Abstract
Objectives
The balance between proinflammatory IFN-γ Th1 vs. the anti-inflammatory allergy-mediating IL-4-heralded Th2 reactions is pivotal in IgE-mediated allergic rhinitis (AR). Hypoxia-Inducible Factor (HIF)-1α is inducible by hypoxia and various cytokines. HIF-1α activates different anti-pathogen and allergic immune cells. This cross-sectional study assessed the changes in serum HIF-1α and its dependent erythropoietin (EPO) levels among hospital-characterized AR patients. Type of the immune reaction, Th1 vs. Th2, was stratified based on the calculated IL-4/IFN-γ direct ratio, after being measured using specific ELISA assays.
Methods
147 AR patients (83 males/64 females), and age-, BMI-, and gender-matched 24 healthy controls (13 males/11 females) were sequentially enrolled at ENT Unit, Prince Muteb General Hospital, Sakaka, Saudi Arabia. Measurement of serum parameters was carried out using specific ELISA assays.
Results
Contrary to the majority of previous publications, all controls and the majority of patients (
Conclusion
These results confirmed a pathogenic implication for HIF-1α and IFN-γ in AR that warranted future bigger and longitudinal studies.
Introduction
The atopic allergic rhinitis (AR) disease manifests as sneezing, nasal congestion, clear rhinorrhea, and nasal pruritus. It is an IgE-mediated immune response induced against inhaled antigens, in the immediate phase, that is followed by a leukotriene-mediated late phase.1,2 Mucosal immunity is controlled by a cocktail of cytokines produced by the naïve CD4+ T helper 0 (Th0) cells upon differentiation either to: (1) Th1 with interferon (IFN)-γ as the predominant cytokine, and cytotoxic T cells, natural killer cells, activated macrophages, and B cells as the cellular machinery, or (2) the antagonistic, Th2 cells, with interleukin (IL)-4 as the signature cytokine, and B cells, eosinophils, and basophils as the cellular armament that coordinate the allergic IgE-mediated or autoimmunity reaction. Allergy-related asthma and its consequences are associated with a Th2-predominance, whereas Crohn's disease and its prognosis are associated with a Th1-predominance. In immunologic diseases, the Th1/Th2 balance is pathogenic, prognostic, and theragnostic. 3 Th1/Th2 balance can be easily and directly determined by comparing the ratio of these antagonistic regulatory cytokines, IFN-γ/IL-4. However, genetic and environmental confounding factors contribute to bias in Th1/Th2 balance.4,5
Obstructive sleep apnea (OSA), which is characterized by sleep-interrupting repeated upper airway obstruction during sleep and intermittent hypoxia, has been linked to rhinitis, and in particular AR. Mouth breathing, decreased pharyngeal diameter, and higher nasal and airway resistance all contribute to this connection. Additionally, OSA and the main proinflammatory mediators found in AR, histamine, IL-1, and IL-4, decrease sleep quality. 6 Intermittent hypoxia increases hypoxia-inducible factor (HIF)-1α that induces pro-oxidant enzymes, reactive oxygen species production and oxidative stress. 7
The dimeric basic helix-loop-helix transcription factor, HIF-1α/β, was first identified for its role in regulation of erythropoietin (EPO) gene expression. HIF-1α/β binds to hypoxia response elements (HREs) in target genes. HIF-1α is the inducible oxygen-regulated subunit of the dimer and its stability determines dimer's transcriptional activity. While airway epithelial cells are exposed to 21% O2, in solid tissue cells, O2 concentration is as low as 1%. Under normoxic conditions, HIF-1α is rapidly degraded by the ubiquitin-proteasome pathway. Growth factors, nitric oxide, various inflammatory cytokines, reactive oxygen species, apoptotic cell debris, infectious pathogens, and allergens control HIF-1α expression, oxygen-independently.8–10 Oppositely, HIF-1 dimer activates different inflammatory immune cells associated with infections and allergies, particularly for nasal allergies—oxygen-dependently and in normoxia.11–15 Upon activation, HIF-1α translocates to the nucleus to reprogram (up- or down-regulation) >2000 genes of cell's transcriptome to be able to adapt to hypoxia. These genes include EPO/erythropoiesis, glucose transporters/glycolytic enzymes, and vascular endothelial growth factor (VEGF)/angiogenesis. This supports energy metabolism in inflammatory cells and promotes pro-inflammatory cytokine expression and immune cell adhesion.11,14 HIF-1α is induced by Th1 cytokines. 16
EPO is essential for erythropoiesis, upon the induction of its expression by the falling tissue O2 pressure under hypoxic physiological, pathological, and experimental conditions—mainly from the peritubular fibroblasts of the renal cortex. 17 However, EPO action is also controlled by cytokines, growth factor, and steroid and peptide hormones. Other than erythropoiesis, EPO is a cytoprotective agent for blood vessels, heart, kidneys, and brain. 18
We planned this study to evaluate the variation in serum levels of HIF-1α and EPO in correlation with IL-4 and IFN-γ and their direct ratio. The ratio is used as the biomarker and classifier for the type of Th cell anti-/pro-inflammatory reaction in AR patients, as compared to healthy controls.
Methods
Setting and patients
Volunteering 147 AR patients (83 males and 64 females), and age-, BMI-, and sex-matching 24 healthy accompany (13 males and 11 females) were sequentially admitted and reviewed at the ENT outpatients' clinic of Prince Muteb General Hospital, Sakaka, Saudi Arabia in the period from September 2021 to June 2022. This cross-sectional study was approved by the permanent committee for Ethics of Scientific Research, Jouf University, Sakaka, Saudi Arabia (21-08-42). Each participant signed a written informed consent, in adherence to the provisions of the Declaration of Helsinki. Physical characterization of AR by ENT specialists was done following the international guidelines. 19 De-identified patients' and healthy control characteristics and medical history were recorded. Patients with anemia of all types, immobility of all causes, endocrine disorders, and chronic inflammatory and renal and liver diseases were excluded. Patients were investigated for the presence of bronchial asthma, nasal polyposis, deviated nasal septum (DNS), hypertrophy of inferior turbinate (HIT), OSA, and/or concha bullosa. Also, tobacco smoking was recorded for patients and controls. Body mass index (BMI) was calculated for each participant (weight in kg/height in m2).
Sample collection and investigations
Five mL of peripheral venous blood were aseptically collected in plain collection tubes and serum was recovered and aliquot frozen at –80oC till used. HIF-1α (pg/mL) and EPO (pg/mL; = x 0.119 mIU/mL) were quantitatively measured using specific quantitative ELISA assays in triplicates as instructed (cat# SL0905 and SL0679Hu, Sunlong Biotech Co. Ltd, Zhejiang, China). Aprotinin (fresh 10x aprotinin to 350 nM) was added to serum. Our lab was the 1st worldwide to characterize circulating HIF-1α as a hypoxia biomarker. We did not detect significant HIF-1α degradation in the extracellular in vitro hypoxic milieu of serum. The patients were stratified as anti-inflammatory–allergic Th2 type reaction at IL-4/IFN-γ ratio ≥5, naïve Th0 type reaction at IL-4/IFN-γ ratio >0.2 to <5, and proinflammatory-tissue damaging Th1 type reaction at IL-4/IFN-γ ratio ≤0.2. 20
Statistical analysis
For the statistical analysis and data presentation, GraphPad Prism 7.00 software was used (GraphPad Software, Inc., La Jolla, CA, USA). Data presented as frequency and mean ± SDM were compared with Student's “t” test and one-way ANOVA with Bonferroni's correction post-test for significance;
Results
Considering the classification of the immune reaction using the direct IL-4/IFN-γ ratio, all healthy controls (
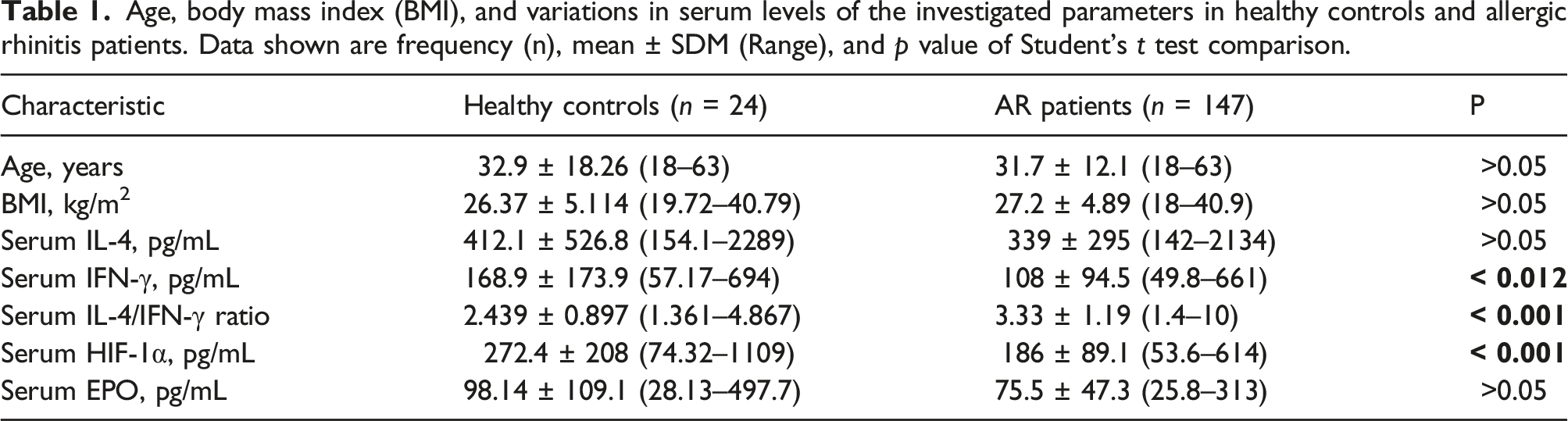
Age, body mass index (BMI), and variations in serum levels of the investigated parameters in healthy controls and allergic rhinitis patients. Data shown are frequency (n), mean ± SDM (Range), and
Comparing AR patients subgrouped for presence of OSA (
Patients suffering from HIT (
Patients with DNS (
Patients' stratification for tobacco smoking, BMI, presence of nasal polyps, or age did not show significant differences in the investigated parameters.
Results of the correlation analysis
Correlations observe among parameters, and AR patients' and disease characteristics. Data presented are Spearman's correlation coefficient r/

Among the healthy controls, BMI correlated positively vs. each of age (r = 0.556/
Discussion
We conducted the current investigation with the expectation that our AR patients would exhibit a predominately Th2 immune response. The data, however, showed that there was a mixed majority of naïve Th0 and a minority of Th2 reactions. All of the controls exhibited a naïve Th0 immune profile with considerably greater IFN-γ levels than AR patients, which resulted in a lower IL-4/IFN-γ ratio. This was not the only unanticipated result we found. Serum HIF-1α and EPO levels, as cellular hypoxia biomarkers, were expected to be higher in AR patients, particularly those suffering from OSA, nasal polyposis, HIT, and DNS than healthy controls. These contrary findings could be reasoned to environmental factors and administration of anti-allergic over-the-counter (OTC) and herbal medications that the patients admitted taking. Controls showed significantly higher levels of HIF-1α. The wide inter-individual variations in IL-4 and EPO levels made their higher contents in the controls non-significantly different from patients. Levels of these cytokines significantly correlated with AR comorbidities. OSA caused significantly higher IL-4 levels but lower IL-4/IFN-γ ratio, HIT had lower IL-4/IFN-γ ratio, and DNS revealed lower EPO and IL-4 levels.
HIF-1α expression is regulated by hypoxia and oxygen-independently by various cytokines and growth factors. Similarly, during both of hypoxia and normoxia, HIF-1α activates different immune cells during the inflammatory processes associated with infections and allergies, particularly for nasal allergies.11–15 In airways of children with allergic asthma, IL-4/IFN-γ ratio in condensate of exhaled breath is increased, consistent with predominance of Th2 cells. Steroid treatment lowers IL-4 but not IFN-γ. 21 After the recognition of antigen, the IL-12/IFN-γ-primed Th1 immunity ensures defense against intracellular pathogenic microorganisms, tumor cells, viruses, and fungi. This employs cytotoxic T cells, NK cells, activated macrophages, and B cell antibodies. The huge amount of IFN-γ produced empowers Th1 shift of the immune reaction while dampening Th2 cytokines and immunity development. The IL-4-primed Th2 immunity ensures immunity against extracellular pathogenic microorganisms, bacteria, and parasites. It utilizes B cell antibodies, eosinophils and basophils. Strongly biased immunity to Th2 arm induces production of large amounts of IL-4, IL-10, and IL-5. This leads to allergic IgE-mediated reaction or specific auto-reactive antibodies-mediated reaction. Further production of IL-4 by these cells enhances Th2 immune pathway while inhibiting the development of Th1 immunity. Therefore, the ratio of these two contra-regulatory cytokines, IL-4 to IFN-γ, dictates which of these two pathways is dominant or depressed.4,5
HIF-1α is induced by Th1 cytokines. 16 Discordance between EPO and HIF-1α in our results may be reasoned to the further regulation of EPO, expression, and action, by cytokines, sex hormones, growth hormone, and IGF-1. 18 Inhibitor of retinoic acid receptor-related orphan was able to relieve experimental AR through reducing the expression of tissue HIF-1α and VEGF, and serum IL-17 level. 9 In support of that, in the same cohort of AR patients, we have previously reported significantly reduced levels of all-trans-retinoic acid, the major physiological ligand for retinoid receptors. 22 HIF1A gene single nucleotide polymorphisms (SNPs) were significantly associated with various diseases. Of which, is the regulatory intronic rs79865957 SNP that was associated with AR, asthma and inflammation, despite the unknown functional consequences of this SNP. HIF-1α inhibition decreases Th2 inflammation as measured by reduced IL-4, IL-5, and IL-13.23,24 In other experimental AR models with Th2 cytokine profile, reduction of HIF-1α and VEGF expression, decreasing the apoptosis-resistant eosinophilia and prevention/neutralizing the induced cytokines were ameliorative, while their induction exacerbates the inflammation.25–29 Similar Th2 profile was not observed in our study that may explain why HIF-1α and EPO expression were rather lower in our patients.
HIF-1α contributes to AR, bronchial asthma, and chronic sinusitis. HIF-1α protein levels in nasal airways correlate with the severity of AR in mice. Induction of HIF-1α sustains the allergic effector cell survival and function, with an important pathogenic role in chronic sinusitis and polyposis. Such unfavorable role of HIF-1 in allergic airway inflammation is attracting more attention, as its inhibition may prove therapeutically useful in AR—through suppressing IgE local and systemic Th2 cytokine expression, airway hyper-responsiveness, and vascular permeability.8,9,11,23,30 HIF-1α plays a crucial role in interferon production by dendritic cells and subsequent cytotoxic T cell activation. 31 There is increased expression of HIF-1α in chronic rhinosinusitis without nasal polyps. 32 Hypoxia due to closure at the ostiomeatal complex is widely considered one of the major pathogenic mechanisms leading to chronic inflammation in chronic rhinosinusitis. 33 A positive relationship is detected between each of HIF-1α and HIF-2α protein expression in epithelial cells and the endoscopic score in nasal polyp samples. However, there is a negative correlation between HIF-1α expression and each of the degree of eosinophil infiltration and IL-17A expression. 34 House dust mite extract induces HIF-1α and its target genes’ expression in human primary cultured nasal epithelial cells and in the nasal mucosa of an AR murine model. 27 In in vitro primary cultures, human eosinophil-infiltrated inferior turbinate chronic rhinosinusitis mucosa expressed higher IFN-γ and HIF-1α compared to control mucosa. Eosinophil-infiltrated mucosa from nasal polyps secreted more IFN-γ than control mucosa under normoxic condition; however, the difference was abolished under hypoxic incubation. Under hypoxia, chronic rhinosinusitis eosinophil-infiltrated and none-infiltrated mucosa from nasal polyps expressed lower nuclear HIF-1α level, whereas mucosa from inferior turbinate of chronic rhinosinusitis patients and controls expressed higher cytoplasmic HIF-1α levels. 35 These findings partially support our results.
We depended on patient surveys to specify the inducing allergens of AR. The survey revealed indoor and out-door exposure to dust, animals, grass, weather changes, and smoke, perfumes, and incense to be the main elicitors. This indicates the possible diagnosis of mixed type AR. The severe symptoms we observed in the overwhelming majority of patients support such a notion. Nevertheless, not determining the specific allergen(s), using skin prick test and/or total and allergen-specific IgE assessment, is a limitation of the current study that will be done in future bigger studies. We also cannot exclude destabilization of HIF-1α by a disease-lifestyle-environment interaction (local dry sandy atmosphere most of the year) and patients administration of OTC and herbal homeopathy. 36 This gets some credit from the observed parallel reduction in the levels of the very stable EPO. The relatively small sample size is another limitation that might have affected our results, too.
Conclusion
This study revealed an overwhelming majority of patients with the naïve Th0 immune profile that was recorded also for all healthy controls. Controls had significantly higher levels of IFN-γ and HIF-1α but lower IL-4/IFN-γ ratio than patients. Controls had non-significantly higher IL-4 and EPO levels compared to AR patients, attributable to the wide inter-individual variations. Levels of these parameters significantly correlated with AR comorbidities. OSA caused higher IL-4 levels but lower IL-4/IFN-γ ratio, hypertrophy of inferior turbinate caused lower IL-4/IFN-γ ratio, and deviated nasal septum caused lower EPO and IL-4 levels. Along with the cytokine and their balance, the significant difference between patients and controls in serum HIF-1α points to a pathogenic role that warranties future bigger and longitudinal studies. The unanticipated immune profile and HIF-1α/EPO levels could be attributed to: (1) life-style/environmental factors, since activation of aryl hydrocarbon receptor competes with HIF-1α for gene responsive sequences, (2) subclinical co-infections, (3) mixed type AR in most of patients, and/or (4) homeopathic administration of AR-relieving herbal decoction and/or OTC medications.
Footnotes
Author contribution
The study conceptualization and design, patients profiling, data rationalization/discussion and final manuscript revision was carried out by Dr. A.N. Al-Rasheedi, A.D. Alotaibi, and Dr. A.H. Almaeen, literature retrieval, lab investigations, data acquisition, analysis, discussion and presentation, and manuscript draft writing was contributed by Dr. A.H. Almaeen, Dr. A.D. Alotaibi, Dr. A.N. Al-Rasheedi, Prof I. Taher, and Prof. T.H. El-Metwally, and, literature retrieval, sample collection, and medical history data were collected by our medical students and interns (A.S.D. Alruwaili, S.H.H. Alruwaili, W.M.A. Alruwaili, A.S. Alsakhry, G.M. Alruwaili, H.M. ALkuwaykibi, NN. Alrawili, R.Z. Alabdullah, and R.S. Alatwi). All authors reviewed the final manuscript and approved it.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
