Abstract
Introduction
C-reactive protein (CRP) is an acute-phase protein of the pentraxin family that is primarily produced by liver hepatocytes and released into the bloodstream in response to inflammatory signals. 1 It’s a non-specific marker for a number of diseases, including infections and chronic inflammatory disorders. It is known that inflammatory factors, tumor necrosis factors, and interleukin-6 (IL-6) influence its level.2–5 C-reactive protein is similar to high-sensitivity C-reactive protein (hsCRP), but hsCRP levels, as the name implies, are more sensitive. 6 High-sensitive C-reactive protein is one of the simplest, most accurate, and least expensive inflammation markers. 7 In healthy people, hsCRP levels are typically less than 10 mg/L. However, its concentrations rise rapidly in the days following an acute phase stimulation, reaching 1,000 mg/L, and then return to baseline with a half-life of 19 h when the stimulus is stopped.3,8 In recent years, hsCRP has been used in a variety of applications, including cardiovascular disease, cancer, and tissue damage caused by various causes.9–11
Slightly higher hsCRP levels are associated with a higher body mass index (BMI) in both adolescents and adults. In obese children, hsCRP rises due to macrophage invasion of enlarged adipose tissue, which emits inflammatory signals and cytokines. Furthermore, fragments of data suggested that slightly higher hsCRP levels were associated with obesity and cardiovascular risk markers such as hypertriglyceridemia and low HDL cholesterol levels.12–18 Furthermore, hsCRP has a predictive value at all levels of serum lipids, blood pressure, the severity of the metabolic syndrome, and in those with or without subclinical atherosclerosis. For instance, even lower hsCRP levels were associated with a higher risk of cardiovascular disease. Pieces of evidence indicate that 1 mg/L, 1–3 mg/L, and 3 mg/L hsCRP levels are related to lower, moderate, and increased cardiovascular risks, respectively. 19 The fact that the degenerative phase of atherosclerosis begins in childhood is now widely accepted.20–22 As a result, hsCRP may be a useful biomarker for detecting and managing people who are at risk of various inflammatory disease conditions. The majority of existing African reference intervals for clinical laboratory parameters, including hsCRP, are based on foreign populations and may not be representative of population values elsewhere.23,24 Because of lifestyle and genetic differences, it is inappropriate to employ Western reference values for Ethiopians. Moreover, there are no published hsCRP reference intervals for children in Ethiopia. 25 Therefore, the purpose of this study was to identify the reference intervals (2.5th and 97.5th percentiles) of serum hsCRP in children who appeared to be healthy. The reference intervals supplied could open the path for hsCRP to be used as a marker for early identification of any inflammatory condition in Ethiopia. Furthermore, the present study adds additional evidence to the existing data from several European, South Asian, and American countries on the distribution of hsCRP.
Materials and methods
Study population and eligibility criteria
The current cross-sectional study was conducted from April 2019 to October 2019 in Addis Ababa, Ethiopia. Addis Ababa is the capital city of Ethiopia and a seat for the African Union. The city was divided into 10 subcities (currently 11) during the study period. A multistage random sampling procedure based on the selection of sub-cities and woredas (the smallest administrative unit) was used to choose study participants. The probability proportionate to size (PPS) sampling method was used, with the size of the sample based on the number of people in the sub-city. Accordingly, four subcities were selected with the help of the Central Statistical Agency of Ethiopia. Then, using a practical sampling strategy, study participants were recruited from randomly selected woredas (the smallest administrative units under subcities) through both school and village-to-village mobilization by health extension workers and well-trained data collectors. Volunteers were requested to come to the mobile specimen collection sites. The study population’s socio-demographic and behavioral characteristics (cigarette smoking, alcohol use, fruit and vegetable consumption, and physical activity), history and physical examination, and anthropometric measurements (blood pressure, weight, and height) were taken. Both the study participants and their parents/guardians, as applicable, provided medical history information. Apparently healthy children living in Addis Ababa for at least 6 months, aged between 5 and 17, who voluntarily participated in the study, were included. Participants with viral (HIV, HBV, and HCV) or bacterial infections (syphilis), intestinal parasites, any acute or chronic health concern, or who were on medication were excluded from participating in the study. Likewise, participants experiencing an acute illness and having chronic diseases like diabetes mellitus, heart disease, anaemia, thyroid disease, liver disease, and cancer of any type were excluded from the study. Participants were partitioned by age into groups of 5–11 years and 12–17 years because of pubertal differences in serum analytes of hsCRP and lipid profile. Based on Clinical Laboratory Standard Institute (CLSI) guidelines, a minimum of 120 samples is needed to establish the 2.5th and 97.5th percentiles with 95% CI by using a non-parametric method. A total of 492 apparently healthy children (ages 5–17 years) participated in the study.
Anthropometric measurements
Well-trained professionals took anthropometric measurements such as body weight and height. Each subject’s weight and height were assessed, and their BMI was computed by dividing their weight (kg) by their height squared (m2). Height was measured in a standing position using well-placed stadiometers without shoes to the nearest 0.1 cm. A digital balance was used to measure the weights to the nearest 0.1 kg. Weights were taken in a standing position with the children barefoot, with no shoes, phones, or other materials in their pockets. After a 5-min break, an electronic sphygmomanometer was used to take blood pressure in the right upper arm, and three measures were averaged to get the final result. The study excluded children with a systolic or diastolic blood pressure of more than 120/80 mmHg. According to the Centres for Disease Control (CDC) recommendations, 26 study participants’ weight status was classified as underweight (5th percentile), healthy weight (5th to 85th percentile), overweight (85th to 95th percentile), and obese (95th percentile).
Sample collection, processing, and biochemical measurements
From 8:30 a.m. to 11:00 a.m., 5 mL of fasting blood samples were taken from the median cubital vein using serum separator tubes (SST) after all children and their families were informed of the study’s purpose. The blood samples were centrifuged at 3000 revolutions per minute for 5 min. The serum was isolated and stored at −80°C until analysis. During the biochemical analysis process, serum samples were exposed to a single freeze-thaw cycle. Serum hsCRP levels were measured in the clinical chemistry laboratory of the Ethiopian Public Health Institute (EPHI) using an immune-turbidimetric test with a Cobas Integra 400® Plus automatic biochemical analyzer (Roche Diagnostics, GmbH, Mannheim, Germany). The amounts of TC, LDL-c, and TG were measured using colorimetric assays. The amino groups of HDL-c were identified enzymatically by cholesterol esterase and cholesterol oxidase, which were combined with polyethylene glycol (which specifically forms complexes with chylomicrons, LDL-c, and VLDL-c). Infectious disease screening tests were conducted at the Ethiopian National Blood Bank Services (NBBS) using enzyme-linked immunosorbent assay (ELISA) testing technologies. All of the samples were frozen and thawed once during the testing process. All of the tests were done in a nationally authorized laboratory, with a measurement uncertainty of 1.4% for TC, 1.3% for LDL-c, 1.8% for TG, 1.9% for HDL-c, and 1.5% for hsCRP at the time of analysis.
Data quality assurance and quality control
The questionnaire was pilot-tested on 10% of the sample size to ensure its completeness, consistency, and applicability, and it was modified as needed. Following the completion of each questionnaire, data collectors and the principal investigator cross-checked the information gathered to ensure its accuracy. The test tube label and the questionnaire’s unique identification number were also compared for similarity. The clinical chemistry analyzer was tested for accuracy using normal and pathological controls after checking the expiration dates of both the reagents and the controls. Furthermore, prior to processing the study participant’s sample, dual quality controls (normal and pathological) and pooled serum samples were always performed, and the study participant’s sample was then analyzed. In general, all pre-analytical, analytical, and post-analytical precautions were taken to ensure the accuracy and precision of the test results.
Statistical analysis
Statistical analyses were performed using the Statistical Package for the Social Sciences (SPSS Statistics, IBM Corporation USA), version 23.0. Histograms and box plots were used to show the distribution of serum hsCRP by age and gender. The skew distribution of hsCRP was represented by the median and 95% Confidence Intervals (CI). Because the data were not normally distributed, a non-parametric Spearman correlation coefficient was used for correlation analysis. Furthermore, bivariate and multivariable logistic regression analyses were performed to assess factors associated with elevated hsCRP levels. The crude and adjusted odds ratios and their corresponding 95% CI were computed. For the purpose of performing regression analysis, the values of hsCRP are classified as normal (hsCRP values <3 mg/L) and high (hsCRP values >3 mg/L). According to sex and age subgroup, the 2.5th, 5th, 10th, 25th, 50th, 75th, 90th, 95th, and 97.5th percentiles were displayed. p-values <0.05 were considered statistically significant.
Result
Characteristics of study population
Socio-demographic characteristics of children aged 5–17 years in Addis Ababa, Ethiopia.

High-sensitive C-reactive protein level distribution
Figure 1 depicts the non-normally distributed hsCRP distribution among study populations. Observation from the figure shows that the values of serum hsCRP were skewed to the right and compacted between 0.00-2 mg/L for both sexes (Figure 1). Moreover, the visual inspection of the box plots shows that the upper whisker values of hsCRP in all study populations were found to be less than 2 mg/L. Meanwhile, a number of outlier values of hsCRP were observed, as indicated in the figure below (Figure 2). Histograms showing sex disaggregated distribution of serum hsCRP concentrations (mg/L) among children in Addis Ababa, Ethiopia. Box plots showing the age and sex specific distribution of hsCRP among children in Addis Ababa, Ethiopia.

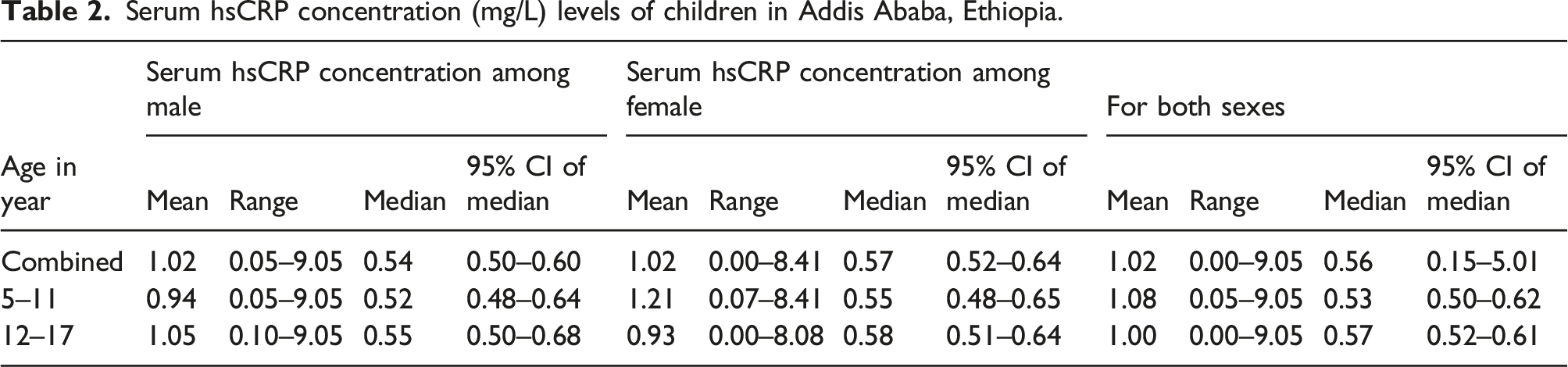
Serum hsCRP concentration (mg/L) levels of children in Addis Ababa, Ethiopia.
Correlation between hsCRP and predictors
Spearman correlation analysis of hsCRP with predictors among study participants in Addis Ababa, Ethiopia.

Note: *p-value less than 0.05.
Factors associated with elevated hsCRP levels among children
Factors associated with high hsCRP levels among children in Addis Ababa, Ethiopia.

Note: BMI, body mass index; TC, total cholesterol; TG, triglycerides; HDL-c, high density lipoprotein cholesterol; LDL-c, low density lipoprotein cholesterol.
*p-value less than 0.05.
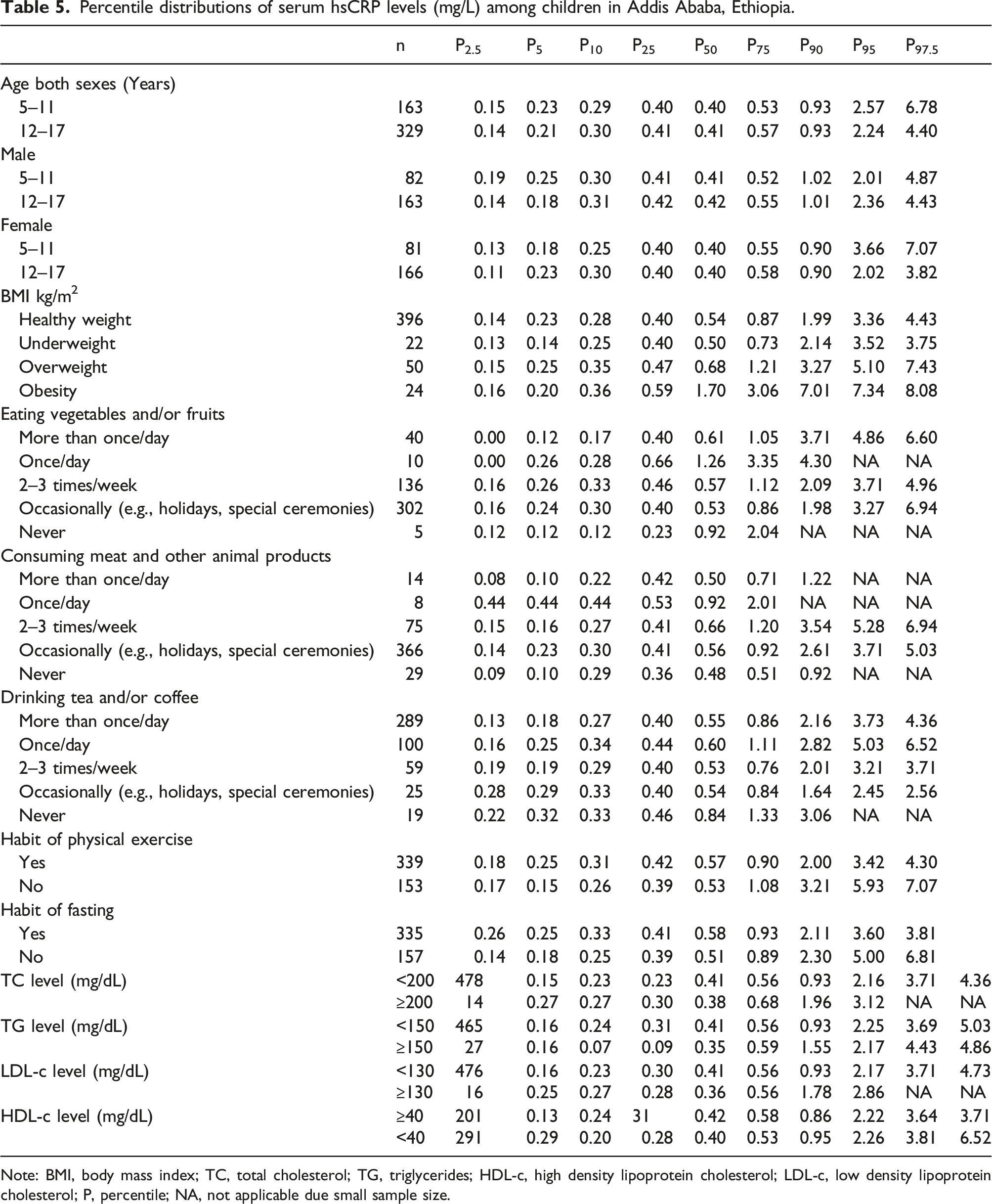
The percentile distributions of serum hsCRP concentrations in apparently healthy children
Percentile distributions of serum hsCRP levels (mg/L) among children in Addis Ababa, Ethiopia.
Note: BMI, body mass index; TC, total cholesterol; TG, triglycerides; HDL-c, high density lipoprotein cholesterol; LDL-c, low density lipoprotein cholesterol; P, percentile; NA, not applicable due small sample size.
Discussion
Feyissa Challa et al. recently described the distribution and risk factors of serum hsCRP levels in a 15–69-year-old Ethiopian population. However, children under the age of 15 were not included in the previous study, and participants were not tested for non-infectious or infectious disorders such as the human immunodeficiency virus, hepatitis B, hepatitis C, syphilis, or malaria. 25 In cardiovascular illnesses and diabetes, hsCRP has recently been identified as a sensitive marker of low-grade inflammation.7,27,28 Based on a large data set, the US CDC and the American Heart Association concluded that hsCRP is a useful marker for predicting the risk of future cardiovascular disease and diagnosing and treating patients with acute coronary syndrome.29,30 A high serum hsCRP level has been linked to obesity and poor lipid metabolism in children in a number of studies.31,32 The distribution of hsCRP concentrations in children has been established in several studies from various countries.33–38 However, little was known about the distribution of hsCRP concentrations in apparently healthy Ethiopian children, and no data on the distribution of hsCRP in apparently healthy Ethiopian children had been published. In Ethiopia, manufacturer provided values that are determined for other populations are being utilized in clinical practice. As a result, this is the first study to characterize the distribution of serum hsCRP concentrations in children who appear to be healthy. Children in our study did not have any infectious diseases (such as HIV, HBV, HCV, syphilis, or malaria), and the percentile modelling took into account non-hypertension, non-diabetes, and the absence of any other known chronic diseases. However, fifty (10.16%) of the study participants were overweight, and twenty-four (4.88%) were obese.
The current work revealed that there were no significant differences in the distribution of hsCRP with age subgroups or sexes in this investigation. This finding was supported by a study done by Rodoo P et al., 39 Blaha MJ et al., 40 and Colantonio DA et al. 41 In contrast, the studies conducted by H Schlenz et al., 42 Thierfelder W et al., 43 and Soldin OP et al. 44 discovered sex differences in serum hsCRP distribution. However, in the present study, the serum hs-CRP levels among females were slightly but not significantly higher than their male counterparts. This could be because the girls had high levels of fat storage, which causes inflammation and raises hsCRP. This finding contradicted the prior two studies done by Tang YB et al. 34 and Raitakari M et al. 45 Thirty-six (7.32%) of the total study participants had a serum hsCRP of >3 mg/L, which was lower than the earlier Ethiopian study by Feyissa et al., which found 18%. 25 Because the prior study included adult participants who were at high risk for numerous factors that could cause blood hsCRP elevation, the discrepancy was expected.
Male children had a median hsCRP value of 0.54 mg/L, while female children had a median hsCRP value of 0.57 mg/L. The present median readings for both sexes were lower than those found in a study by Tang Yan Bin et al. in China, which found that the average hsCRP concentration in the urban population was 0.68 mg/L for males and 0.65 mg/L for females. 34 Adult participants in the China study may be more exposed to traditional risk factors, including cardiovascular disease and smoking, which could explain the differences. On the other hand, the current median value of hsCRP is higher than in several previous studies.46–49 Another comprehensive study done by H Schlenz et al. observed that the median hsCRP values in eight European nations (Sweden, Germany, Hungary, Italy, Cyprus, Spain, Belgium, and Estonia) varied from 0.2 to 0.3 mg/L, with the exception of Italy, where the median hsCRP levels were 0.5 mg/L. 42
For the age groups 5–11 and 12–17 years, the established 2.5th and 97.5th percentiles for combined sexes were 0.15–6.78 mg/L and 0.14–4.40 mg/L, respectively. For 5–11 and 12–17 years, the derived 2.5th and 97.5th percentiles were 0.19–4.87, 0.14–4.43 mg/L for males, and 0.13–7.07, 0.11–3.82 mg/L for females, respectively. A study done by Soldin et al. 44 reported slightly higher 97.5th percentiles of hsCRP, which varied from 7.9 mg/L in males to 10.0 mg/L in females. Likewise, H Schlenz et al. 42 reported 97.5th percentiles of hsCRP 2.8 and 6.3 mg/L in males and females, respectively. Moreover, the highest 97.5th percentile of 11.3 mg/L hsCRP was reported in Swedish children. 41 The hsCRP concentrations reported by Ford ES et al. ranged from 0.1 to 90.8 mg/L, 47 which was extremely higher in the upper limit than the current reports of 0.00–9.05 mg/L. This variability may be explained by the difference in the study population and the applied exclusion criteria.
In this study, the 95th percentiles of hsCRP were 0.15 and 5.01 mg/L for 5–17-year-old males and females, respectively. This range is slightly different from the one reported by the large European cohort study. 42 The 97.5th percentiles of hsCRP in the current study ranged from 2.56 to 8.08 mg/L, which is comparable with other previous findings (2.8–6.3 mg/L).45,50. In contrast to the above reports, a Canadian study 41 revealed the lowest 97.5th hsCRP percentile of 1 mg/L for both sexes. The discrepancy in hsCRP concentrations may be due to differences in health status (such as the presence of chronic and acute illness conditions), other behavioral habits, differences in methodologies, or the number of study participants.
According to the current findings, children who never eat vegetables and/or fruits and those who are obese were more than three times more likely to have elevated serum hsCRP concentrations. Obese children also had lower 2.5th hsCRP values of 0.16 mg/L and higher 97.5th hsCRP values of 8.08 mg/L. A number of studies13–15,42,51–54 found a link between childhood obesity and high hsCRP levels. Previous studies have revealed that adipose tissue is a secondary source of hsCRP, with hsCRP mRNA expression in human adipose tissue.55–57 In the regression analysis, the hsCRP levels were found to be directly related to not consuming fruit and/or vegetables. Fruits and vegetables’ antioxidant effects may be mediated by changes in fat mass and decreased adipocyte IL-6 production.58,59 Previous studies25,35 discovered a direct link between hsCRP and physical activity. In contrast, no association was found between hsCRP and physical activity, fasting behaviors, meat and/or animal product consumption, or coffee and/or tea consumption in this study. There was no clear explanation for these findings; as the data was self-reported, there could be some bias. Children who did not fast had higher upper-limit hsCRP levels (6.81 mg/L) than those who did (3.81 mg/L). Similarly, those who did not engage in regular physical activity had higher upper limit hsCRP levels (7.07 mg/L) than those who did (4.30 mg/L). Participants who were overweight or obese had significantly higher levels of hsCRP, implying that physical activity and fasting could accelerate peripheral fat oxidation, resulting in less fat deposition and weight loss.60,61
In the current study, no significant association was found between lipid profiles (such as TC, TG, LDL-c, and HDL-c) and hsCRP levels. In line with the current study, several other previous studies have shown that hsCRP has no association with serum triglyceride levels25,31,36,62 or LDL cholesterol. 25 In contrast, prior cross-sectional and longitudinal investigations have found that high hsCRP levels are associated with low HDL-c levels,36,63–65 and other studies have found an association with triglyceride levels.66–68 Even though HDL-c levels were not significantly related to hsCRP, participants with low HDL-c had greater hsCRP concentrations (6.52 mg/L) at the upper limit than those with normal HDL-c (3.71 mg/L). We recruited apparently healthy children for this study. For instance, more than 95% of the participants had normal serum lipid levels, with only a tiny percentage of children having aberrant serum lipid levels, which could explain the absence of connections. The study has some limitations, such as those under-five-year-old children, non-native Ethiopian children, and migrants were not taken into account due to resource constraints, and the study was limited only to Addis Ababa. Furthermore, the sample size was determined only using the CLSI guidelines; no power calculations were done.
Conclusion
This study is the first to determine the reference intervals of serum hsCRP among apparently healthy Ethiopian children. The study detected no significant differences in hsCRP distribution across age groupings or sexes in this investigation. Meanwhile, notable differences were observed in the established reference intervals of hsCRP in comparison with published literature. The hsCRP levels were found to be linked to increased BMI and never or occasionally eating vegetables and/or fruits. Furthermore, we confirmed that the serum hsCRP concentration in obese children was considerably greater than in non-obese children. Since hsCRP has recently been identified as a sensitive marker of low-grade inflammation, this finding highlights the importance of improving weight status in childhood. Moreover, based on our findings, we suggest using these population-based hsCRP reference values as a screening tool for the early detection of a variety of inflammatory diseases.
Supplemental Material
Supplemental Material - Determination of high-sensitivity C-reactive protein reference intervals for apparently healthy children in Addis Ababa, Ethiopia
Supplemental Material for Determination of high-sensitivity C-reactive protein reference intervals for apparently healthy children in Addis Ababa, Ethiopia by Ousman Mohammed, Melkitu Kassaw, Endalkachew Befekadu, Letebrhan G/Egzeabher, Abebaye Mekonen, Demiraw Bikila, Feyissa Challa, Abiy Belay, Mistire Wolde, Kassu Desta and Aster Tsegaye in European Journal of Inflammation
Footnotes
Acknowledgements
We appreciate the study participants’ tolerance during the study period. We would especially like to express our gratitude and appreciation to the data collectors.
Authors contribution
All authors made significant contributions to the manuscript. All authors have read and given their final approval of the version of the manuscript submitted for publication.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ethiopian Public Health Institution and the Ministry of Innovation and Technology, which were the sources of the budget.
Ethical consideration
The study was granted permission by the Department of Medical Laboratory Science’s departmental research and ethical review committee (DRERC), with protocol number DRERC/439/2019/MLS, in accordance with the Declaration of Helsinki. Moreover, written informed consent was obtained from all subjects and/or their legal guardian(s). An information sheet was distributed to all study participants and their parents or guardians to explain the purpose of the study. The names of the participants were kept confidential, and only codes were used on the questionnaire and biological sample containers to maintain confidentiality.
Availability of data and materials
All the datasets used and/or analyzed during the current study are available in the manuscript.
Informed consent
Written informed consent was obtained from all subjects and/or their legal guardian(s). Likewise, the author obtained written informed consent from the legally authorized representative of the subjects for the publication of this study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
