Abstract
Background:
Canada has historically been among the world leaders in hypertension care, but hypertension treatment and control rates have regressed in recent years. This guideline is intended to provide pragmatic primary care-focused recommendations to improve hypertension management in adults at the population level.
Methods:
We employed Grading of Recommendations Assessment, Development and Evaluation and ADAPTE frameworks in accordance with Appraisal of Guidelines for Research and Evaluation (AGREE II) quality and reporting standards to develop recommendations on managing hypertension for adults aged 18 years and older. We used the HEARTS framework—a model of care developed by the World Health Organization to improve hypertension control and reduce cardiovascular burden—to integrate these recommendations into streamlined, pragmatic, and evidence-based algorithms. The guideline committee predominantly comprised primary care providers and also included patient, methodology, and hypertension specialist representatives. Our process for managing competing interests adhered to Guidelines International Network principles.
Recommendations:
The 9 recommendations for managing hypertension in adults are grouped under the categories of diagnosis and treatment. Diagnostic recommendations include a standardized approach to measuring blood pressure (BP) and confirming hypertension, as well as providing a uniform definition for hypertension of BP ≥130/80 mmHg. Treatment recommendations include targeting a systolic BP <130 mmHg, implementing healthy lifestyle changes, and providing stepwise guidance on optimal medication choices for patients requiring pharmacotherapy.
Interpretation:
Our aim is to enhance the standard of hypertension care in the Canadian primary care setting. Accurate diagnosis and optimal treatment of hypertension can reduce adverse cardiovascular events and risk of death.
Hypertension is the most common modifiable risk factor for cardiovascular disease and death, 1 prevalent in about 1 in 4 adults in Canada. 2 Historically, Canada has been among the world’s leaders in hypertension treatment and control rates. 3 However, declining trends in hypertension treatment and control across Canada in recent years have raised concerns.4,5 This decline has been proposed to relate to discrepancies in optimal blood pressure (BP) targets, overly complex guideline recommendations, inadequate implementation strategies, and suboptimal engagement with front-line health care providers. 6
Most hypertension is managed in primary care; therefore, improving hypertension care at the population level necessitates prioritizing primary care. To this end, Hypertension Canada has adopted a new 2-part guideline approach. 7 As an adjunct to its forthcoming comprehensive guideline, Hypertension Canada has developed this primary care-focused hypertension guideline that comprises pragmatic recommendations for efficient implementation in everyday practice.
We used the World Health Organization’s (WHO) HEARTS (Healthy lifestyle counselling, Evidence-based treatment protocols, Access to essential medicines and technology, Risk-based cardiovascular disease management, Team-based care, Systems for monitoring) framework to integrate these recommendations into streamlined, pragmatic, and evidence-based algorithms for use in primary care in Canada. Designed to improve population-wide hypertension control and reduce cardiovascular disease burden, HEARTS outlines principles for optimal diagnostic procedures and simplified directive treatment algorithms along with monitoring and evaluation. 8 HEARTS was initially developed within the Kaiser Permanente system in the United States, where hypertension control rates improved from 44% to 90% in just over a decade.9,10 HEARTS was not designed as a rigid protocol; rather, nations are encouraged to adapt the framework to meet their unique needs, and the framework has been successfully implemented in a host of countries to improve hypertension care. 11
Herein, we present the Hypertension Canada guideline recommendations and HEARTS-adapted algorithms for the diagnosis and treatment of hypertension in adults in Canadian primary care.
Scope
The target users for this guideline are primary care providers (family physicians, nurse practitioners, nurses, and pharmacists), policy makers, and patients and caregivers affected by hypertension. Whereas this guideline is intended as a framework for managing most cases of hypertension in primary care, it is not intended to be applied directly to all clinical scenarios. Specifically, this guideline is not to be used to manage hypertension in children or in people who are pregnant or trying to become pregnant; separate guidelines are available for these patient populations.12,13 This guideline is designed as an adjunct to the forthcoming Hypertension Canada comprehensive guideline, to enhance implementation in the primary care setting. The upcoming comprehensive guideline will serve as a resource for more complex and nuanced aspects of hypertension management, such as resistant hypertension.
Recommendations
We formulated the guideline recommendations using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) framework (Table 1) 14 ; they are summarized in Table 2. Along with each recommendation, we provide the supporting rationale and the values and preferences that the guideline committee prioritized, based on the existing literature and perspectives shared by primary care providers and patients.
Interpretation of strength of recommendation and certainty of evidence using the Grading of Recommendations Assessment, Development and Evaluation approach 14

Recommendations on the diagnosis and treatment of hypertension in primary care
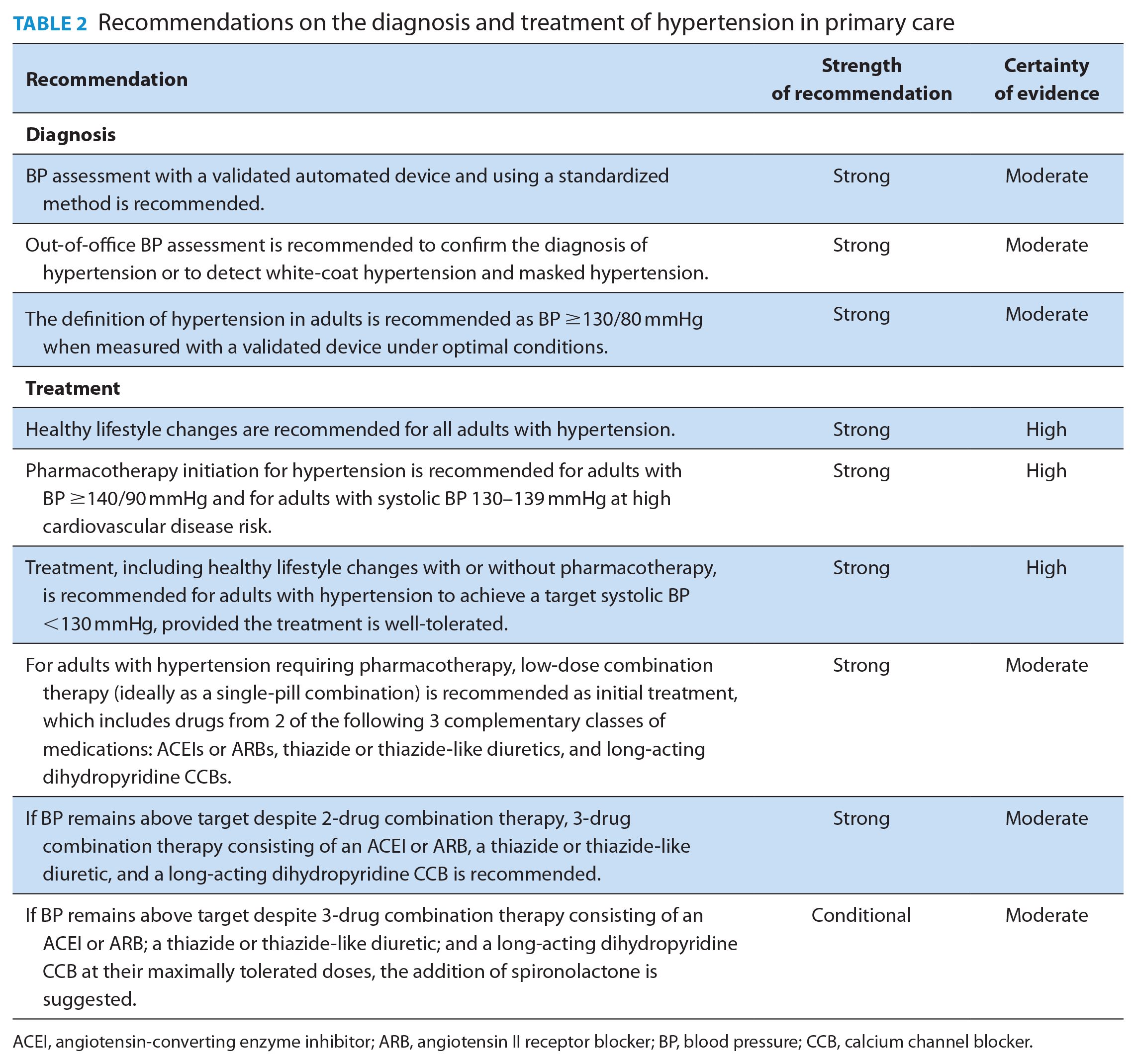
ACEI, angiotensin-converting enzyme inhibitor; ARB, angiotensin II receptor blocker; BP, blood pressure; CCB, calcium channel blocker.
Diagnosis
Blood pressure assessment with a validated automated device and using a standardized method is recommended (strong recommendation, moderate-certainty evidence).
Rationale
Because detection and management of hypertension rely on accurate BP measurement, it is important to use a device that has been validated and confirmed for accuracy. Validated automated oscillometric devices are preferred to auscultatory sphygmomanometers as they are easier to use, less prone to human error and end-digit preference (i.e., where the observer rounds off the last digit), and have better reproducibility. 15 Validation demonstrates relative equivalency between the tested device and rigorously performed manual auscultatory measurements. 16 Globally, only 10% of devices have evidence of validation for accuracy. 17 In Canada, 90% of BP devices sold at pharmacies are validated compared with only 45% of BP devices sold by online retailers. 18 Exceptions where automated devices are inaccurate and manual BP measurement is preferred include in patients with persistent or high burden of arrhythmias, and populations in which an automated device has not been validated (e.g., children and pregnant people, for whom this guideline is not intended). 16
Even when a validated automated device is used, the accuracy of BP measurement may be influenced by many factors.19-21 A standardized procedure with proper preparation and positioning, appropriate equipment, and multiple averaged measurements reduces variability (Figure 1). 22 Meta-analyses show that standardized automated office BP provides BP measurements that closely approximate daytime ambulatory BP monitoring (ABPM) and home BP monitoring (HBPM).23,24 Non-standardized office BP measurements result in readings that are on average 5–10 mmHg higher than standardized measurements. 25 Measuring BP more than once with all values averaged reduces short-term variability. 26 The optimal number of measurements is uncertain, although most recent clinical trials have employed a protocol consisting of a 5-minute seated rest period, followed by 3 measurements at 1-minute intervals.27-30
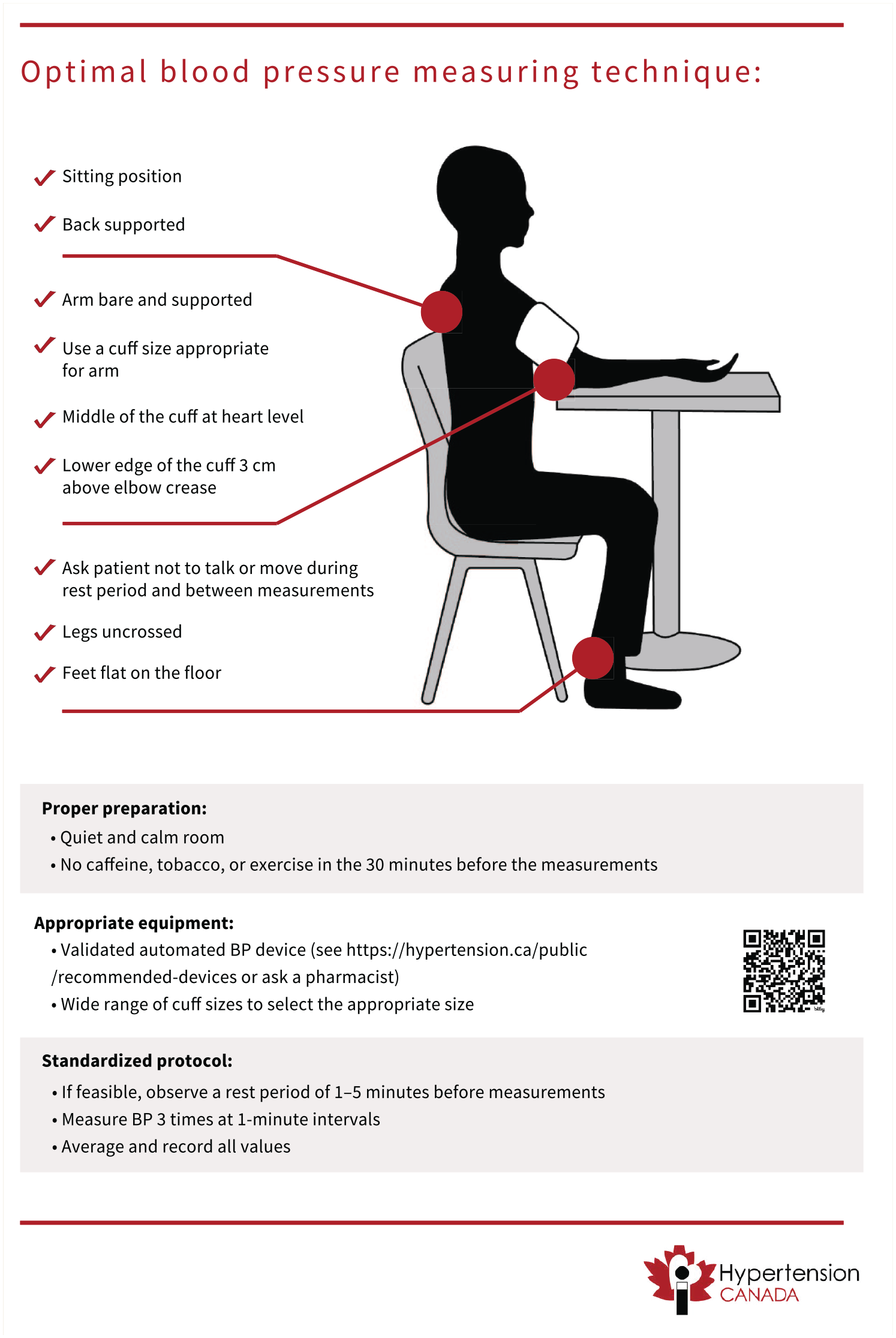
Optimal blood pressure measuring technique
Values and preferences
The guideline committee placed a high value on using proper BP measurement technique and equipment to ensure accurate readings. As such, the recommendation prioritizes precision in BP assessment to ensure appropriate diagnosis and management. While recognizing that access to validated devices and standardized methods may be limited in some settings, this recommendation underscores the importance of maintaining measurement quality to reduce errors and improve clinical decision-making.
Out-of-office BP assessment is recommended to confirm the diagnosis of hypertension or to detect white-coat hypertension and masked hypertension (strong recommendation, moderate-certainty evidence).
Rationale
Out-of-office BP measurements (ABPM or HBPM) are useful to confirm the diagnosis of hypertension when office BP is elevated. Ambulatory BP monitoring measures BP at 20- to 30-minute intervals during both day and night. 31 The standard protocol for HBPM involves measuring BP in duplicate twice daily for a week. Out-of-office BP measurements (particularly ABPM) correlate more closely with cardiovascular events and death than office BP measurements do. For example, a large observational study of about 60,000 primary care patients found that 24-hour ambulatory systolic BP was strongly associated with cardiovascular death (hazard ratio [HR] 1.51, 95% confidence interval [CI] 1.41–1.62) and all-cause death (HR 1.43, 95% CI 1.37–1.49), even after adjusting for office BP. 32 Out-of-office BP assessment is also required to identify the common BP phenotypes of white-coat hypertension (BP elevated in-office but not out-of-office, which is present in 15%–30% of people with elevated office BP) 33 and masked hypertension (BP elevated out-of-office but not in-office; prevalence of 10%–15%).33,34
When out-of-office BP measurements are not feasible owing to lack of accessibility, affordability, or adequate training for patients or caregivers, the diagnosis of hypertension can be confirmed with repeat office BP measurement using the standardized technique. Although reliance on single-visit office BP measurements to diagnose hypertension reduces specificity relative to ABPM, 35 it may need to be considered in certain circumstances, such as for patients with infrequent office visits who are unable or unwilling to perform out-of-office measurements.
Values and preferences
The guideline committee prioritized the importance of accurate hypertension diagnosis by emphasizing out-of-office BP assessment. The recommendation places a high value on minimizing misdiagnosis from white-coat hypertension or masked hypertension, which could lead to unnecessary treatment or missed cases of hypertension. Although accessibility and feasibility of out-of-office BP monitoring may vary across different settings, this recommendation places greater importance on diagnostic precision over potential challenges in implementation.
The definition of hypertension in adults is recommended as BP ≥130/80 mmHg when measured with a validated device under optimal conditions (strong recommendation, moderate-certainty evidence).
Rationale
Starting as low as with a systolic BP of 90 mmHg, observational data have shown a continuous relationship between higher BP and risk for adverse cardiovascular outcomes. 36 Yet for clinical care and public health purposes, it is helpful to establish a categorical threshold to define hypertension. In the present guideline, we set the BP threshold to define hypertension in adults at 130/80 mmHg, a threshold below what was previously recommended by Hypertension Canada. 31
The rationale behind this change is based on observational and randomized controlled trial (RCT) data on the relationship between BP and the magnitude of cardiovascular risk. Meta-analysis of prospective study data has shown that the relative risk for major adverse cardiovascular events for people with BP ≥130–139/85–89 mmHg is 1.5-fold to 2.0-fold higher than for people with BP <120/80, and a risk substantially higher than for those with BP 120–129/80–84 mmHg. 37 Similarly, RCT data on the effects of more intensive BP-lowering treatments have consistently shown their effectiveness in reducing the risk for major adverse cardiovascular events for people with BP ≥130/80 mmHg (and in some cases for people with BP <130/80 mmHg), as discussed in the “Treatment” recommendations section.27,28,30,38-44
In adults with confirmed hypertension, routine testing should be performed to assess cardiovascular disease risk and screen for end-organ damage (Appendix 1, Supplementary Table 1, available online under Supplementary Materials).
Values and preferences
The guideline committee placed high value on early detection and intervention by defining hypertension at a lower threshold (BP 130/80 mmHg) than in previous guidelines. This reflects a high value placed on aligning with emerging evidence that associates cardiovascular risk with lower BP levels. Although this lower threshold will increase the number of people labelled as having hypertension, the recommendation emphasizes the benefits of earlier management in preventing long-term complications.
Treatment
Healthy lifestyle changes are recommended for all adults with hypertension (strong recommendation, high-certainty evidence).
Rationale
Unhealthy lifestyle habits play a major role in the development of hypertension and its associated cardiovascular morbidity and mortality. Thus, lifestyle modification should be advised for all people with hypertension. Dietary salt intake displays a nearly linear dose-response relationship with BP. 45 Data from RCTs show that reducing dietary sodium via a salt substitute among people aged 60 years or older with hypertension and a history of stroke led to a 13% decrease in major adverse cardiovascular events (rate ratio 0.87, 95% CI 0.80–0.94) and a 12% decrease in all-cause death (rate ratio 0.88, 95% CI 0.82–0.95) over about 5 years. 46 The WHO advises restriction of dietary sodium intake to less than 2 g per day. 47 Notably, more than 70% of dietary sodium intake from the typical Western diet comes from processed foods rather than table salt. 48
Increased dietary potassium intake (i.e., from a diet rich in fruits and vegetables) is linked to lower BP and reduced cardiovascular risk.46,49-51 The WHO advises dietary potassium intake of more than 3.5 g per day. 52 For patients with chronic kidney disease or those prescribed medications that may raise potassium (e.g., angiotensin-converting enzyme inhibitors [ACEIs], angiotensin II receptor blockers [ARBs], and mineralocorticoid receptor antagonists), potassium should be monitored.
Hypertension and obesity (body mass index ≥30) are commonly comorbid conditions, and weight loss in these cases can improve BP control. A meta-analysis of RCTs on weight-reducing diets in adults with hypertension and obesity found a mean decline in systolic and diastolic BP of 4.5 (95% CI 1.8–7.2) and 3.2 (95% CI 1.5–4.8) mmHg, respectively, although the reduction in BP occurred in a dose-dependent relationship with the magnitude of weight reduction. 53 Weight-reducing medications such as glucagon-like peptide 1 receptor agonists are also effective in improving BP. For instance, a RCT of semaglutide in adults with overweight or obesity showed that in addition to a 12% reduction in weight, mean systolic BP was also reduced by 5.1 (95% CI 3.9–6.3) mmHg. 54
Regular exercise can improve BP control. Dynamic aerobic exercise has been the most well-studied form of exercise, with meta-analysis data showing that among people with hypertension, mean systolic and diastolic BP improved by 6.9 and 4.9 mmHg, respectively. 55 The WHO advises at least 150–300 minutes of moderate aerobic activity per week.31,56
Reducing alcohol consumption lowers BP in a dose-dependent fashion with a suggested threshold effect. Meta-analysis data demonstrate that among adults who consume 2 or fewer drinks per day, reducing alcohol intake had no effect on BP. However, for adults consuming 3, 4–5, or 6 or more drinks per day, reducing alcohol intake is associated with a mean reduction in systolic BP of 1.2 (95% CI 0.0–2.3), 3.0 (95% CI 2.0–4.0), and 5.5 (95% CI 4.3–6.7) mmHg, respectively.31,57,58
Finally, although the effects of smoking on BP are only modest, 59 all people with hypertension should be counselled to stop smoking to reduce its associated risks of cardiovascular morbidity and death. 60
Values and preferences
The guideline committee prioritized the foundational role of healthy lifestyle changes in managing hypertension, recognizing their broad benefits beyond BP control. This recommendation reflects a high value placed on non-pharmacologic interventions that can improve overall health and reduce cardiovascular risk. Given that various healthy lifestyle changes have been shown to potentially reduce BP, the committee chose not to specify particular interventions in the recommendation, recognizing that people may have different values and preferences regarding which changes to prioritize.
Pharmacotherapy initiation for hypertension is recommended for adults with BP ≥140/90 mmHg and for adults with systolic BP 130–139 mmHg at high cardiovascular disease risk (strong recommendation, high-certainty evidence).
Rationale
When an adult is diagnosed with hypertension and their BP is ≥140/90 mmHg, pharmacologic treatment should be initiated. This is based on RCT data demonstrating that such individuals benefit from pharmacologic treatment, including 28% lower odds of stroke (odds ratio [OR] 0.72, 95% CI 0.55–0.94) and 22% lower odds of all-cause death (OR 0.78, 95% CI 0.67–0.92) over 5 years, irrespective of their baseline cardiovascular disease risk.39,61
Pharmacologic treatment should also be started for adults with systolic BP 130–139 mmHg who are at high-risk of cardiovascular disease (Box 1). This threshold is based on meta-analysis data of RCTs showing that adults at high baseline risk for cardiovascular disease with BP within this range benefit from a reduction in risk for major adverse cardiovascular events with pharmacologic treatment, primarily driven by a 60% decrease in stroke risk for every 10 mmHg decrease in systolic BP achieved.39,62
High cardiovascular disease risk conditions*

Clinicians should emphasize that pharmacologic treatment is to be used in combination with (and not as a replacement for) lifestyle modification, and that successful lifestyle modification may allow for down-titration or discontinuation of medications in the future. For adults with systolic BP 130–139 mmHg and not at high cardiovascular disease risk, healthy lifestyle changes alone should be emphasized, with BP reassessment within 3–6 months. If systolic BP remains at 130–139 mmHg and the person remains not at high cardiovascular disease risk, we advise BP reassessment every 6–12 months.
Values and preferences
The guideline committee prioritized timely initiation of pharmacotherapy to reduce cardiovascular risk, placing a high value on preventing complications associated with elevated BP by targeting people at the greatest risk. The committee recognizes that the benefit of pharmacotherapy initiation in people at lower risk (i.e., systolic BP 130–139 without any high-risk conditions) is uncertain. This recommendation places lower value on pharmacotherapy avoidance, costs, and tolerability, as most agents are now available as low-cost generics and are well-tolerated.
Treatment, including healthy lifestyle changes with or without pharmacotherapy, is recommended for adults with hypertension to achieve a target systolic BP <130 mmHg, provided the treatment is well-tolerated (strong recommendation, high-certainty evidence).
Rationale
The recommended treatment target of systolic BP <130 mmHg is supported by current RCT evidence showcasing the benefits of more intensive BP control.27,28,30,39-44 A recent large meta-analysis of RCTs evaluating lower BP targets with pharmacotherapy (>70,000 participants) found that compared with a systolic BP target of ≥130 mmHg, a systolic BP target of <130 mmHg led to a 22% reduction in major adverse cardiovascular events (HR 0.78, 95% CI 0.70–0.87) and an 11% reduction in all-cause death (HR 0.89, 95% CI 0.79–0.99). 38 Notably, this meta-analysis also showed that compared with a systolic BP target of <140 mmHg, a systolic BP target of <120 mmHg led to an 18% reduction in major adverse cardiovascular events (HR 0.82, 95% CI 0.74–0.91) and a possible reduction in all-cause death (HR 0.85, 95% CI 0.71–1.01). 38 The Systolic Blood Pressure Intervention Trial (SPRINT; comparing a systolic BP target of <120 mmHg vs <140 mmHg) reported a number needed to treat (NNT) of 61 for the primary cardiovascular composite outcome (myocardial infarction, other acute coronary syndromes, stroke, heart failure, or death from cardiovascular causes) and an NNT of 90 for all-cause death. 30
Rather than recommending the more intensive BP target of systolic BP <120 mmHg, we chose to recommend a more conservative systolic BP target of <130 mmHg, given that research-quality BP measures are generally 5–10 mmHg lower than BP measures in the routine clinical care setting 25 ; modern-day RCTs studying intensive BP control included only people with baseline systolic BP ≥130 mmHg 38 ; most participants in large RCTs targeting a systolic BP <120 mmHg did not achieve this target27,29,30; and feedback from primary care providers and patients supported the recommendation. We do not recommend a specific diastolic BP target, given evidence that adults with a systolic BP <130 mmHg are at relatively low cardiovascular risk even when diastolic BP is 70–90 mmHg. 65
Although RCTs on intensive BP control have proven benefit in reducing cardiovascular events and risk of death,27,28,30,40-44 implementation of intensive BP control must be balanced against potential harms. A recent meta-analysis of RCTs on intensive BP targets showed significantly increased rates of the following adverse events (although the absolute risks as shown by the numbers needed to harm [NNH] were low): hypotension (NNH 508, 95% CI 309–1425), syncope (NNH 1701, 95% CI 991–5999), injurious falls (NNH 2941, 95% CI 1479–258 938), electrolyte abnormalities (NNH 3222, 95% CI 1150–4013), and acute kidney injury or acute renal failure (NNH 1657, 95% CI 693–4235). 38
Exceptions to targeting a systolic BP <130 mmHg include patient-specific factors such as goals of care, frailty, fall risk, and orthostatic hypotension. In these scenarios, a higher systolic BP target may be required to minimize adverse effects, and we advise targeting a systolic BP as low as is reasonably achievable, although this threshold varies on a case-by-case basis, necessitating use of clinical discretion.
Values and preferences
The guideline committee placed a relatively high value on a simplified approach to hypertension management by adopting a single treatment target for all, irrespective of cardiovascular risk and comorbidities. While we recognize that some people may benefit from a lower target and some may tolerate only a higher target, this recommendation prioritizes ease of implementation and is a response to requests for more pragmatic and streamlined guidance to hypertension management in primary care.
For adults with hypertension requiring pharmacotherapy, low-dose combination therapy (ideally as a single-pill combination) is recommended as initial treatment, which includes drugs from 2 of the following 3 complementary classes of medications: angiotensin-converting enzyme inhibitors or angiotensin II receptor blockers, thiazide or thiazide-like diuretics, and long-acting dihydropyridine calcium channel blockers (strong recommendation, moderate-certainty evidence).
Rationale
On average, ACEIs or ARBs, thiazide or thiazide-like diuretics, dihydropyridine calcium channel blockers (CCBs), and β-blockers reduce BP to a similar degree 66 and effectively lower cardiovascular risk when used at optimal doses. 67 However, the magnitude of reduction of cardiovascular risk (specifically for stroke) is less with β-blocker use for hypertension, and β-blockers are more likely to be discontinued because of adverse effects. 68 Given their less favourable benefit-to-risk ratio, β-blockers are not recommended as first-line therapy for hypertension unless a specific clinical indication is present, such as heart failure, angina, postmyocardial infarction, or heart rate or rhythm control.69,70 Angiotensin-converting enzyme inhibitors or ARBs, thiazide or thiazide-like diuretics, and dihydropyridine CCBs are well-tolerated, provide similar cardiovascular risk protection, and should all be considered first-line agents for hypertension.67,71
However, ACEIs and ARBs are teratogenic, particularly when taken in the second or third trimester of pregnancy, and should be avoided or discontinued in all people who are pregnant or trying to become pregnant. 72 Moreover, all people of childbearing age should be counselled on the teratogenic risk of ACEIs and ARBs as part of the shared decision-making process.
Although thiazide-like diuretics were previously preferred over thiazide diuretics, 31 an RCT comparing chlorthalidone and hydrochlorothiazide found similar efficacy in mitigating adverse cardiovascular events (HR 1.04, 95% CI 0.94–1.16), whereas hypokalemia incidence was higher with chlorthalidone (6.0% vs 4.4%, p < 0.001). 73
We recommend upfront combination therapy (ideally as a single-pill combination) with an ACEI or ARB plus either a thiazide or thiazide-like diuretic or a dihydropyridine CCB for adults with hypertension requiring pharmacologic treatment. About 70% of adults with hypertension will require more than 1 class of medication to achieve BP control, 74 a proportion projected to increase as BP targets are lowered.
Combining lower doses of different classes provides additive BP-lowering effects while minimizing adverse effects. 66 Meta-analysis data show that single-pill combinations reduce mean systolic BP by 4.0 mmHg (95% CI 0.1–7.9) beyond that achieved by free-equivalent combination therapy. 75 Single-pill combinations achieve BP control in about one-third more patients than with standard monotherapy (65% vs 48%, risk ratio [RR] 1.32, 95% CI 1.20–1.45). 76 Compared with free-drug combinations, single-pill combinations are associated with significantly better drug adherence (OR 1.21, 95% CI 1.03–1.43) and a possible trend toward better drug persistence (OR 1.54, 95% CI 0.95–2.49). 77 An observational study of more than 100,000 patients with hypertension showed that upfront combination therapy reduced therapeutic inertia, as these patients were over 2-fold more likely to be on a multidrug prescription at 3 years than those started on monotherapy. 78 Notably, the patients started on combination therapy in this study also had a 16% (95% CI 10%–21%) reduction in hospital admission for cardiovascular events and a 20% (95% CI 11%–28%) reduction in all-cause death. 78
Single-pill combinations also yield substantial cost savings compared with their free-drug equivalents, with a 2009 Canadian study estimating a yearly cost savings of $27–45 million. 79 With regard to potential harms from use of single-pill combinations in the initial management of hypertension, meta-analysis data showed no difference in withdrawal from adverse events for single-pill combinations compared with free-drug combination use (OR 0.80, 95% CI 0.58–1.11)76,77 or standard-dose monotherapy (RR 1.19, 95% CI 0.83–1.69). 76 However, there is a higher incidence of dizziness with single-pill combinations than with standard-dose monotherapy (RR 1.54, 95% CI 1.08–2.19). 76
Values and preferences
The guideline committee placed a high value on initiating a combination of effective and well-tolerated drugs to reduce therapeutic inertia and increase time in target range. Single-pill combination agents are often available at lower cost than their individual components. While recognizing the potential uncertainty regarding which agent may cause intolerance in a combination pill, we placed lower value on this concern than on the benefits of timely and efficient treatment.
If BP remains above target despite 2-drug combination therapy, 3-drug combination therapy consisting of an angiotensin-converting enzyme inhibitor or angiotensin II receptor blocker, a thiazide or thiazide-like diuretic, and a long-acting dihydropyridine calcium channel blocker is recommended (strong recommendation, moderate-certainty evidence).
Rationale
Given the aforementioned benefits regarding effective BP and cardiovascular risk reduction relative to other medication classes (discussed under previous recommendation), we recommend using ACEIs or ARBs, thiazide or thiazide-like diuretics, and long-acting dihydropyridine CCBs preferentially in combination in adults with hypertension requiring pharmacotherapy.67,80,81 When 3 drugs are required to achieve BP control, we recommend combining all 3 of these complementary medication classes. Notably, ACEIs and ARBs should not be used in combination, as they increase the risk for adverse events including hyperkalemia, acute kidney injury, hypotension, and syncope, with no added clinical benefit.82,83 Recently, single-pill triple-combination therapy with ACEI or ARB, thiazide or thiazide-like diuretic, and dihydropyridine CCB has been shown to be effective in improving hypertension control84-86; however, this single pill is not yet available in Canada.
Values and preferences
The guideline committee prioritized the use of medications with well-established cardiovascular benefits over those for which such benefits have not been clearly demonstrated.
If BP remains above target despite 3-drug combination therapy consisting of an angiotensin-converting enzyme inhibitor or angiotensin II receptor blocker, a thiazide or thiazide-like diuretic, and a long-acting dihydropyridine calcium channel blocker at their maximally tolerated doses, the addition of spironolactone is suggested (conditional recommendation, moderate-certainty evidence).
Rationale
Resistant hypertension is present when BP remains above target despite adherence to the combination of an ACEI or ARB, thiazide or thiazide-like diuretic, and CCB at their maximally tolerated doses. 64 In this setting, the addition of spironolactone results in substantially greater BP reduction than with alternative fourth-line agents. The PATHWAY-2 RCT found that among adults with resistant hypertension, the mean reduction in systolic BP was greater with spironolactone than with placebo (8.7, 95% CI 7.7–9.7 mmHg), doxazosin (4.0, 95% CI 3.0–5.0 mmHg), and bisoprolol (4.5, 95% CI 3.5–5.5 mmHg). 87 Mechanistic subanalyses later showed that these findings related to a high proportion of dysregulated aldosterone production among people with resistant hypertension. 88 However, no prospective data exist that demonstrate improved cardiovascular outcomes with spironolactone compared with other antihypertensive agents.
The PATHWAY-2 trial showed no difference in serious adverse events or withdrawal for adverse events (including hyperkalemia and gynecomastia) with spironolactone versus doxazosin, bisoprolol, or placebo. 87 However, spironolactone is known to increase the risk of hyperkalemia, particularly in patients with chronic kidney disease (in whom spironolactone use is associated with a 3-fold higher risk of hyperkalemia-associated hospital admission) 89 and those taking other medications that raise potassium levels (e.g., ACEIs or ARBs). 90 We advise monitoring serum potassium 2–4 weeks after spironolactone initiation and with any dose adjustment. Given the antiandrogen effects of spironolactone, males should be counselled on the risk of gynecomastia. For instance, in the Randomized Aldactone Evaluation Study (RALES) trial, which randomized patients with heart failure with reduced ejection fraction to spironolactone 25 mg versus placebo, 10% of males randomized to spironolactone developed gynecomastia, compared with 1% of males randomized to placebo. 91
Patients with resistant hypertension should be considered for screening for secondary causes of hypertension and referral to specialist care. Specifically, given the high prevalence of primary aldosteronism among patients with resistant hypertension,92-95 screening with aldosterone and renin measurements (ideally before the introduction of spironolactone) should be performed. 96
Values and preferences
The guideline committee placed high value on the proven BP-lowering benefits of spironolactone compared with other fourth-line agents, while assigning lower value to potential adverse effects, including the risk of hyperkalemia, that may arise with its use. The lower strength of evidence for this recommendation reflects the limited data available on the long-term cardiovascular benefits of the treatment.
Methods
This guideline was developed by the volunteer Primary Care Guideline Committee of Hypertension Canada and was supported by Hypertension Canada. We followed GRADE 14 and ADAPTE 97 frameworks to develop the recommendations in accordance with the Appraisal of Guidelines for Research and Evaluation (AGREE II) quality and reporting standards. 98
Composition of participating groups
The Primary Care Guideline Committee was selected by the Hypertension Canada Guideline Executive Committee. The guideline committee consisted of family medicine physicians (K.A.T., J.H., and G.C.), pharmacists (R.T.T. and S.C.G.), a nurse practitioner (J.B.), hypertension specialists (R.G. [co-chair], G.L.H. [co-chair], R.T.T., A.A.L., N.R.C.C., and E.L.S.), and a methodologist (N.S.) with expertise in guideline development and GRADE methodology. Additionally, a working group of 4 patient-partners with lived experience with hypertension (listed in Acknowledgements) was formed, which provided feedback throughout the guideline development process and led the creation of the associated patient support tool.
Selection of priority topics
During an in-person open forum session at the 2023 Canadian Hypertension Congress on Oct. 23, 2023, with more than 200 primary care providers, hypertension providers, and patients with lived experience, there was a consensus request for the development of a streamlined, pragmatic, and evidence-based hypertension guideline specifically focused on primary care.
With input from the primary care providers on the guideline committee and the patient-partner working group, and after review of existing hypertension guidelines (discussed below), the committee came to consensus on the priority topics for inclusion, encompassing diagnosis and management of hypertension. Additionally, in response to the provider request made at the congress, the committee decided to limit the number of recommendations to only those deemed most relevant to primary care.
Literature review and quality assessment
The guideline committee used the ADAPTE 97 framework to select international hypertension guidelines published over the previous 10 years. The committee considered the quality and availability of evidence reviews, use of GRADE or similar grading frameworks to rate the strength of recommendations and certainty of evidence systematically and explicitly, clarity of recommendations, and publication timeframes when selecting the source guidelines: American Heart Association, 99 WHO, 71 and the European Society of Cardiology (ESC). 64 The committee used the existing evidence syntheses and GRADE levels from each of these source guidelines when developing the recommendations. The ESC guideline had the most recently updated recommendations (published in August 2024), with evidence tables updated to January 2024, with the exception of the Effects of Intensive Blood Pressure Lowering Treatment in Reducing Risk of Cardiovascular Events (ESPRIT) trial, which was published in June 2024.28,64
Development of recommendations
The guideline committee meetings were held virtually on a monthly basis from July 25, 2024, to Jan. 20, 2025. The committee reviewed the evidence from the source recommendations and made judgments about the effects of the interventions, feasibility, resources, patient values and preferences, and availability or accessibility issues. Each recommendation was discussed and either adapted or adopted. Consensus was desired; however, when a unanimous decision could not be reached, the committee voted, with consensus defined as more than 70%. We assigned the strength of each recommendation according to the GRADE framework, as strong (applying the terminology “recommended”) or conditional (applying the terminology “suggested”). 14 Further information on how each recommendation was adapted from the source guidelines is available in Appendix 1, Supplementary Tables 2–10.
In addition, the committee used HEARTS to develop streamlined algorithms for hypertension diagnosis and treatment to optimize implementation into the Canadian primary care setting, which incorporate and align with the recommendations in this guideline. 8
External review
The preliminary guideline was posted on the Hypertension Canada website from Sept. 27, 2024, through Oct. 25, 2024, for external review. Family physicians, pharmacists, nurses, hypertension specialists, and patients and caregivers from across Canada were invited to review the guideline and provide feedback via an online survey with both English and French versions. Requests for feedback were emailed using mailing lists from major national organizations representing these stakeholders (listed in the Acknowledgements section), including requests for patient-specific feedback from established patient-partners from Hypertension Canada and the Heart and Stroke Foundation of Canada. We received 143 responses (45 pharmacists, 26 family physicians, 25 other physicians, 19 nurse practitioners, 14 nurses, 10 patients and 4 researchers). The feedback was collated, reviewed, and incorporated to update both the recommendations and HEARTS-derived algorithms (as appropriate) upon collective review by the guideline committee. The responses to frequent comments and questions received are detailed in Appendix 2, available online under Supplementary Materials.
Management of competing interests
Competing interests for all guideline committee members were managed according to Hypertension Canada policies, which are informed by Guidelines International Network principles (Appendix 3, available online under Supplementary Materials). 100 On appointment, all members agreed to avoid direct competing interests with companies that could be affected by the guideline and provided written disclosure of all financial and non-financial interests relevant to the guideline topic. Verbal updates on competing interests were requested at the beginning of every committee meeting. The conflict-of-interest oversight committee reviewed the disclosures and made judgments about conflicts. Guideline chairs were required to be free of relevant financial competing interests throughout the guideline development process. Two committee members with potentially relevant disclosures related to pharmaceutical companies were permitted to join in the discussion about pharmacotherapy recommendations but were excluded from voting on these topics. Notably, only generic medications were discussed.
Funding came solely from Hypertension Canada, which receives its funding from the following sources: device endorsement revenue (63%); investment income (18%); certification program revenue (7%); grants and sponsorships (4%), including from a health technology company; registration fees (4%); membership dues (4%); and donations (<1%). No funding is received from pharmaceutical companies. Hypertension Canada funded the guideline and assembled the committee; however, it had no role in developing the recommendations or supporting resources. No sponsorship was accepted for this guideline, and no companies were allowed to participate in the guideline process.
Implementation
Consistent with the guideline recommendations, the committee adapted the HEARTS framework to develop suggested algorithms for the diagnosis (Figure 2) and treatment (Figure 3) of hypertension in the Canadian primary care setting. 8 Standardized protocols are effective in improving population-wide BP control. A recent meta-analysis of RCTs demonstrated that standardized treatment protocols reduce mean systolic and diastolic BP by 6.7 mmHg (95% CI 3.7–9.8) and 2.6 mmHg (95% CI 1.2–4.1), respectively, compared with usual care. 101 With broad adoption, specific algorithms can reduce drug costs through bulk drug purchasing and make standardized education and task sharing more efficient and less costly. 11
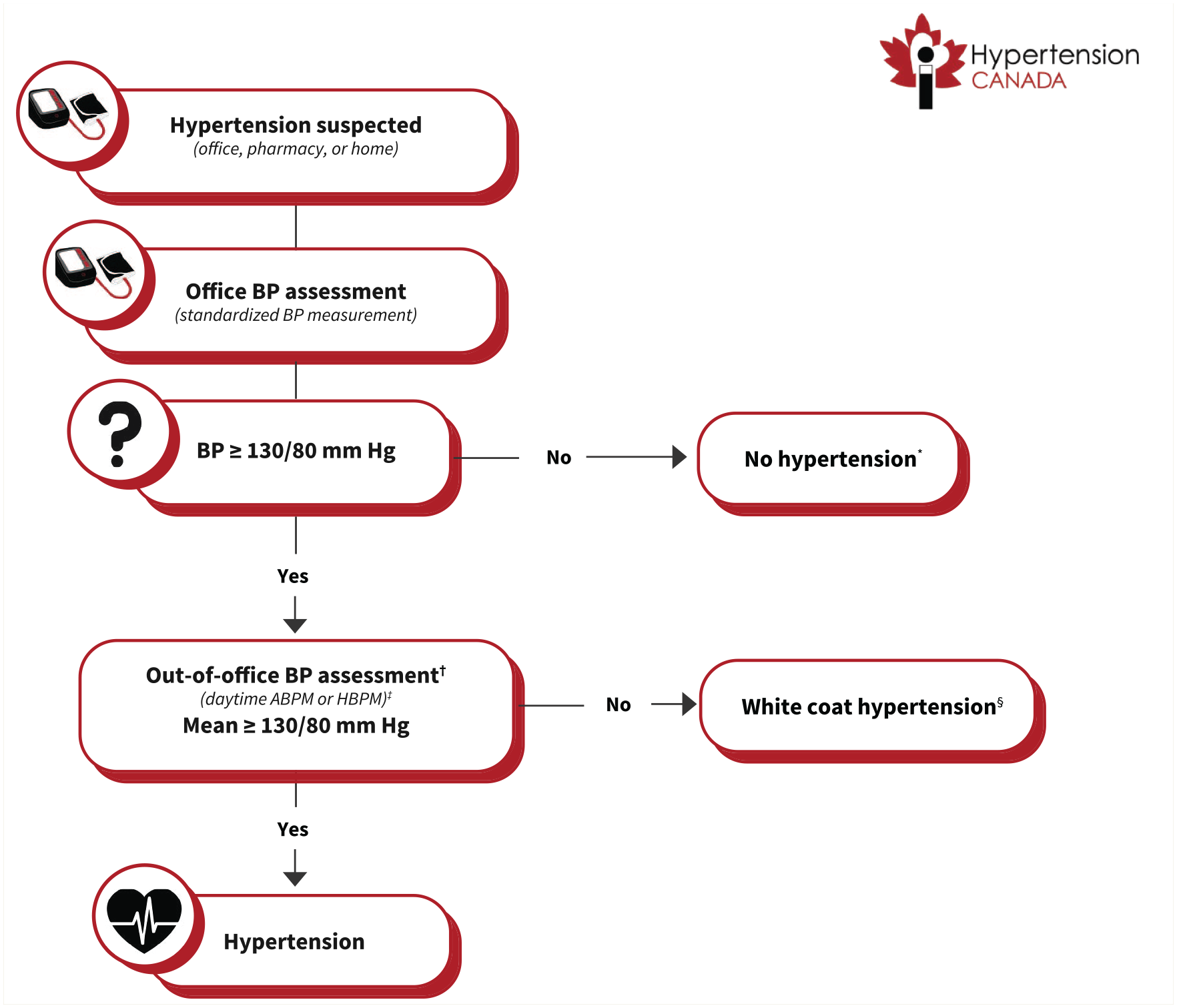
Suggested algorithm for the diagnosis of hypertension in primary care
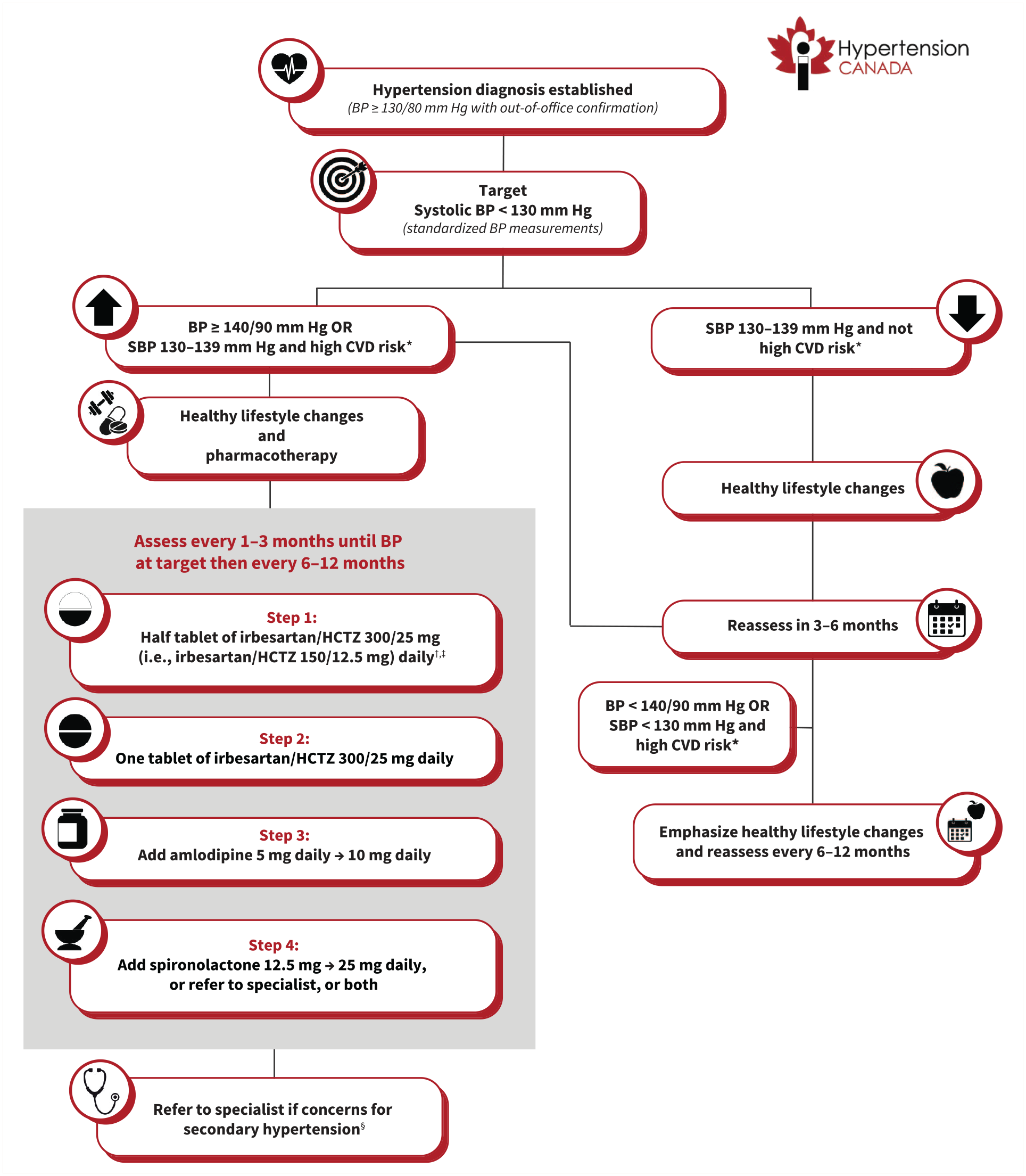
Suggested algorithm for the treatment of hypertension in primary care (not intended for use in pregnancy)
Factors we considered for the recommended drug selection within the algorithm included efficacy, tolerability, cost, coverage, availability, protection from future drug shortages, and ability to split pills (Appendix 2). Based on these, we selected a combination pill with irbesartan and hydrochlorothiazide as the preferred initial combination therapy at this time, although the initial combination will be periodically reconsidered given availability and new evidence to suggest more cost-effective choices. Acceptable alternative single-pill combination therapies currently available in Canada are listed in Table 3.
Available single-pill antihypertensive medication combinations in Canada and associated costs
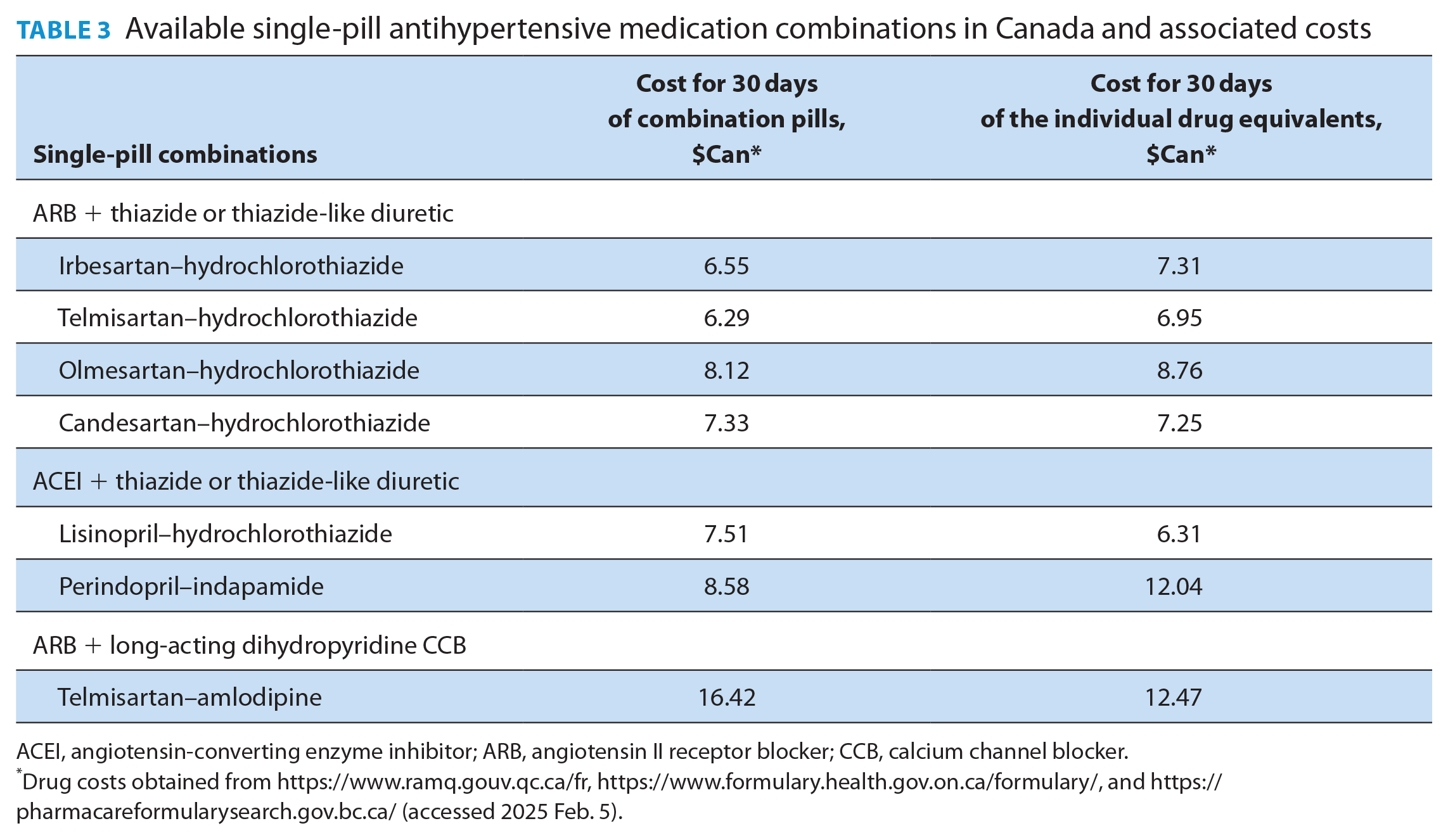
ACEI, angiotensin-converting enzyme inhibitor; ARB, angiotensin II receptor blocker; CCB, calcium channel blocker.
Drug costs obtained from https://www.ramq.gouv.qc.ca/fr, https://www.formulary.health.gov.on.ca/formulary/, and https://pharmacareformularysearch.gov.bc.ca/ (accessed 2025 Feb. 5).
Importantly, the cost of most single-pill combinations is generally lower than the cost for the equivalent individual components. It should be noted that the lower BP thresholds adopted for both defining hypertension and treatment targets relative to previous Hypertension Canada guidelines means that a higher number of people will be labelled as having hypertension, which may have meaningful personal implications (e.g., stigma, insurance). 31 However, the committee thought that the contemporary evidence strongly supports the lower thresholds to promote early detection of, and intervention for, hypertension, which will translate into mitigating long-term cardiovascular complications at the population level.
The HEARTS framework has a well-established implementation policy to engage primary care and optimize guideline uptake, which will be leveraged for this purpose within Canada. 8 This includes active engagement with primary care centres, along with the development of knowledge transfer tools to support health care providers and patients. These tools include a downloadable patient support tool, to be made widely available for posting in clinics and provision to patients as educational handouts (Appendix 4, available online under Supplementary Materials). Additional free online resources for health care providers and patients are listed in Appendix 5, available online under Supplementary Materials.
Timely dissemination of the guideline content and implementation tools will take place via the Hypertension Canada website (https://hypertension.ca) and network, social media, podcasts, and national primary care conferences. Successful implementation will be tracked via downloads of the patient support tool, traffic to the guideline website, and prospective trends on hypertension treatment and control rates, which Hypertension Canada has traditionally captured via the Canadian Health Measures Survey. 5
Hypertension Canada will update this guideline when new data pertinent to the diagnosis and treatment of hypertension emerge or when new cost-effective antihypertensive combination pills become available in Canada.
Other guidelines
Hypertension Canada has adopted a new 2-part guideline approach. This primary care-focused guideline was developed at the request of primary care providers for more pragmatic guidance on managing hypertension in routine clinical practice. An upcoming comprehensive guideline will be updated on a rolling topic-by-topic basis beginning later this year. 7 Currently, we are undertaking a prioritization exercise to determine the topics to be updated and the order in which these updates will occur. The comprehensive guideline will serve as a resource for more complex and nuanced aspects of hypertension management.
The recommendations within this guideline were developed based on evidence syntheses and adaptation of recommendations from the American Heart Association, 99 ESC, 64 and WHO hypertension guidelines. 71 Compared with the 2020 Hypertension Canada guideline, 31 the present guideline encourages initial low-dose combination therapy. Additionally, a common request from primary care providers in developing this guideline was to provide a single BP threshold definition for hypertension and a single BP treatment target. Therefore, in contrast to the 2020 Hypertension Canada guideline 31 but consistent with other international guidelines64,99 and based upon updated evidence on the benefits of more intensive BP-lowering,27-30,38 we adopted the definition of hypertension as BP ≥130/80 mmHg, and the treatment target of systolic BP <130 mmHg.
Gaps in knowledge
These recommendations are informed by the best level of evidence available to date. Ongoing research will continue to inform and advance hypertension care. Although a large armamentarium of antihypertensive medications already exists, a number of new drug classes have recently been developed.102-104 Furthermore, renal denervation provides a potential future interventional approach to improve BP control. 105 The role of these novel management approaches in future hypertension algorithms remains to be seen.
Limitations
Although we did not conduct new evidence reviews to support these recommendations, numerous reviews have been conducted by different organizations. Therefore, to reduce duplication of research and to expedite creation of a new primary care-focused guideline, we reviewed the evidence from high-quality, recently published guidelines. Correspondingly, the strength and certainty of the evidence of the recommendations were based on literature available at the time the source guidelines performed their evidence syntheses (the most recent being the 2024 ESC guideline). We acknowledge that the source guidelines were not developed using a Canadian context; however, we considered the feasibility, acceptability, resources, and other issues in Canada when adapting the recommendations.
Further, some aspects of the recommendations lack Canada-specific evidence. For instance, when using cardiovascular risk calculators to determine whether an individual is at high cardiovascular disease risk, several risk calculators are available.63,106-109 However, none are Canada-specific and it is uncertain which is most accurate within Canada.
Our guideline committee also attempted to incorporate NNT and NNH to demonstrate the risks and benefits relevant to each recommendation. These metrics were available for intensive BP targets, but not for all recommendations, and instead we used syntheses of descriptive summaries of effects. Finally, with the goal of developing pragmatic primary care-specific management recommendations, more nuanced details of hypertension management are not included in this guideline. For instance, topics such as how best to integrate care with ever-evolving complementary treatments (e.g., statins, sodium-glucose cotransporter 2 inhibitors, and glucagon-like peptide 1 receptor antagonists) to lower cardiovascular risk among patients with hypertension and other comorbidities were beyond the scope of this guideline. Such topics will be addressed with the forthcoming Hypertension Canada comprehensive guideline.
Conclusion
We aimed to provide pragmatic, evidence-based recommendations and algorithms to improve the standard for care for hypertension management in the Canadian primary care setting. Successful uptake of this guideline will serve to improve hypertension treatment and control at the population level.
A French version of these Hypertension Guidelines will be published in CPJ at a later date. ■
Supplemental Material
sj-pdf-1-cph-10.1177_17151635251343907 – Supplemental material for Hypertension Canada guideline for the diagnosis and treatment of hypertension in adults in primary care
Supplemental material, sj-pdf-1-cph-10.1177_17151635251343907 for Hypertension Canada guideline for the diagnosis and treatment of hypertension in adults in primary care by Rémi Goupil, Ross T. Tsuyuki, Nancy Santesso, Kristin A. Terenzi, Jeffrey Habert, Gemma Cheng, Stephanie C. Gysel, Jill Bruneau, Alexander A. Leung, Norman R.C. Campbell, Ernesto L. Schiffrin and Gregory L. Hundemer in Canadian Pharmacists Journal / Revue des Pharmaciens du Canada
Supplemental Material
sj-pdf-2-cph-10.1177_17151635251343907 – Supplemental material for Hypertension Canada guideline for the diagnosis and treatment of hypertension in adults in primary care
Supplemental material, sj-pdf-2-cph-10.1177_17151635251343907 for Hypertension Canada guideline for the diagnosis and treatment of hypertension in adults in primary care by Rémi Goupil, Ross T. Tsuyuki, Nancy Santesso, Kristin A. Terenzi, Jeffrey Habert, Gemma Cheng, Stephanie C. Gysel, Jill Bruneau, Alexander A. Leung, Norman R.C. Campbell, Ernesto L. Schiffrin and Gregory L. Hundemer in Canadian Pharmacists Journal / Revue des Pharmaciens du Canada
Supplemental Material
sj-pdf-3-cph-10.1177_17151635251343907 – Supplemental material for Hypertension Canada guideline for the diagnosis and treatment of hypertension in adults in primary care
Supplemental material, sj-pdf-3-cph-10.1177_17151635251343907 for Hypertension Canada guideline for the diagnosis and treatment of hypertension in adults in primary care by Rémi Goupil, Ross T. Tsuyuki, Nancy Santesso, Kristin A. Terenzi, Jeffrey Habert, Gemma Cheng, Stephanie C. Gysel, Jill Bruneau, Alexander A. Leung, Norman R.C. Campbell, Ernesto L. Schiffrin and Gregory L. Hundemer in Canadian Pharmacists Journal / Revue des Pharmaciens du Canada
Supplemental Material
sj-pdf-4-cph-10.1177_17151635251343907 – Supplemental material for Hypertension Canada guideline for the diagnosis and treatment of hypertension in adults in primary care
Supplemental material, sj-pdf-4-cph-10.1177_17151635251343907 for Hypertension Canada guideline for the diagnosis and treatment of hypertension in adults in primary care by Rémi Goupil, Ross T. Tsuyuki, Nancy Santesso, Kristin A. Terenzi, Jeffrey Habert, Gemma Cheng, Stephanie C. Gysel, Jill Bruneau, Alexander A. Leung, Norman R.C. Campbell, Ernesto L. Schiffrin and Gregory L. Hundemer in Canadian Pharmacists Journal / Revue des Pharmaciens du Canada
Supplemental Material
sj-pdf-5-cph-10.1177_17151635251343907 – Supplemental material for Hypertension Canada guideline for the diagnosis and treatment of hypertension in adults in primary care
Supplemental material, sj-pdf-5-cph-10.1177_17151635251343907 for Hypertension Canada guideline for the diagnosis and treatment of hypertension in adults in primary care by Rémi Goupil, Ross T. Tsuyuki, Nancy Santesso, Kristin A. Terenzi, Jeffrey Habert, Gemma Cheng, Stephanie C. Gysel, Jill Bruneau, Alexander A. Leung, Norman R.C. Campbell, Ernesto L. Schiffrin and Gregory L. Hundemer in Canadian Pharmacists Journal / Revue des Pharmaciens du Canada
Footnotes
Acknowledgements
The authors thank Matthew Ford, Anna Stoutenberg, Asif Ahmed, and Laurel Taylor from Hypertension Canada for providing administrative support and project coordination. The authors thank the following patient-partners who provided feedback from the patient perspective on the guideline and led development of the patient support tool: Kerri Eady, J.K. Laughton, Karen Moffat, and Raveenie Rajasingham. The authors thank the following patient-partners who provided feedback during the external review: Catherine Allard, Linda Edwards, Rosemary Lyman Lohaza, Duncan Macpherson, and R.M. Wiebe. The authors thank the following organizations for assisting with eliciting member feedback during the external review phase: Hypertension Canada, Canadian Association of Cardiovascular Prevention & Rehabilitation, Canadian Cardiovascular Harmonized National Guidelines Endeavour, Canadian Cardiovascular Society, Canadian Council of Cardiovascular Nurses, Canadian Medical Association, Canadian Nurses Association, Canadian Society of Internal Medicine, College of Family Physicians of Canada, Co-op Pharmacy, Diabetes Canada, Heart & Stroke Foundation of Canada, Kidney Foundation of Canada, Obesity Canada, Public Health Physicians of Canada, and Société québécoise d’hypertension artérielle.
Author Contributions:
All of the authors contributed to the conception and design of the work, and the acquisition, analysis, and interpretation of data. Rémi Goupil and Gregory Hundemer drafted the manuscript, and all authors revised it critically for important intellectual content. All authors gave final approval of the version to be published and agreed to be accountable for all aspects of the work.
Funding:
Funding for this initiative was provided by Hypertension Canada.
Statement of Conflicting Interests:
Rémi Goupil reports receiving research grants from the Canadian Institutes of Health Research (CIHR), the Kidney Foundation of Canada, and Fonds de recherche du Québec–Santé, Université de Montréal. Dr. Goupil also reports holding unpaid positions as a board member of the Canadian Society of Nephrology and vice president of the Société québécoise d’hypertension artérielle. Ross Tsuyuki reports receiving a research grant from Sanofi Canada and an editor-in-chief stipend from the Canadian Pharmacists Journal, as well as consulting fees from Shoppers Drug Mart. Dr. Tsuyuki also reports holding (volunteer) positions as president and past-president of the Board of Directors of Hypertension Canada. Kristen Terenzi reports consulting fees, honoraria, and travel support from, as well as participation on data safety monitoring boards with, AstraZeneca, Astella, Amgen, AbbVie, Aspen, Allergan, Bayer, Boehringer Ingelheim, Lundbeck, Novartis, Novo Nordisk, Pfizer, Moderna, GlaxoSmithKline, Eli Lilly, and Takeda. Jeffrey Habert reports consulting fees from Otsuka, Eli Lilly, Amgen, Idorsia, Eisai, and Elvium; speaker fees, and participation on scientific planning committees with, Boehringer Ingelheim, Eli Lilly, Elvium, Bausch and Lomb, AstraZeneca, Novartis, Lundbeck, Novo Nordisk, Janssen, Eisai, HLS, Otsuka, Idorsia, Pfizer, Amgen, AbbVie, GlaxoSmithKline, Bayer, and Valeo; and meeting support from Eli Lilly and Idorsia. Dr. Habert was co-chair for the Thrombosis Canada clinical guidelines, coauthor of the 2023 Canadian Network for Mood and Anxiety Treatments (CANMAT) guideline, and co-author of the Canadian sleep consensus on insomnia. He is the physician lead of the Holland-Davis Family Health Organization and a director for the CPD Network Physician Organization. Gemma Cheng received an honorarium from the Montreal Chinese Hospital Foundation for a lecture on type 2 diabetes and hypertension. Stephanie Gysel reports funding from Novo Nordisk and University Health Foundation; consulting fees from Novartis Canada, Amgen Canada, Pfizer Canada, and Kye Pharma; honoraria from Novartis Canada, Amgen Canada, and Pfizer Canada; and travel support from Amgen Canada. Jill Bruneau reports receiving research grants from CIHR, and travel support from Memorial University of Newfoundland to attend the Canadian Women’s Heart Health Alliance Conference, and from Hypertension Canada to attend the Nurse Practitioner Conference of Canada. Dr. Bruneau also reports participating in the Knowledge Translation Working Group, Canadian Women’s Heart Health Alliance, and is a member of the Hypertension Canada Board of Directors (both positions unpaid). Dr. Bruneau reports use of intrauterine devices (IUD) simulator, expertise, and sponsorship from Bayer for the purposes of Nurse Practitioner Education only (paid to institution). Alexander Leung reports receiving the Heart and Stroke Foundation of Canada’s National New Investigator Award and Petro Canada’s Young Innovator Award in Community Health, as well as the Calgary Centre for Clinical Research, Cumming School of Medicine, and Alberta Health Services Clinical Research Fund Seed Grant (all paid to institution). Dr. Leung also reports receiving an honorarium as a clinical expert for the Canadian Agency for Drugs and Technologies in Health Scientific Advice Program. Norman Campbell reports serving as chair of Alberta Health Professionals for the Environment, past-chair of the Canadian Hypertension Coalition, special advisor to the World Hypertension League Board, consultant for the Pan American Health Organization’s HEARTS in the Americas program, consultant with Resolve to Save Lives, and consultant for the Crowfoot Primary Care Clinic. Ernesto Schiffrin reports receiving a project grant from CIHR, consulting fees from Boehringer Ingelheim International, and payment for participating in a speaker’s bureau from Idorsia. Dr. Schiffrin has also served on the Hypertension Canada Budget Committee. Gregory Hundemer reports receiving research grants from CIHR, the Kidney Foundation of Canada, and The Ottawa Hospital Academic Medical Organization, and is the Lorna Jocelyn Wood Chair for Kidney Research (Ottawa Hospital Research Institute). No other competing interests were declared.
This article has been peer reviewed.
Ethics Statement:
Not applicable.
Content Licence:
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC-BY-NC-ND 4.0) licence, which permits use, distribution, and reproduction in any medium, provided that the original publication is properly cited, the use is non-commercial (i.e., research or educational use), and no modifications or adaptations are made. See: ![]()
Editor’s Note:
This guideline was first published in the Canadian Medical Association Journal, and is being republished simultaneously with permission in Canadian Family Physician and Canadian Pharmacists Journal, with a synopsis in Canadian Nurse. Requests for permissions and reprints should be sent to CMAJ.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
