Abstract
Objective
To examine whether perinatal factors are associated with low-grade inflammation measured by high-sensitivity C-reactive protein levels during adolescence.
Methods
Nested case-control study of 125 teenagers who were born by Cesarean delivery and had high-sensitivity C-reactive protein levels determined at 15–17 years. Data on obstetric and perinatal factors were recorded prospectively at the time of their birth.
Results
Median values of high-sensitivity C-reactive protein were significantly higher in teenagers, who were born as large for gestational age or with maternal diabetes compared to others (2.54 vs 0.34 mg/L; p < 0.024), and born during spring or summer compared to those born during winter or autumn (0.48 vs 0.27 mg/L; p < 0.023). No other perinatal associations were detected (for ex. such as electivity of operation, onset of labor, rupture of fetal membranes, cervical dilatation at delivery, gestational age, Apgar scores at 5 min, umbilical blood pH value, administration of neonatal antibiotics or need of neonatal intensive care treatment) in CRP levels. Further, teenagers with current body mass index in the highest tertile, regular medication for chronic disease and girls using oral contraceptives had significantly higher high-sensitivity C-reactive protein levels than others.
Conclusion
Prenatal exposures such as maternal metabolic environment and seasonality may have longterm effects on the low-grade inflammation and later cardiometabolic risks. Seasonality might be partly explained by maternal vitamin D levels during pregnancy, and thus future efforts are warranted to ensure sufficient vitamin D availability during pregnancy. Surprisingly, no other significant associations were detected between perinatal characteristics and high-sensitivity C-reactive protein levels.
Keywords
Introduction
Several maternal and perinatal factors have an important effect on the infant’s life later on. During last years, the most evaluated variables in this association have been maternal obesity and stress, malnutrition and high-fat diet during pregnancy, specific pregnancy complications such as gestational diabetes and pre-eclampsia, and various specific and commonly used drugs, which all may impair fetal and neonatal immunological development, increase individual risk for metabolic and neurological disorders and cardiovascular risks later.1–4 In addition, impaired intrauterine fetal growth, preterm birth, and mode of birth as well as early neonatal treatments and therapies may have further ground effects on the future lifetime health.
We have shown earlier that the possible lack of natural birth processes in elective Cesarean deliveries and treatment in the neonatal intensive care unit after birth might lead to impaired immune responses, which were detected later during teenage years. 5 Others have shown that young adults who had intrauterine growth restriction, had clearly later elevated levels of triglycerides, C-reactive protein, low-density lipoprotein cholesterol as well as high systolic blood pressure, and carotid intima-media thickness and impaired brachial flow-mediated dilatation indicating increased future risk for cardiovascular diseases. 6 Further, Antikainen and associates have reported that prepubertal children whose mothers had had gestational diabetes during their pregnancy, had more low-grade inflammation, which was detected by increased levels of high-sensitivity C-reactive protein (hs-CRP). 7 Season of birth and vitamin D levels have been studied in the association of future cardiovascular risk as well.8–10 Season of birth has been strongly associated with the vitamin D levels at birth or early childhood in many studies,11,12 and in umbilical samples low vitamin D levels has been related to higher CRP values. 13 Among adults, however, Tornhammer and associates showed no association between levels of earlier measured neonatal vitamin D and adult C-reactive protein, but in contrast increased risk for obesity among females with higher neonatal vitamin D levels. 8 Thus far we know, no other perinatal variables have been evaluated in the relation to the low-grade inflammation in follow-up studies.
In general, high-sensitivity C-reactive protein levels are used to measure low-grade subclinical inflammation commonly in the evaluation of the individual risk of cardiovascular disease or other inflammation-related disorder. In this nested case-control study our primary aim was to evaluate whether other maternal or prenatal, as well as intrapartum and early perinatal factors do play any role in the levels of low-grade inflammation in teenage years.
Methods
Patients
The final study population consists of 125 teenagers with hs-CRP levels, ages 15–17 years, and their mothers who underwent Cesarean section at Kuopio University Hospital between March 1990 and February 1992. This group belonged to the larger study population consisted of 805 singleton consecutive pregnant women, who enrolled in the Cesarean delivery birth cohort study. Nearly 98% of all women with singleton pregnancies and Cesarean deliveries at Kuopio University Hospital during the study period were included in the Cesarean delivery birth cohort study.14,15 When their children were 15–17 years old, all live children whose mothers were alive and lived in Finland with known mailing addresses were sent a questionnaire (n = 749) in autumn 2006 (KEISARI-study). The children living in the area of the Hospital District of Northern Savo (n = 688) were invited to clinical examinations such as skin prick tests and further, all children living in the area of Kuopio city (n = 136) were invited to the blood tests during a 4-week period in February to March 2007 (Figure 1). The methods and results of initial Cesarean delivery birth cohort study, as well as longitudinal KEISARI-study have been described more in detail in our earlier reports.5,14–16 The Cesarean delivery birth cohort study, as well as KEISARI-study was approved by the Research Ethics Committee, Hospital District of Northern Savo, Kuopio, Finland (12/1989 and 143/2006), and written consent was obtained from the mothers or legally authorized representatives and teenagers at the time of sampling. All clinical examinations were voluntary. Flow chart of women with Cesarean deliveries (1990–1992) and their children during different part of recruitments and 125 teenagers finally included in hs-CRP analysis.
The type of Cesarean delivery was divided into three classes: elective, unelective and emergency. Season of birth was recorded as followed: spring (March, April and May), summer (June, July and August), autumn (September, October and November) and winter (December, January and February). We divided the birth weights into three subgroups according to birthweight for gestational age: small gestational age (SGA), appropriate gestational age (AGA) and large gestational age (LGA). SGA and LGA were defined as birthweights corrected for gestational age beyond ± 2 standard deviations (SDs) from the mean, according to the sex-specific Finnish fetal growth curve. 17 Birth asphyxia was defined as umbilical arterial pH < 7.05 and Apgar scores at 5 min ≤ 7 and immediate need for neonatal intensive care treatment after birth. Maternal diabetes was defined from hospital records by ICD-9 codes such as 6488A (Abnormal glucose tolerance of mother) or 6480A (Diabetes Mellitus complicating pregnancy). Regular medication, use of oral contraceptives (OC) and time of menarche was asked in postal questionnaires. Current smoking was checked at the time of blood sampling without parents, and it was divided into three groups: never, less than three times a week and every day. All study women and children were Finnish Caucasians.
The blood tests included for example evaluation of specific immunoglobulins, and high sensitivity C-reactive protein (hs-CRP) levels. Blood tests were accomplished in 129 teenagers. Serum hs-CRP levels were measured by ISLAB in Kuopio, Finland according to the manufacturer’s instructions by an immunoturbidimetric assay. The detection limit was 0.20 mg/L. Four children who had values over 8 mg/L (values between 13.2 and 38.0 mg/L) were left out of the study to exclude children with acute infections.
Statistical analysis
Relationships between median hs-CRP levels and perinatal factors and current demographic factors of the offspring were first analyzed using Mann-Whitney U and Kruskall-Wallis tests. Hs-CRP values were then divided into two groups using the 75th percentile cutoff point (1.00 mg/L). Pearson chi-square and Fisher’s exact tests were used to estimate p-values for the relationships between different variables and hs-CRP levels higher than the 75th percentile. Logistic regression analysis was used to investigate relationships between hs-CRP levels in teenagers and adjusted effects of various predictors of high hs-CRP levels. Predictors chosen for the logistic regression analysis were at least marginally associated (p < 0.20) with hs-CRP levels in teenage offspring in univariate analysis. These predictors were season of birth, LGA/maternal diabetes, teenager’s body mass index (BMI), use of regular medication for chronic disease or contraception at the age of 15–17 years. Values of p < 0.05 were considered statistically significant.
Results
All studied 125 teenagers were born by Cesarean delivery (elective [38.4%], unelective [40.8%] and emergency [20.8%]). Season of birth divided quite equally in the study population (22.4–27.2%). Four mothers (3.2%) had gestational diabetes and one (0.8%) had pregestational diabetes. Five children were born LGA.
Among all, 96.8% of the teenagers were at excellent or good general health. Nineteen (15.2%) had regular medication for some chronic disease (seven [5.6%] for asthma, three [2.4%] for migraine and ten [8.0%] for other causes). Among females, 98.2% already had regular menses and five (8.9%) used OC. Four out of five girls with OC had also medication for chronic diseases. Most teenagers had not ever smoked, but nearly 10% smoked daily. Median hs-CRP level was 0.35 mg/L (range 0.20–7.97 mg/L).
Demographic and clinical factors of studied 125 mothers at the time of delivery and their associations with hs-CRP levels among teenage offspring.
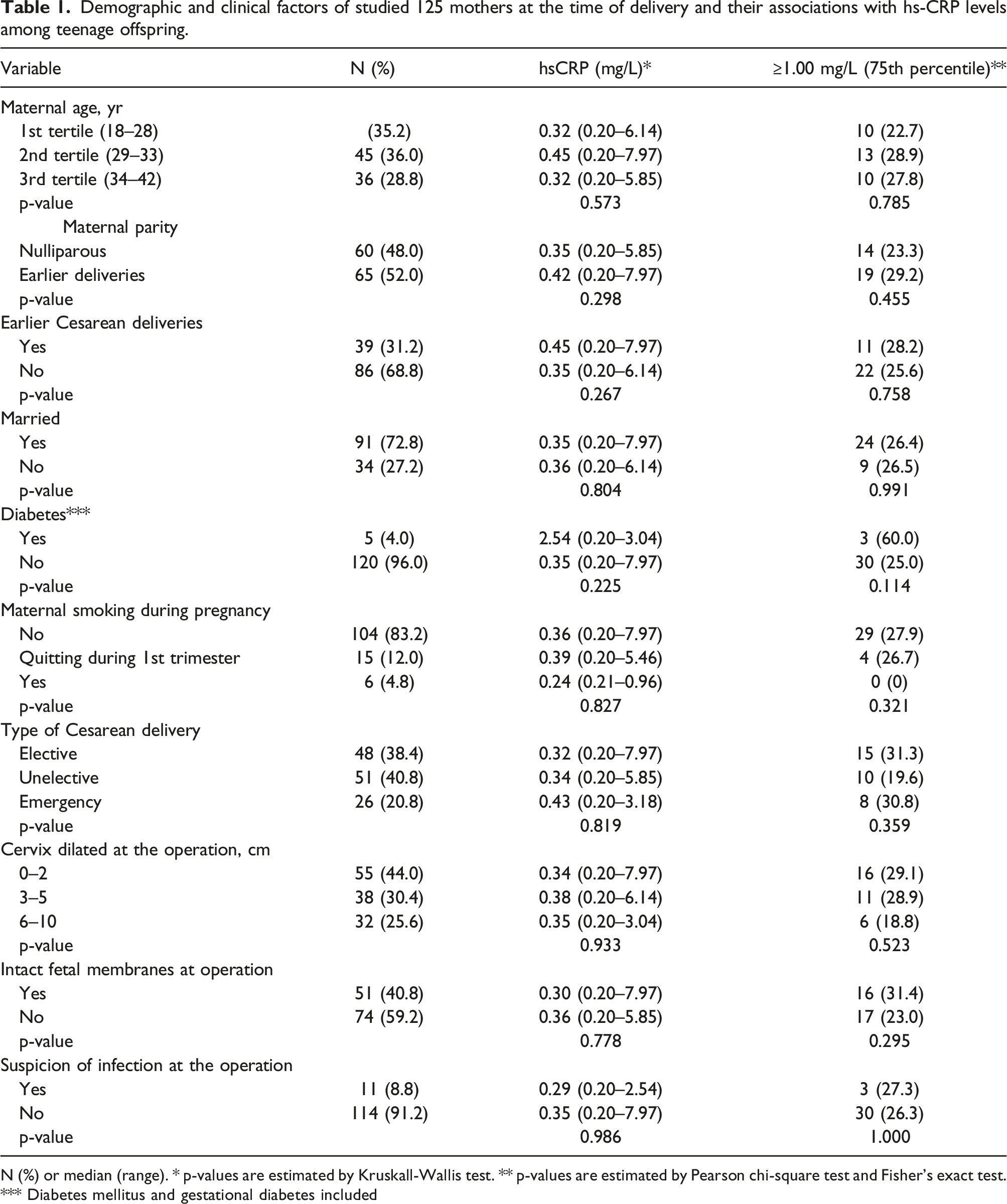
N (%) or median (range). * p-values are estimated by Kruskall-Wallis test. ** p-values are estimated by Pearson chi-square test and Fisher's exact test. *** Diabetes mellitus and gestational diabetes included
Perinatal factors of studied 125 children and their associations with hs-CRP levels at 15–17 years.
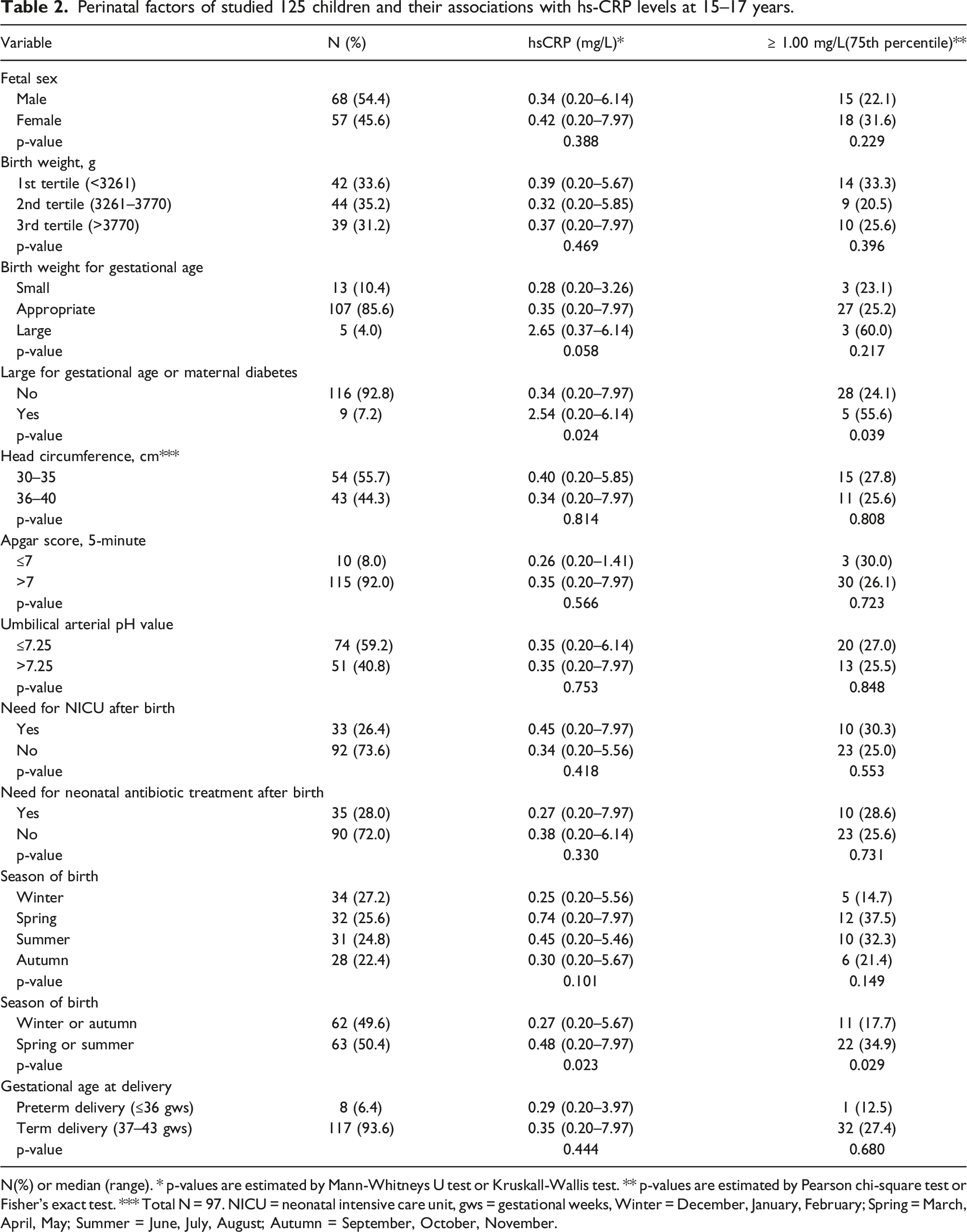
N(%) or median (range). * p-values are estimated by Mann-Whitneys U test or Kruskall-Wallis test. ** p-values are estimated by Pearson chi-square test or Fisher's exact test. *** Total N = 97. NICU = neonatal intensive care unit, gws = gestational weeks, Winter = December, January, February; Spring = March, April, May; Summer = June, July, August; Autumn = September, October, November.
Current demographic factors of studied 125 children at the age of 15−17 years and their associations with hs-CRP levels.
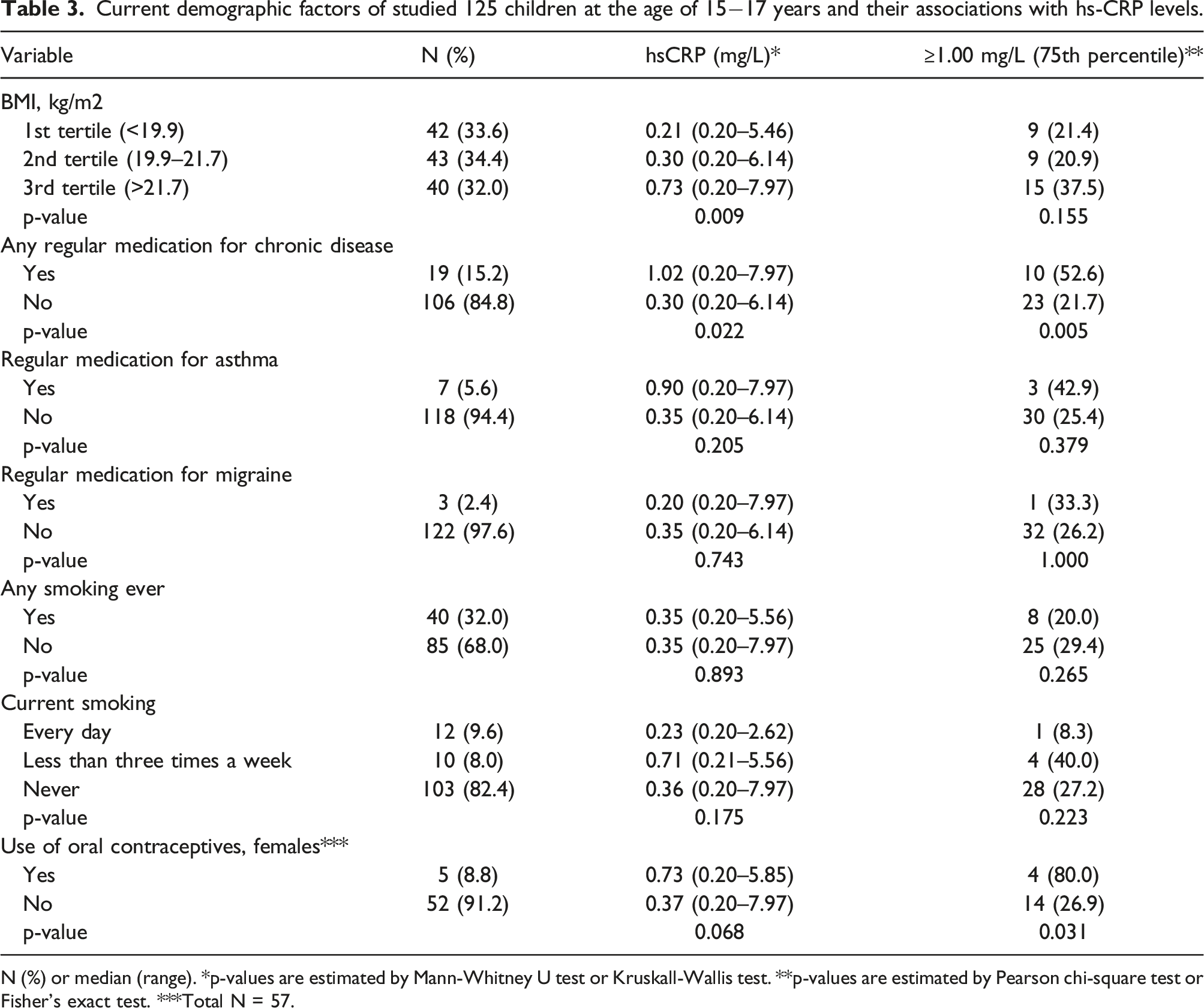
N (%) or median (range). *p-values are estimated by Mann-Whitney U test or Kruskall-Wallis test. **p-values are estimated by Pearson chi-square test or Fisher's exact test. ***Total N = 57.
In multivariate logistic adjusted analysis, maternal diabetes or LGA was associated with the 5.67-fold (95% CI 1.14−28.13) higher risk for increased hs-CRP-values (p < 0.034). Furthermore, birth during spring or summer was associated with the 2.55-fold higher risk (95% CI 1.03−6.28; p < 0.043) and use of any regular medication 4.10-fold higher risk for increased hs-CRP values (95% CI 1.39−12.07; p < 0.010). Current BMI in the highest tertile tended to associate with the increased hs-CRP levels 2.63-fold (95% CI 0.90−7.69; p < 0.077), consecutively. These characteristics explained 22.8% of the variation in hs-CRP in teenage offspring.
Discussion
We showed here first time that there were no association between various maternal, intrapartum and perinatal clinical factors and hs-CRP levels during adolescence. Since all of our children were born by different modes of Cesarean (elective, unelective or emergency), it made us possible to compare whether onset of labor (related to cervical dilatation at the operation) or ruptured fetal membranes or umbilical arterial pH or Apgar scores were related to hs-CRP levels. However, maternal diabetes or being born large for gestational age (LGA) and season of birth, seemed to be an individual risk factor for higher hs-CRP levels: children who were born in spring or summer had higher median hs-CRP levels than those born in winter or autumn. Besides, we confirmed earlier findings of the association of current factors and hs-CRP levels such as BMI, use of OC and chronic diseases.18–20
We showed that maternal diabetes or being born LGA seemed to be associated with higher hs-CRP levels among teenagers. It is known that maternal diabetes increases the risk of LGA, and further metabolic syndrome, obesity and impaired glucose tolerance in offspring.1,21 These factors are also associated with high hs-CRP levels in individuals. Higher hs-CRP levels have been found to be an individual risk factor for developing diabetes.22,23 Obese individuals with high BMI also have higher hs-CRP levels than those with a lower BMI. 18 We also showed a significant association between current BMI and hs-CRP levels in teenage offspring. Fink and associates reported that maternal prenatal levels of hs-CRP were related with the levels of hs-CRP of their six-month-old infants. 24 Our present analysis did not include the maternal hs-CRP levels, so the association cannot be confirmed in this report. Further, since we had only five mothers with diabetes during pregnancy (according to the hospital records) and five children being born LGA, further studies are needed to confirm the association between maternal glycemic environment and higher hs-CRP levels in offspring. Nearly 30 years ago, diagnostic criteria for gestational diabetes were not defined as well as nowadays, even though transient hyperglycemia during pregnancy has been recognized more than 50 years.
It has long been known that season of birth has some still unresolved effects on the risk of developing certain diseases such as multiple sclerosis, rheumatoid arthritis, ulcerative colitis, systemic lupus erythematosus, celiac disease, autoimmune Addison’s disease, schizophrenia and type 1 diabetes.25–30 We found that median levels of hs-CRP were highest in those teenagers that were born during spring and the second highest in those born during summer. In line with present finding Disanto et al. have showed earlier that the risk of immune-mediated diseases is inversely associated with the ultraviolet B exposure in second and vitamin D levels in third trimester, consequently. 27 In Finland, maternal vitamin D levels are strongly related to the seasonality and exposure to sunlight, thus that pregnant women during late autumn and winter have the lowest levels.31,32 Mothers of winter-born children are likely to have the greatest exposure to the sunlight during second trimester (during gestational weeks 13–28 occurring between summer and autumn) comparing to mothers of spring-born children (gestational weeks 13–28 occurring between autumn and winter). In this study adolescents born in winter had a median hs-CRP level of 0.25 mg/mL whereas those born in spring had nearly three-times higher median hs-CRP levels of 0.74 mg/mL.
We also discovered that four out of five teenage girls who used oral contraceptives had hs-CRP levels more than 75th percentile. In contrast, 14 out of 52 girls (26.9%) who did not use OC had hs-CRP levels over 75th percentile. The number of girls using OC was low in our study, but the findings go well along with the results of previous studies.18,20 Usage of menopausal hormone replacement therapy has a similar increasing effect on hs-CRP levels. 33 All five girls using OC were also born in spring or summer, so their time of birth is also a factor that might affect their higher hs-CRP levels.
The strength of this study is that the perinatal data was collected prospectively. The long follow-up time (15−17 years) is also a strength of this study. Prospectively collected pre- and perinatal data is rarely evaluated as early environmental exposure in follow-up studies. A weakness of our study is that we unfortunately did not perform power calculations and thus had a quite small number of mother-child pairs, which resulted in small subsets for example in girls who use OC and in mothers who had maternal diabetes. Estimation of the sample size would help us in the future to plan study better. A further challenge of this study was the inability to recall other possible early factors that might associate with the low-grade inflammation during childhood and adolescence, that is breastfeeding, diet and eating manners. Further, since we used composed variables, partly due to the small sample size of 125 adolescents and rarity of maternal diabetes, the type I error cannot be excluded. However, we think that our results give new important information of the early life variables that should be better evaluated in future studies with the low-grade inflammation. In addition, our results show that most evaluated perinatal factors were not associated with the low-grade inflammation among teenagers.
Conclusion
In conclusion, maternal diabetes or being born large for gestational age (LGA) and season of birth such as born in spring or summer were associated with higher hs-CRP levels among teenagers. However, these findings might be exaggerated since women with pregestational, or gestational diabetes were not recognized as well as nowadays and our population included only those who were born by Cesarean delivery. Surprisingly, other perinatal factors, such as onset of labor or stage of cervical dilatation at the operative delivery were not associated with hs-CRP levels among teenage offspring reflecting the minor role of the birth itself and the birth-associated factors in the regulation and development of low-grade inflammation later in life.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
The Cesarean delivery birth cohort study, as well as KEISARI-study was approved by the Research Ethics Committee, Hospital District of Northern Savo, Kuopio, Finland (12/1989 and 143/2006).
Informed consent
Written informed consent was obtained from all subjects before the study. Yes, from mothers and children before blood sampling. All blood tests were voluntary.
