Abstract
Introduction
Coronavirus disease (COVID-19) has spread rapidly to more than 200 countries and regions worldwide. Although several options have been implemented in attempts to control the rate of COVID-19 infection, the mortality rates remain high. The aim of this study is to evaluate whether patients with severe COVID-19 will benefit from the combination of anticoagulant and glucocorticoid therapy.
Methods
A single-center retrospective cohort study of patients with COVID-19 was performed. Several factors influencing patients with COVID-19 have been analyzed retrospectively, including basic conditions, inflammatory factors, coagulation parameters, medications, and outcomes. Continuous and categorical variables have been evaluated via binary logistic regression analysis to predict mortality. The association between anticoagulant treatment and outcomes has also been evaluated, stratifying patients according to glucocorticoid treatment.
Results
61 patients with severe COVID-19, including 23 critically ill patients have been enrolled in the study. In those with severe and critical illness COVID-19, mortality rate was lower in those who received combined anticoagulant and glucocorticoid with odds ratio of 0.003 (p = 0.042) and 0.031 (p = 0.046), respectively. In cases of severe COVID-19, those who were treated with both anticoagulant and glucocorticoid had lower mortality (17.6%) than those treated with glucocorticoid alone (31.8%), although this did not reach statistical significance (p = 0.321). In the critically ill COVID-19 patients who received both anticoagulant and glucocorticoid, there was significantly lower mortality (30%) than those who received glucocorticoid alone (77.8%) (p = 0.047). Seventy-five percent of non-survivors of COVID-19 met the International Society on Thrombosis and Haemostasis criteria of disseminated intravascular coagulation (total points ≥5).
Conclusion
Patients with severe COVID-19 or critically ill patients with COVID-19 may benefit from the combination therapy of anticoagulant and glucocorticoid. As the disease getting severe, more benefits may be obtained from the combination therapy.
Keywords
Introduction
Coronavirus disease (COVID-19), which is caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection, has rapidly spread to more than 200 countries and regions worldwide. By October7, 2021, the global cumulative number of diagnoses has exceeded 237 million, and the number of confirmed cases of COVID-19 is continuing to rise. Several options have been implemented to control the mortality rate of COVID-19, including using glucocorticoids and anti-viral drugs (interferon, lopinavir/ritonavir, ribavirin, chloroquine phosphate, arbidol).1,2 However, antiviral treatment which specifically targets COVID-19 is not available. The global cumulative number of deaths due to COVID-19 has exceeded four million, and the mortality rate is approximately 2%. Multiple organ dysfunction syndrome (MODS), disseminated intravascular coagulation (DIC), and venous thromboembolism (VTE) are considered as possible causes to the death in patients with severe COVID-19.3,4 There is an urgent need to learn from existing experience to reduce the patient fatality rate of COVID-19. The aim of the study is to evaluate whether patients with severe COVID-19 will benefit from the combination of anticoagulant and glucocorticoid therapy.
Materials and Methods
Patient characteristics
This retrospective study includes individuals who were diagnosed as COVID-19 infected persons from 10 February 2020 to 28 March 2020, at Sino-French New City Branch of the Tongji Hospital of Huazhong University of Science and Technology in Wuhan, China. Eligible patients are aged 18 years or older with severe COVID-19. This study has been approved by the Ethics Committee of the Third Xiangya Hospital of Central South University (NO. 2020-S585), and the written informed consent is waived due to the retrospective nature of the study. All patients have been anonymized to protect the confidentiality of individual participants.
The diagnosis of COVID-19is based on the World Health Organization interim guidance. Severe COVID-19 has been defined as meeting any of the following items, according to the Diagnosis and Treatment Plan of COVID-19 suggested by the National Health Commission of China: respiratory rate ≥30 breaths/min; arterial oxygen saturation ≤93% at rest; PaO2/FiO2 ≤300 mmHg; pulmonary imaging showing that lesions have significantly progressed by 50% within 24–48 h; age >60 years with severe chronic disease including hypertension (HTN), diabetes mellitus (DM), coronary heart disease (CHD), cancer, structural lung disease, or pulmonary heart disease or receiving immunosuppressant therapy. Critically ill patients with COVID-19 have been defined as meeting any of the following items on admission: respiratory failure requiring mechanical ventilation, shock, organ failure, or dysfunction. Anticoagulant treatment group was defined as receiving heparins for 7 days or longer, mainly with prophylactic dose of low molecular weight heparin (LMWH, 40–60 mg enoxaparin/day) 5 which was the most commonly used anticoagulant therapy for COVID-19 at our hospital.
Observation indexes
Five indices are collected from patients with severe COVID-19 1 : basic conditions, including sex, age, smoking history, chronic diseases (HTN, DM, CHD, heart failure, chronic obstructive pulmonary disease, cancer, chronic liver dysfunction, and chronic renal dysfunction) 2 ; inflammation indicators, including white blood cell (WBC) and lymphocyte counts; levels of tumor necrosis factor (TNF), interleukin (IL)-1, IL-2R, IL-6, IL-10, albumin, procalcitonin (PCT),C-reactive protein (CRP); and the erythrocyte sedimentation rate 3 ; coagulation parameters, including the platelet count, prothrombin time (PT), activated partial thromboplastin time (APTT), fibrinogen level, and D-dimer level 4 ; medications, 5 ; outcomes (28-days mortality).
Statistical analysis
Between survivors and non-survivors, normally and abnormally distributed quantitative variables have been compared using the Student’s t-test and Mann-Whitney U test, respectively. Categorical variables are compared by using the chi-squared test or Fisher’s exact test according to sample size. The results are presented as the mean ± standard deviation, median (interquartile range), or number (percentage), as appropriate. Consecutive and categorical variables have been evaluated by using binary logistic regression analysis to predict the mortality. A p-value of <0.05 is considered statistically significant. Data are analyzed by using SPSS 22.0 for Windows.
In addition, we calculate the power at online websites of Power and Sample Size (http://powerandsamplesize.com/Calculators/Compare-2-Proportions/2-Sample-Non-Inferiority-or-Superiority). This calculator uses the following formulas to compute power:
Results
Sixty-one patients (30 males and 31 females) were diagnosed with severe COVID-19, including 23 critically ill patients (6 males and 17 females). Completed clinical and laboratory test data were available for all patients. The average age of the patients with severe COVID-19 is 65.67 years, and that of the critically ill patients with COVID-19 is 67.65 years. By 28 March 2020, 12 patients died; all other patients have recovered and been discharged. The death rate of our research is 19.6%.
The grade of DIC in patients with severe COVID-19.

PT: prothrombin time,DIC:disseminated intravascular coagulation.
Characteristics of patients with severe COVID-19.

DM: diabetes mellitus, HTN:hypertension, CHD:coronary heart disease, CHF: chronic heart failure, COPD: chronic obstructive pulmonary disease, WBC: white blood cell, TNF: tumor necrosis factor, IL: interleukin, PCT: procalcitonin, CRP: C-reactive protein,ESR:erythrocyte sedimentation rate, PT: prothrombin time, APTT: activated partial thromboplastin time.
Characteristics of critically ill patients with COVID-19.

DM: diabetes mellitus, HTN:hypertension, CHD: coronary heart disease, CHF: chronic heart failure, COPD: chronic obstructive pulmonary disease, WBC: white blood cell, TNF: tumor necrosis factor, IL: interleukin, PCT: procalcitonin, CRP: C-reactive protein,ESR: erythrocyte sedimentation rate, PT: prothrombin time, APTT: activated partial thromboplastin time.
Among the patients with severe COVID-19, 28.6% and 25.0% of patients have received the combination of anticoagulant and glucocorticoid treatment in the survivor and non-survivor groups, respectively. There is no significant difference in the number of patients who have received the combination of anticoagulant and glucocorticoid treatment between the two groups (p = 0.805). In critically ill patients with COVID-19, 63.7% and 25.0% of patients have received the combination of anticoagulant and glucocorticoid treatment in the survivor and non-survivor groups, respectively. There is no significant difference in the number of patients who have received the combination of anticoagulant and glucocorticoid treatment between the two groups (p = 0.100).
Multivariate correlative factors of mortality in patients with severe COVID-19.
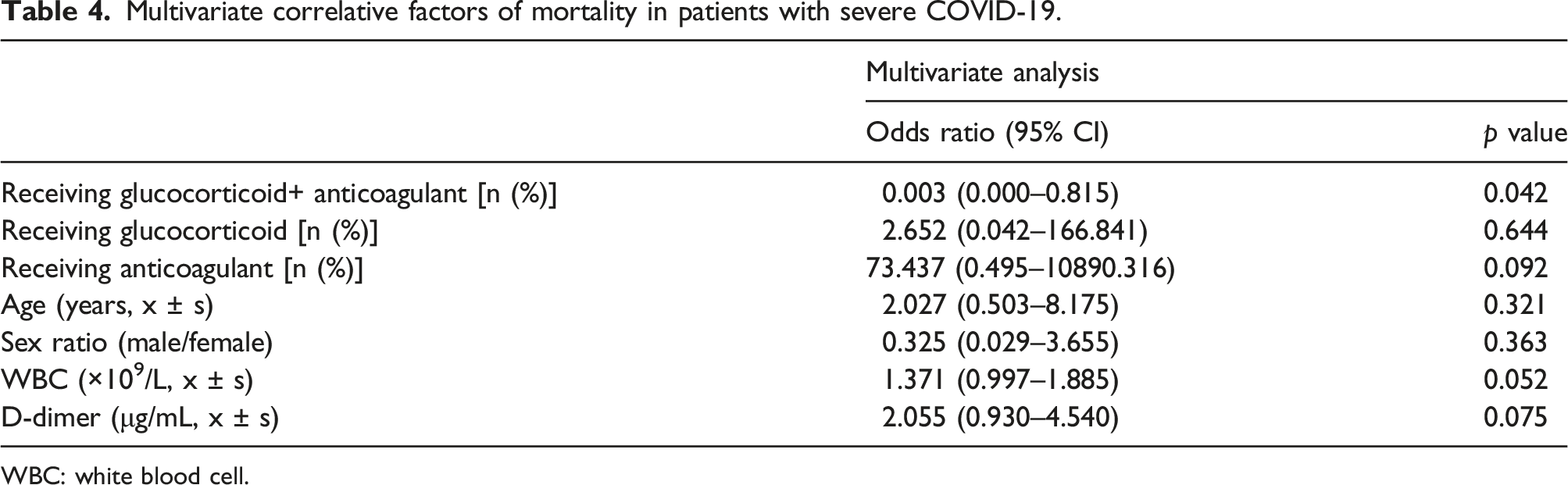
WBC: white blood cell.
Multivariate correlative factors of mortality in critically ill patients with COVID-19.

WBC: white blood cell,PCT: procalcitonin, APTT: activated partial thromboplastin time.
The association between anticoagulant therapy and outcomes in patients with severe COVID-19.

The association between anticoagulant therapy and outcomes in critically ill patients with COVID-19.

Discussion
There are often two sides of the body’s inflammatory response to the invasion of pathogenic microorganisms. An appropriate inflammatory response can eliminate the invading pathogenic microorganisms, but an excessive inflammatory response leads to tissue and organ damage in the body. Under normal circumstances, inflammatory cytokines and the anti-inflammatory system keep a relatively balanced in the internal environment. Once the body is infected with SARS-CoV-2, the inflammatory response dominates in the battle with the anti-inflammatory system. As a result, many inflammatory factors (TNF-α and IL-1, among others) are released and trigger a chain reaction, namely “the inflammation waterfall effect”. It results in systemic inflammatory response syndrome (SIRS), which may rapidly lead to multiple organ dysfunction syndrome (MODS). At present, the most effective and preferred method to inhibit SIRS is the use of glucocorticoids. 7 However, glucocorticoids, as a double-edged sword, inhibiting both the inflammatory response and the body’s ability to clear the invading virus.8,9 The timing of glucocorticoid use appears to be important. Perhaps early administration (within a few days of the onset of symptoms) may not be helpful or even harmful compared to glucocorticoids given 7–10 days from the onset of symptoms. The large RECOVERY trial from the UK of 175 NHS hospitals showed that of the 2104 subjects who received dexamethasone (beginning ≥7 days after onset of disease) had a lower incidence of death than the 4321 subjects who received placebo. The use of dexamethasone resulted in lower 28-days mortality among those who were receiving either invasive mechanical ventilation or oxygen alone at randomization but not among those receiving no respiratory support. 10 In our study, glucocorticoid treatment has been employed in 59.2% and 83.3% of survivors and non-survivors, respectively, among patients with severe COVID-19, with a p value of 0.118. Meanwhile, glucocorticoid treatment has been employed in 81.8% and 83.3% of survivors and non-survivors, respectively, among critically ill patients, with a p value of 1.000. In our study, glucocorticoids were given after 7 days from the onset of symptoms.11-15 Our results show that glucocorticoid administration did not reduce the mortality in patients with severe COVID-19 or critically ill patients with COVID-19.
Studies find that changes in coagulation and anticoagulation systems play a crucial role in the initiation and development of inflammatory responses in patients with SIRS and MODS.16-18 DIC is considered to be part of the overall pathological course of SIRS or MODS. DIC is always discovered in patients with COVID-19 that develops into SIRS and MODS. 19 It has been reported that 71% of COVID-19-related deaths met the DIC diagnostic criteria of the International Society on Thrombosis and Haemostasis (ISTH). 20 In line with these reports, 75% of COVID-19-related deaths meet the DIC diagnostic criteria from ISTH in this study. Viral invasion activates the complement system and causes endothelial injury, which in turn leads to coagulopathy. The study has confirmed that coagulation parameters, including the platelet count, PT, and D-dimer level, were significantly different in the survivor and non-survivor groups among patients with severe COVID-19.
Heparin is the main anticoagulant for patients with severe disease. Studies have demonstrated that heparins can reduce the rate of viral infection and increase viral clearance. 18 The underlying mechanism remains unclear, which may be related to the occupation of viral spike proteins and down-regulation of virus invasion-related receptors. Although at the theoretical level, patients with COVID-19 may benefit from anticoagulant therapy, the actual effect needs clinical verification. A retrospective study including 449 patients with severe COVID-19 has found that prophylactic dose of heparins can reduce the 28-days mortality of patients with sepsis-induced coagulopathy scores >4 or D-dimer levels >6 × ULN. It is worth noting that no difference in 28-days mortality is observed between heparin- and non-heparin-treated patients overall. 5 Another study has demonstrated that pre-admission anticoagulant therapy did not show a protective effect in patients with severe COVID-19. 21 The American Hematology Association does not recommend routine empirical anticoagulation therapy for patients with COVID-19. The results of this paper also suggest that anticoagulant therapy alone is unlikely to be protective against COVID-19-related mortality.
Glucocorticoids inhibit inflammatory responses and may increase the risk of VTE.22,23 A retrospective study of 38,765 patients with VTE and 387,650 age- and sex-matched controls has found a higher risk of VTE in patients who have undergone systemic glucocorticoid administration, 24 suggesting that glucocorticoids use is associated with increased prevalence of VTE. A case–control study has identified that low doses (daily dose of prednisolone <5 mg) of glucocorticoids would double the risk of pulmonary embolism (PE), while the highest dose of glucocorticoids (daily dose >30 mg) would confer a 10-fold increased risk of PE. 25 The increased VTE risk caused by glucocorticoids, may be associated with their effects on stimulating thrombosis and inhibiting mononuclear macrophages to eliminate activated coagulation factors. 26 Also, dexamethasone (6 mg per day) tends to increase clotting factor and fibrinogen concentrations. This possible effect of steroids in the procoagulant inflammatory environment of patients with COVID-19, in absence of a reasonable anticoagulant treatment may be catastrophic at times in severe COVID-19 patients contributing to mortality. 27
Patients with severe COVID-19 often undergo central venous catheterization and exhibit diminished mobility, which further increase the risk of VTE. 28 According to computed tomography pulmonary angiography and/or ultrasonic testing, the proportion of patients with VTE among critically ill patients is as high as 27%. 29 Cui et al. 30 also report that the incidence of VTE is as high as 25% among patients with severe COVID-19. It is easy to infer that the risk of VTE is very high in both critically ill patients with COVID-19 and patients with severe COVID-19, and anticoagulant treatment can prevent the occurrence of VTE.
Therefore, whether patients with severe COVID-19 and critically ill patients with COVID-19 can benefit from the combination of anticoagulant and glucocorticoid treatment is the primary aim in this study. Neither the chi-square test nor single-factor binary logistic regression analysis has shown statistically significant differences between survivors and non-survivors, which may have been caused by the small sample size or masked by the confounding factors. After controlling for confounding factors by adjusting for sex, age, the WBC count, and the D-dimer level, we have found that glucocorticoids combined with anticoagulant therapy can improve the survival rate in patients with severe COVID-19 and critically ill patients with COVID-19. According to the analysis of the outcomes in stratified patients, we have found that the combination of anticoagulant and glucocorticoid treatment exhibited a tendency to reduce mortality in patients with severe COVID-19, but the difference was not statistically significant (17.6% vs. 31.8%, p = 0.321). Importantly, the combination of anticoagulant and glucocorticoid therapy significantly reduced the mortality of critically ill patients with COVID-19 (30.0% vs 77.8%, p = 0.047), which suggests that with increasing disease severity, more benefits may be obtained from the combination treatment.
There have been two recent studies published in the New England Journal of Medicine on full-dose anticoagulation in patients with non-severe and severe COVID-19, reaching different conclusions. One open-label, adaptive, multiplatform, randomized clinical trial included 1098 critically ill patients with severe COVID-19 (534 assigned to therapeutic-dose anticoagulation and 564 assigned to usual-care thromboprophylaxis) found that an initial strategy of therapeutic-dose anticoagulation with heparin did not result in a greater probability of survival to hospital discharge or a greater number of days free of cardiovascular or respiratory organ support than did usual-care pharmacologic thromboprophylaxis. 31 Another open-label, adaptive, multiplatform, randomized clinical trial included 2219 patients with COVID-19 who were not critically ill, revealed that therapeutic-dose anticoagulation with heparin increased the probability of survival to hospital discharge with reduced use of cardiovascular or respiratory organ support as compared with usual-care thromboprophylaxis. 32 This prompts us that noncritically ill patients with COVID-19 may be obtained more benefits than critically ill patients. Regrettably, our study did not distinguish between therapeutic-dose anticoagulation therapy and pharmacologic thromboprophylaxis.
This research, however, is subject to several limitations. The primary limitation to the generalization of these results is the nature of retrospective study. Secondly, the sample size is small. In order to allow valid conclusions, we calculate the power at online websites of Power and Sample Size. Although the sample size is small, it still has sufficient power, which may be related to the large difference between the two groups. In spite of that, further prospective randomized controlled clinical trial is needed to validate the effect of combining anticoagulant and glucocorticoid therapy on mortality in patients with severe COVID-19.
Conclusions
The study has demonstrated that patients with severe COVID-19 or critically ill patients with COVID-19 can benefit from the combinationtherapyof anticoagulant and glucocorticoid. As disease severityincreasing, more benefits may be obtained from the combination treatment.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The research is supported by Natural Science Foundation of Hunan Province (No. 2018JJ2611), Project of Hunan Provincial Health Commission (No. 20190967) and the Educational Reform Research Project of Central South University (No. 2018jy182).
Data Availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics approval
Ethical approval for this study was obtained from the ethics committee of the Third Xiangya Hospital of Central South University (NO. 2020-S585).
Informed consent
The written informed consent is waived due to the retrospective nature of the study. All patients have been anonymized to protect the confidentiality of the individual participants.

