Abstract
Objectives
The objective is to assess the performance of the Eluvia polymer coated drug eluting stent (DES) compared to a bare metal stent (BMS) platform in patients with femoropopliteal arterial disease.
Methods
This is a retrospective, single-center analysis. Patients treated with the Eluvia DES (group Eluvia) or the EverFlex BMS (group BMS) for femoropopliteal disease between January 2013 and December 2019 were included. Primary measure outcome of this analysis was the overall mortality. The PTX specific mortality, the primary patency, the amputation free survival (AFS), and the target lesion revascularization (TLR) rates were additionally evaluated.
Results
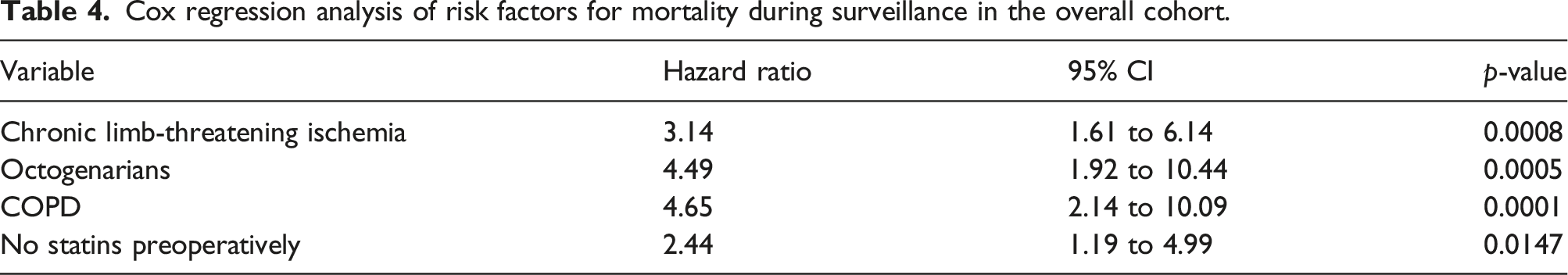
A total of 124 patients were treated by BMS deployment, while the Eluvia platform was preferred in 75 subjects. In both groups the majority presented with lifestyle limiting claudication (BMS: 84% vs Eluvia: 73%, p = 0.73). Chronic total occlusions were more frequent in patients treated by BMS (BMS: 71% vs Eluvia: 84%, p = 0.027), whereas the calcification burden (BMS: 81% vs Eluvia: 76%, p = 0.43) and the median lesion length (in mm, IQR) (BMS: 160 (100 to 240) vs Eluvia: 140 (80 to 229), p = 0.17) were comparable. At 24 months, the overall survival (BMS: 93% vs Eluvia: 89%, hazard ratio (HR): 1.20, 95% confidence interval (CI): 0.55 to 2.64, p = 0.64) and the PTX specific survival (BMS: 95% vs Eluvia: 95%, HR: 1.28, 95% CI: 0.41 to 4.02, p = 0.67) did not differ significantly between the two platforms. No significant difference was observed regarding the 24 months primary patency rate (BMS: 66% vs Eluvia: 78%, HR: 0.65, 95% CI: 0.37 to 1.15, p = 0.18), the freedom from TLR (BMS: 83% vs Eluvia: 89%, HR: 0.81, 95% CI: 0.39 to 1.68, p = 0.572), and the AFS (BMS: 93 vs Eluvia: 89%, HR: 1.20, 95% CI: 0.55 to 2.64). The Cox regression analysis revealed a higher mortality risk among patients with chronic limb-threatening ischemia (CLTI) (HR: 3.14, 95% CI: 1.61 to 6.14, p = 0.008), chronic obstructive pulmonary disease (COPD) (HR: 4.65, 95% CI: 2.14 to 10.09, p = 0.001), in octagenerians (HR: 4.40, 95% CI: 1.92 to 10.44, p = 0.005), and in patients not on statins at baseline (HR: 2.44, 95% CI: 1.19 to 4.99, p=0.014).
Conclusions
In this cohort, the use of the Eluvia DES did not increase the risk for mortality compared to BMS deployment. CLTI, COPD, advanced age, and the lack of statin therapy at baseline were associated with a higher risk for death.
Introduction
Endovascular therapy has evolved rapidly over the last years and is currently considered the initial approach for the majority of patients with peripheral arterial disease (PAD). 1 Historically, plain angioplasty was the treatment of choice for infrainguinal atherosclerosis. 2 However, the risk for neointimal hyperplasia and consequently for restenosis, clinical failure, and repeated interventions was rather high.3,4 Paclitaxel (PTX), a lipophilic antineoplastic agent, can be locally delivered in the arterial wall either with drug coated balloons (DCB) or drug eluting stents (DES) in order to inhibit neointimal hyperplasia. Numerous randomized control trials (RCTs) showed higher patency rates after the application of PTX coated devices compared to their non-coated competitors. 5 Additionally, the use of DCBs and DES was associated with promising results in challenging real-world cohorts with a long-term sustained clinical benefit.1,6
In December 2018, a study-level meta-analysis of 28 RCTs with 4663 patients questioned the safety of PTX for peripheral interventions. Katsanos et al. 7 reported an increased risk for all-cause mortality, which manifests ≥2 years after the use of DCB or DES. Following the initial publication, the Food and Drug Administration (FDA) performed its own meta-analysis showing that PTX-related devices were associated with increased mortality 3 years after the index procedure. 8 Moreover, an individual patient data meta-analysis was published from the Vascular InterVentional Advances (VIVA) physicians and found an absolute 4.6% risk for mortality after PTX exposure. 9 On the other hand, data from pragmatic real-world cohorts could not confirm this finding and more recently the interim analysis of the randomized registry-based SWEDEPAD trial did not reveal any difference between PTX coated and uncoated devices.10-13An important parameter of the FDA analysis is that a mortality class effect could not be determined. 8 Thus, the safety profile of the different PTX coated devices has to be evaluated separately.
The Eluvia (Boston Sci. MA, USA) is a DES platform which uses a dual-layer coating, which enables the sustained release of PTX over the first 12 months after the stent deployment. 14 Initial reports showed promising mid-term results for the treatment of complex femoropopliteal lesions.1,14 However, the Eluvia platform was included neither in the meta-analysis published from Katsanos et al. nor in the analysis of the FDA, while the device received an FDA-approval based on a noninferiority trial compared with the Zilver PTX DES (Cook Medical, Bloomington, IN).15,16 In this context, there is a relevant gap of data regarding the safety profile of this device compared to non-coated scaffolds. The aim of this study is to assess the safety and the efficacy of the Eluvia DES compared to a bare metal stent (BMS) platform in patients with symptomatic femoropopliteal disease.
Methods
This is a single-center, retrospective analysis, performed in line with the requirements of the local ethics committee and adhering to the Declaration of Helsinki. All patients with symptomatic PAD who underwent endovascular treatment of a femoropopliteal lesion between January 2013 and December 2019, with the Eluvia DES or with the EverFlex (Medtronic, Plymouth, Minn) BMS were included into this study. The main criterion for the stent implantation was recoil or flow-limiting dissection after plain angioplasty. The selection of the deployed scaffold (DES or BMS) was based on the preference of the treating physician. This study followed the reporting guidelines from the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) statement for cohort studies. 17
In order to solely evaluate the impact of the Eluvia platform on the mortality risk we excluded all cases with bailout stenting after DCB angioplasty. Moreover, all patients with previous or concomitant PTX DCB/DES treatment for additional lesions or the same lesion as well as patients with PTX treatment for coronary disease were excluded from our analysis. Patients previously or simultaneously treated with non-PTX drug coated devices for below the knee disease (Sirolimus, Everolimus etc.) were not excluded.
All patients underwent a thorough clinical examination at baseline. Patient demographics, imaging, and clinical data were prospectively collected and retrospectively analyzed. Follow-up examinations were scheduled at 6 and 12 and 24 months after the index procedure. The patency of the treated vessels was assessed by duplex ultrasound at each follow-up. In cases of clinical worsening, an angiography was performed.
Dual antiplatelet therapy with aspirin (ASA) (100 mg/d) and clopidogrel (75 mg/d) was routinely prescribed for 3 months, followed by ASA or clopidogrel monotherapy lifelong. Patients previously taking warfarin or oral anticoagulants were maintained on the anticoagulant with an additional antiplatelet therapy for 3 months after the procedure. A statin therapy was suggested in all patients.
Endpoints and definitions
The primary measure outcome of this study was the overall mortality. A PTX specific survival analysis was also performed. For the specific survival evaluation, the analysis was censored in the event of PTX treatment at follow-up regardless indication. Secondary outcomes were primary patency rate, freedom from clinically driven target lesion revascularization (CD-TLR), and freedom from major amputation. Primary patency was defined as freedom from significant restenosis or occlusion without any re-intervention.
Significant restenosis was indicated by a >2.0 peak systolic velocity ratio calculated as the peak systolic flow velocity in the lesion divided by the peak systolic velocity 1 cm proximal to the lesion. Amputation-free survival was defined as the time until a major amputation of the index limb and/or death of any cause, whichever occurred first. A major amputation was defined as any above-ankle amputation. The degree of calcification was graded based on the arterial wall calcium deposits observed during fluoroscopy based on the suggested Peripheral Arterial Calcium Scoring Scale (PACSS). Grade 0 represents the lack of visible calcium at the target lesion, grade 1 refers to unilateral calcification shorter than 5 cm, grade 2 refers to unilateral wall calcification longer than 5 cm, grade 3 shows the presence of bilateral wall calcification shorter than 5 cm, and finally grade 4 is defined as bilateral wall calcification with calcium extension longer than 5 cm. 18 The cutoff of 15 cm was used to define long lesions based on the Consensus Definitions From Peripheral Academic Research Consortium (PARC) 19
Statistical analysis
For the statistical analysis and graphics, the MedCalc Statistical Software (version 12.4.0.0; MedCalc Software, Ostend, Belgium) was used. Continuous variables are presented as means ± standard deviation or median (interquartile range), while categorical data are given as the counts. Continuous numeric variables were compared by Student t test for paired samples or Wilcoxon test according to their distribution (D-Agostino-Pearson test). Cumulative survival, PTX specific survival, primary patency, as well as freedom from TLR and amputation-free-survival were estimated using the Kaplan-Meier method. A univariate analysis was performed for patients with patent stents versus patients with patency loss to identify statistically significant differences between the groups. Similar analysis was performed in the total cohort regarding mortality; these variables were included in Cox regression analyses to determine risk factors for patency loss and death, respectively. The threshold of statistical significance was p ≤ 0.05.
Results
Demographics.
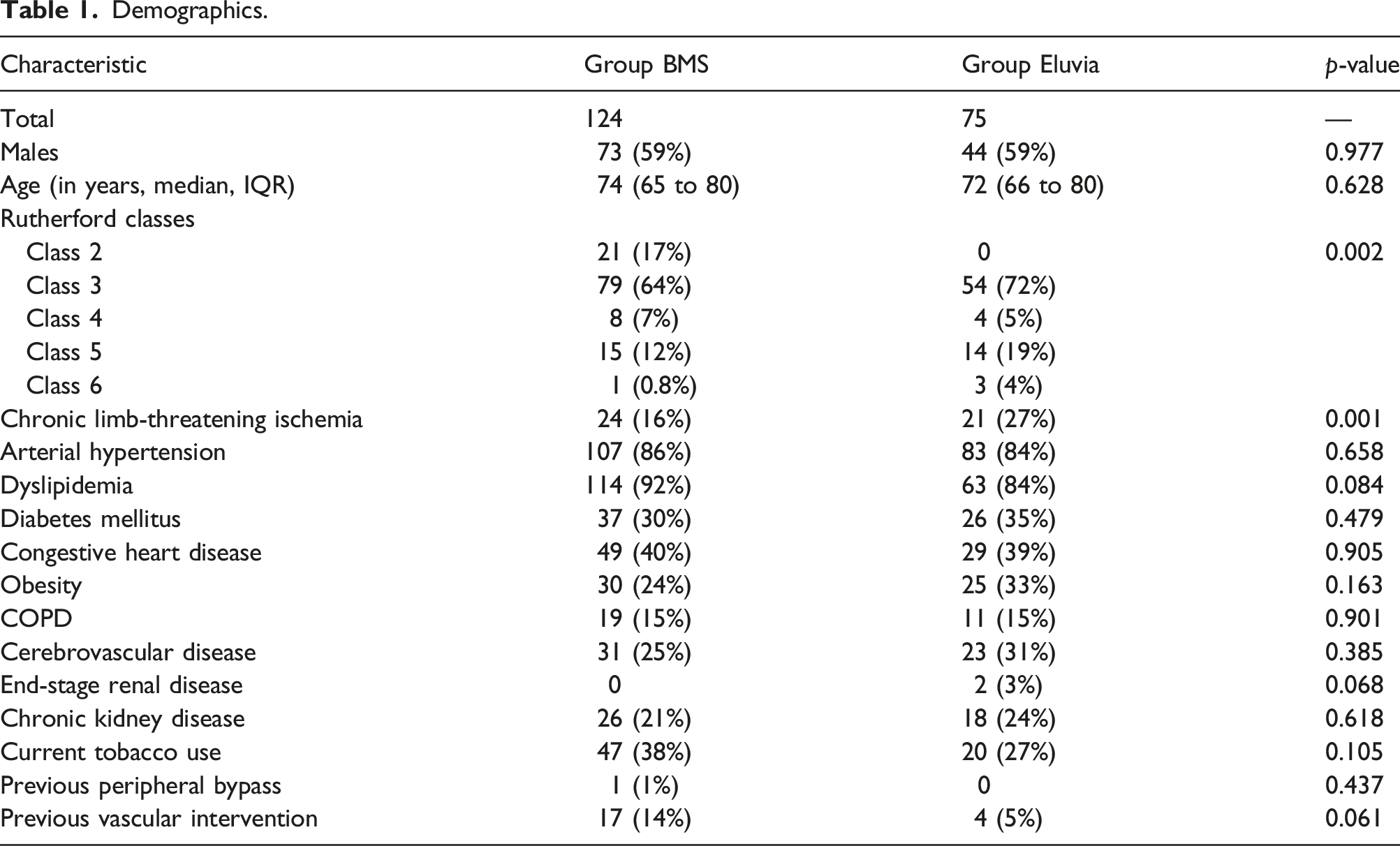
Anatomic and procedural characteristics.
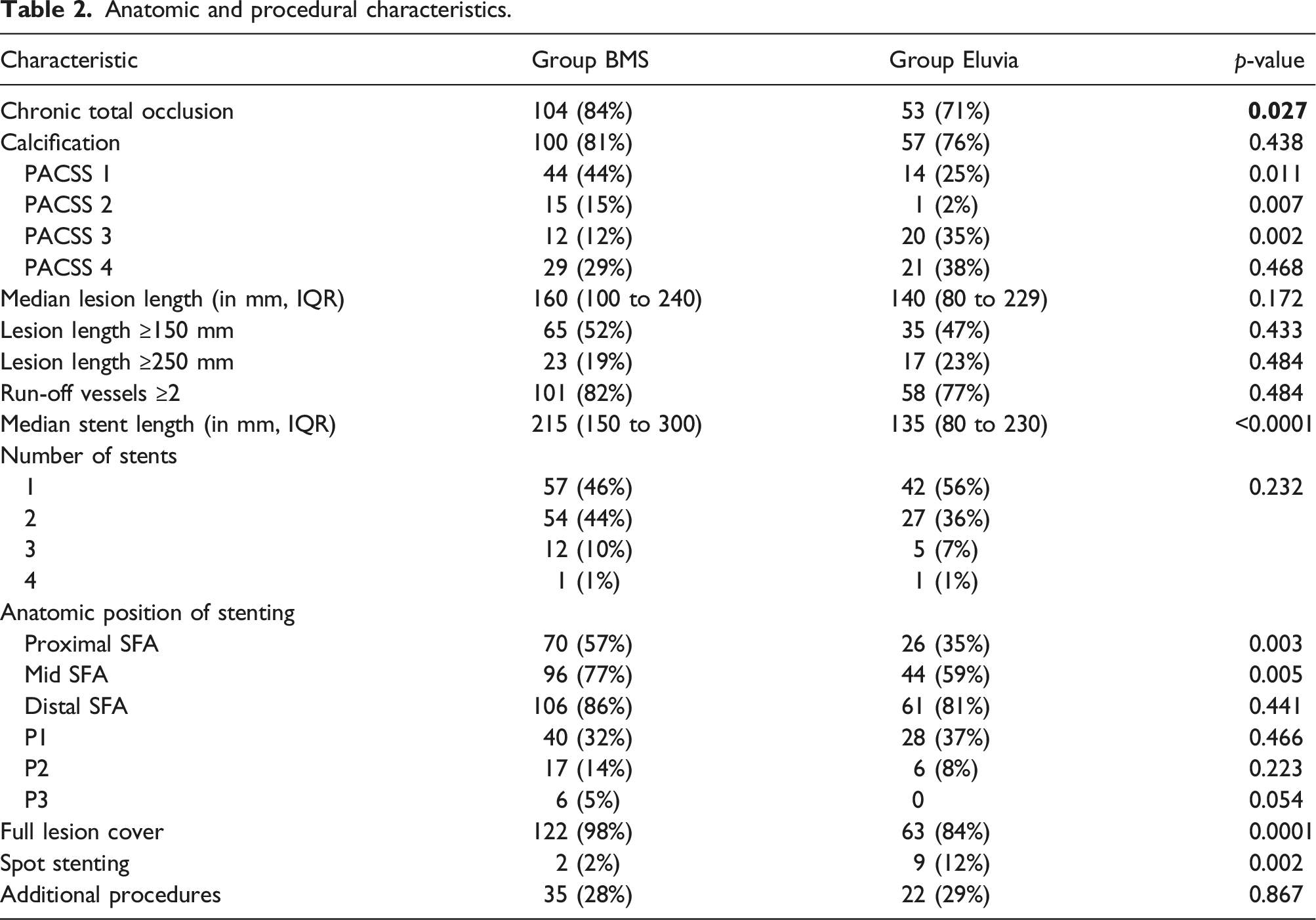
Overview of the 24-months outcomes between the two groups.
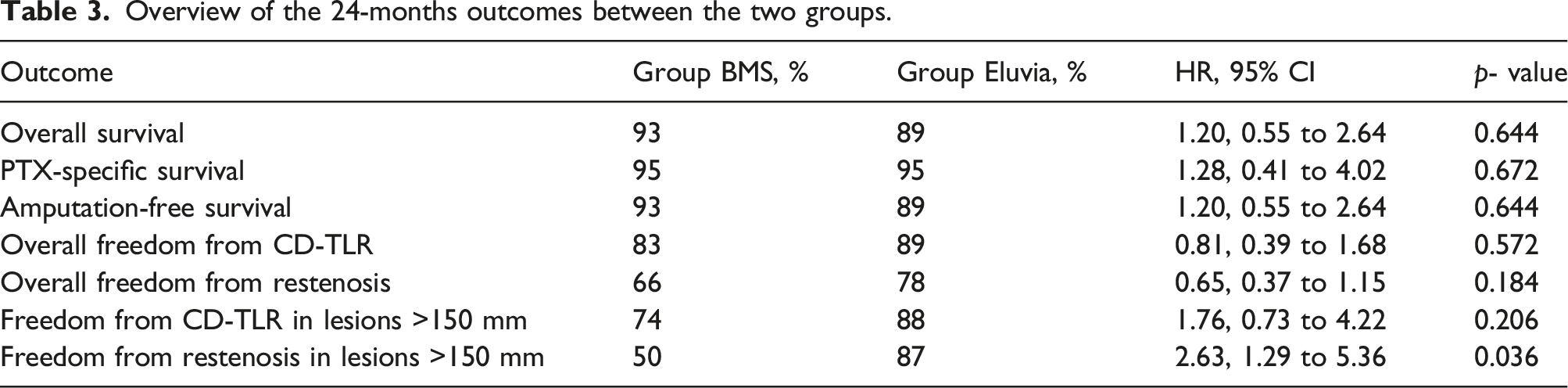
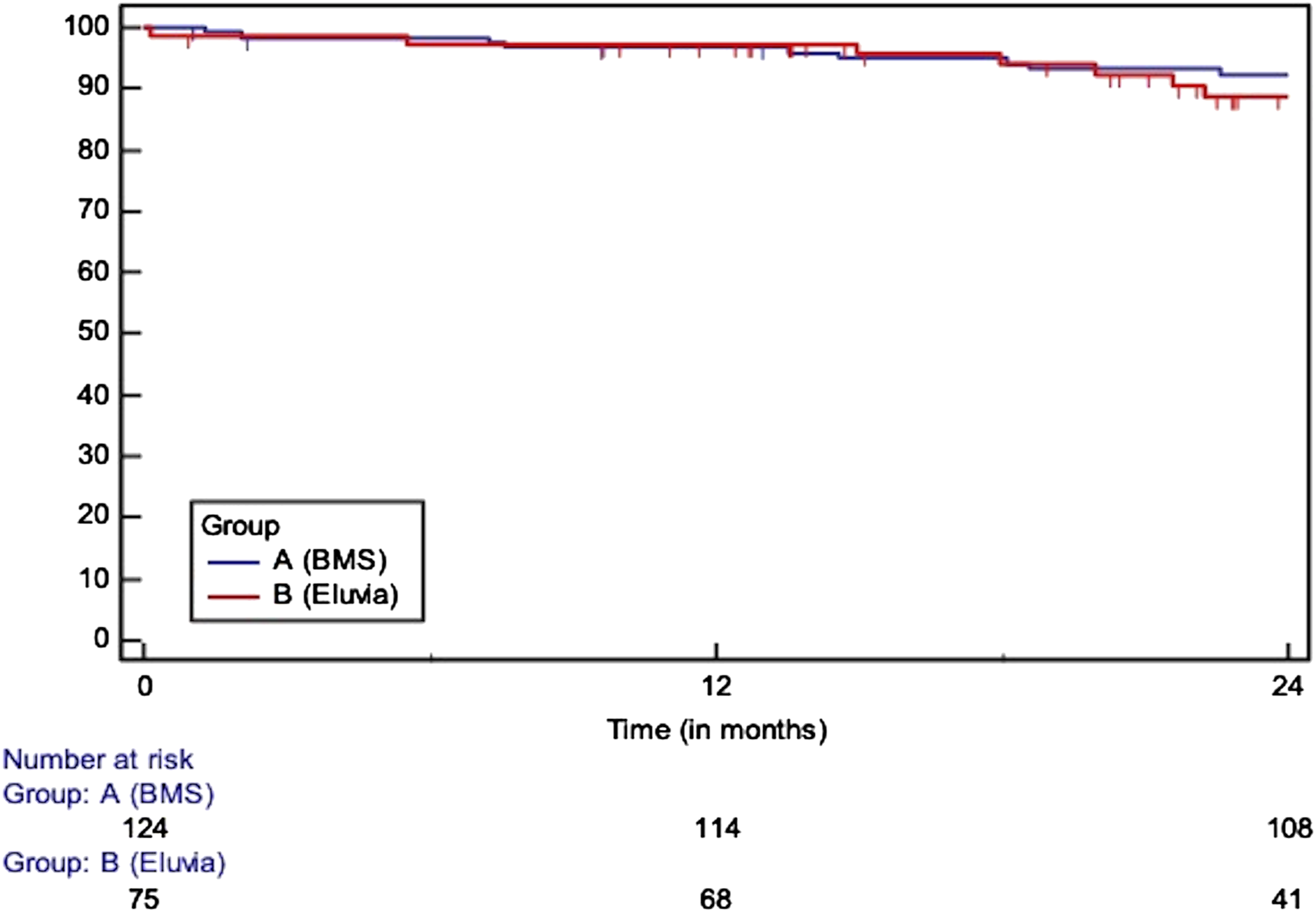
Survival (HR: 1.20, 95% CI: 0.55 to 2.64, p = 0.644), at 12 months: 97 vs 98%, at 24 m: 93% vs 89%)/SE>10% at 25.18 months.
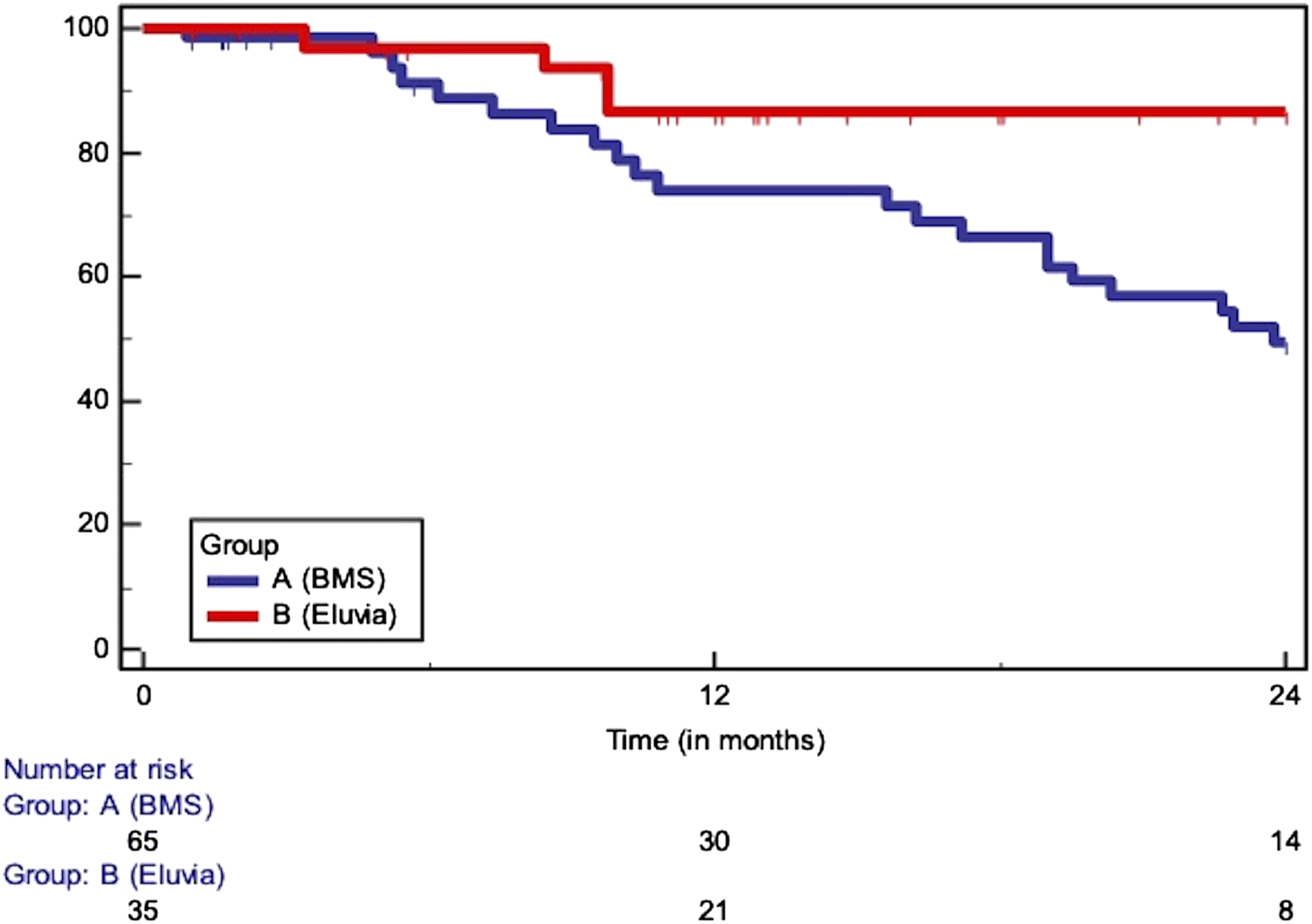
Primary patency in lesions longer than 150 mm (at 12 m: 73 vs 87%, at 24 m: 50 vs 87%, HR: 2.63, 95% CI: 1.29 to 5.36)/SE>10% at 24.79 months.
Cox regression analysis of risk factors for mortality during surveillance in the overall cohort.
Cox regression analysis of risk factors for restenosis in lesions longer than 150 mm.
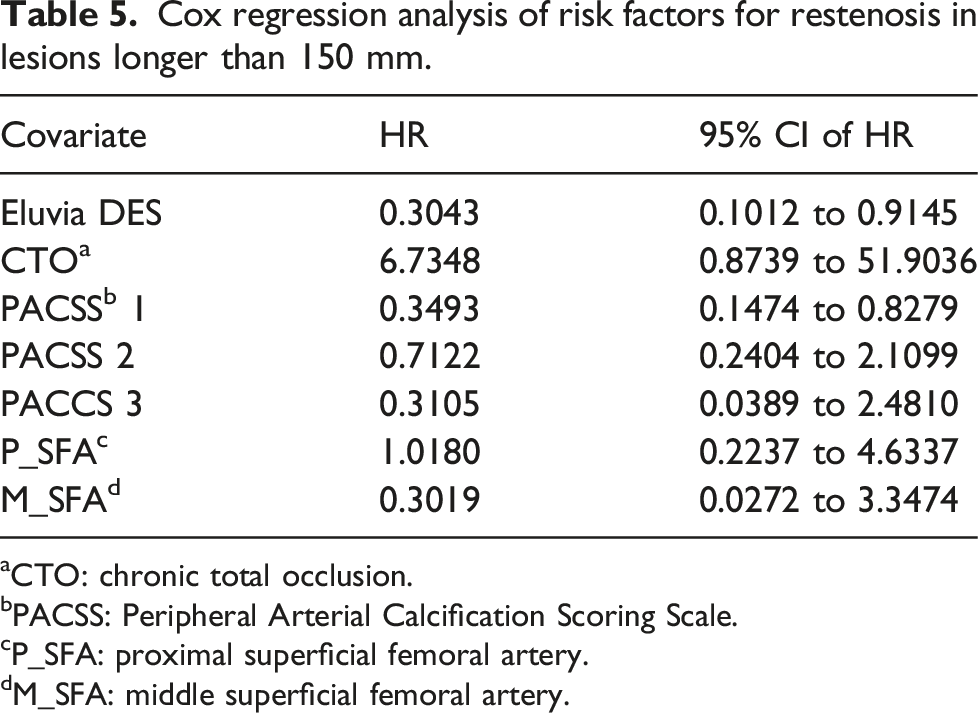
aCTO: chronic total occlusion.
bPACSS: Peripheral Arterial Calcification Scoring Scale.
cP_SFA: proximal superficial femoral artery.
dM_SFA: middle superficial femoral artery.
Discussion
The increased mortality risk, observed after the use of PTX coated devices, had a significant impact on the treatment of patients with PAD and prompted a debate among vascular specialists. 16 The Eluvia DES was not included in the initial meta-analysis and till now there is no available data regarding the safety profile of this polymer coated DES compared to non-PTX coated devices.1,7 In this cohort, the use of the Eluvia DES was not associated with a higher risk for death, while its clinical performance was comparable to the EverFlex BMS. A benefit in terms of increased patency was shown in lesions longer than 150 mm. CLTI, COPD, advanced age, and the lack of statin therapy at baseline were identified as independent risk factors for mortality.
In December 2018, a meta-analysis of 28 RCTs described an increased mortality rate after the use of PTX coated DCBs and DESs for femoropopliteal PAD. 7 Many concerns were raised regarding the design of this study, the heterogeneity of the mortality association across the trials and the lack of patient level data. However, two additional studies from the FDA and the VIVA physicians also reported a higher risk for death after the application of PTX.8,9 On the contrary, recent meta-analyses, studies of large-scale registries and single-center experiences could not confirm this finding.10–13,20–23
Although RCTs are considered the most scientifically rigorous clinical study design, patients included in prospective trials are highly selected, carry a different atherosclerotic profile and have a decreased comorbidity compared to real-world pragmatic cohorts.22,24 Unlike RCTs, registries and insurance claim data provide information on a more generalizable group of patients and physician practices. 22 Nonetheless, the published registries were not without limitations. No device specific recommendations could be made, the lesions characteristics could not be analyzed, and the use of PTX for coronary or peripheral interventions prior to the index procedure was unknown, while in case of TLR or repeated intervention, the secondary application of PTX was not assessed. In our study, we excluded patients previously or concomitantly treated by PTX and we additionally performed a PTX specific analysis for repeated interventions. Both the overall mortality and the PTX specific mortality did not differ between the two groups.
The discrepancy between the initially published meta-analysis, the VIVA and FDA evaluation and the registry data, mirrors the differences between the treated cohorts. In this study, although the Eluvia did not influence the risk for death, the presence of CLTI, COPD, and advance age were independent risk factors for mortality. Moreover, like previous observations, the lack of statin therapy was identified as risk factor highlighting the need for aggressive secondary prophylaxis. 25 Of note, these groups of patients are usually excluded from prospective trials, while the studies included in the meta-analysis of Katsanos et al. 7 did not have a standardized secondary prophylaxis protocol and could not provide any information regarding the medications of the included patients.
Regarding the indication for DES deployment in femoropopliteal lesions, a benefit was shown in this cohort only for lesions longer than 150 mm. In the Zilver PTX randomized trial, higher patency rates for relatively short lesions (average lesion length was ≈65±40 mm) were observed following the deployment of the Zilver PTX DES (Cook Medical, Bloomington, IN); however, in the Bare Metal Stent versus Paclitaxel Eluting Stent in the Setting of Primary Stenting of Intermediate-Length Femoropopliteal Lesions (BATTLE) trial, the Zilver PTX stent failed to show superiority over a BMS platform stent in freedom from restenosis at 1 year (mean lesion length was 7.6 ± 4.1 cm).3,26 In a mixed cohort of polymer and non-polymer coated DES, Saratzis et al. 27 reported comparable results between DES and interwoven stents for femoropopliteal disease. The ongoing EMINENT trial (NCT02921230) will assess the performance of the Eluvia DES over various commercially available BMS. Patients will be followed for 5 years. Although the study will provide valuable information regarding the efficacy of DES to inhibit restenosis, the exclusion of individuals with advanced ischemia (Rutherford class 5 and 6) might limit its applicability in a real-world scenario.
Given the promising performance of the Eluvia DES in complex disease, the high risk for restenosis with BMS in long lesions, and the results of this cohort, the preferable use of the Eluvia in longer lesions when a scaffold is needed seems reasonable.1,28 This approach has been adopted from many physicians as data from the Vascular Quality Initiative registry from 2010 to 2017 showed that DES were preferred over BMS for the treatment of longer lesions. 29
Limitations
This study carries the well-known limitations of retrospective registries. Despite the comparable baseline characteristics of the two groups, the lack of randomization remains an important limitation of this analysis. Finally, a higher patient volume and a longer follow-up (up to 5 years) might be needed to reveal a late mortality signal. Regarding the efficacy of both devices to inhibit restenosis, the median stent length in the BMS group was 215 mm vs 135 mm in the PTX eluting stent group. Also, chronic total occlusion frequency was significantly higher in the BMS group. Similarly, the BMS group had significantly higher rates of proximal and mid SFA stenting versus PTX stent. This might lead to a possible bias against the BMS group. Additionally, the selection of the used platforms was at the discretion of the treating physicians. The Eluvia DES was introduced in 2016 and patients treated prior to 2016 were mainly treated with BMS deployment. Nonetheless, after the introduction of the Eluvia platform, this scaffold was preferred. Finally, we did not perform any PTX dose dependent analysis, although the current body of literature does not suggest any dose dependent mortality effect.
Conclusions
Among patients undergoing femoropopliteal peripheral endovascular intervention with the Eluvia DES, there was no increased risk of 24 months, all-cause mortality. CLTI, COPD, advance age, and the lack of statin therapy were identified as independent risk factors for death. Comparable clinical outcomes, patency, and CD-TLR rates were observed following the use of the Eluvia and the EverFlex platforms in the overall cohort, while the use of DES reduced the restenosis rate in lesions longer than 150 mm.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
