. 17 But, whether OMT affects the HIF-1a signaling and hemangiomagenesis is uncertain. In the present study, OMT significantly reduced cell proliferation and induced apoptosis followed by blockade of HIF-1a signaling, and OMT combined with other agents such as NM-3 exerts synergistic inhibitory effect on cancer cells, 18 suggesting that OMT inhibition of HIF-1a signaling might provide a novel therapeutic strategy for the treatment of HA.
Bcl-2, a protooncogene product associated with inhibition of cellular apoptosis, is correlated with the proliferation and apoptosis of HA.19,20 Bcl-2 expression is increased, but p53 is decreased in HA. 21 Bcl-2 may be an important therapeutic target in the treatment of HA. 22 Overexpression of cyclinD1 is reported in many tumors including sporadic hemangioblastomas, and plays a critical role in control of the cell cycle. 23 CyclinD1 gene amplification has been verified to promote abnormal endothelial cell proliferation and angiogenesis in proliferating HAs. 24 Importantly, it has been shown that knockdown of HIF-1a downregulates Bcl-2 expression, but upregulates p53 expression in cancer cells.25,26 Our present study indicated that OMT decreased the expression of Bcl-2 and CyclinD1, and increased p53 expression, suggesting that OMT might participate in the antitumor effect of HA via HIF-1a-mediated downregulation of Bcl-2 and CyclinD1 and upregulation of p53 expression.
In conclusion, our findings demonstrate that the expression of HIF-1a and VEGF is increased in proliferating phase HA and OMT suppresses cell proliferation and induces cell apoptosis and cycle arrest in proliferative phase HA through inhibition of the HIF-1a signaling, suggesting OMT may represent a novel therapeutic strategy for the treatment of HA.
Footnotes
Declaration of conflicting interests
The author(s) declared that no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the Science and Technology Commission of Shanghai Municipality, PR China (No. 13140903802) and the Medicine and Engineering Cross Foundation of Shanghai Jiaotong University (No. YG2012MS33).
