Abstract
Objective:
This study aimed to investigate the effect of rapamycin on inflammatory pain in rats.
Introduction:
Inflammatory pain is a kind of pathological pain caused by inflammatory mediators or factors such as TNF-α (Tumor Necrosis Factor-α), IL-1β (Interleukin-1β), and IL-6 (Interleukin-6). NSAIDs and opioid analgesics are commonly used for relieving inflammatory pain, but the side effects limit their clinical application. New drugs based on new mechanisms for inflammatory pain are urgently needed. Autophagy is an evolutionarily conserved homeostatic process for lysosomal degradation of intracellular components. Recent reports indicate the involvement of autophagy in the development and maintenance of neuropathic pain, but the role of autophagy in inflammatory pain still needs to be explored.
Methods:
The pain-related behaviors of rats were studied by paw withdrawal threshold and paw withdrawal latency. The autophagy level of the rat spinal cord was detected by western blots. The concentrations of TNF-α, IL-1β, and IL-6 were detected by ELISA.
Results:
We found that the paw withdrawal threshold and paw withdrawal latency were both significantly decreased after CFA (Complete Freund’s Adjuvant) injection, accompanied by the activation of mTOR signaling pathway and the inhibited autophagy flux in the spinal cord. And inflammatory cytokines were increased in the spinal cord after CFA injection. Then, we studied the effect of rapamycin on CFA-induced inflammatory pain in rats, and found that rapamycin restored the autophagy flux and significantly reduced mechanical allodynia and thermal hyperalgesia. In addition, rapamycin significantly decreased the levels of TNF-α, IL-1β, and IL-6 after CFA injection in the spinal cord.
Conclusion:
Our results suggested that rapamycin might be a promising candidate for the treatment of inflammatory pain by restoring the autophagy flux in the spinal cord.
Introduction
As one of the most common health issues, chronic pain has become a worldwide medical problem due to its refractoriness. 1 Inflammatory pain is a kind of pathological pain caused by inflammatory mediators or factors, such as TNF-α, IL-1β, and IL-6. These pro-inflammatory cytokines acidify the tissue, and activate nociceptive primary afferent neurons, leading to increased pain sensation both peripherally and centrally. 2 As many pain disorders with uncertain causes are often caused by tissue inflammation, controlling inflammation, and neuroinflammation may be the most effective measure to treat inflammatory pain.3,4 There are some drugs used clinically for relieving inflammatory pain. Nonsteroidal anti-inflammatory drugs (NSAIDs) are the preferred initial pharmacology treatment for inflammatory pain, but their clinical utility is restricted due to associated adverse effects (such as gastrointestinal tolerance and cardiovascular risks) and inadequate efficacy. 5 Opioid analgesics is also used for severe pain, however, the risk of adverse reactions, tolerance and addiction after long-term use limits their application. 6 Nowadays, we still lack drugs to treat inflammatory pain effectively. 7 Therefore, new drugs are urgently needed to treat inflammatory pain, perhaps from new mechanisms underlying the disease.
Autophagy is an evolutionarily conserved homeostatic mechanism, and interplays with inflammation. The impairment of autophagy may mediate susceptibility to inflammatory diseases. 8 There is evidence that autophagy is implicated in neuropathic pain processing. After spinal ligation (SNL), the expression of microtubule associated protein 1 light chain 3 (LC3)-II (a form of autophagy associated with LC3) was significantly increased, and ubiquitin and LC3 binding protein p62 (Sequestosome 1, SQSTM1) was also accumulated in the spinal cord, indicating that autophagy flux was blocked in the spinal cord of mice after SNL. 9 Although the expression levels of LC3-II and p62 were elevated in the spinal cord of SNL mice, there was no change in the expression of LC3-II and p62 in the spinal cords of chronic constriction injury (CCI) mice, and only the expression of Beclin1 was increased. In the spinal cord of mice after nerve injury (SNI), LC3-II expression was increased, but p62 expression was unchanged. All these results suggest that autophagy within the spinal cord is differentially regulated in different models of neuropathic pain. 10 However, until now the roles of autophagy in inflammatory pain is still elusive.
Since autophagy was functionally limited in neuropathic pain, researchers used some drugs to alleviate neuropathic pain via modulating autophagy, and rapamycin was one of these drugs. 11 Rapamycin is a typical inducer of autophagy which can inhibit mTOR, a serine/threomine kinase.11,12 In mice with CCI-induced neuropathic pain, treatment with intraplantar injection of rapamycin into the ipsilateral hind paw produces long-lasting analgesic and anti-inflammatory effects, as well as facilitating nerve regeneration and preventing pain chronicity. 13 Intrathecal rapamycin administration significantly alleviated neuropathic pain after SNL in rats. 14 Generally speaking, rapamycin can relieve neuropathic pain via enhancing autophagy function. However, the role and the underlying mechanism of rapamycin in treating inflammatory pain still lacks comprehensive investigation. Our study found that mTOR signaling pathway was activated and autophagy flux was blocked in the spinal cord of rats with CFA-induced inflammatory pain. Intrathecal injection of rapamycin restored the blocked autophagy flux and relieved CFA-induced mechanical allodynia and thermal hyperalgesia in rats, perhaps through decreasing proinflammatory factors in the spinal cord. In conclusion, our results suggested that rapamycin might be a promising candidate for the treatment of inflammatory pain.
Materials and methods
Animals
We obtained male Sprague-Dawley (SD) rats weighing between 220 and 250 g from the Animal Research Center of Anhui Medical University. Animal treatment procedures were conducted in compliance with international guidelines (NIH Publications No. 8023, revised in 1978) and the Chinese Council’s Guides for the Care and Use of Laboratory Animals. The protocols in this study were approved by the Animal Care Committee of Anhui Medical University (LLSC20231002). The animals were housed under controlled conditions with ad libitum access to food and water. The housing environment maintained a 12-h light/dark cycle, a temperature of 23 ± 1°C, and a relative humidity of 50% ± 10%.
Intrathecal catheter implantation
Rats were anesthetized with intraperitoneal pentobarbitone (40 mg/kg, Hengrui, China, given intraperitoneally). 15 A PE-10 catheter (BD America) was inserted into the rat’s L4-L5 vertebral space, directed towards the head, and advanced 3.5 cm rostrally to reach the level of the spinal cord lumbar enlargement segments, as described in previous studies. 16 After recovery for 3 days, 10 μL 2% lidocaine was administered intrathecally to confirm the success of intrathecal catheter implantation. The rats that postoperatively exhibited paralysis or non-responding to lidocaine test were excluded from the study. Successful intrathecal catheter implantation was determined by the onset of paralysis within 30 s and subsequent recovery within 30 min. Rats demonstrating these signs were selected for further study. 17
CFA-induced inflammatory pain model
After recovery from the implantation (on the seventh day after implantation), 18 100 μL Complete Freund’s Adjuvant (CFA, #SLCD6299, non-diluted, Sigma, America) was injected into the intraplantar surface of the left hindpaw, according to previous research. 19 In the Control group, a subcutaneous injection of normal saline (100 µL, 0.9%) was administered to exclude the influence of needle puncture and subcutaneous injection. 20
Behavioral test
The mechanical and thermal pain thresholds were measured at different time after CFA injection.21,22 The mechanical pain threshold, which represents the threshold of foot withdrawal in response to a mechanical stimulus, was measured using the Electronic Von Frey apparatus (IITC Life Science, USA). The force inducing the withdrawal reflex was recorded and averaged, which represented the mean value of each hind paw. 21 Thermal pain threshold was tested by measuring foot withdraw in response to heat stimulus. 22 Each rat was housed in an individual plastic cage and the mid-plantar surface of the hind paw was exposed to radiant light using the IITC Model 336 apparatus. To prevent tissue damage, the cut-off time was 20 s. Thermal pain threshold test was repeated three times. 22
Experimental design
To investigate the impact of autophagy on inflammation-induced pain caused by CFA, 24 rats were randomly allocated to either the Control group or the CFA group (n = 12). Baseline behavior test before CFA administration (day 0) was performed. In the following 1, 3, 5, 7, and 14 days after CFA injection, pain behavior tests were also performed. On the third day after modeling, the lumbar spinal cord (L4-6) was obtained, and the levels of autophagy-related proteins LC3-II and p62 in the dorsal horn of the spinal cord were analyzed by Western blotting.
In order to assess the impact of rapamycin on inflammation-induced pain by CFA, 48 rats were randomly allocated to the Control (plantar injection of saline), CFA (plantar injection of CFA), CFA + Normal saline (CFA + NS), and CFA + Rap (Rapamycin, #AY22989, MCE, America) groups (n = 12). The rats in CFA + NS group received 100 μL normal saline (0.9%), and rats in CFA + Rap group were given rapamycin through intrathecal catheter (10 μL, 0.1 μg), on the 7th day after intrathecal catheter implantation. Saline or rapamycin was delivered through PE-10 catheter once a day from day 1 to day 3 after CFA injection. 10 The dosage of rapamycin was determined based on previous literature.14,23 Following the administration of rapamycin or normal saline, the PE-10 catheter was flushed with 10 μL of normal saline after rapamycin or normal saline administration. Pain behaviors were assessed at baseline (before CFA injection) and 1 h after drug application on day 1, 3, 5, 7, and 14. 10 We injected rapamycin intrathecally into rats for three consecutive days after CFA injection, and took the lumbar spinal cord (L4-6) 1 h after rapamycin injection on the third day to monitor the expression of spinal dorsal horn-related proteins by WB experiment.
Western blot analysis
On day 3 following CFA injection, the L4-L6 spinal cord segments were extracted and homogenized in RIPA (Radio Immunoprecipitation Assay) buffer (50 mM Tris-HCl, 150 mM NaCl, 1% Triton X-100, 0.1% SDS, and 1X protease inhibitor). The total protein concentration of each sample was determined using a Bicinchoninic Acid (BCA) kit (Beyotime Biotechnology, China). After normalization, total protein extracts were subjected to SDS-polyacrylamide gel electrophoresis, and subsequentil transferred onto a polyvinylidene fluoride (PVDF) membrane (Millipore, Germany). The membrane was blocked with 5% skimmed milk power for 1 h, and was incubated with primary antibodies (LC3, 1:1000, #14600-1-AP, Proteintech Biologicals; p62, 1:1000, #18420-1-AP Proteintech Biologicals; mTOR, 1:1000, #2972, Cell Signaling Technology; Phospho-mTOR, 1:1000, #2971, Cell Signaling Technology; p70s6k, 1:1000, #14485-1-AP Proteintech Biologicals; Phospho-p70s6 kinase, 1:1000, #9234, Cell Signaling Technology; β-actin, 1:1000, #66009-1-Ig, Proteintech Biologicals) at 4°C overnight. Next, the membrane was incubated with a horseradish peroxidase conjugated secondary antibody (1:10000, #W401B, #W402B, Promega) for 1 h. The membrane was visualized with an ECL kit (#BL520A, Bioshap, Hefei, China).
Enzyme-linked immunosorbent assay (ELISA)
The spinal cord (L4-6) samples were collected on day 3 after administration. Samples was homogenized in RIPA buffer. 24 Then the concentrations of TNF-α, IL-1β, and IL-6 in the supernatant were detected using a human specific ELISA kit (CSB-E04741m, CSB-E08054m, CSB-E04639m, CUSABIO, Wuhan, China). The resulting cytokines levels were expressed as picograms per 100 mg of tissue.
Statistical analyses
The data were analyzed by SPSS software version 19.0. Descriptive statistics for the data were expressed as mean ± SD (Standard Deviation). Differences between groups were assessed using one-way ANOVA followed by Tukey’s Post-hoc test or unpaired two-tailed Student’s t-test. Statistical significance was determined at the level of *p < 0.05.
Results
Mechanical allodynia and thermal hyperalgesia after intraplantar injection of CFA
Mechanical allodynia was observed by paw withdrawal threshold (PWT) and thermal hyperalgesia was observed by paw withdrawal latency (PWL). We tested the PWT and PWL of rats before model establishment and on days 1, 3, 5, 7, and 14 after model establishment. Consistent with the findings of previous studies, intraplantar (i.pl.) injection of CFA in rats induced mechanical allodynia and thermal hyperalgesia (Figure 1(a and b)).25–27 Therefore, we believe that the inflammatory pain model was successfully established.

CFA-induced mechanical allodynia and thermal hyperalgesia on the ipsilateral hind paw. (a) Mechanical allodynia was observed with PWT. (b) Thermal hyperalgesia was observed with PWL.
Activation of mTOR signaling pathway and inhibition of autophagy flux in the spinal cord after CFA injection
To elucidate the mechanisms involved in inflammatory pain, we next studied the autophagy in the spinal cord following CFA injection. Autophagy involves the conversion of cytoplasmic LC3 (LC3-I) into the lc3-phosphatidyl ethanolamine conjugate (LC3-II), which specifically associates with the elongated autophagosome membrane. Consequently, the measurement of LC3-II levels serves as a reliable marker for monitoring autophagy. 28 P62 (SQSTM1), a crucial adaptor protein for selective autophagy, plays a pivotal role in linking ubiquitin and autophagosomes. Upon binding to LC3, p62 is transported to autophagosomes where it is efficiently degraded. Consequently, impaired autophagy results in the accumulation of p62. 29 Building upon previous findings of inhibited autophagy flux in neuropathic pain, our study aimed to investigate the autophagy flux in of the spinal cord of rats with inflammatory pain. On the third day after modeling, we took the lumbar spinal cord (L4-6) and performed WB experiments to detect the expression of autophagy-related proteins p62 and LC3II in the dorsal horn of the spinal cord. In the CFA group, there was a significant increase in the expression of p62 and LC3-II compared to the Control group (Figure 2(a–c)), indicating the inhibition of autophagy flux in the context of inflammatory pain. As the same time, our results showed that p62 in the spinal dorsal horn increased on the side of CFA injection, but not on the contralateral side. We also found that p62 was mostly co-labeled in neurons and only partially co-labeled with microglia and astrocytes (Figure S1).

The expression of mTOR, p-mTOR, p70s6k, p-p70s6k, p62, and LC3-ӀӀ in the ipsilateral spinal cord after CFA injection. (a, b) The relative expression of p-mTOR was increased after CFA injection (n = 4 per group). (a, c) The relative expression of p-p70s6k was increased after CFA injection (n = 4 per group). (d, e) The relative expression of LC3-ӀӀ was increased after CFA injection (n = 4 per group). (d, f) The relative expression of p62 was increased after CFA injection (n = 4 per group).
Next, we studied the changes of mTOR signaling pathway in the spinal cord after CFA administration. the expression of mTOR and p70s6k (70-kDa) ribosomal protein S6 kinase) in the spinal cord of CFA group had no significant change compared to the Control group, but the expression of p-mTOR and p-p70s6k was significantly increased (Figure 2d–f), suggesting the activation of the mTOR signaling pathway in rats with inflammatory pain. Our data suggested activated mTOR signaling pathway and inhibited autophagy flux in the spinal cord of rats after CFA injection.
Inhibition of mTOR signaling pathway and recovery of autophagy flux by intrathecal rapamycin administration
To evaluate the restorative effect of intrathecal rapamycin on autophagic flux, we injected rats with intrathecal rapamycin for three consecutive days after model establishment, and obtained the lumbar (L4-6) spinal cord 1 h after drug injection on the third day. Western blot experiments were used to detect the expression of LC3-II and p62 in the spinal cord. The CFA group showed a significant increase in the expression of LC3-II and p62compared to the Control group (p < 0.05) (Figure 3a–c). Normal saline did not affect the expression of LC3-II and p62 in spinal cord. But rapamycin treatment significantly reduced the expression of p62 (p < 0.05) (Figure 3a–c), indicating that rapamycin treatment could recover the autophagy flux in the spinal cord in rats with CFA-induced inflammatory pain.

Inhibition of mTOR signaling pathway and recovery of autophagy flux by intrathecal administration of rapamycin. (a) Western blot analysis of p70s6k and p-p70s6k expression. (b) Densitometric analysis of the protein levels of p70s6k and p-p70s6k (n = 3 per group). (c) Western blot analysis of LC3-ӀӀ and p62 expression, with β-actin serving as an internal reference. (d, e) Densitometric analysis of the protein levels of LC3-ӀӀ and p62 (n = 5 per group).
We then evaluated whether intrathecal injection of rapamycin inhibited mTOR signaling pathway in the spinal cord in rats with CFA-induced inflammatory pain. Compared with the Control group, the expression of p-p70s6k in CFA group was significantly increased (Figure 3d and e). Normal saline did not affect the expression of p-p70s6k, but rapamycin treatment significantly reduced the expression of p-p70s6k in the spinal cord (Figure 3d and e). It is suggested that rapamycin can restore autophagy flux in the spinal cord in rats with inflammatory pain by inhibiting mTOR signaling pathway.
Suppression of proinflammatory factors by intrathecal rapamycin administration after CFA injection
Autophagy interacts with inflammation in some diseases. We then revealed weather rapamycin could suppress the inflammation in rats with CFA-induced inflammatory pain. To evaluate the effect of rapamycin on some proinflammatory factors, we injected rapamycin intrathecally to rats for three consecutive days after model establishment, and obtained the lumbar (L4-6) spinal cord 1 h after drug injection on the third day to monitor the levels of TNF-α, IL-1β, and IL-6 in the spinal cord by ELISA. As shown in Figure 4, CFA resulted in a remarkable increasement of IL-1β, IL-6, and TNF-α, compared with Control group (Figure 4a–c). Intrathecal administration of rapamycin significantly reduced these proinflammatory factors, and intrathecal NS (normal saline) showed no effect (Figure 4a–c). In the Western Blot experiment, similar to the results of ELISA, the expression of TNF-α, IL-1β and IL-6 is increased after CFA injection, and the expression of these inflammatory factors is decreased after Rap administration (Figure S2). These results demonstrated that intrathecal rapamycin administration effectively suppressed inflammation in the context of inflammatory pain.

Effect of intrathecal administration of rapamycin and NS on the expression of proinflammatory factors in the spinal cord after CFA injection. (a) ELISA analysis of TNF-α. (b) ELISA analysis of IL-1β. (c) ELISA analysis of IL-6.
Amelioration of CFA-induced mechanical allodynia and thermal hyperalgesia by intrathecal rapamycin administration
As shown above, intrathecal rapamycin administration restored the autophagy flux and decrease the inflammation in rats with inflammatory pain, we then explored the changes of mechanical allodynia and thermal hyperalgesia after intrathecal rapamycin administration. We injected rapamycin intrathecally into rats for three consecutive days after modeling, and performed PWT and PWL experiments before modeling and on days 1, 3, 5, 7, and 14 after modeling. Following CFA injection, there was a significant decrease in both the PWT and PWL (p < 0.05), as observed in the CFA group compared to Control group. Intrathecal rapamycin administration in the CFA + Rap group led to a significant increase in both PWT and PWL, when compared to both the CFA and CFA + NS groups (p < 0.05). (Figure 5a and b). But NS did not exert a significant effect on PWT and PWL (Figure 5a and b). The findings demonstrated that rapamycin ameliorated CFA-induced mechanical allodynia and thermal hyperalgesia.
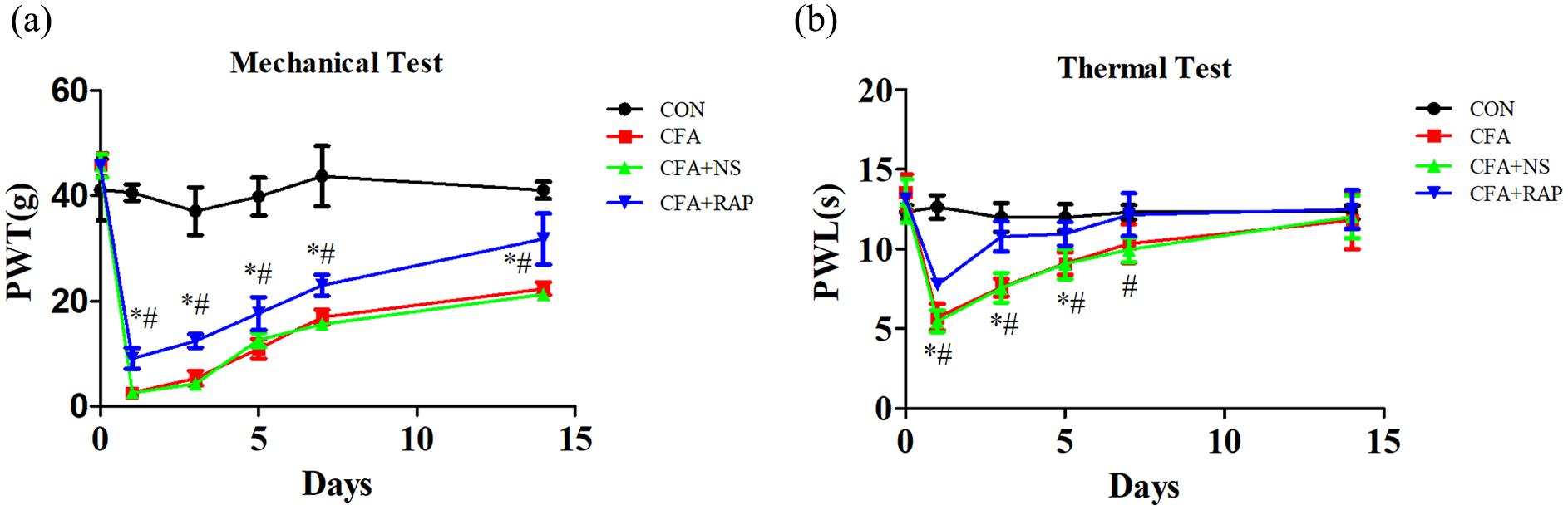
Amelioration of CFA-induced mechanical allodynia and thermal hyperalgesia by intrathecal rapamycin. (a) Effect of intrathecal administration of rapamycin on CFA-induced mechanical allodynia. (b) Effect of intrathecal administration of rapamycin on CFA-induced thermal hyperalgesia.
Discussion
Pain is a self-protection mechanism and a key feature of many diseases, especially inflammation-related diseases. More than 30% of people suffer from pain worldwide, 30 whose lives are disturbed by persistent pain problem. According to the etiology and clinical manifestations, the most prevalent forms of pain encompass nociceptive pain, inflammatory pain, and neuropathic pain. 31 Besides, all types of pain are closely related to inflammation or neuroinflammation, especially inflammatory pain. 30 Thus, control of inflammation and neuroinflammation is an effective management for inflammatory pain treatment. Additionally, prolonged utilization of analgesic medications, such as NSAIDs and opioid, presents serious side effects, and affects patients’ quality of life. 32 So, these is an urgent need for new drugs based on novel mechanisms to treat inflammatory pain. The present study aimed to investigate the analgesic and anti-inflammation potential of rapamycin in inflammation pain. In our study, inflammatory pain activated the mTOR signaling pathway in the dorsal horn of the rat spinal cord and blocked autophagic flux, thereby increasing the production of inflammatory factors in the spinal cord and causing mechanical allodynia and thermal hyperalgesia. It is worth noting that rapamycin can restore blocked autophagy flow, reduce the production of inflammatory factors, and relieve mechanical and thermal hyperalgesia by inhibiting the mTOR signaling pathway in the rat spinal cord (Figure 6). These results also provide some theoretical support for the use of rapamycin to treat inflammatory pain.

Schematic diagram: Intrathecal rapamycin administration restored the autophagy flux by inhibiting mTOR signaling pathway in spinal cord, and had analgesia and anti-inflammation effect in rats with CFA-induced inflammatory pain.
Recent investigations have studied the association between autophagy and neuropathic pain, revealing that autophagy dysfunction underlay neuropathic pain and modulating autophagy could attenuate pain-related behaviors.33–35 For instance, rapamycin could alleviate SNL-induced neuropathic pain by activation of autophagy and inhibition the expression of IL-1β in rat. 14 Nevertheless, how autophagy influences inflammatory pain remains poorly understood. Therefore, the objective of this study was to investigate the contributions of autophagy to CFA-induced inflammatory pain. Our findings indicated that CFA-induced inflammatory pain was associated with impaired autophagy in the spinal cord. However, intrathecal administration of rapamycin successfully alleviated CFA-induced inflammatory pain and restored the inhibited autophagy flux in the spinal cord. Following intrathecal rapamycin administration, CFA-activated mTOR signaling pathway was inhibited, suggesting that rapamycin restored autophagy by suppressing the mTOR signaling pathway in the spinal cord of rats with inflammatory pain. Our findings provided evidence for the significant involvement of autophagy dysfunction in CFA-induced inflammatory pain, and suggested that targeting mTOR might hold promise as a molecular intervention for the treatment of inflammatory pain.
Neuroinflammation arises as a localized inflammatory response within both the peripheral nervous system (PNS) and central nervous system (CNS) in response to various factors such as trauma, bacterial or viral infection, autoimmunity, and toxin, etc.36,37 Neuroinflammation is characterized by activation and infiltration of leukocytes, as well as activation of glial cells, and increasement of inflammatory mediators, all of which play a critical role in the maintenance of pain.36,37 Previous researches have established the significant involvement of neuroinflammation in various neuropathic diseases, Parkinson’s disease, 38 and Alzheimer’s disease. 39 Neuroinflammation plays an indispensable role in inflammatory pain, serving as a hallmark of this condition. Lu 40 demonstrated the efficacy of intrathecal administration of docosahexaenoic acid in alleviating carrageenan-induced inflammatory pain by suppressing neuroinflammation within the spinal cord. In the present work, we observed an increase in TNF-α, IL-1β, and IL-6 levels within the spinal cord after CFA application, consistent with prior investigations. 41 These inflammatory mediators were improved to take vital part in pain pathway via activating nociceptors sensitization directly or triggering production of other pain mediators indirectly.42–44 After intrathecal administration of rapamycin, the expression of CFA-increased TNF-α, IL-1β, IL-6 was decreased, compared with intrathecal administration of normal saline. The results suggested that rapamycin might alleviate inflammatory pain via inhibiting the expression of pro-inflammation factors within the spinal cord.
Currently, the main treatment for inflammatory pain is pharmacologic approaches, a complex process requiring professional clinical training and supervision. Commonly employed pharmacological interventions for the management of inflammatory pain in clinical practice are: non-steroidal anti-inflammatory drugs (NSAIDs), weak opioids (tramadol), opioids (morphine), antidepressants (chlorpromazine) and antiepileptic drugs (carbamazepine). 32 Nevertheless, these pharmacological agents are associated with a range of adverse events, including but not limited to nausea, vomiting, dizziness, drowsiness, blurred vision, gastrointestinal bleeding, sleep disorders, etc. 32 Considering the serious side effects of existing drugs, there is a pressing need for the development of novel therapeutics to treat neuropathic pain. Rapamycin, as an autophagy inducer, exerts a protective role in the context of neuropathic pain. Intrathecal injection of rapamycin could increase the autophagy level of microglia cells in spinal dorsal horn, inhibiting the release of IL-1, and alleviating the neuropathic pain symptoms. 14 Our results suggested that rapamycin exerted an anti-inflammation effect and analgesic effect in the context of CFA-induced inflammatory pain. In our opinion, this study represents the initial exploration of rapamycin’s analgesic effect in inflammatory pain. Intensive studies about how autophagy affects neuroinflammation are needed.
There are still limitations in this study. Firstly, the sample sizes selected in this study are based on similar studies, and the statistical results indicate that the sample sizes are large enough. Secondly, there are three primary types of autophagy: macroautophagy, microautophagy, and chaperone-mediated autophagy (CMA), 45 and we didn’t distinguish specific type of autophagy in this study. Investigating the efficacy of other drugs or techniques that activate autophagy in the context of inflammatory pain represents an essential avenue for future research.
Conclusions
We found that autophagy flux was inhibited in the spinal cord of rat experiencing CFA-induced inflammatory pain. Rapamycin treatment recovered autophagy flux by inhibiting the mTOR signaling pathway, and suppressed the production of TNF-α, IL-1β, and IL-6 in the spinal cord, as well as mitigated CFA-induced mechanical allodynia and thermal allodynia (Figure 6). These findings suggest that rapamycin holds promise as a potential therapeutic candidate for inflammatory pain.
Supplemental Material
sj-docx-1-iji-10.1177_03946320251317284 – Supplemental material for Rapamycin ameliorates inflammatory pain via recovery of autophagy flux mediated by mammalian target of rapamycin (mTOR) signaling pathway in the rat spinal cord
Supplemental material, sj-docx-1-iji-10.1177_03946320251317284 for Rapamycin ameliorates inflammatory pain via recovery of autophagy flux mediated by mammalian target of rapamycin (mTOR) signaling pathway in the rat spinal cord by Jiawei Zhang, Shi Chen, Rongyi Zhang, Xiaoting Zheng, Chang Liu, Jiqian Zhang, Lei Zhang, Zhilai Yang and Likui Wang in International Journal of Immunopathology and Pharmacology
Supplemental Material
sj-docx-2-iji-10.1177_03946320251317284 – Supplemental material for Rapamycin ameliorates inflammatory pain via recovery of autophagy flux mediated by mammalian target of rapamycin (mTOR) signaling pathway in the rat spinal cord
Supplemental material, sj-docx-2-iji-10.1177_03946320251317284 for Rapamycin ameliorates inflammatory pain via recovery of autophagy flux mediated by mammalian target of rapamycin (mTOR) signaling pathway in the rat spinal cord by Jiawei Zhang, Shi Chen, Rongyi Zhang, Xiaoting Zheng, Chang Liu, Jiqian Zhang, Lei Zhang, Zhilai Yang and Likui Wang in International Journal of Immunopathology and Pharmacology
Supplemental Material
sj-docx-3-iji-10.1177_03946320251317284 – Supplemental material for Rapamycin ameliorates inflammatory pain via recovery of autophagy flux mediated by mammalian target of rapamycin (mTOR) signaling pathway in the rat spinal cord
Supplemental material, sj-docx-3-iji-10.1177_03946320251317284 for Rapamycin ameliorates inflammatory pain via recovery of autophagy flux mediated by mammalian target of rapamycin (mTOR) signaling pathway in the rat spinal cord by Jiawei Zhang, Shi Chen, Rongyi Zhang, Xiaoting Zheng, Chang Liu, Jiqian Zhang, Lei Zhang, Zhilai Yang and Likui Wang in International Journal of Immunopathology and Pharmacology
Supplemental Material
sj-docx-4-iji-10.1177_03946320251317284 – Supplemental material for Rapamycin ameliorates inflammatory pain via recovery of autophagy flux mediated by mammalian target of rapamycin (mTOR) signaling pathway in the rat spinal cord
Supplemental material, sj-docx-4-iji-10.1177_03946320251317284 for Rapamycin ameliorates inflammatory pain via recovery of autophagy flux mediated by mammalian target of rapamycin (mTOR) signaling pathway in the rat spinal cord by Jiawei Zhang, Shi Chen, Rongyi Zhang, Xiaoting Zheng, Chang Liu, Jiqian Zhang, Lei Zhang, Zhilai Yang and Likui Wang in International Journal of Immunopathology and Pharmacology
Footnotes
Acknowledgements
We thank professor Xuesheng Liu and Er-wei Gu for proof readings and valuable comments.
Author contribution
Zhilia Yang and Likui Wang are both corresponding authors and designed the research. Jiawei Zhang, Shi Chen, Rongyi Zhang, Xiaoting zheng, Chang Liu, Jiqian Zhang and Lei Zhang performed the research and analyzed the data. All authors contributed to the writing and critical assessment of the manuscript. Jiawei Zhang, Shi Chen, and Rongyi Zhang contributed equally to this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by Scientific Research Project of Anhui Provincial Health Commission (No. AHWJ2023A20031), Fund Project of Anhui Medical University (No. 2023xkj150), National Natural Science Foundation of China (No. 81500949) and Excellent Young Talents Fund of Higher School in Anhui Province (No. gxyq2018009) to Zhilai Yang.
Ethics approval
The protocols in this study were approved by the Animal Care Committee of Anhui Medical University.
Animal welfare
Animal treatment procedures were conducted in compliance with international guidelines (NIH Publications No. 8023, revised in 1978) and the Chinese Council’s Guides for the Care and Use of Laboratory Animals.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
