Abstract
Obesity is associated with low-grade inflammation that originates mainly from adipose tissue. This is implicated in the pathogenesis of type-2 diabetes and cardiovascular diseases. Strong evidence indicates that chronically elevated systemic low-grade lipopolysaccharide (LPS), elicits low-grade inflammation. However, evidence on LPS effect on adipokines’ level, such as leptin, is scarce, and it has never been investigated ex vivo in human subcutaneous adipose tissue (SAT) and therefore we aim to investigate this. To achieve our aim, SAT explants were obtained from 12 patients (50% males) and were treated with/without LPS. Protein concentration was determined by enzyme-linked immunosorbent assay (ELISA). We found that the average age and body mass index (BMI) of included patients were 58.6 years and 28.6 kg/m2, respectively. LPS induced significantly (~3×,
Introduction
Obesity has reached epidemic proportions and is still escalating at an alarming rate worldwide. In 2014, the World Health Organization reported that more than 1.9 billion adults (~39%) were overweight of these, over 600 million were obese (~13%). 1 In Palestine, the prevalence of obesity has been shown to be approximately four times among women (49%) and two times among men (30%) higher than the worldwide observed prevalence. 2 Obesity is associated with chronic diseases such as cardiovascular, type-2 diabetes and some cancers, which are the leading causes of death. Chronic low-grade inflammation has been shown to be implicated in the pathogenesis of these chronic diseases. 3
Recently, high-fat feeding of mice has been shown to induce a low-grade inflammatory tone through increasing both the proportion of a lipopolysaccharide (LPS)-containing microbiota in the gut and LPS in plasma. This suggests that LPS could be a molecular link between high-fat feeding, microbiota and inflammation triad. Indeed, comparison of mice that were chronically infused subcutaneously with LPS and those that were fed a high-fat diet has shown that both gained the same amount of body weight and elicited a similar inflammatory response in various tissue including, adipose tissue.3,4
Adipose tissue is the primary site of obesity-induced chronic low-grade inflammation. 5 It is well known that once LPS reaches the systemic circulation, infiltrates adipose tissue and consequently triggers an innate immune response,3,4 such as tumor necrosis factor (TNF)-α and interleukin-1 (IL-1). Nevertheless, the effect on other adipokines such as leptin is not clear. This prompted us to determine the role of LPS not only on leptin, but also on TNF-α release from human abdominal subcutaneous adipose tissue (SAT), which has never been investigated earlier.
Materials and methods
Materials and reagents
Gentamicin, glucose, LPS, phosphate-buffered saline (PBS), and bovine serum albumin (BSA) were purchased from Sigma and M199 media from Invitrogen (USA). Human leptin DuoSet enzyme-linked immunosorbent assay (ELISA) kit, substrate solution were purchased from R&D systems (USA), and ELISA plates were purchased from Greiner Bio-One (Germany). Tween 20 was purchased from Sun Pharmaceutical Industries Ltd (Palestine).
Human SAT culture
Human SAT explants were obtained from 12 patients, who underwent surgeries for various reasons as described in Table 1. Body mass index (BMI), gender, age, and other criteria of these patients were described in Table 1. The study was approved by the local medical ethical committee (An-Najah National University IRB committee). Patients were invited to participate in our study and samples were collected after they signed a consent form. Adipose tissue culture was performed as described previously6–8 with few modifications. After the last washing step, tissue explants in triplicate were incubated for 2 and 20 h with and without 10 µg/mL LPS and cultured at 37°C and 5% CO2. Subsequently, the media was stored in a freezer at −20 C prior to ELISA measurements.
Criteria of the patients.
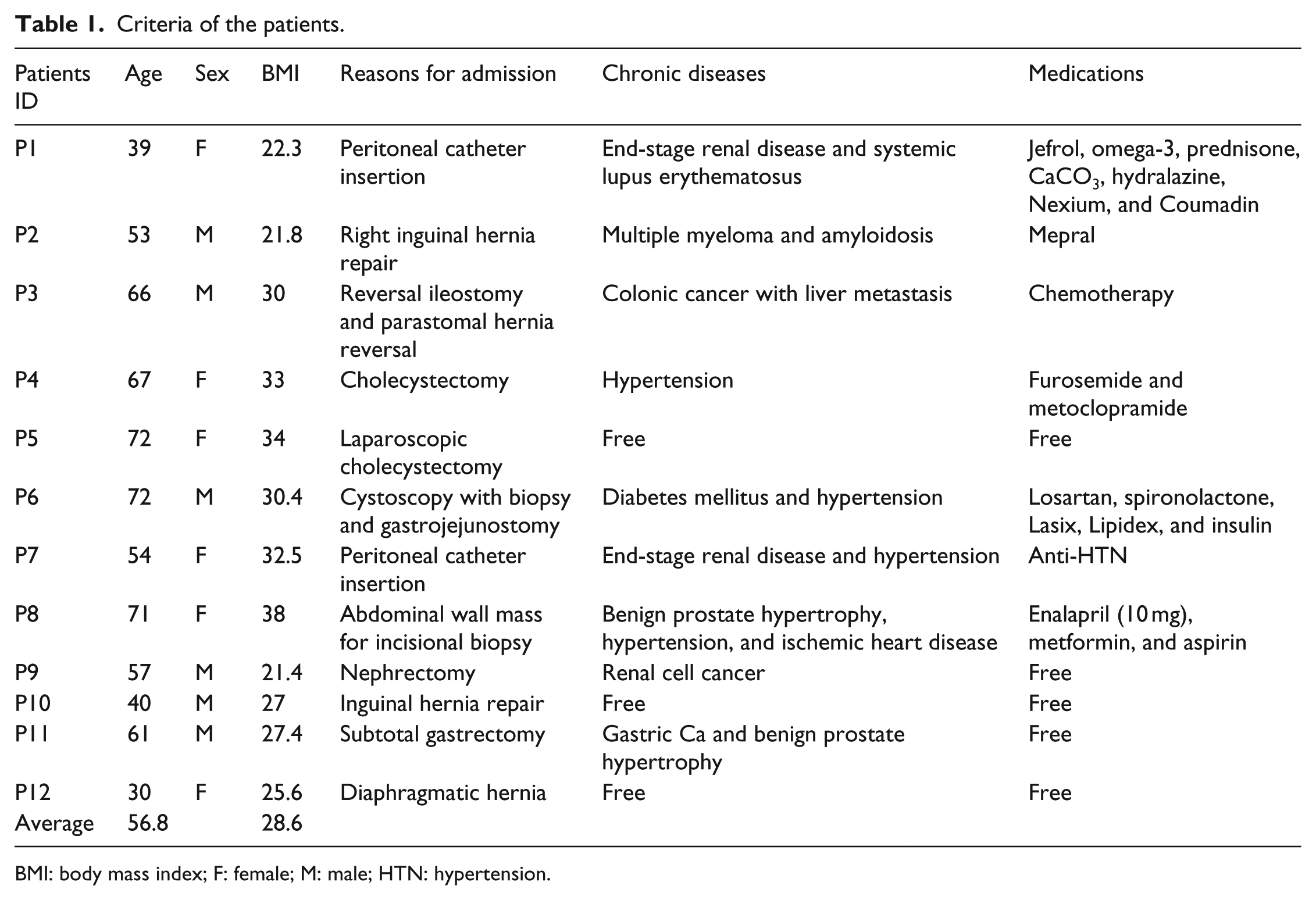
BMI: body mass index; F: female; M: male; HTN: hypertension.
Leptin and TNF-α quantification by ELISA
Secreted leptin and TNF-α quantities were measured in culture media by DuoSet ELISA kit according to the manufacturer’s instructions (R&D systems).
Statistics
Statistical analysis was performed employing GraphPad Prism software version 6.01. To determine whether the values came from a Guassian distribution, D’Agostino and Pearson omnibus normality tests were performed. Comparison between two groups was performed by Student’s
Results
Criteria of patients
The number of patients participated in our study was 12, with male to female ratio of 1 (M/F = 1), and 50% were females. Their age ranged from 30 to 72 (mean = 56.8) years, while their BMI range from 21.4 to 38 (mean = 28.6) kg/m2. Patients were admitted to both Rafidia and An-Najah National University Hospitals in Nablus district for various surgical operations, including hernia repair, nephrectomy, peritoneal catheter insertion, cholecystectomy, subtotal gastrectomy, abdominal wall mass for incisional biopsy, and gastrojejunostomy as shown in Table 1. They were under chronic medications that included jefrol, omega-3, prednisone, CaCO3, hydralazine, Nexium, Coumadin, Mepral, furosemide, metoclopramide, losartan, spironolactone, Lasix, Lipidex, insulin, enalapril, metformin, and aspirin.
LPS effect on leptin release from human abdominal SAT
As demonstrated in Figure 1(a), 10 µg/mL of LPS treatment for 2 h had no effect on leptin release from SAT culture, while it inhibited its release slightly (~20%), but significantly (
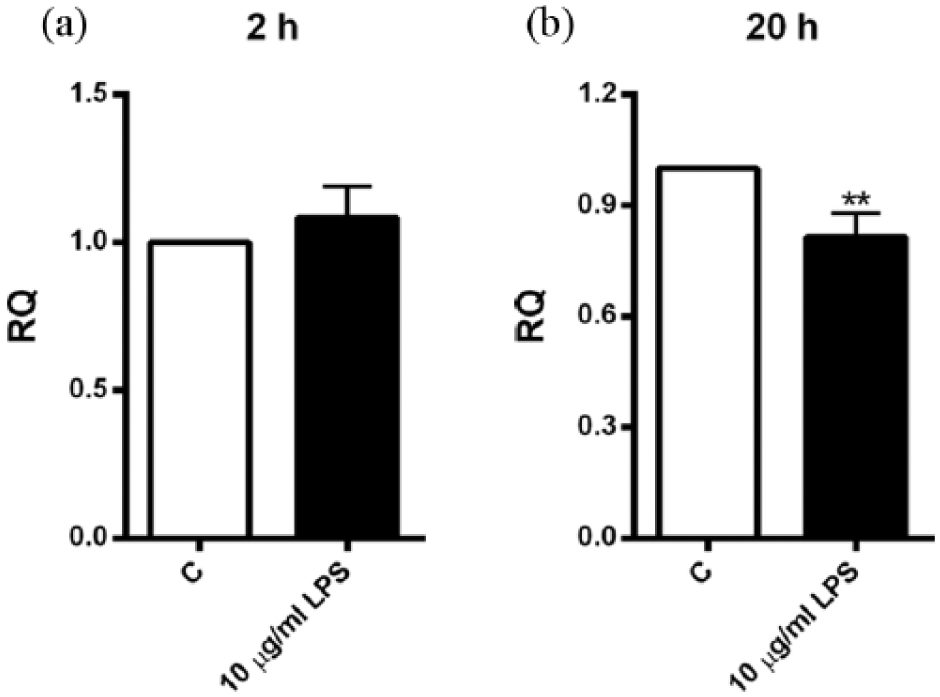
LPS influence on leptin secretion. SAT explants derived from each patient were incubated in triplicate with or without 10 µg/mL of LPS for (a) 2 h (N = 10) and (b) 20 h (N = 9). Secreted quantities of leptin in media were determined by ELISA. Results were depicted as relative quantities (RQs) compared to the control (without LPS; C). **
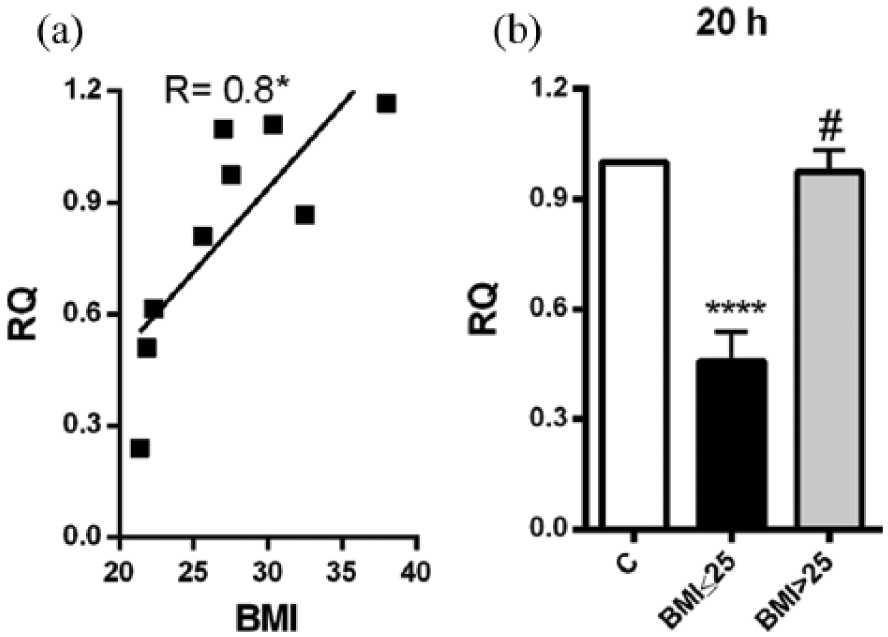
Role of BMI in leptin response. SAT explants derived from each patient were incubated in triplicate for 20 h with or without 10 µg/mL of LPS. (a) The correlation between BMI and leptin response to LPS was determined employing Pearson correlation test. Pearson correlation coefficient was 0.8 and *

Role of (a) age and (b) gender in leptin response. SAT explants derived from each patient were incubated in triplicate for 20 h with or without 10 µg/mL of LPS. (a) Role of age, the correlation between age and leptin response to LPS was determined employing Pearson correlation test. Pearson correlation coefficient was 0.3 (
LPS effect on TNF-α release from human SAT
As shown in Figure 4(a) and (b), LPS treatment for 2 and 20 h significantly (

LPS influence on TNF-α secretion. SAT explants derived from each patient were incubated in triplicate with or without 10 µg/mL of LPS for (a) 2 h and (b) 20 h. Secreted quantities of leptin in media were determined by ELISA. Results were depicted as relative quantities (RQs) compared to the control (without LPS; C). ****

Role of (a) BMI and (b) age in TNF-α response. SAT explants derived from each patient were incubated in triplicate for 20 h with or without 10 µg/mL of LPS. Correlation between BMI and age on one hand and TNF-α response to LPS on the other hand was determined employing Pearson correlation test. Pearson correlation coefficients were −0.4 (
Discussion
Strong evidence in rodents’ studies indicates that chronically elevated systemic LPS, below levels that activate the immune system, elicits low-grade inflammation as well as metabolic dysregulation of adipose tissue and consequently its associated chronic diseases.9,10 Evidence from human studies is scarce and less clear. In vivo, LPS has been shown to have no effect or to increase slightly leptin release,11,12 while in vitro, LPS has been shown to either induce 13 or to suppress 14 leptin release from human adipocytes.
As adipose tissue plays a major role in obesity-induced low-grade inflammation, we investigated the effect of LPS on the release of leptin and TNF-α from human SAT, and it appeared that leptin was slightly reduced, while TNF-α was induced. Interestingly, we found that LPS specifically and highly suppressed leptin release from SAT explants derived from lean patients only and not from explants derived from overweight and obese patients. This may suggest that LPS release from microbiota to the circulation will inhibit the release of leptin from adipose tissue of lean subjects and consequently increases food intake; since leptin is the satiety hormone. 15
In this study, we found, for the first time, that LPS specifically suppresses the release of leptin hormone from human SAT obtained from lean patients only and induces TNF-α release from SAT derived from both lean and obese patients. Our findings provide extra evidence and further confirm earlier studies regarding the roles of LPS and adipose tissue in eliciting low-grade inflammation and obesity and consequently associated chronic diseases. Therefore, further investigations are necessary to identify factors that can prevent or attenuate LPS passage through intestinal barrier. This may prove useful in prevention and/or medical management of obesity and its associated chronic diseases.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received financial support for the research from An-Najah National University, but not for authorship, and/or publication of this article.
