Abstract

Atrial fibrillation (AF) is the most common sustained cardiac arrhythmia. 1 AF is rarely life-threatening; however, it is associated with significant impairment in quality of life and increases the risk of stroke and left ventricular (LV) dysfunction. Furthermore, it is a substantial economic burden on the health care system through emergency room visits, hospitalizations and indirect costs. 2 The Canadian Cardiovascular Society (CCS) AF Guidelines program was developed to aid clinicians in the management of these complex patients. 2 The 2020 iteration of the CCS AF Guidelines is a comprehensive document that integrates the past decade of guideline updates and recommendations. It was developed in accordance with the Grading of Recommendations, Assessment, Development and Evaluation (GRADE) approach, 3 which separates the quality of evidence (very low, low, moderate or high quality) from the strength of recommendations (strong or weak) while allowing acknowledgment of values and preferences in the provision of clinical care. A strong recommendation is one in which the desirable effects of an intervention clearly outweigh the undesirable effects or clearly do not. A weak recommendation is one in which the tradeoffs are less certain, either because of low-quality evidence or because the desirable and undesirable effects are closely balanced. The 2020 CCS AF guidelines provide 129 recommendations. We endeavour to summarize the key recommendations that are most relevant to pharmacists caring for AF patients.
What is AF and what are the goals of therapy?
AF is a complex condition that ranges from being an isolated electrical problem in the heart to, more commonly, a manifestation of other cardiac and noncardiac pathologies. During normal heart rhythm, the heartbeat is initiated in the sinus node, travels through the atria causing atrial contraction, then travels to the AV node and His-Purkinje system to cause coordinated ventricular contraction. In a person with AF, there are a number of ectopic foci or “extra beats” that generate electrical impulses. As a result, the atria experience rapid irregular and uncoordinated contractions. Because these electrical impulses reach the AV node erratically, the ventricular rhythm is also irregular with variable lengths of time between ventricular beats (Appendix 1, available in the online version of the article). Throughout a person’s lifetime, AF can progress from self-terminating episodes of paroxysmal AF (e.g., continuous episode lasting longer than 30 seconds but terminating within 7 days of onset) to longer-lasting episodes of persistent AF (e.g., episodes lasting longer than 7 days) that may require intervention (e.g., cardioversion) for termination. 2
For pharmacists involved in the care of patients with AF, the goals of therapy can be broadly grouped into relieving patients’ symptoms, improving functional capacity and quality of life, preventing complications (e.g., stroke and heart failure) and reducing avoidable health care utilization. These goals can be achieved by participating in an integrated, multidisciplinary, patient-focused approach aimed at risk factor modification, pursuing rate and/or rhythm control and identification of patients in need of oral anticoagulation for stroke prevention 2 (Figure 1).
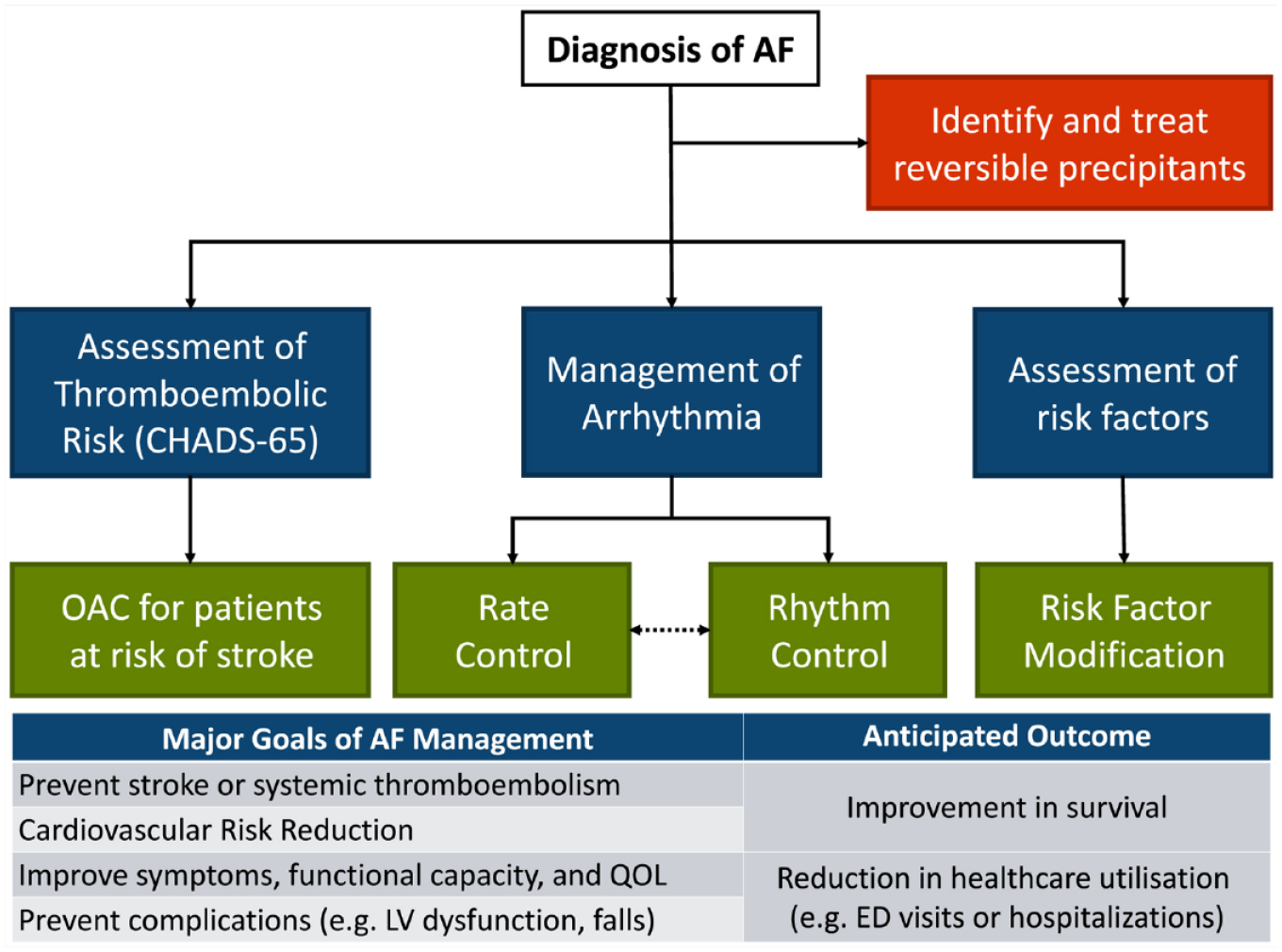
Overview of the management of atrial fibrillation and goals of treatment
Takeaway 1: Importance of risk factor modification
In patients with established AF or at high risk of developing AF, the 2020 CCS guidelines recommend a systematic approach to the identification of traditional modifiable cardiovascular risk factors and conditions associated with AF, with strict guideline-adherent management to reduce major cardiovascular events (Strong Recommendation; High-Quality Evidence). The mechanisms by which these risk factors predispose patients to AF are multifactorial and include neurohormonal activation, structural changes and electrical remodelling.1,2 There is a linear relationship between incident AF and blood pressure (BP) readings, with systolic BP a better predictor than diastolic BP. 4 The risk of AF associated with diabetes increases with the duration of diabetes and worsening glycemic control. 2 Current smokers are more likely to develop AF than former smokers are, with AF risk being associated with higher cumulative exposures to firsthand tobacco use.2,5 In general, the risk of developing AF increases with the severity and number of modifiable cardiovascular risk factors.
Pharmacists, particularly those in community and primary care practice, are well positioned to affect key risk factors that increase the lifetime prevalence of AF, with evidence to support pharmacist-led medication management in improving outcomes.6,7 Aggressive management of hypertension, diabetes, tobacco use, obstructive sleep apnea and lifestyle interventions targeting weight loss for overweight patients; regular exercise; and limiting alcohol intake should be implemented in patients with established AF, with goals as outlined in Figure 2. 2
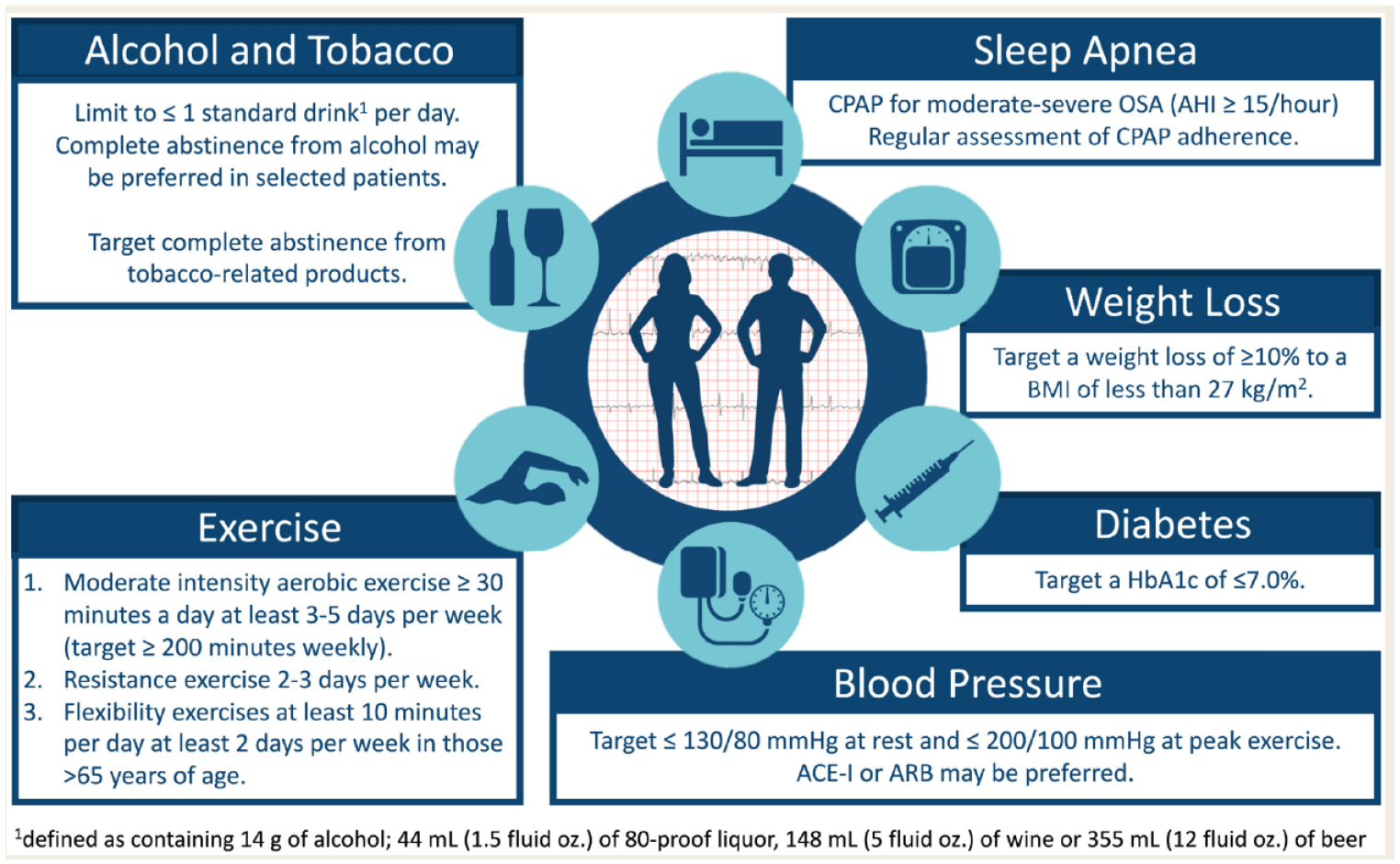
Modifiable risk factor targets for patients with atrial fibrillation
In addition, drug-induced AF should be ruled out in patients with new-onset or worsening burden of AF. Several classes of medications can induce AF, including stimulants, alcohol, antipsychotics, bisphosphonates, immunomodulators and anticancer therapies such as tyrosine kinase inhibitors like sunitinib, anthracyclines like doxorubicin, alkylating agents like cisplatin and cyclophosphamide, HER2 receptor blockers like trastuzumab, antimetabolites like 5-fluorouracil and microtubule agents like paclitaxel. 8 Management of drug-induced AF does not necessarily require removal of the causative agent if the benefits of ongoing treatment of the comorbid condition outweigh the risks associated with AF.
Takeaway 2: How to approach stroke prevention in AF patients—risk assessment
AF is an independent risk factor for stroke. This risk is influenced by patient characteristics and comorbidities. Valvular disease significantly increases the risk of stroke with AF; this has commonly been referred to as “valvular” AF. The 2020 CCS AF Guidelines consider valvular AF to be “AF in the presence of any mechanical heart valve or in the presence of moderate to severe mitral stenosis.” 2 Patients with valvular AF should be anticoagulated unless contraindications exist. Patients without these valvular conditions (those with nonvalvular AF) require an assessment of stroke risk to determine who should be offered anticoagulation (see below). The 2020 CCS guidelines recommend that warfarin be used for patients with a mechanical prosthetic valve or those with AF and moderate to severe mitral stenosis (Strong Recommendation, Moderate Quality Evidence). This is based on the fact that these patients were excluded from the randomized controlled trials comparing direct oral anticoagulants (DOACs) vs warfarin, the findings of the REALIGN trial comparing dabigatran to vitamin K antagonist (VKA) in patients with mechanical heart valves (which was stopped early due to excess embolic and bleeding events with dabigatran) 9 and the long experience and clinical reports of the use of warfarin in the setting of rheumatic mitral stenosis. 2 Like valvular AF, there are other patient conditions in which oral anticoagulation is indicated. Examples include patients requiring cardioversion (see Takeaway 6) and those with congenital heart disease and cardiomyopathies. Please refer to the full guidelines for details.
Patients with nonvalvular AF require stroke risk stratification based on their stroke risk factors. These include age, previous history of stroke or transient ischemic attack, concomitant heart failure, diabetes mellitus, hypertension and presence of vascular disease. There are a number of risk stratification tools, such as the CHADS2 10 and CHA2DS2VASc, 11 that consider the patient’s stroke risk factors to come up with a score that predicts an annual risk of stroke. As age can be reliably determined in all patients, the guidelines committee agreed that age should be a starting point for a practical algorithm that includes all of the CHADS2 and CHA2DS2VASc criteria, arranged to easily identify those patients who should receive anticoagulation. The 2020 CCS guidelines recommend that the “CCS Algorithm” (CHADS-65) be used to guide the choice of appropriate antithrombotic therapy for the purpose of stroke and systemic embolism prevention in NVAF (Strong Recommendation, High-Quality Evidence; Figure 3). Further, they recommend that patients with atrial flutter be stratified and treated in the same manner. 2 The key message of the CHADS-65 algorithm and a recommendation of the guidelines is that oral anticoagulants be prescribed for most patients with AF aged 65 years or older or with a CHADS2 score of 1 or more (Strong Recommendation; Moderate-Quality Evidence). This recommendation is based on the effect of oral anticoagulation on reducing the absolute risk of stroke compared with the increase in major bleeding. In patients aged 65 years or older and without other risk factors for stroke, the use of VKAs decreased the annual risk of stroke from 2.1% to 0.7%, while it increased the risk of major bleeding by approximately 0.5% per year to 1.0%. 12 Further, the 2020 guidelines recommend that most patients should receive a DOAC (apixaban, dabigatran, edoxaban or rivaroxaban) in preference to warfarin when oral anticoagulation therapy is indicated for patients with nonvalvular AF (Strong Recommendation; High-Quality Evidence). This recommendation is based on the results of several large randomized trials demonstrating that DOACs are either as or more effective than warfarin in preventing stroke; that they have similar or less risk of major bleeding and less intracranial hemorrhage compared with warfarin and on the greater ease of use of DOACs compared with dose-adjusted warfarin.13 -16
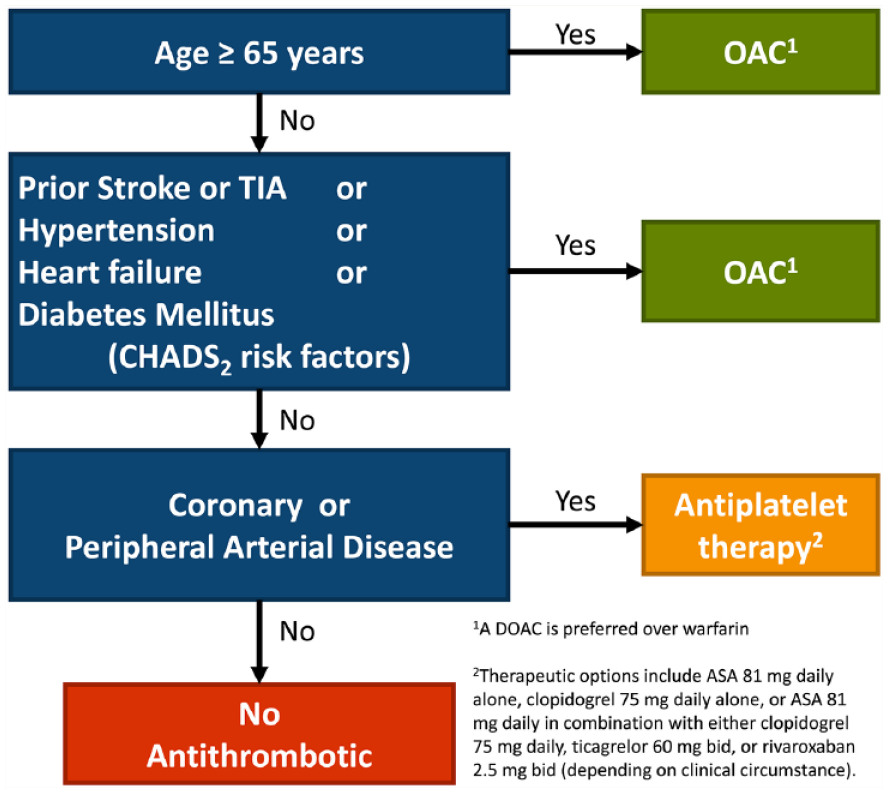
The “CCS Algorithm” (CHADS-65) to guide antithrombotic therapy decision making for patients with nonvalvular atrial fibrillation or atrial flutter
Importantly, the benefit of oral anticoagulation must be weighed against the risk of major bleeding. Shared decision making with patients is key to ensuring an understanding of the risks and the importance of lifelong therapy. AF-related stroke is fatal in 1 in 5 patients and disabling in 3 in 5, while most anticoagulant-associated bleeds do not result in death or permanent disability.17,18
Takeaway 3: Acetylsalicylic acid should not be used solely for stroke prevention in AF patients
The 2020 guidelines suggest that no antithrombotic therapy be prescribed for stroke prevention for most patients with NVAF who are younger than 65 years with no CHADS2 risk factors (Weak Recommendation; Moderate-Quality Evidence) or vascular disease. When the annual risk of stroke is <1%, the risk of increased major bleeding, even with acetylsalicylic acid (ASA), outweighs its benefit. 19 For patients with NVAF who are younger than 65 years with no CHADS2 risk factors, the current evidence does not support antiplatelet monotherapy for stroke prevention. 2 Further, the superior benefit:risk ratio of DOAC therapy over ASA is consistent across age and CHADS2 scores, such that even in patients with higher stroke risk, there is no role for ASA monotherapy. 20
Takeaway 4: Stroke prevention in patients with AF and coronary artery disease
A significant proportion of AF patients have concomitant coronary artery disease (CAD), many of whom will also require percutaneous coronary intervention (PCI) for revascularization. These patients typically require antiplatelet therapy for the prevention of coronary events and oral anticoagulation for the prevention of AF-related stroke. Treating these patients with antithrombotic therapy requires a balance of the relative efficacy benefit, since dual antiplatelet therapy is more effective than anticoagulation alone in reducing cardiovascular death, myocardial infarction, stent thrombosis and ischemic coronary events in patients who have experienced an acute coronary syndrome (ACS), but it is less effective than anticoagulation for the prevention of stroke/systemic embolism in AF. 21 As such, patients with AF and CAD often require combination antithrombotic therapy, while recognizing that it increases the risk of fatal and nonfatal bleeding and death. 22 The key is to carefully assess the individual risks of bleeding against the anticipated coronary and cerebral thrombotic risk, recognizing that some of these factors, particularly the angiographic ones, may not be readily available to us as pharmacists (Appendix 2, available in the online version of the article).
Patients with stable CAD (e.g., those without previous ACS or who are at least 12 months out from it)
For patients with NVAF who are younger than 65 with no CHADS2 risk factors, any antithrombotic therapy should be directed at the underlying CAD. In other words, these patients do not need anticoagulation specifically for stroke prevention related to their AF.
For AF patients with stable CAD who are 65 years or older or who have a CHADS2 score of 1 or more, the guidelines recommend using oral anticoagulation alone (Strong Recommendation; Moderate-Quality Evidence). In the trials supporting this recommendation, combining anticoagulation (warfarin or DOAC) with ASA did not decrease thrombotic endpoints but did increase major bleeding.22,23 The most recently published study was terminated early because of increased all-cause mortality in the patients receiving combined rivaroxaban plus ASA vs those receiving rivaroxaban alone. 22 While the guidelines committee recognizes that there may be highly selected patients with high-risk features for ischemic coronary outcomes who are also at low risk of bleeding in whom a combination strategy may be considered, for the most part, patients with stable CAD who require anticoagulation for stroke prevention should not receive concomitant ASA.
Patients with recent ACS and/or PCI (e.g., within 12 months)
Combination antithrombotic therapy is required for patients who are at greater risk of ischemic thrombotic events, such as those with recent ACS or PCI. There has been a growing body of evidence to help us balance this risk with the risk of bleeding.24 -27 In general, what we have learned is the following:
A dual pathway strategy such as combining anticoagulation with a P2Y12 inhibitor (preferably clopidogrel) causes less bleeding than combining anticoagulation with ASA plus a P2Y12 inhibitor (triple antithrombotic therapy).
Using a DOAC is safer than using warfarin. 28
ACS and high-risk PCI patients need “some” ASA.
The 2020 CCS AF guidelines differentiate between 1) ACS without PCI, 2) elective PCI without high-risk features and 3) ACS with PCI or elective PCI with high-risk features. Essentially, if your patient has any of the high-risk features (Figure 4), the guidelines recommend triple antithrombotic therapy with oral anticoagulant, clopidogrel and up to 1 month of ASA (Strong Recommendation, High-Quality Evidence). If they do not, the guidelines recommend dual pathway therapy with oral anticoagulant and clopidogrel (Strong Recommendation, Low-Quality Evidence). It is important to note that even in the setting of dual pathway therapy, ASA is given around the time of the PCI procedure.
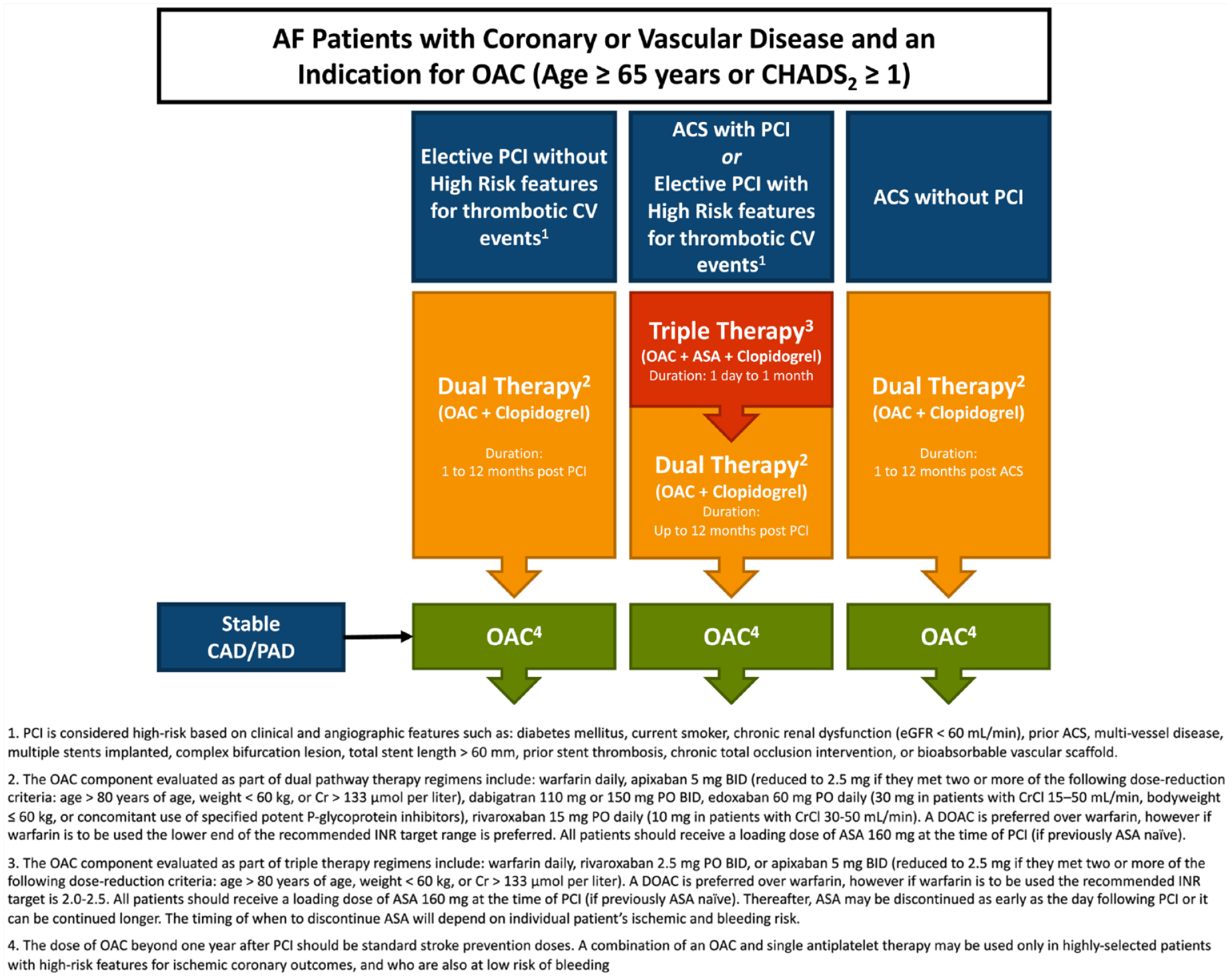
Risk factors associated with increased ischemic coronary events and/or bleeding
Clopidogrel is recommended as the preferred P2Y12 inhibitor when dual pathway or triple therapy is used (Strong Recommendation; Moderate-Quality Evidence).
Anticoagulants that have been studied as part of the dual pathway strategy and that are recommended in the guidelines are adjusted-dose warfarin targeting an international normalized ratio (INR) of 2 to 3, apixaban 5 mg twice daily (unless criteria for a reduced dose are met), dabigatran 110 mg twice daily, dabigatran 150 mg twice daily, edoxaban 60 mg daily (unless dose-reduction criteria are met) and rivaroxaban 15 mg daily (10 mg daily for patients with creatinine clearance [CrCl] 30-50 mL/min).
Anticoagulants that have been studied as part of the triple antithrombotic strategy include adjusted-dose warfarin targeting an INR of 2 to 3, apixaban 5 mg twice daily (unless dose-reduction criteria are met) and rivaroxaban 2.5 mg twice daily (note that this dose is not currently approved in Canada for this indication).
Key considerations include attention to modifiable bleeding risk factors, particularly uncontrolled systolic BP (see Takeaway 7), consideration of proton pump inhibitor use, avoidance of prasugrel or ticagrelor in combination with oral anticoagulation, use of a lower INR target range (2-2.5) when warfarin is used in this setting and recommending a walking aid for those with gait or balance disorders. Finally, the antithrombotic strategy should be reassessed regularly, and the dose of anticoagulant, at a minimum of 1 year after PCI (or when the antiplatelet therapies are stopped), should be standard stroke prevention in AF dosing.
Takeaway 5: Stroke prevention in patients with renal dysfunction
AF and chronic kidney disease (CKD) share multiple risk factors and are frequent comorbid conditions, with 30% of AF patients having at least moderate CKD.29,30 Renal function is a key consideration for selection and dosing of oral anticoagulants and affects their relative benefit, with increasing severity of CKD increasing the risks of both stroke and bleeding. 30 The 2020 CCS AF guidelines recommend the following:
1. Patients with AF who are receiving oral anticoagulation should have their renal function assessed at baseline and at least annually to detect latent kidney disease, determine anticoagulant eligibility and support drug dosing (Strong Recommendation; Moderate-Quality Evidence).
2. CrCl, as estimated using the Cockcroft-Gault method, should be used to support dosing decisions of anticoagulant medications (Strong Recommendation; High-Quality Evidence).
3. Antithrombotic therapy in AF patients with CKD should be provided according to their risk of stroke/systemic embolism and the severity of renal dysfunction:
• Stage 3 CKD or better (eGFR >30 mL/min): We recommend that such patients receive antithrombotic therapy as determined by the “CCS algorithm” (Strong Recommendation; High-Quality Evidence).
• Stage 4 CKD (eGFR 15-30 mL/min): We suggest that such patients receive antithrombotic therapy as determined by the “CCS algorithm” (Weak Recommendation; Low-Quality Evidence).
• Stage 5 CKD (eGFR < 15 mL/min or dialysis dependent): We suggest that such patients do not routinely receive antithrombotic therapy for stroke prevention in AF (Weak Recommendation; Low-Quality Evidence).
Current evidence favours anticoagulation with DOACs given their safety, efficacy and convenience.2,31 There are additional concerns that VKA therapy contributes to further renal decline, whereas DOACs are associated with less progression of renal dysfunction. 32 The 4 currently approved DOACs have varying reliance on renal clearance, with specific dosing based on renal function (Table 1).
Health Canada–approved dosing for direct oral anticoagulants
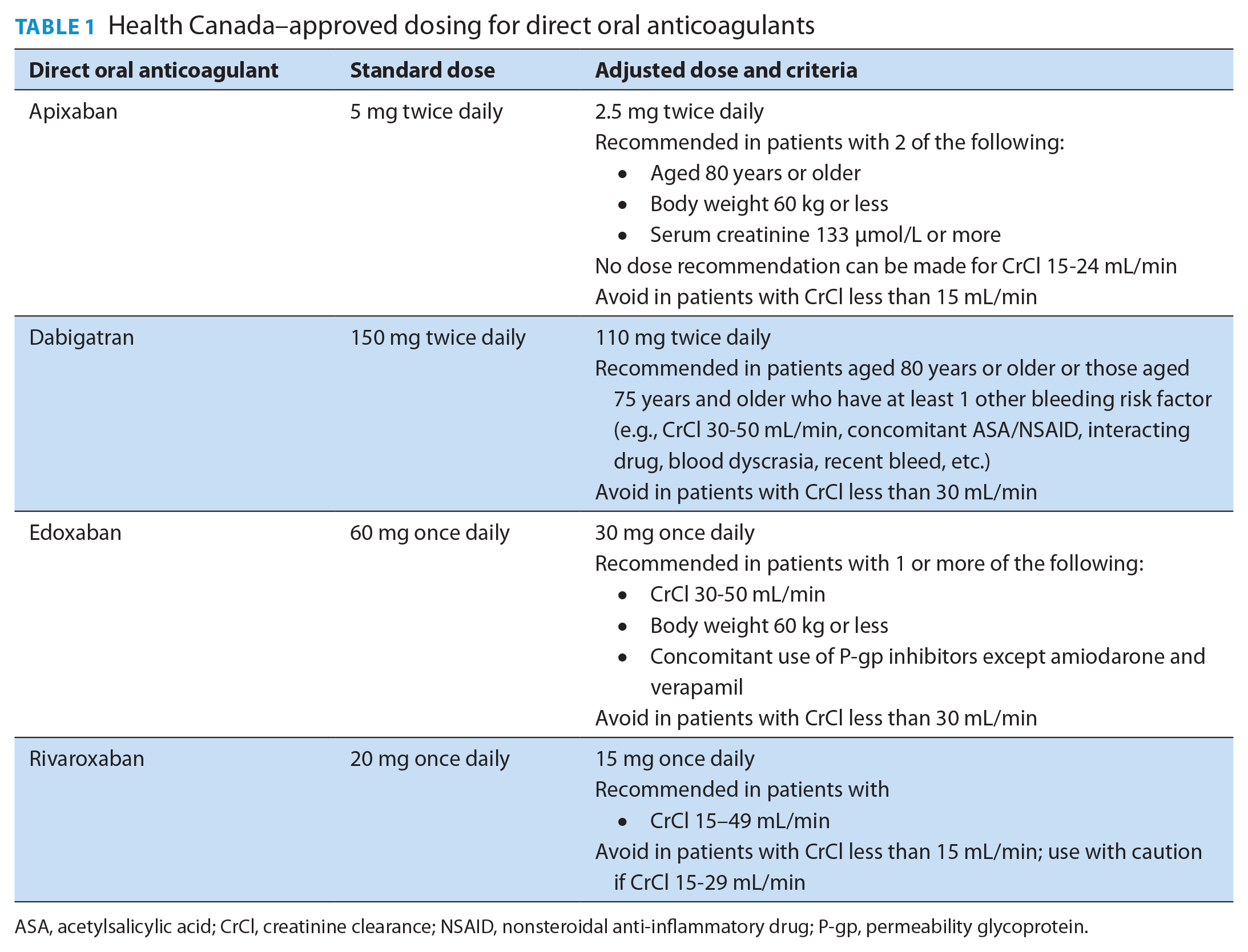
ASA, acetylsalicylic acid; CrCl, creatinine clearance; NSAID, nonsteroidal anti-inflammatory drug; P-gp, permeability glycoprotein.
As patients with more advanced stages of CKD (stage 4-5) have been excluded from landmark trials, the benefit of anticoagulation in these populations is less clear. DOACs (rivaroxaban and apixaban) are favoured in stage 4 CKD patients, although their approvals are largely based on pharmacokinetic and pharmacodynamic data. 30
It remains unclear whether patients with severe renal dysfunction (eGFR <15 mL/min) and those on dialysis benefit from anticoagulation, although the risk of harm is clear. 30 DOACs are currently contraindicated in these patients, and the use of VKA is associated with increased bleeding, vascular calcification and calciphylaxis. Studies are underway in patients on dialysis that should provide further guidance in this population.
Takeaway 6: Stroke prevention in other challenging populations
Obese and overweight patients
Overweight (body mass index [BMI] 25-29.9) and obese patients (BMI 30 or greater) were included in the 4 key trials comparing DOACs to warfarin. Post hoc analyses of these patients indicate that DOACs are at least as good as warfarin for stroke prevention in patients with a BMI of 40 or less, with a similar incidence of major bleeding.13,33 -35 In addition, a meta-analysis of these trials suggests that DOACs are more effective than warfarin and safer for overweight patients (BMI 25-30: odds ratio [OR], 0.87; 95% confidence interval [CI], 0.76-0.99 for stroke/systemic embolism; OR, 0.83; 95%CI, 0.71-0.96 for major bleeding) and similarly effective and safe in obese patients (BMI >30: OR, 0.87; 95% CI, 0.76-1.00 for stroke/systemic embolism; OR, 0.91; 95% CI, 0.81-1.03 for major bleeding). 36 Although it is important to note that severely obese patients were not well represented, these data provide reassurance that DOACs are at least as effective as warfarin for stroke prevention in AF patients with a BMI less than 40. 2 DOACs should be avoided in the post–bariatric surgery population because of the lack of studies in this population and the potential for altered absorption.
Patients with liver disease
Patients with liver disease are at increased risk of bleeding due to disruption of the coagulation process and increased risk of embolic events due to a prothrombotic state. 37 Advanced liver disease can also affect the metabolism and elimination of oral anticoagulants. The Child-Pugh score, which provides a scoring system for liver disease severity based on clinical and lab parameters, can be used to guide the choice of anticoagulant in patients with AF and liver disease. 38 The 2020 CCS AF guidelines recommend that oral anticoagulation not be routinely prescribed for patients with AF and advanced liver disease (Child-Pugh grade C or liver disease associated with significant coagulopathy; Strong Recommendation; Low-Quality Evidence). If it is deemed appropriate in these patients, VKA is recommended as the preferred agent in patients with a baseline INR of 1.7 or less with frequent and careful monitoring. 2 In these cases, OAC decisions should be made in collaboration with specialists.
Frail elderly patients
Advanced age is a well-established risk factor for both stroke and bleeding in AF patients. Despite the higher risk of stroke in the frail, elderly population and the known benefits of anticoagulation, it remains underused.39,40 Secondary analyses of the landmark trials comparing DOACs to VKAs showed similar or better efficacy outcomes in elderly patients (>75 years old).41 -44 Further, meta-analysis showed no significant difference in rates of major or clinically relevant nonmajor bleeding in DOAC-treated vs non–DOAC-treated elderly patients. 45 The 2020 CCS AF guidelines recommend that oral anticoagulation be prescribed for most frail elderly patients with AF (Strong Recommendation; Moderate-Quality Evidence). Selection of a particular agent should consider the risk for drug-drug and drug-disease interactions. In addition, any modifiable bleeding risk factors should be addressed, including providing appropriate walking aids for those at risk of falls and reassessing medications that can lead to falls or hypotension. 2
Patients with recent cardioversion
Pharmacologic and electrical cardioversion are used to restore sinus rhythm. There is a known increased thromboembolic risk around cardioversion related to embolization of preexisting thrombi after restoration of sinus rhythm and a period of “atrial stunning” that might be responsible for the development of new thromboemboli after cardioversion. 2 The 2020 AF guidelines recommend that every patient for whom elective electrical or pharmacologic cardioversion is planned should receive therapeutic anticoagulation for at least 3 weeks before cardioversion (Strong Recommendation; Moderate Quality Evidence). In addition, they recommend that all patients should receive at least 4 weeks of anticoagulation after cardioversion (Weak Recommendation; Low-Quality Evidence). The need for ongoing anticoagulation after 4 weeks postcardioversion should be on the basis of the risk of stroke as determined by the CCS Algorithm (CHADS-65; Strong Recommendation; Moderate-Quality Evidence). As such, there may be occasional cases in a pharmacist’s practice in which patients’ anticoagulation may be appropriately stopped 4 weeks after cardioversion. However, any discontinuation of an oral anticoagulant in an AF patient warrants pharmacist review.
Takeaway 7: Bleeding prevention strategies and management of OAC interruptions
Bleeding is a risk with any antithrombotic therapy. In the pivotal clinical trials comparing DOACs with warfarin, major bleeding occurred in 2% to 4% of patients. Pharmacists can employ bleeding-reduction strategies to optimize the risk-benefit of oral anticoagulation for patients. The 2020 AF guidelines recommend initial and ongoing evaluation of bleeding risk for all patients with AF whose stroke risk warrants antithrombotic therapy, with the use of strategies to mitigate the increased risk of bleeding (Strong Recommendation; Low-Quality Evidence). A practical approach to this is to use a validated, easy-to-use bleeding risk algorithm, such as the HAS-BLED (Hypertension, Abnormal renal/liver function, Stroke, Bleeding history or predisposition, Labile INR, Elderly [>65 years], Drugs/Alcohol Concomitantly) algorithm 46 at each point of contact, including prescription refills. Pharmacists should focus on identification of risk factors that can be modified. These include BP control, avoidance of unnecessary ASA and nonsteroidal anti-inflammatory therapy, treatment of anemia, limiting alcohol intake and ensuring that the patient is receiving the most appropriate dose of anticoagulant. Underdosing of DOACs has been associated with increased cardiovascular hospitalization and overdosing, with increased all-cause mortality. 47 The guidelines also suggest the additional use of a proton pump inhibitor to decrease the risk of gastrointestinal adverse effects for patients who require daily antithrombotic therapy that includes ASA (Weak Recommendation; Moderate-Quality Evidence). Patients receiving oral anticoagulant therapy should be counselled to review with their prescriber and/or pharmacist when changes are made to their medications to check for drug-drug interactions and should be provided with guidance regarding self-monitoring for bleeding, including when to seek medical help.
AF patients may need to undergo medical procedures that require temporary interruption of their anticoagulation. In these situations, clinicians should use a practical approach to balance the risk of a stroke with a periprocedural bleeding event. Of note, there are a number of procedures with minimal risk of bleeding that do not generally require interruption of anticoagulation, including cataract surgery, skin biopsies, colonoscopy without biopsy and simple dental extractions. 2 The Thrombosis Canada Perioperative Anticoagulant Management algorithm available at thrombosiscanada.ca serves as a useful tool to consult regarding the risk of bleeding associated with different procedures, recommendations regarding the duration of the interruption and whether bridging anticoagulation is needed in patients receiving warfarin. Pharmacists should work with patients and their medical providers to ensure timely reintroduction of anticoagulation postprocedure. The 2020 CCS AF guidelines suggest that such therapy be restarted when hemostasis is established (within 24 hours for a procedure with a low risk of bleeding and within 48-72 hours for a procedure with a high risk of bleeding; Weak Recommendation; Low-Quality Evidence). 2
In the case of life-threatening bleeding and/or in patients requiring emergency surgery, specific reversal agents for DOACs exist. Idarucizumab, which is available in Canada, is a humanized monoclonal antibody fragment that works by binding to dabigatran with high affinity to neutralize it and its metabolites, reversing its anticoagulant effect. 48 Andexanat alfa is a recombinant human factor Xa decoy protein used to sequester factor Xa inhibitors in the vascular space by binding with high affinity to the active site of any factor Xa inhibitor, including apixaban, edoxaban and rivaroxaban. 49 It is currently in use in the United States but is not yet available in Canada. The 2020 CCS AF guidelines recommend both of these agents for patients experiencing uncontrollable or potentially life-threatening bleeding. 2
Takeaway 8: Optimizing patient symptoms and quality of life—Rate control
Strategies for managing the symptoms of AF, improving functional status and preventing complications such as LV dysfunction, include rate and rhythm control. 2 Managing AF typically starts with rate control, as most patients will require this at some point. The main factor influencing the decision of which rate control medication to select is the patient’s LV function. The 2020 CCS AF guidelines recommend evidence-based beta-blockers (bisoprolol, carvedilol, metoprolol) be first-line agents for rate control of AF in patients with significant LV dysfunction (LV ejection fraction of 40% or less; Strong Recommendation; Moderate-Quality Evidence). For patients with preserved systolic function (LV ejection fraction of more than 40%), the guidelines recommend beta-blockers or nondihydropyridine calcium channel blockers (diltiazem or verapamil) as first line (Strong Recommendation; Moderate-Quality Evidence). Digoxin may be added if first-line therapy is ineffective or may be considered as monotherapy in sedentary patients or those who are unable to take first-line options 2 (Figure 5). Diltiazem or verapamil would be favoured over beta-blockers in patients with concomitant hypertension, with reactive airway disease or in those who might be bothered by beta-blocker–induced exercise intolerance. Rate control therapy should be titrated to achieve symptom control as well as an average resting heart rate below 100 beats per minute while in AF. 2 Monitoring of patients should include heart rate, symptoms and functional status. Home-monitoring strategies can include BP monitors, FitBit-type monitors, Apple watches or Kardia devices; however, it is important to note that these “pulse-based” heart rate monitors often underestimate the true heart rate.
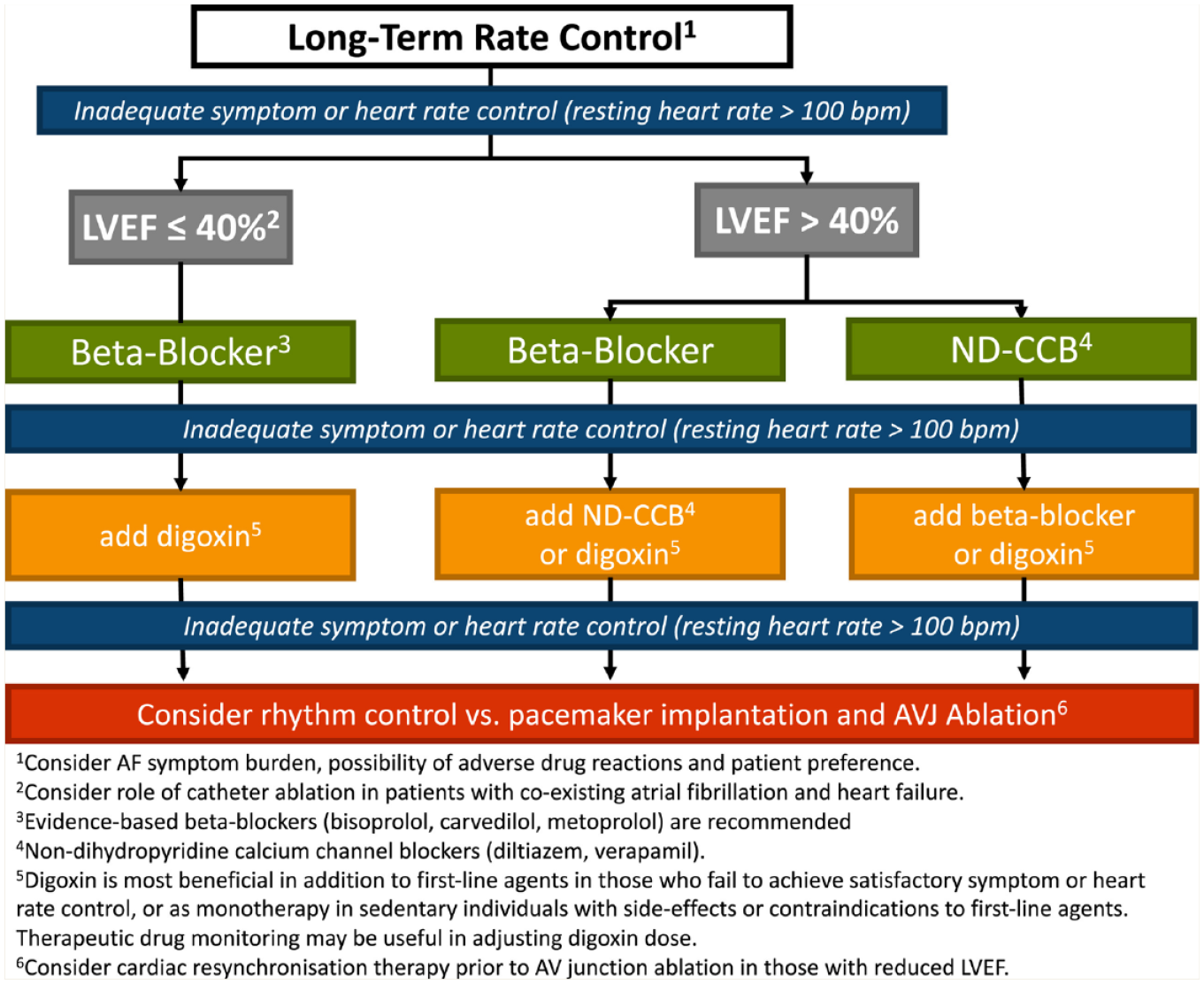
Approach to long-term rate control
Takeaway 9: Optimizing patient symptoms and quality of life—Rhythm control
In patients who continue to have symptoms despite achieving reasonable rate control or when rate control is challenging, a rhythm control strategy is often employed. Rhythm control using antiarrhythmic drugs is also favoured in patients with recent-onset AF, paroxysmal AF episodes that are frequent, those with highly symptomatic AF and in those with arrhythmia-induced cardiomyopathy. 2 Currently, 5 agents are available for maintenance of sinus rhythm in patients with AF (amiodarone, dronedarone, flecainide, propafenone, sotalol). The choice of antiarrhythmic agent is dependent on whether the patient also has heart failure, LV dysfunction or underlying ischemic CAD. For patients with LV dysfunction (ejection fraction of 40% or less), amiodarone is the safe option. Class IC antiarrhythmics (flecainide and propafenone) should not be used in patients with ischemic heart disease given their association with increased mortality in the post–myocardial infarction setting.50,51 In addition, they may cause AF to be converted to rapid atrial flutter and should be combined with an AV node blocker (beta blocker, diltiazem or verapamil) to prevent this. In the absence of structural heart disease or heart failure, any of the antiarrythmics can be used, although amiodarone is typically reserved for refractory cases given the potential for significant side effects (Figure 6).
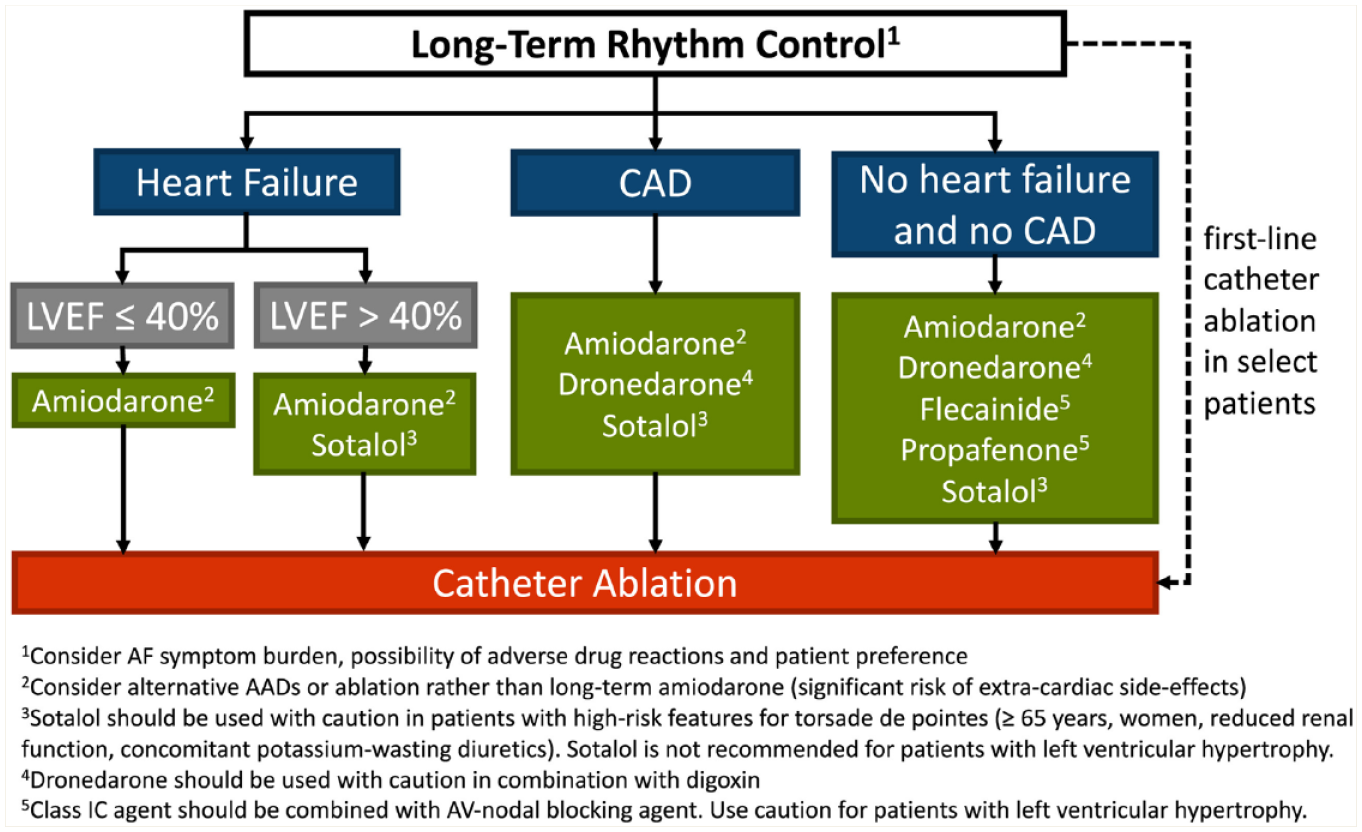
Approach to long-term rhythm control
Monitoring of patients should include periodic rhythm monitoring to ensure ongoing efficacy as well as assessment of symptom burden and adverse effects of therapy. We should help patients understand that the goal of rhythm control therapy is not complete elimination of AF but a decrease in the overall burden (episodes and symptoms). Patients should be encouraged to follow up with their care providers if they notice an increasing frequency of their episodes.
Takeaway 10: Adherence and persistence with medical therapies
The World Health Organization broadly classifies medication nonadherence factors into the following categories: socioeconomic, therapy related, patient related, condition related and health system/health care related. 52 Contemporary patient care should ensure that individual patient values and preferences are considered when making decisions about medical therapy in a shared decision-making environment. 2 Nonadherence to oral anticoagulant therapy in patients with AF is well described. Up to 30% of patients are not adherent to their anticoagulant, which translates into worse patient outcomes including preventable strokes, increased medical costs and death. 53 In one study, a 10% decrease in adherence translated into a 13% increase in stroke and mortality. 54 The assessment of adherence to VKA therapy is indirectly accomplished via INR monitoring, although there is no equivalent monitoring for DOACs. While DOACs increase the convenience of Stroke Prevention in Atrial Fibrillation (SPAF) therapy, assessment of adherence remains an important aspect of care.
Pharmacists play an essential and impactful role in assessing adherence as well as addressing underlying issues that contribute to medication nonadherence. 55 A pharmacist-developed checklist 56 for DOAC monitoring is available at https://thrombosiscanada.ca/wp-content/uploads/2016/03/ENG-Pharmacist-Checklist-V21-13July18-1.pdf. The timing of refill requests can be reviewed, reminder calls can be used to offer patient counselling on the importance of adherence and persistence to SPAF therapy, complex medication regimens should be simplified when possible and patients offered convenience packaging when appropriate. In one study of DOAC-treated patients, pharmacist involvement in patient selection, monitoring and provision of more intensive care to nonadherent patients was associated with significantly improved adherence. 55 Reviewing the affordability of medications with patients should be undertaken by prescribing clinicians as well as pharmacists to avoid any undue financial burden. Ensuring prior authorizations are in place where required remains challenging and often requires a team-based approach (pharmacist, patient, prescriber).
Conclusion
Being among the most accessible health care providers, pharmacists play a vital role in all elements of AF management, including prevention. At every encounter with a patient, pharmacists are able to monitor the effectiveness and adverse effects of stroke prevention and rate/rhythm control therapies, ensuring the appropriateness of drug selection and dose, employing bleeding-reduction strategies, monitoring for drug interactions and reinforcing the importance of adherence. The 2020 CCS AF guidelines target audience includes all pharmacists involved in caring for patients with or at risk for AF. We encourage readers to refer to the full guidelines published in the Canadian Journal of Cardiology 2 for more details and supplemental material. ■
Supplemental Material
sj-pdf-1-cph-10.1177_17151635211058160 – Supplemental material for The 2020 CCS atrial fibrillation guidelines for pharmacists: Top 10 takeaways
Supplemental material, sj-pdf-1-cph-10.1177_17151635211058160 for The 2020 CCS atrial fibrillation guidelines for pharmacists: Top 10 takeaways by Kori Leblanc, Jenny MacGillivray, Amanda Carroccia, Laurent Macle and Jason G. Andrade in Canadian Pharmacists Journal / Revue des Pharmaciens du Canada
Footnotes
Author Contributions:
All authors approved the final version of the article.
Declaration of Conflicting Interests:
Our author team has the following potential conflicts of interest: Kori Leblanc discloses having received speaking fees from Servier, Bayer, Boehringer Ingelheim, Pfizer, Bristol Myers Squibb, Astra Zeneca and Novartis; participation on advisory boards for Servier, Bayer, Boehringer Ingelheim, Pfizer, Bristol Myers Squibb and Novartis; and research grant funding from Servier Canada. Jason Andrade has received speaking fees from Medtronic, BMS-Pfizer, Servier, Biosense-Webster and Bayer and grants from Medtronic and Baylis. Laurent Macle has received speaking fees from Medtronic,BMS-Pfizer, Servier Biosense-Webster and Bayer; has received grants from Medtronic, BMS-Pfizer, Servier, Abbott and Biosense-Webster; and has participated on advisory boards for Medtronic and Biosense-Webster.
Funding:
The authors received no financial support for the research, authorship and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
