Abstract
Bleeding-induced multiple organ dysfunction syndrome (MODS) is one of the major causes of death in pregnant women. MODS is thought to result from an inappropriate generalized host inflammatory response to a variety of acute insults. In this study we established a MODS model in postpartum rats, in which MODS was induced by the combination of induced hypotension for 60 min and clamping of the superior mesenteric artery for a period of 40 min. We sacrificed all the rats 24 h after dexmedetomidine (DEX) treatment. Thymus, spleen, and mesenteric lymph node tissue were collected to detect interferon-γ (IFN-γ) and interleukin-4 (IL-4) protein expression; lung and intestine tissue were collected to measure IFN-γ and IL-4 gene expression. In the present study, IFN-γ and IL-4 mRNA were increased in the lungs and intestines of the MODS rats. DEX administration decreased IFN-γ and IL-4 mRNA expression. IFN-γ and IL-4 expression for the thymus, spleen, and mesenteric lymph nodes were higher in the MODS postpartum rats relative to control rats, and these expression levels decreased upon DEX administration, But there were no significant differences between DEX doses. In conclusion DEX administration appeared to reduce IFN-γ and IL-4 protein expression in thymus, spleen, and mesenteric lymph node tissue and reduce IFN-γ and IL-4 gene expression in the lungs and intestines in the MODS postpartum rats but was not dose-dependent.
Introduction
Multiple organ dysfunction syndrome (MODS) refers to severe wounds, shock, infection, complex surgery, and similar insults that result in organ dysfunction or failure in two or more systems appearing 24 h after the initial insult. The resulting multiple organ dysfunction is manifested by the inability to maintain systemic homeostasis in acutely injured patients. MODS is the major cause of death in critically ill patients, and the associated MODS mortality rate has been reported to be as high as 70%. 1 MODS is a major research focus in the international medical community. 2 From January 2006 to December 2011, a total of 19,015 patients were treated in the Department of Obstetrics in Peking University Third Hospital. Seventy-three cases had multiple organ dysfunctions, the frequency of MODS in obstetric patients was 38.39/10,000; 3 and recent results indicate an increase of sepsis-related maternal mortality from 0.85 deaths/100,000 pregnant women in 2003–2005 to 1.13/100,000 in 2006–2008, which is concerning. 4 The major causes of MODS in obstetric patients are pre-eclampsia, postpartum hemorrhage, and amniotic fluid embolism. Bleeding-induced multiple organ dysfunction syndrome (BMODS) is a rapidly progressive disease that occurs in critically ill obstetric patients. The treatment for postpartum BMODS differs from the treatment for MODS induced by other causes. BMODS is one of the major causes of death in pregnant women. 5 Thus, finding an agent that can resuscitate a patient from hemorrhagic shock is urgently required.
Dexmedetomidine (DEX) is an α2-adrenergic agonist that selectively binds to presynaptic α2-adreneric receptors (α2AR) in the locus coeruleus and decreases the amount of norepinephrine released resulting in the reduction of postsynaptic adrenergic activity. 6 Administration of DEX causes different effects in different organs.7,8 DEX can act as a sedative, an analgesic, and an anxiolytic, and experimental studies have indicated that DEX can provide renal, coronary, and neural protection.9,10 In animal studies, DEX has been shown to reduce the mortality rate and inhibit the inflammatory response in rats exposed to endotoxin without obvious adverse side effects such as respiratory inhibition.11,12 Mice subjected to renal ischemia reperfusion (IR) incur significant remote lung injury; however, intraperitoneal DEX administered before or after ischemia effectively prevented ischemic damage. 9 DEX, if used appropriately, maintains psychomotor function, which could help participation in the laboring process. 13 It has also been shown to enhance spontaneous contractions in human myometrium, which may have relevance to its use before and during labour. 14 In the isolated, perfused human placenta, DEX passes less readily into fetal circulation. 15 In the present study, we used postpartum rats to establish an animal model of multiple organ dysfunction induced by the combination of hypotension and clamping of the superior mesenteric artery to observe the effects of DEX on cytokine release in postpartum MODS rats.
Materials and methods
Experimental animals and care
Twenty male and forty female Sprague-Dawley rats (weight, 220–250 g; Animal Center of Guangdong Province, PR China) were housed in the Guangzhou Animal Center in accordance with specific pathogen animal standards under a 12-h light/dark cycle. The mice were housed in specially prepared heat- and humidity-controlled cages and were given free access to food and water. The animals used in this study were approved by the Animal Use and Care Committee of Guangzhou Medical University. The care and handling of the animals were conducted in accordance with the National Institutes of Health guidelines.
Animal model of multiple organ dysfunction syndrome induced by postpartum bleeding
Postpartum female rats were used for experiments within 24 h after delivery. Forty experimental female rats were anesthetized by intraperitoneal administration of 10% hydration chlorine aldehyde (0.4 mL/100 g) and were placed on an operating table in a supine position, then were randomly allocated into four groups of 10 rats each in the following manner: a control group and three experimental groups (groups A, B and C). A rectal temperature probe was inserted for monitoring. The rectal temperature was maintained at 37°C by using a heating blanket during the induction of ischemia. A polyethylene catheter (PE-50) filled with sterile heparinized 0.9% saline (50 U/mL) was inserted into the right femoral artery. The mean arterial pressure (MAP) was continuously monitored (BIOPAC System, Santa Barbara, CA, USA). Another PE-50 was inserted into the left femoral vein for blood sampling and drug administration. All catheters used in our experiments were filled with heparinized saline to prevent blood clotting. For group A, after completing the above procedure, hypotension was induced by drawing blood from the left femoral vein into a heparinized glass syringe for temporary storage until the MAP reached a target level of 40–50 mmHg. 16 The MAP was maintained at this low but constant mean arterial pressure for 60 min by the withdrawal and re-infusion of the stored blood obtained from the left femoral vein to induce artificial hypotension in the postpartum rats. The normal MAP was restored by infusing the full amount of blood that was previously drawn from the left femoral vein. After a normal arterial pressure was maintained for less than 30 min, the abdomen of the rats was opened under sterile conditions, and then the superior mesenteric artery was clamped for 40 min and then was released. For groups B and C, 1 µg/kg DEX (Hospira Inc., Lakeforest, IL, USA) was administered by intravenous infusion over approximately 10 min before drawing blood, after which the procedure described above for group A was performed, and then 2.5 µg/kg/h or 5.0 µg/kg/h DEX, respectively, was administered via the left femoral vein for 4 h.
After the operations above were performed, the incision in the abdomen was sutured and the catheters were removed. The rats were caged individually after the surgery and were provided food and drinking water
RNA isolation and real-time reverse transcriptase-PCR (RT-PCR)
RT-PCR was performed using the left lung and intestine tissue samples. Total RNA was isolated with an RNA isolation kit (Takara Co., Ltd.). The total RNA concentration and 260:280 nm absorbance ratios were calculated using a NanoDrop ND-1000 Spectrophotometer (Nano-Drop Technologies, Montchanin, DE, USA). The residual genomic DNA was removed by incubation with RNase-free DNase (Toyobo Biotech Co., Ltd, Osaka-shi, Japan). For the initial strand cDNA synthesis, RNA (2 μg) was converted to cDNA using Superscript II reverse transcriptase (Invitrogen, Carlsbad, CA, USA). The reaction mixture was inactivated by heating the reactions to 70°C for 15 min. One microliter of the reaction mixture was amplified by Taq DNA polymerase (Takara Co., Ltd.) in a thermal cycler (GeneAmp PCR system 2700, Applied Biosystems, Foster City, CA, USA). The initial denaturation was performed at 94°C for 3 min followed by 30 cycles of 30 s at 95°C (denaturation), 1 min (specific annealing temperature for each primer), and 1 min at 72°C (extension). A final elongation step was performed at 72°C for 5 min (final extension). The sequences for the primers used in present study are listed in Table 1. The PCR products were separated by 1% agarose gel electrophoresis. After staining with ethidium bromide, the amplified DNA bands were analyzed by using the image analysis software ScionImage (Scion Corp., Frederick, MD, USA).
qRT- PCR primers and conditions.

Immunohistochemistry (IHC)
IHC analysis was performed by using the streptavidin-biotin amplification method using the Histofine Kit (West Tang Biotech Co., Ltd., Shanghai, PR China) based on a previously described protocol. 17 Thymus, spleen, and mesenteric lymph nodes were removed and post-fixed for 48 h in 4% paraformaldehyde at 4°C. After deparaffinization, slides were heated in a microwave oven (500 W) for 15 min in citric acid buffer (1 mM citric acid and 9 mM trisodium citrate dehydrate, pH 6.0). The primary antibody for IFN-γ and IL-4 was applied at a dilution of 1:200. The antigen-antibody complex was visualized with 3,3′-diaminobenzidine (DAB, Biosynthesis Biotechnology Co., Ltd., Beijing, PR China) solution (1 mM DAB, 50 Mm Tris-HCl buffer [pH 6.0], and 0.006% H2O2) and was counterstained with hematoxylin. Immunoreactivity absorption tests for IFN-γ and IL-4 were performed by incubating the antibody–antigen mixture containing equal volumes of optimally diluted anti-serum (Biosynthesis Biotechnology Co., Ltd., Beijing, PR China) to IFN-γ and IL-4 and the corresponding IFN-γ and IL-4 peptide.
The positive cells were measured from every 100 cell randomly in each experimental condition and counts were replicated using 10 different fields of view on each coverslip in four independent cultures. Expression of IFN-γ and IL-4 was determined by two independent observers by assessing semi-quantitatively the percentage of stained cells and the staining intensity. The percentage of positive cells was rated as follows: 0, <10% positive cells; 1 point, 10–25% positive cells; 2 points, 25–50% positive cells; and 3 points, >50% positive cells. The staining intensity was rated as follows: 0, completely negative; 1 point, weak intensity; 2 points, moderate intensity; and 3 points, strong intensity. Points for expression and percentage of positive cells were added, and specimens were attributed to four groups according to their overall score: 0, negative; 1–2 points, weak expression; 3–4 points, moderate expression; and 5–6 points, strong expression.
Statistical analysis
Statistical analyses were performed using the Statistical Package from Social Sciences (SPSS) Version 13.0 for Windows. Data are presented as the mean ± standard deviation. The two-way ANOVA with a stringent post hoc test were used for statistical analysis. The t-tests were used for BMODS model in postpartum rats between the control group and group A. The significance threshold was set at 0.05.
Results
Bleeding-induced MODS model in postpartum rats
The results showed that the processing treatment established BMODS model in postpartum rats, similar to our previous studies.
18
There were significantly (
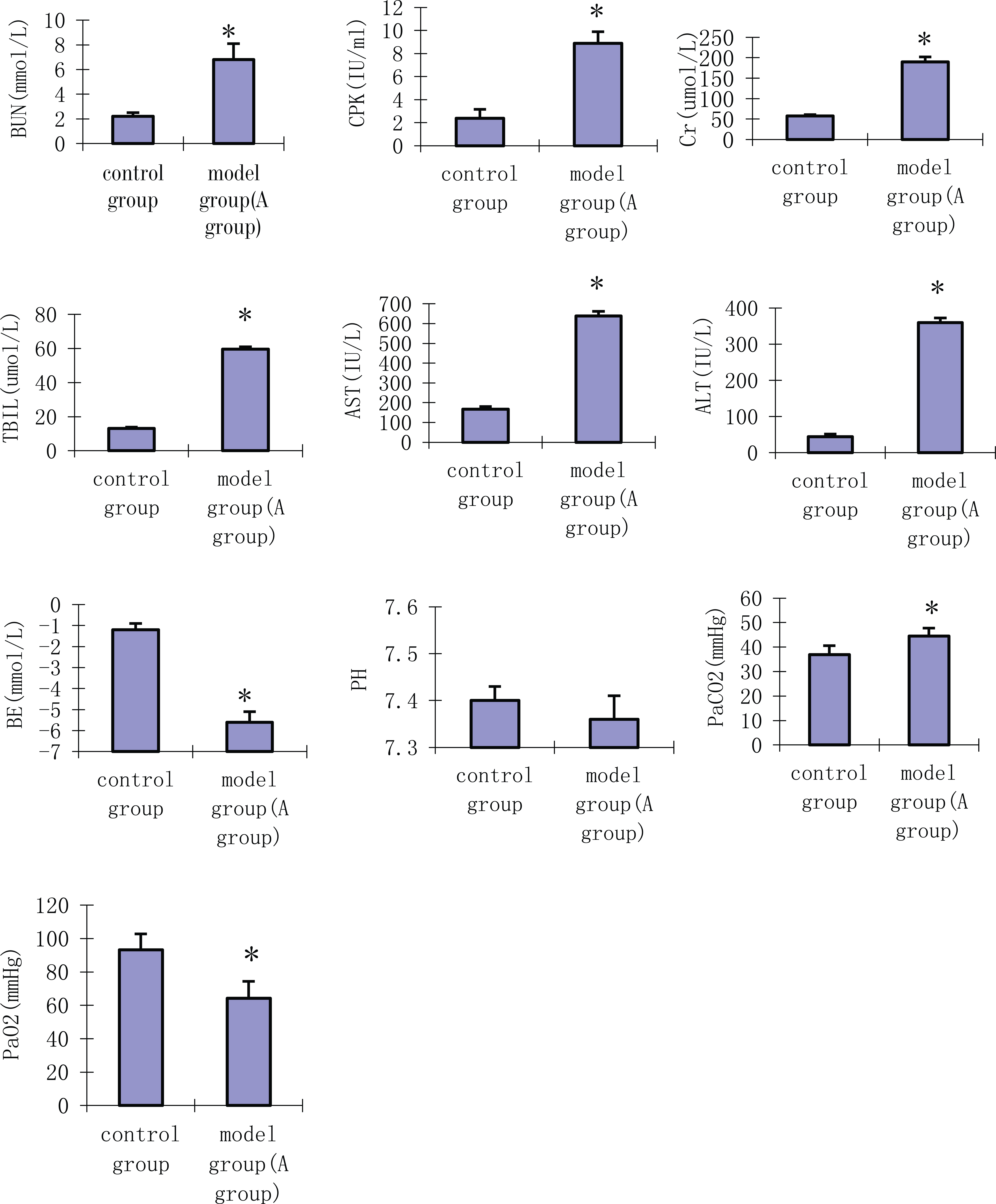
Liver, kidney function and arterial blood gas during postpartum BMODS in rats. The experiments were performed three times. Values shown are mean ± SD. A difference of P < 0.05 was considered statistically significant. *Compared with control group p<0.05.
The IFN-γ and IL-4 mRNA expression in the lungs and small intestines of group A were significantly (

The RT-PCR analysis of effect of DEX administration on IFN-γ and IL-4 mRNA expression relative to b-action in lung tissue. The experiments were performed three times. Values shown are mean ± SD. A difference of P < 0.05 was considered statistically significant. *Compared with control group p<0.05; # Compared with A group P <0.05; $ compared with B group P <0.05.

The RT-PCR analysis of effect of DEX administration on IFN-γ and IL-4 mRNA expression relative to b-action in small intestine. The experiments were performed three times. Values shown are mean ± SD. A difference of P < 0.05 was considered statistically significant. *Compared with control group p<0.05; # Compared with A group P <0.05; $ compared with B group P <0.05.
Immunohistochemical staining detected IFN-γ and IL-4 expression in the spleen, thymus, and mesenteric lymph nodes and was used for a more comprehensive evaluation of the rat organs. The IFN-γ and IL-4 IHC staining total score in the thymus, spleen, and mesenteric lymph nodes in group A increased significantly relative to the control group (

Effect of DEX administration on IFN-γ and IL-4 IHC staining in spleen tissue. immunohistochemistry analysis was performed by using the streptavidin-biotin amplification method, overall score were measured and analyzed statistically in four groups. Values shown are mean ± SD. A difference of P < 0.05 was considered statistically significant. *Compared with control group p<0.05; # Compared with A group P <0.05; $ compared with B group P <0.05.

Effect of DEX administration on IFN-γ and IL-4 IHC staining in thymus tissue. Immunohistochemistry analysis was performed by using the streptavidin-biotin amplification method, overall score were measured and analyzed statistically in four groups. Values shown are mean ± SD. A difference of P < 0.05 was considered statistically significant. *Compared with control group p<0.05; # Compared with A group P <0.05; $ compared with B group P <0.05.
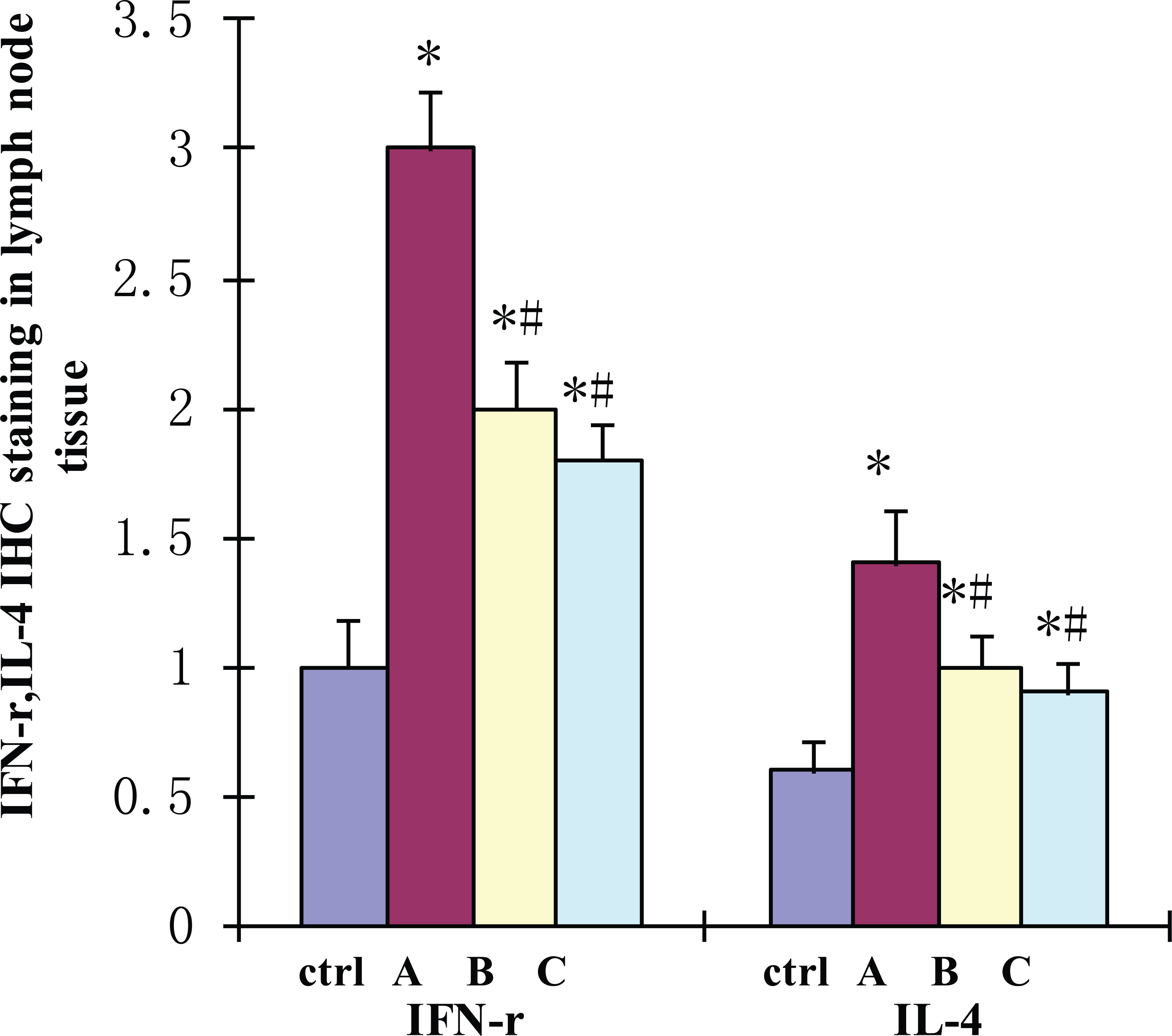
Effect of DEX administration on IFN-γ and IL-4 IHC staining in lymph node tissue. Immunohistochemistry analysis was performed by using the streptavidin-biotin amplification method, overall score were measured and analyzed statistically in four groups. Values shown are mean ± SD. A difference of P < 0.05 was considered statistically significant. *Compared with control group p<0.05; # Compared with A group P <0.05; $ compared with B group P <0.05.

IFN-γ and IL-4 positive expression in different group of immune organs.
Discussion
Gestational hypertension, pre-eclampsia or eclampsia, placental abruption, postpartum hemorrhage, and infection are major causes of MODS in obstetric patients. Obstetric MODS due to obstetric complications increased blood volume adds to the burden of various organs during the pregnancy period and can adversely affect the safety of the mother and the fetus. There have been several hypotheses regarding the mechanism of MODS, including IR, inflammation, intestinal bacteria, toxin shifts, two strikes double pre-excitation syndrome, and stress genes. Mortality associated with pregnancy or childbirth originates from three main causes: embolism (21.4%), pre-eclampsia/ hypertensive disorders (19.4%), or hemorrhage (13.4%). 19 Evidence shows that each of these diseases is linked to endothelial dysfunction. This leads to systemic vascular alterations, which may promote a systemic inflammatory response. 20
Th1 cells mainly secrete interleukin-2 (IL-2), interferon-γ (IFN-γ), and tumor necrosis factor-α (TNF-α), whereas Th2 cells secrete IL-4, IL-5, IL-6, IL-9, IL-10, and IL-13. The Th2 cytokines are responsible for B lymphocyte proliferation, antibody production, immune tolerance, and humoral immunity.21,22 Therefore, we chose IFN-γ and IL-4 to evaluate changes in the immune function in postpartum rats with MODS. In the present study, according to the diagnosis criteria of MODS, we sacrificed all the rats 24 h after the experiment. Thymus, spleen, mesenteric lymph nodes, lung, and intestine tissue were collected for further testing. IFN-γ and IL-4 mRNA in lung and intestine tissue were increased in the postnatal rats with MODS.
DEX is an α2-adrenergic agonist that selectively binds to presynaptic α2-adreneric receptors (α2AR) in the locus coeruleus and decreases the amount of norepinephrine released resulting in the reduction of postsynaptic adrenergic activity. DEX administration decreased IFN-γ and IL-4 mRNA expression but not dose dependent in the postnatal rats with MODS. IFN-γ and IL-4 expression scores for thymus, spleen, and mesenteric lymph node tissue were increased in postpartum rats with MODS and were decreased upon DEX administration. Our results show that DEX reduced IFN-γ and IL-4 expression in the lungs and small intestines, which were included as target tissues. A lack of immune response can often lead to septic shock, but septic shock was not observed in our study, which is consistent with previous studies that have shown that DEX administration reduces the mortality rate of septic shock patients. 23 Other reports have demonstrated the suppressing effect of DEX on inflammatory mediator production in human whole blood after LPS stimulation. DEX suppressed TNF-α, IL-6, IL-8, and high-mobility group box 1 protein production in human whole blood. The suppressing effects of DEX on pro-inflammatory mediator production were reversed by yohimbine.
Cytokine release plays an important role in the pathogenesis of MODS. IL-6 secretion is closely related to the severity of inflammation in inflammatory bowel disease and can accurately reflect the extent of tissue damage. 24 In our study, DEX decreased IFN-γ and IL-4 release in MODS induced by postpartum hemorrhage in rats. Some reports have demonstrated that DEX inhibits cytokine release in rats following endotoxin administration 25 based on a decrease in the total levels of cytokines and a reduction in the concentration of MIP-2 and IL-β as well as a decrease in the ratio of dry/wet tissue and lung inflammation in mice. 23 The mechanisms responsible for the effect of DEX on the reduction of cytokine secretion remains a matter of debate. Our results suggest that DEX has strong anti-inflammatory effects in postpartum MODS rats. These findings suggest that DEX suppresses humoral and cellular immune responses. A limited number of reports exist regarding the mechanism of DEX at the cellular level.
Our study has shown that DEX exerts an anti-inflammatory effect by reducing levels of inflammatory factors, however, the upstream mechanism is still unknown. Lung tissues of septic rats induced by Cecal Ligation and Puncture (CLP) was designed to investigate the effects of DEX on inflammatory reaction. After induction of sepsis, the rats were treated with normal saline or DEX (5, 10, or 20 µg/kg). The concentrations of IL-6 and TNF-α in bronchoalveolar lavage fluid (BALF) and plasma were measured. The expressions of toll-like receptor 4 (TLR4) and MyD88 were measured and the activation of NF-κB in rat lung tissues was assessed. The results suggest that DEX may inhibit inflammatory reaction in lung tissues of septic rats by suppressing the TLR4/MyD88/NF-κB pathway. 26 Huang and his colleagues established a rat model that simulates a clinical surgical procedure to investigate the anti-inflammatory effect of perineural administration of DEX and the underlying mechanism. DEX reduced the sciatic nerve levels of IL-6 and TNF-α at both the mRNA and protein level. DEX also inhibited the translocation of activated NF-κB to the nucleus and the binding activity of NF-κB. In conclusion, DEX inhibited the nuclear translocation and binding activity of activated NF-κB, thus reducing inflammatory cytokines. 27
In summary, the present study demonstrates that DEX administration reduces the release of cytokines IFN-γ and IL-4 in the spleen, thymus, and mesenteric lymph nodes as well as in the target organs (lungs and small intestines) in postpartum MODS rats but is not dose-dependent. Our results provide a strong theoretical basis for DEX administration in patients with MODS induced by postpartum hemorrhage.
Footnotes
Acknowledgements
We thank Chen Tao for technical support of surgical preparations. We also thank American Journal Experts (AJE) for editing the English version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the Youth Foundation of Guangzhou Medical University in China (grant no. 2012A10) and by the Science and Technology Foundation of the Guangdong Province in China (grant no. 2012B020700007).
