Abstract
Introduction
Infectious diarrhea is a common disease frequently occurring in pediatrics. Long-term diarrhea can lead to pediatric malnutrition, poor growth and development, severe dehydration or electrolyte imbalance and is even life- and health-threatening. Its prevention and treatment have been widely drew attention from clinical pediatricians. 1 At present, fluid replacement, antidiarrheal, antibacterial or antiviral methods are mostly adopted to treat pediatric infectious diarrhea in western medicine. However, some children still present some symptoms of digestive disorders after treatment, affecting growth and development, and the reasons may be related to intestinal microecological disorders and dysbacteriosis caused by multiple factors such as diarrhea and treatment of anti-infection. 2 Therefore, in the treatment of infectious diarrhea in children, it is of vital importance to supplement probiotics, regulate bacterial metabolism, correct intestinal microecological disorders, and reconstruct intestinal natural protective barriers. 3 In addition, zinc, as an essential trace element for human body, has multiple effects, including promoting growth and development, improving immune function and mucosal function. 4 Studies have pointed out that blood zinc levels in children with diarrhea are significantly lower than those in healthy ones, suggesting that diarrhea is closely related to zinc deficiency. Diarrhea can lead to zinc deficiency, and vice versa, so zinc supplementation is particularly important in the treatment of diarrhea. 5
Clostridium butyricum powders have multiple effects such as promoting intestinal mucosal repair and regeneration, nourishing intestinal tract, eliminating inflammation, and regulating intestinal bacteria balance, which is beneficial for restoring normal physiological function of the intestine. 6 Study of Fu et al. has shown that the application of Clostridium butyricum powder plus antidiarrheal in the treatment of infectious diarrhea in children can significantly improve efficacy, accelerate the improvement of symptoms, effectively reduce the level of serum inflammatory factors, and is safer. 7 Zinc gluconate can meet the needs of the human body for zinc, promote the repair of damaged intestinal mucosa, enhance intestinal absorption of water and nutrients, improve immunity, and enhance disease resistance. 8 Study of Zhang et al. has shown that conventional treatment combined with zinc gluconate in the treatment of infantile diarrhea can improve clinical efficacy, relieve symptoms, and strengthen the function of humoral immunity, cellular immunity, and red blood cell immunity. These results suggest that adjuvant therapy with the drugs mentioned above can improve the clinical efficacy in children with diarrhea. 9 A study by Guo et al. has shown that Clostridium butyricum powders combined with zinc gluconate have a prominent effect in treating acute diarrhea in children, which can effectively improve symptoms and reduce inflammatory responses, thereby reducing the pain of children. 10 However, its effects on intestinal mucosal function and intestinal bacteria have not been reported. It has also been reported in a study that the treatment of antibiotic-associated diarrhea with Combined Clostridium Butyricum and Bifidobacterium powders can effectively shorten the diarrhea and hospital stay of children, significantly increase the number of intestinal bacteria in children, and reduce inflammatory response. 11 The therapeutic effect of Clostridium Butyricum powder combined with zinc gluconate on infantile diarrhea is significant, and whether the synergistic effect of the two has a regulatory effect on intestinal bacteria has become the topic of this study. To find out this, we analyzed the clinical data of 86 children with infectious diarrhea to explore the clinical value of Clostridium butyricum powders combined with zinc gluconate.
Materials and methods
Clinical data
Comparison of clinical data between the two groups [n%; (

Inclusion and exclusion criteria
Inclusion criteria 12 : Patients were included in the study if they (1). were clinically diagnosed with infectious diarrhea and presented with symptoms including diarrhea, abdominal pain, vomiting and fever; (2). had an acute onset, with a course of disease ≤3 days; (3). did not take probiotics or zinc within 1 week before enrollment. Exclusion criteria: Patients were excluded if they (1). presented with severe fever or dehydration; (2). had dysfunction in the heart, liver, or kidney; (3). combined with disorders of the immune system, endocrine system, and blood system; (4). adhered poorly to or could not cooperate with the treatment during study.
Treatment regimen
Both groups were given conventional symptomatic and supportive treatments including fluid replacement, montmorillonite powder (Simcere Pharmaceutical Co., Ltd., China) for diarrhea, anti-infective therapy, and dietary control. Based on the treatments above, children in the control group were given Clostridium butyricum powders (Qingdao Eastsea Pharmaceutical Co., Ltd.; Approval No. S200440088; specification: 500 mg/sachet, each sachet containing at least 7.5 * 106 CFU of viable bacteria), and each time, those under 1 year old were given half a sachet while those aged one year old or above were given 1 sachet, three times a day. Besides the same regime as the control group, children in the experiment group were also given zinc gluconate oral solution (Henan Tongyuan Pharmaceutical Co., Ltd.; Approval No. H41025018; specification: 10 mL per bottle, each bottle containing 3.5 mg of active ingredient per milliliter), and those under 1 year old were given 5 mL each time, while those aged one year old or above were given 10 mL each time, once a day. Both groups were treated for 5 days.
Outcome measures
(1) Clinical efficacy
After treating for 5 days, the clinical efficacy was defined as follows: a. Ineffective: the children’s symptoms and signs were not significantly improved, and stool frequency and consistency were not recovered; b. Effective: the children’s symptoms and signs were significantly improved, and the stool frequency and consistency were significantly recovered; c. Markedly effective: the children’s symptoms and signs basically disappeared, and the stool frequency and consistency returned to normal.
12
Overall response rate = (the number of effective cases + the number of markedly effective cases)/total number of cases * 100%. (2) Recovery time of symptoms and signs
The improvement of diarrhea, abdominal pain, vomiting and body temperature was recorded during treatment, and the time of recovery from these symptoms and to normal body temperature were compared between the two groups. (3) Inflammatory factors
Serum interleukin-6 (IL-6), interleukin-17 (IL-17), and C-reactive protein (CRP) levels were compared between the two groups before and after treatment: 4 mL of blood samples were drawn from the patient and centrifuged at 3000 rpm for 10 min (centrifugal radius: 6.5 cm). Then the serum was collected for the detection of inflammatory factors using an enzyme-linked immunosorbent assay (Shanghai Enzyme-linked Biotechnology Co., Ltd).
13
(4) Intestinal mucosal function
Before and after treatment, the levels of diamine oxidase (DAO) and D-lactate were compared between the two groups: 4 mL of fasting blood was drawn at the cubital vein from the patients in the morning. Subsequently, the serum was separated using the same method as above. Then the levels of DAO and D-lactate were measured and compared by spectrophotometry.
14
(5) Intestinal microecological indicators
Before and after treatment, 0.2 g of fresh stool was obtained and detected by viable plate counting for the contents of intestinal bifidobacteria and lactobacillus, which were compared between the two groups. (6) Safety
The incidences of nausea, vomiting, constipation and other adverse reactions were statistically compared between the two groups. Incidence of adverse reactions = number of adverse reactions/total number of cases * 100%. 10
Statistical methods
SPSS 22.0 software was used to process all data. Measurement data were analyzed by Bartlett's test of homogeneity of variance and Kolmogorov-Smirnov test, and those confirmed for homogeneity of variance and following approximately normal distribution were expressed as (
Results
Clinical data
There was no significant difference in clinical data between the two groups, including gender, age, disease course, and degree of dehydration (all p > .05), which were comparable. See Table 1.
Clinical Efficacy
Comparison of clinical efficacy between two groups [n (%)].

Recovery time of symptoms and signs
Recovery time of symptoms and signs in the two groups (

Inflammatory factor
Comparison of serum IL-6, IL-17, and CRP levels between the two groups (

Note: IL-6: interleukin-6; IL-17: interleukin-17; CRP: C-reactive protein. Compared with that before treatment, ***p < .001.
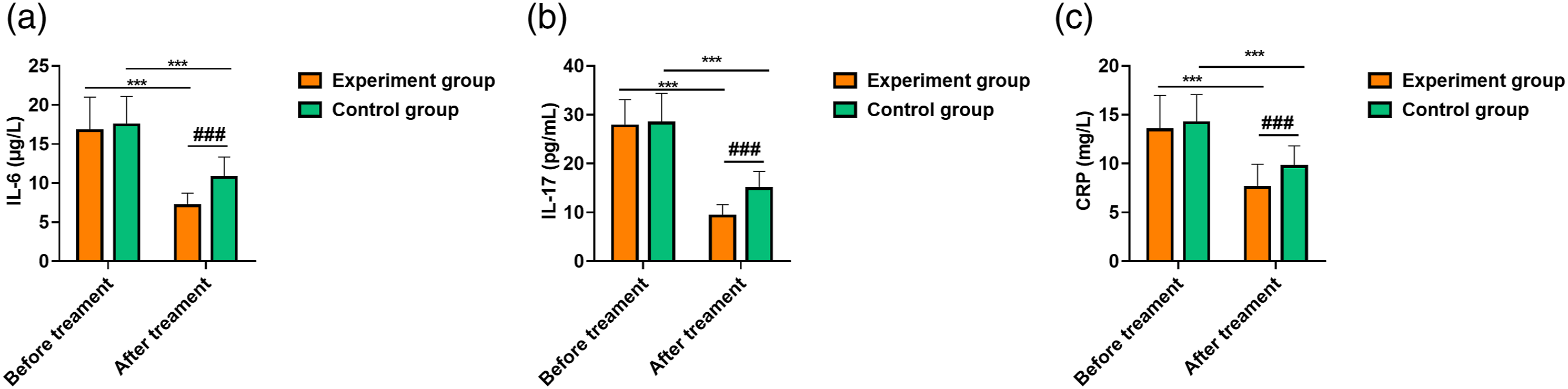
Comparison of serum IL-6, IL-17, and CRP levels between the two groups. Note: A: IL-6 (μg/L); B: IL-17 (pg/mL); C: CRP (mg/L). Compared with that before treatment, ***p < .001; compared with the control group after treatment, ###p < .001.
Intestinal mucosal function
Comparison of DAO and D-lactate levels between the two groups (

Note: DAO: diamine oxidase. Compared with that before treatment, ***p < .001.
Indicators to intestinal microecology
Comparison of intestinal bifidobacteria and lactobacillus levels between the two groups (

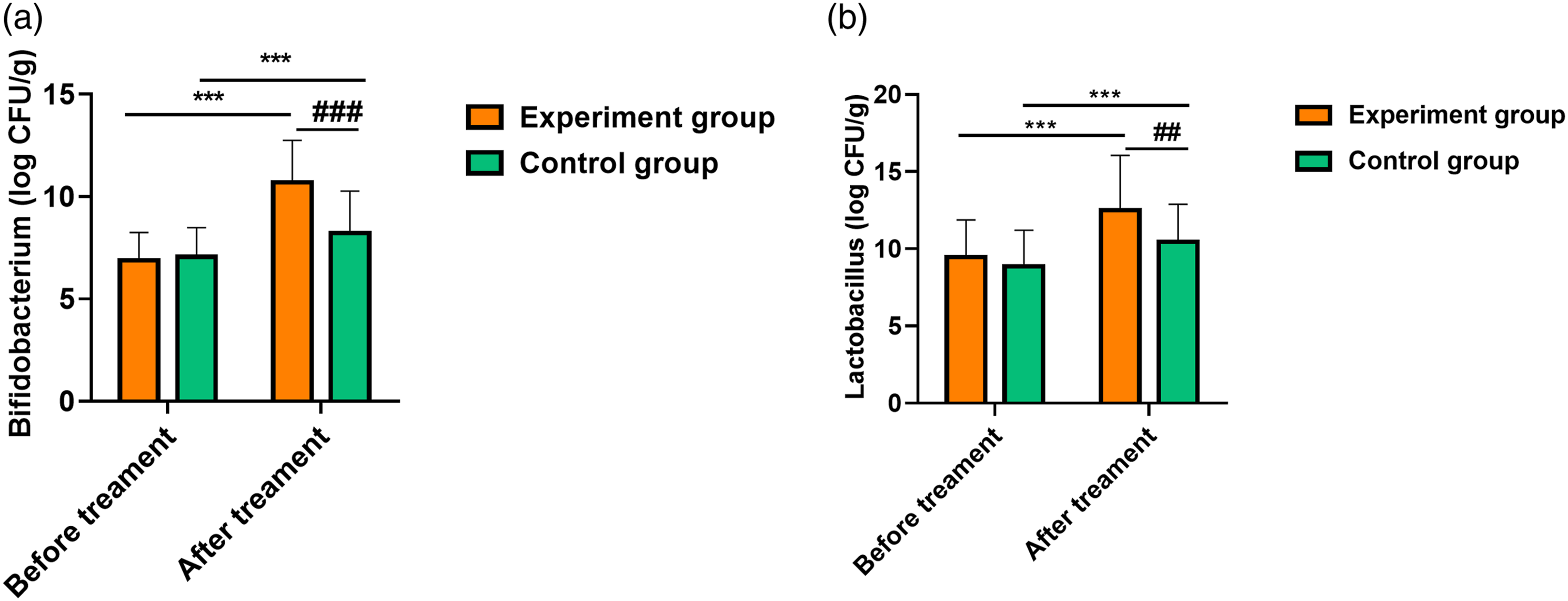
Comparison of intestinal bifidobacterial and lactobacillus levels between the two groups. Note: A: Bifidobacterium (log CFU/g); B: Lactobacillus (log CFU/g). Compared with that before treatment, ***p < .001; compared with the control group after treatment, ##p < .01, ###p < .001.
Safety
Comparison of incidence of adverse reactions between the two groups [n (%)].

Discussion
Presently, fluid replacement, antidiarrheal and antiinfective therapies are the most basic methods for clinical treatment of infectious diarrhea in children. Although these measures can correct electrolyte imbalance, improve dehydration and relieve diarrhea symptoms, some children are still difficult to completely get their benefits, leading to poor efficacy and clinical results. 15 Therefore, it has become a research priority for clinical pediatricians to select a safe and effective regimen for the treatment of pediatric infectious diarrhea.
The results of this study showed that the clinical efficacy, recovery time of symptoms and signs of the experiment group were better than those of the control group, and there was no significant difference in the incidence of adverse reactions between the two groups. The study by Lu Haimei et al. showed that the combination of probiotics and zinc supplements in the treatment of diarrhea in children could significantly shorten the length of hospital stay, and recovery time of stool frequency, stool property and body temperature, which was consistent with the results of this study. 16 These results suggest that Clostridium butyricum powders combining zinc gluconate in treating pediatric infectious diarrhea can effectively improve diarrhea symptoms and promote disease outcomes.17–20
The results of this study showed that after treatment, the serum IL-6, IL-17 and CRP levels, DAO, D-lactate levels, intestinal bifidobacteria, and lactobacillus levels in the experiment group were lower than those in the control group, suggesting that this regimen is beneficial to reduce inflammatory responses, improve intestinal mucosal function, and promote recovery of intestinal microecology. A study by Zhang Lianfeng et al. has revealed that Clostridium butyricum powders combined with ribavirin can significantly reduce the levels of TNF-α, IL-6, and CRP in children with hand-foot-mouth disease and improve clinical efficacy, which is consistent with the results of this study. 21 This result suggests that Clostridium butyricum powders combined with zinc gluconate can synergistically exert a prominent therapeutic effect through a multi-target action. This may be related to the fact that, on the one hand, Clostridium butyricum powders combined with zinc gluconate activates the immune system and improves immunity as well as the body’s anti-inflammatory ability. Meanwhile, this regime can promote the reproduction of intestinal bifidobacteria and lactobacilli, enhance bacteria activity and selectively remove intestinal harmful bacteria, thus promoting the growth of probiotics, and rapidly balances intestinal environment. On the other hand, Clostridium butyricum powders can directly promote the absorption of trace elements such as calcium, iron and zinc, vitamin K, folic acid and other nutrients in children, while appropriate supplementation of zinc gluconate helps to improve malnutrition caused by diarrhea and facilitates the recovery of the disease.
Also, the results of our study demonstrated that there was no significant difference in the incidence of adverse reactions between the experiment group (8.70%) and the control group (2.50%), which suggests that the combination of the two drugs does not increase adverse reactions and is safe and effective in clinical practice. However, tolerable side effects were found in both groups, this may be related to the fact that the sample size is rather small, which needs to be expanded in further studies.
This study is a preliminary study, which is limited to small sample size. At the same time, the follow-up time of this study is short, and we did not conduct a multicenter, double-blind, randomized controlled trial requiring a large sample size. Therefore, we will focus on a multicenter study using a larger sample size to further clarify the effects of Clostridium butyricum powders combined with zinc gluconate in the treatment of pediatric infectious diarrhea.
In addition, this study also has certain limitations. The potential confounding factors in the initial selection of treatment were not specified, and the initial content of zinc was not measured. Besides, only the children who refused zinc supplementation were selected as the control, which may lead to some bias in the results and will be considered in future study.
Conclusion
The use of Clostridium butyricum powders combined with zinc gluconate in the treatment of children with infectious diarrhea can effectively relieve clinical symptoms, down-regulate the level of serum inflammatory factors, regulate intestinal microecology, improve intestinal mucosal function, and enhance overall therapeutic effect without increasing the risk of adverse reactions, which guarantees medication safety of the patients.
Footnotes
Acknowledgements
The authors thank the reviewers for their useful comments.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Ethical approval for this study was obtained from Lanzhou University Second Hospital, (Approval Number 2020A-209).
Informed consent
Written informed consent was obtained from legally authorized representatives before the study.
