Abstract
Diclofenac (DIC) is administrated to treat pain, inflammatory disorders, and dysmenorrhea but kidney problems are the main worries of the agent. The literature has revealed that quercetin (QR) has anti-inflammatory and antioxidant attributes. This study aims to highlight the possible nephroprotective effects of QR on DIC-exposed rats.
In this study, the animals after exposure to DIC (50 mg/kg, i.p) were administrated to QR (100 mg/kg, p.o). Then, the levels, as well as the activity of several oxidant and anti-oxidant mediators, were evaluated.
Our results showed that DIC treatment was coupled with the elevation in the levels of malondialdehyde (MDA), nitric oxide (NO), and some pro-inflammatory factors such as TNF-α, NF-κB, and IL-1β, suggesting that probably this agent exert its toxicity in the kidney tissue through inducing both oxidative stress and inflammation. Interestingly, QR was successful in restoring the activity of antioxidant compounds such as GSH, GPx, SOD, and CAT in the kidney tissue of DIC-treated rats. Moreover, in the presence of QR, DIC was unable to increase the expression of pro-inflammatory cytokines, suggesting that perhaps QR might have anti-inflammatory properties. In agreement with this, the results of the histopathological evaluation also showed that while DIC increased the lymphocyte infiltration into the kidney tissue, QR reduced the number of lymphocytes in DIC-treated rats.
The results revealed that QR exerted a supportive effect against diclofenac-induced renal injury in male rats through modulation of oxidative stress and mitigation of inflammatory response.
Introduction
Diclofenac (DIC) is the most prescribed global non-steroidal anti-inflammatory drug (NSAID) that is administrated for alleviating pain and treating a wide array of inflammatory disorders. 1 Nevertheless, its harmful impact on the kidney represents a real obstacle to its clinical application. 2 As a potent inhibitor of cyclooxygenase, it reduces the prostaglandin biosynthesis from arachidonic acid. 3
DIC triggers mitochondrial permeability transition pores liberating cytochrome c which produces excessive free radicals and, in this manner, induces caspase cascade and lipid peroxidation culminating at cellular apoptosis. 4 It also hinders the mitochondrial electron transport chain from ATP formation in the renal cells. 5 It is bio-transformed to benzoquinineimines which are involved in shifting redox homeostasis towards the pro-oxidant side leading to severe renal damage. 6 Accumulating evidence that arises from laboratory animal studies emphasizes its nephrotoxicity manifested by induction of DNA fragmentation and oxidative stress and appearance of several histopathological lesions, along with an increase in renal damage markers.7,8 Therefore, the application of cytoprotective antioxidant compounds to alleviate drug-induced nephrotoxicity is worthwhile, especially the phytochemical ones in light of their safety and bioavailability.
Quercetin (QR), a flavonoid found in different plants, vegetables, and seeds, 9 is widely used as an ingredient in many foods and beverages. However, what has attracted attention to QR from the medical perspective was the anti-oxidant ability of the agent. 10 The polyphenolic compound within the QR exerts its anti-oxidant properties through scavenging free radicals and seizing oxidative chain reactions. 11 Moreover, by targeting the PI3K/Akt signaling axis, it has been suggested that QR could reduce the expression of anti-apoptotic-related proteins such as Bcl-w.12,13 It has also been indicated that QR might have the ability to non-specifically suppress a wide range of protein kinases.14,15
Identification of the functions of naturally occurring antioxidants in counteracting the damages caused by NSAID has utmost significance in formatting promising complementary nutritional approaches inconjunct with chemotherapy to attenuate its undesirable effects on vital organs as the kidney. Thus, the objective of the present investigation is to examine whether QR could attenuate DIC-induced renal toxicity in male rats.
Material and methods
Animals
To evaluate the effect of DIC and QR (Sigma, St. Louis, MO) on renal function, 6–8 weeks old male Wistar rats (200 ± 20 g) were chosen (JondiShapour University, Iran) and were housed in the animal facility within the optimal condition. The procedure of this study was conducted according to the approved guidelines of Iran University of Medical Sciences Ethics Committee, Tehran, Iran (Ethic number IR. IUMS. REC. 99. 246).
Study design
After housing for about a week in the laboratory condition, 32 male rats were divided into four groups. The sample size for each group (n = 8) was selected based on previous studies that described by Akca et al. and El-Boshy et al.16,17 Group 1 which was the control group only were intraperitoneally treated with 1 ml/kg of normal saline. Group 2 of rats were intraperitoneally injected with 50 mg/kg of DIC.18,19 Group 3 or QR-treated rats were orally exposed to 50 mg/kg. And, group 4 were simultaneously treated with DIC (50 mg/kg, i.p.) and QR (50 mg/kg, p.o.).20,21 All the treatments were continued for 5 consecutive days. Then, for evaluating the effects of the compounds, the 12-h fasted rats were sacrificed by anesthetizing and both whole blood and the kidney tissues were collected. For preparing the serum, whole blood samples were centrifuged for 10 min at 3000 r/min. Of note, the serum samples were stored at −20°C until use. For histopathological examination, a part of the collected kidney tissue was fixed in 10% neutral buffered formalin. And, for evaluating the alteration in the activity of some oxidative as well as an anti-oxidant compound, a part of the kidney tissues were also stored at −20°C.
Weight and organ index assays
To could compare the weight of the rats in response to the treatments, this parameter was measured both at the beginning of the experiments and after 5 days of drug exposure. Moreover, the kidney of the sacrificed rats was also weighed.
Biochemical analysis
In the present study, an auto-analyzer system (BT3000, Rome, Italy) was used for evaluating the effects of the compound on the serum levels of Uric acid (UA), creatinine (Cr), GOT, GPT, and urea (Pars Azmun Co., Iran).
In Pars Azmon’s uric acid colorimetric assay kit, uricase acts on uric acid to produce allantoin, carbon dioxide, and hydrogen peroxide. Hydrogen peroxide in the presence of peroxidase reacts with a chromogen (amino-antipyrine and Tribromo-hydroxybenzoic acid) to form a colored complex. The absorbance measured at 505 nm is proportional to the amount of uric acid in the specimen.
In Pars Azmon’s Creatinine (Cr) colorimetric assay kit, Cr forms a color complex with alkaline picrate. The intensity of the color created is proportional to the amount of creatinine in the sample and the color is measured at 520 nm.
In Pars Azmon’s urea colorimetric assay kit, urea is enzymatically hydrolyzed by urease to yield ammonia and carbon dioxide. The ammonia and α-oxoglutarate are converted to glutamate in a reaction catalyzed by L-glutamate dehydrogenase (GLDH). Simultaneously, a molar equivalent of reduced NADH is oxidized. Two molecules of NADH are oxidized for each molecule of urea hydrolyzed. The rate of change in absorbance at 340 nm, due to the disappearance of NADH, is directly proportional to the urea concentration in the sample.
Pars Azmon’s GOT Colorimetric Assay Kit is a rapid and simple assay in which GOT catalyzes the transfer of an amino group from L-aspartate to 2-oxoglutatare and the products of this reversible transamination reaction are oxoglutarate and L-glutamate. The oxoglutatare and NADH are converted to malate and NAD by the enzyme malate dehydrogenase. The reduction in NADH absorbance at 340 nm is proportionate to GOT activity.
In Pars Azmon’s GPT Colorimetric Assay Kit, an amino group is transferred from L-alanine to 2-oxoglutatare. The products of this reversible transamination reaction include pyruvate and L-glutamate. GPT activity is measured by monitoring the rate of NADH oxidation in a coupled reaction system applying lactate dehydrogenase. The oxidation of NADH to NAD+ is accompanied by a reduction in absorbance at 340 nm.
Plasma antioxidant capacity assay
Ferric reducing/antioxidant power (FRAP) method was used to evaluate whether the compounds could alter PAC levels. The procedure of this test has been described in the previous study of Nouri et al. 22 The plasma of drugs-treated rats was subjected to a reagent consisting of 300 mm acetate buffer, 10 mm TPTZ (trypyridyl-s-triazine), 40 mm HCl, and 20 mm FeCl3 for 10 min at 37°C. Then, the optical absorbance (OD) was measured at 593 nm. FeSO4 was used for normalizing the serum levels of FRAP.
Preparation of tissue homogenate
To prepare 10% w/v homogenate, 0.1 g of the kidney was homogenized in 1 ml of 0.1 M phosphate buffer using a homogenizer (Disperser, Germany). The homogenates were centrifuged at 10,000 r/min for 15 min and the supernatants were kept frozen at −20°C for subsequent oxidant/antioxidant analysis.
Evaluating the nitrite content
Greiss reagent (Sigma, St. Louis, MO) was used for evaluating the level of nitrite in the kidney samples. 23 The mixture of kidney supernatant and Greiss reagent (sulfanilamide 1.0%, orthophosphoric acid 2.5%, and N-(1-naphthyl) ethylenediamine 0.1%) was incubated at 37°C for 30 min. The optical absorbance (OD) was evaluated at 548 nm. The standard curve was obtained and the levels of nitrite in the kidney tissue were assessed according to the standard curve and reported as μM/mg.
Measurement of serum malondialdehyde
MDA concentration was measured by HPLC device (Agilent, USA) and according to Agarwal method. 24 In this method, 50 μl of blood serum or homogenate along with 50 μl of 0.05% solution of BHT, 400 μl of 0.44 mm phosphoric acid, and 100 μl of 42 mm thiobarbituric acid solution were poured into the microtube. After incubating the samples at 100°C, they were cooled and 250 μl of butanol was added to the microtube to extract the malondialdehyde-thiobarbituric acid complex and vortexed for 5 min at 14,000 r/min. 20 μl of the supernatant is directly injected into an HPLC apparatus.
Evaluating the intracellular levels of glutathione
The Ellman method was used to evaluate the GSH levels in the kidney tissue of the rats after exposure to the drugs. 25 In the presence of GSH in the tissue, DTNB (5,5-dithiobis-2-nitrobenzoic acid) would undergo reduction to produce a yellow color product that is detectable at 412 nm. The alteration in GSH levels was evaluated by Shimadzu spectrophotometer (Japan) and the results are presented as μmol/g.
Evaluating of Tissue glutathione peroxidase activity
For evaluating the activity of GPx, GSH (Sigma, St. Louis, MO), H2O2 (Merck, Germany), and NaN3 were added to samples and the reduction of GSH was measured. 26
Determination of tissue catalase and superoxide dismutase activities
To determine whether QR and DIC could alter the activity of CAT and SOD in the kidney tissue, the same procedure described by Heidarian et al. 27 was applied. In brief, homogenate and H2O2 solutions were mixed. The amount of H2O2 reduction was evaluated at the wavelength of 240 nm. For SOD evaluation, the inhibition of nitro blue tetrazolium (NBT) (Sigma, St. Louis, MO) was examined in the kidney tissues at 560 nm. 28 In all tests, the Bradford assay was used for protein extraction, and data were illustrated as U/mg protein. 29
Determination of serum IL-1β, NF-κB, and TNF-α
To determine the serum levels of IL-1β, NF-κB, and TNF-α, an ELISA-based kit (BT-Laboratory, China) was used (Cat.No: E0119Ra, E0290Ra, E0764Ra, respectively). All reagents, samples, and standards were prepared, and then samples and ELISA reagents were added into each well and incubated for 1 h at 37°C. In the next step, the plate was washed five times, substrate solutions A and B were added, and the plate was incubated for 10 min at 37°C. In the end, after the addition of the stop solution, the color would be developed. The OD value within 10 min was read.
Gene expression analysis (Quantitative reverse transcription PCR Method)
Primer sequences in this study.
Histopathological examination
After fixing the kidney tissue of the rats in a 10% formalin buffer solution, embedding them paraffin, and slicing them by a microtome (AMR 400, Amos Scientific, Australia), each 3 μm section were dyed with hematoxylin and eosin (H&E). 30 The changes in the kidney tissue upon drug exposure were evaluated using a microscope (Olympus Tokyo, Japan).
Statistical analysis
Statistical significance was determined with the One-way analysis of variance (ANOVA) and Tukey’s post hoc test. The presented data are the mean ± SD of three independent experiments. SPSS software (version 20.0) SPSS Inc., Chicago, Illinois, USA) was used for data analysis. The P-value less than 0.05 was considered statistically significant.
Results
Changes in body weight and organ index
Effect of quercetin on changes in body mass and kidney index.

Data are expressed as mean ± SD (n = 8) and analyzed by one-way ANOVA followed by Tukey post hoc test. Control: received normal saline; DIC-only: treated by diclofenac; QR-only: treated by quercetin; and DIC + QR: treated by diclofenac plus quercetin. Kidney index (mg/g) = Kidney weight (mg)/Bodyweight (g).
ap < .05 versus control group.
bp < .05 versus DIC-only treated group.
Effect of quercetin on indicators of serum renal function
Effect of quercetin on serum biochemical parameters.

Data are expressed as mean ± SD (n = 8) and analyzed by one-way ANOVA followed by Tukey post hoc test. Control: received normal saline; DIC-only: treated by diclofenac; QR-only: treated by quercetin; and DIC + QR: treated by diclofenac plus quercetin.
ap < .05 versus control group.
bp < .05 versus DIC-only treated group.
Anti-oxidant effects of quercetin was mediated through increasing plasma antioxidant capacity, and diminishing nitrite content and malondialdehyde levels
Effect of quercetin on ferric reducing/antioxidant power, nitrite content, and malondialdehyde levels in the experimental groups.

Data are expressed as mean ± SD (n = 8) and analyzed by one-way ANOVA followed by Tukey post hoc test. Control: received normal saline; DIC-only: treated by diclofenac; QR-only: treated by quercetin; and DIC+QR: treated by diclofenac plus quercetin.
ap < .05 versus control group.
bp < .05 versus DIC-only treated group.
cp < .05 versus QR-only treated group.
Effects of quercetin on antioxidant activity markers and GSH level
Figure 1 shows the effects of quercetin on CAT, SOD, GPx activities, and GSH levels. A significant depletion was observed in CAT and SOD activities in group 2 (DIC-treated rats) compared to the control group (p < .05) (Figure 1(a) and (b)). Moreover, the activity of GPx and GSH levels were also remarkably declined in DIC-treated rats in comparison with the untreated control rats (p < .05) (Figure 1(c) and (d)). On the other hand, while the single agent of QR did not have any effect on the activity and the level of the aforementioned molecules when this agent was administrated to the rats after exposure to DIC (group 4), it was able to remarkably increase CAT, SOD, GPx activities, and GSH levels as compared to group 2 (p < .05). Effect of quercetin on activities of CAT (A), SOD (B), GPx (C), and level of GSH (D) in renal tissue. Each value represents the mean ± SD of eight rats. Control: received normal saline; DIC-only: treated by diclofenac; QR-only: treated by quercetin; and DIC+QR: treated by diclofenac plus quercetin. ap < .05 versus control group, bp < .05 versus DIC-only treated group, cp < .05 versus QR-only treated group.
Effect of quercetin on inflammatory cytokines
Our results showed that unlike QR which did not alter the expression level of TNF-α, IL-1β, and NF-kB as compared to the control group, DIC treatment was remarkably coupled with the elevation in the mRNA expression levels of the inflammatory cytokines Figure 2(a)–(c). Interestingly, in the rats treated with DIC and QR, we observed a decrease in the expression levels of TNF-α, IL-1β, and NF-κB as compared to group 2 (p < .05), suggesting that probably QR could ameliorate inflammatory effects of DIC. In agreement with the results obtained from the gene expression analysis, we also found that treatment with DIC led to increment in the serum levels of TNF-α, IL-1β, and NF-κB (P < .05) (Table 5). Moreover, in comparison with the untreated rats, the levels of these factors were not altered in the rats treated with the single-agent of QR. The protective effects of QR on kidney tissue became more evident when we found that the levels of TNF-α, IL-1β, and NF-κB declined significantly in rats that were treated with both DIC and QR as compared to DIC-treated rats (p < .05) (Table 5). Effect of quercetin on genes expression of TNF-α (A) IL-1β (B) and NF-κB (C). Each value represents the mean ± SD of eight rats. Control: received normal saline; DIC-only: treated by diclofenac; QR-only: treated by quercetin; and DIC+QR: treated by diclofenac plus quercetin. ap < .05versus control group, bp < .05 versus DIC-only treated group, cp < .05 versus QR-only treated group. Effects of quercetin on serum levels of TNF-α, IL-1β, and NF-κB. Data are expressed as mean ± SD (n = 8) and analyzed by one-way ANOVA followed by Tukey post hoc test. Control: received normal saline; DIC-only: treated by diclofenac; QR-only: treated by quercetin; and DIC + QR: treated by diclofenac plus quercetin. Kidney index (mg/g) = Kidney weight (mg)/Bodyweight (g). ap < .05 versus control group. bp < .05 versus DIC-only treated group. cp < .05 versus QR-only treated group.

Histopathological findings
Histopathological findings suggested that the morphology of kidneys was normal in untreated control rats and QR-treated rats (Figure 3(a)and (c)). However, in DIC-treated rats, the evidence of lymphocyte infiltration was evident (Figure 3(b)). In group 4, the results showed that QR depletes the kidney tissue from the DIC-induced lymphocyte infiltration (Figure 3(d)). Effects of quercetin on the renal histology of experimental groups. (A) Control group with normal structure; (B) diclofenac-only administered rats show lymphocyte infiltration (the black arrows); (C) quercetin-only administered rats; (D) diclofenac-administered rats supplemented with quercetin.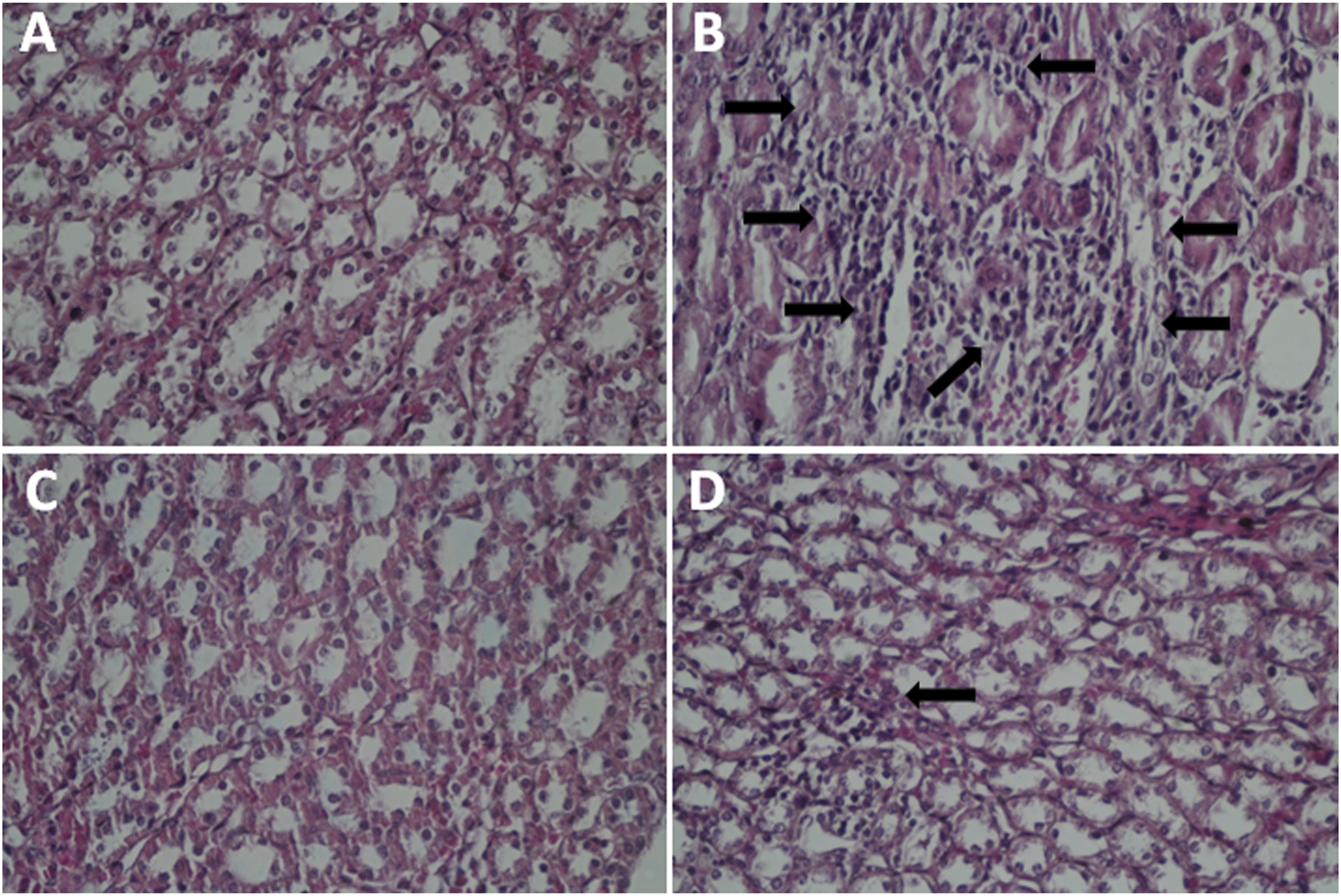
Discussion
Although being the most important NSAID used as a pain-killer, the enthusiastic for clinical administration of DIC has been muted due to renal complications. It has been claimed that through the production of free radicals, DIC could impair the function of mitochondria in the kidney tissue and thereby induce renal injuries.1,2 It is well-established that elevation in the serum levels of sGOT, sGPT, Cr, UA, and urea is an indicator of renal damage. 18 In accordance, our results also showed that DIC could increase the serum levels of the aforementioned markers in the rats, which is in agreement with the results of the previous investigations.18,19 The protective effects of QR on DIC-induced renal damage became evident when we found that QR could decrease the serum levels of sGOT, sGPT, Cr, UA, and urea in DIC-treated rats. Multiple numbers studies indicated that anti-oxidant agents could diminish oxidative stress through scavenging the free radicals and maintaining the structure of the cell membrane.31,32 Given these, it is reasonable to assume that perhaps the reductive effects of QR on the biochemical factors could be due to its anti-oxidant properties and this agent could protect the renal tissue from DIC-induced free radicals.
Another oxidative stress marker that its level could be indicative of tissue damage is MDA. 33 Interestingly, we found that DIC not only was able to elevate MDA levels but also could diminish FRAP levels in the rats. The results of the previous studies also indicated that DIC recruited free radicals to produce MDA.34,35 In DIC-plus-QR treated rats, we found a decline in the renal MDA and an elevation in the TRAP levels, indicating that this is the anti-oxidant activity of the agent that suppresses the lipid peroxidation. In agreement with our founding, previous studies also declared that anti-oxidant agents could shift the balance of the redox system through eliminating the free radicals.36,37
We also found that DIC could accumulate nitrite in the kidney tissue and thereby induce kidney damage. In this vein, previous investigations also suggested that DIC engages NO for exerting its devastating effects on the organs.34,38 Notable diminutions of nitrite level in DIC plus QR treated group implied that this agent exerted renal protection in DIC exposed animals through the diminution of NO levels and nitrosative stress.
It is well-established that some anti-oxidant compounds, including SOD, CAT, and GPx could counteract the toxic effects of ROS by degrading this free radical to its components. Moreover, among the long list of anti-oxidant compounds perhaps GSH is one the most important ones that could bind to ROS through its gamma peptide linkage and decline its toxicity.39,40 In accordance with these and due to the oxidative property of DIC,18,41 we also observed that DIC could diminish the intracellular levels of GSH, CAT, SOD, and GPx in the kidney tissue of the rats. On the other hand, QR was successful in restoring the activity and the levels of GSH, CAT, SOD, and GPx in DIC-treated rats. The striking therapeutic potential of QR on DIC-induced renal toxicity is not restricted only to these findings, as the results of the histopathological evaluation also revealed that QR could diminish the DIC-induced infiltration of lymphocytes into the kidney tissue and clear the inflammatory responses in the tissue. A mounting body of evidence indicated that by eliminating oxidative stress, some anti-oxidant agents could restore antioxidant capacity.8,42 Given these, we postulated that the kidney protective effects of QR are mediated through its anti-oxidant properties.
When it comes to tissue damage, the role of inflammation should not be underestimated and many studies also accused inflammatory responses as a mechanism through which DIC induced tissue injuries. Thus far, NF-κβ, TNF-α, and IL-1β are suggested to be the main players in the regulation of inflammatory responses. It has been claimed that the expression of these cytokines reduced the survival of kidney cells by inducing either apoptosis or necrosis.18,43 In the present study, we found that when rats were treated with DIC, the expression levels of NF-κβ, TNF-α, and IL-1β in the kidney tissue elevated more significantly as compared to the control group, which this finding was in accordance with the previous investigations.44,45 However, the expression of all these cytokines diminished robustly, when DIC-treated rats were exposed to QR. Since some previous studies enumerated the anti-inflammatory property for the anti-oxidant agents,46,47 it could be suggested that perhaps the protective effects of QR against kidney tissue would be due to its anti-inflammatory effects. In this vein, even the histopathologic investigation also showed that QR could diminish the percentage of infiltrated lymphocytes in the kidney tissue upon exposure to DIC, shedding more light on the anti-inflammatory property of the agent.
In this work, we did not assess the effects of DIC and QR on the other oxidative biomarkers and biochemical parameters, for example, immunohistochemistry for some proteins and or genes that are associated with the renal function such as TNF-α, NF-κB, caspase‐3, glutathione reductase, and glutathione‐S‐transferase. These factors are involved and altered in oxidative stress and we suggest they be evaluated in future studies.
Conclusion
The results show that by restoring the anti-oxidant reservoir for the kidney cells, QR is able to diminish DIC-induced oxidative stress as well as inflammatory responses and thereby protect the kidney tissue from the toxicity of DIC. Given these, it could be suggested that QR could be a valuable candidate to be tested in the further evaluation as an ameliorating agent for decreasing renal toxicity of drugs, in particular DIC.
Footnotes
Acknowledgements
The authors gratefully acknowledge the help of our department’s staff.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Iran University of Medical Sciences, Tehran, Iran [Grant no. 19273].
Ethics approval
Ethical approval for this study was obtained from Iran University of Medical Sciences Ethics Committee, Tehran, Iran (Ethic number IR. IUMS. REC. 99. 246). The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
Data availability
All relevant data are within the paper and its supporting information files.
