Abstract
The aim of the BIOPAC trial was to determine long-term safety and efficacy of a novel microcrystalline paclitaxel-coated balloon (mcPCB) with a biocompatible polymer as an excipient in the treatment of occlusive femoropopliteal lesions. In this first-in-human prospective controlled randomized trial, 66 patients with femoropopliteal, symptomatic (Rutherford stages 2B to 5) occlusive arterial disease were randomized to either mcPCB (study group) or POBA (plain old balloon angioplasty) (control group) on a 1:1 basis. Late lumen loss (LLL) at 6 months was the primary endpoint of the study and serious adverse events (SAE: death, amputation, repeated revascularization) were considered a composite secondary endpoint. Routine angiography was scheduled for all study subjects at 6-month follow-up; outpatient appointments were scheduled at 12 and 36 months after intervention. At 6 months, the LLL was 63% lower in the mcPCB group compared to the POBA group (0.52 ± 1.2 vs 1.39 ± 1.1 mm; psup < 0.01). Binary restenosis occurred in 23% vs 52% of patients (p = 0.02). At 3 years, the prevalence of SAE was significantly lower in the mcPCB group (33.3 vs 63.3%; p = 0.02), which mainly resulted from a twofold reduction in target vessel revascularization rate (28.6 vs 59.3%; p = 0.02). The difference in mortality was nonsignificant (7.4 vs 14.3%; p = 0.42). Patients with mcPCB were less symptomatic and less likely to adhere to secondary prevention measures. In this pivotal trial, a novel mcPCB proved superior to POBA concerning LLL at 6-month follow-up, and SAE at 12 months. This result was sustained up to 3 years. There was no difference between groups regarding mortality.
Introduction
Peripheral artery disease (PAD) remains the major cause of morbidity, mortality, and disability. 1 Endovascular angioplasty has become a preferred strategy for lower extremity artery revascularization as it offers several advantages over open surgical repair. 2 Plain old balloon angioplasty (POBA) successfully restores vessel patency; however, high long-term rates of restenosis (40–60%) leading to repeated revascularization and treatment failure remain a clinical and economic challenge.2,3 Stenting, on the other hand, due to fracture risk, should mainly be considered as a bailout solution. 4
Paclitaxel-coated balloons (PCB) have emerged as a therapeutic option for revascularization of femoropopliteal arterial disease, which is deemed superior to POBA.5–7 This is mainly due to reduction of restenosis and repeated revascularization rates. The main advantage of PCB technology is long-term tissue retention of paclitaxel after a single application. 8 On the other hand, safety concerns have been raised regarding paclitaxel macro-particle shedding during intervention and distal embolization, particularly in critical limb ischemia (CLI) or below-the-knee lesions. 9 Most recently, Katsanos et al. 10 reported an increased risk of death following application of PCBs at long-term follow-up. Although subsequent publications have not confirmed these conclusions,11,12 any potential increase in long-term mortality rates raises legitimate concern. Hence, further analyses of this issue are undoubtedly well justified.
Second-generation technologies offer more homogeneous coatings achieved with semi-automatic syringe deposition as well as either amorphous or microparticles of crystalline paclitaxel. 13 In the pre-clinical setting, these improvements have resulted in improved vessel healing, 14 reduced particle shedding, and distal embolization.14,15 Nevertheless, clinical data on the safety and efficacy of these novel coating developments remain limited.
We therefore decided to present our 3-year outcomes of a second-generation, novel microcrystalline 3 µg paclitaxel/mm2 balloon with a Poligrade™ polymer as an excipient (microcrystalline paclitaxel-coated balloon (mcPCB), PAK; Balton Sp. z o.o., Warszawa, Poland), and compare these to POBA in a randomized clinical trial setting.
Methods
Study design and population
The BIOPAC trial is a prospective, controlled, single-blind, randomized clinical trial comparing late lumen loss (LLL) as a primary endpoint in patients with femoropopliteal lesions treated with the mcPCB versus uncoated control. The study was performed from 2014 through 2019 in five cardiovascular centers (list of the centers is provided in the online supplementary Appendix) and received unrestricted funding from the Balton company. The inclusion criteria consisted of occlusive or total single de novo or nonstent restenotic lesions (operator-determined > 50% stenosis; length > 4 cm and < 15 cm; reference vessel diameter > 3 mm and < 7 mm) and successful guidewire crossing. Eligible participants were > 18 years old with intermittent claudication or chronic CLI (i.e. Rutherford stages 2B to 5). Only patients with diameter stenosis of ⩾ 50% were included, with typical symptoms suggestive of limb ischemia.
The exclusion criteria were age > 80 years, Rutherford stage 6, acute coronary syndrome, heart failure with reduced ejection fraction, below-the-knee lesions, chronic kidney disease, history of stroke within the preceding 6 months, and life expectancy < 2 years.
The study complied with the Declaration of Helsinki and the protocol was approved by the institutional and local ethics committees – decision No. 2014/03/13/5. The trial was registered at ClinicalTrials.gov (NCT02145065). All patients provided written, informed consent for participation.
Enrollment
Patients were considered eligible for the study following successful guidewire crossing of the lesion. Patients who had met the guidewire crossing criterion were randomized with a 1:1 allocation ratio to either mcPCB or uncoated balloon only (control group; Lovix®; Balton) using a computerized system. The randomization was carried out following completion of a case report form.
Primary endpoint
LLL was the primary outcome measure in the target lesion segment (entire length of the balloon inflation area plus 5 mm proximal and distal margins) at 6 months as assessed by blinded angiographic core lab analysis (affiliated with the Center for Cardiovascular Research and Development, American Heart of Poland, Katowice). LLL was defined as the difference between minimal lumen diameter (MLD) after baseline procedure and at follow-up angiography.
Secondary endpoint
Serious adverse events (SAE) at 1, 6, 12, and 36 months were the composite secondary endpoint consisting of all-cause death, any amputation (major and minor) or repeat revascularization including target lesion revascularization (TLR) and target vessel revascularization (TVR). Additionally, Duplex Doppler evaluation including ankle–brachial index was performed prior to and post-procedure, and at 6, 12, and 36 months. The Rutherford stage was reported at each follow-up. Primary patency rates were based on freedom from TLR and angiographic binary restenosis > 50% at 6 months or Doppler ultrasound peak systolic velocity ratio > 2.5 at 6 months. Ischemia-driven revascularization was caused by symptomatic restenosis (> 50% diameter stenosis). CLI was defined as signs and symptoms falling within Rutherford stages 5 and 6.
SAE were independently adjudicated by the Clinical Events Committee; Data Safety and Monitoring Board, Center for Cardiovascular Research and Development, American Heart of Poland, Katowice; independently from clinical sites. None of the investigators was a member of these committees. The external, independent monitoring evaluated the progress of the study and data quality.
Device description
The test device was an over-the-wire balloon catheter coated with a proprietary biocompatible polymer film (Poligrade) serving as an excipient and microcrystalline form of paclitaxel at a dose of 3 μg/mm2. The coating of this device is applied by semi-automatic microsyringe surface drug deposition; a proprietary drying process allows more consistent, uniform, and microparticle paclitaxel coverage with minimal or no particle shedding during inflation in the in vitro setting (data on file at Balton). The control devices were over-the-wire 0.035” balloons (Lovix; Balton). Nitinol self-expandable uncoated stents were used when necessary. All coated balloons were 5–7 mm in diameter and 40–150 mm in length.
Procedures
Lesion eligibility was assessed on angiography performed via radial approach. Patients who had met the clinical and angiographic criteria received dual antiplatelet therapy with 150 mg aspirin and 300 mg clopidogrel before intervention unless they had previously received those medications. Heparin (50–90 IU/kg) was administered during the procedure. Antegrade or contralateral retrograde common femoral artery access was obtained using a 6 or 7 French sheath. Digital subtraction angiography with live qualitative vascular angiography (QVA) of the region of interest was performed. The lesion was then crossed with a hydrophilic 0.035” wire (Terumo Glidewire, Tokyo, Japan or AqWire, Medtronic, Fridley, MN, USA). Following randomization, the primary intervention started. POBA was performed in all participants. In the study group, following predilation, mcPCB (diameter equivalent to that of POBA) was inflated for no less than 120 seconds. The operators were instructed to ensure PCB placement proximally and distally beyond the margins of the predilated injury segment, to inflate within 3 minutes of insertion, and to maintain inflation for 120 seconds. Bailout stenting was recommended in both groups in case of severe C-type dissections and/or > 50% residual stenosis. Dual antiplatelet therapy was recommended for 1 month after POBA or mcPCB, and for 3 months if bailout stenting was performed.
Quantitative vascular angiography analysis
Baseline, post-procedural, and follow-up angiograms were digitally recorded and assessed off-line in the quantitative angiographic core laboratory (AHP Center for Cardiovascular Research and Development) using QAngio XA software (version 7.1.14.0, Medis, Medical Imaging Systems, Leiden, The Netherlands) by two independent experienced operators unaware of treatment allocation and independent from clinical sites. Measurements performed in projection images demonstrating the highest degree stenosis were included in the analysis. The contrast-unfilled, nontapered catheter tip was used for calibration. Neither the principal investigators nor the authors participated in core lab adjudication.
Statistics and study hypothesis
The study hypothesis and 6- and 12-month results have already been published. 16 Normally distributed parametric data were expressed as a mean and SD. In cases of skewed distribution, data were expressed as median and interquartile range. Nonparametric data were presented as an absolute number and percentage. A chi-squared test was used for comparison of nonparametric data, while the Student’s t-test was used for normally distributed parametric data. Survival and event-free survival curves were estimated using Kaplan–Meier analysis with a log-rank test for comparison of curves. These analyses were performed and graphs were made using Statistica 12 (StatSoft, Tulsa, OK, USA) and Stat MedCalc v.9 (MedCalc Software, Ostend, Belgium).
Results
Study flowchart and follow-up
The study scheme is presented in Figure 1. The mean follow-up was 1095 days (SD 7.03). Follow-up was completed from 29 patients (including two deaths, 27 contacted) of the study group (87.8%) and 29 (including four deaths) of the control group (87.8%). Ambulatory follow-up with duplex Doppler ultrasound was available in 16 patients of each group (48.4%).
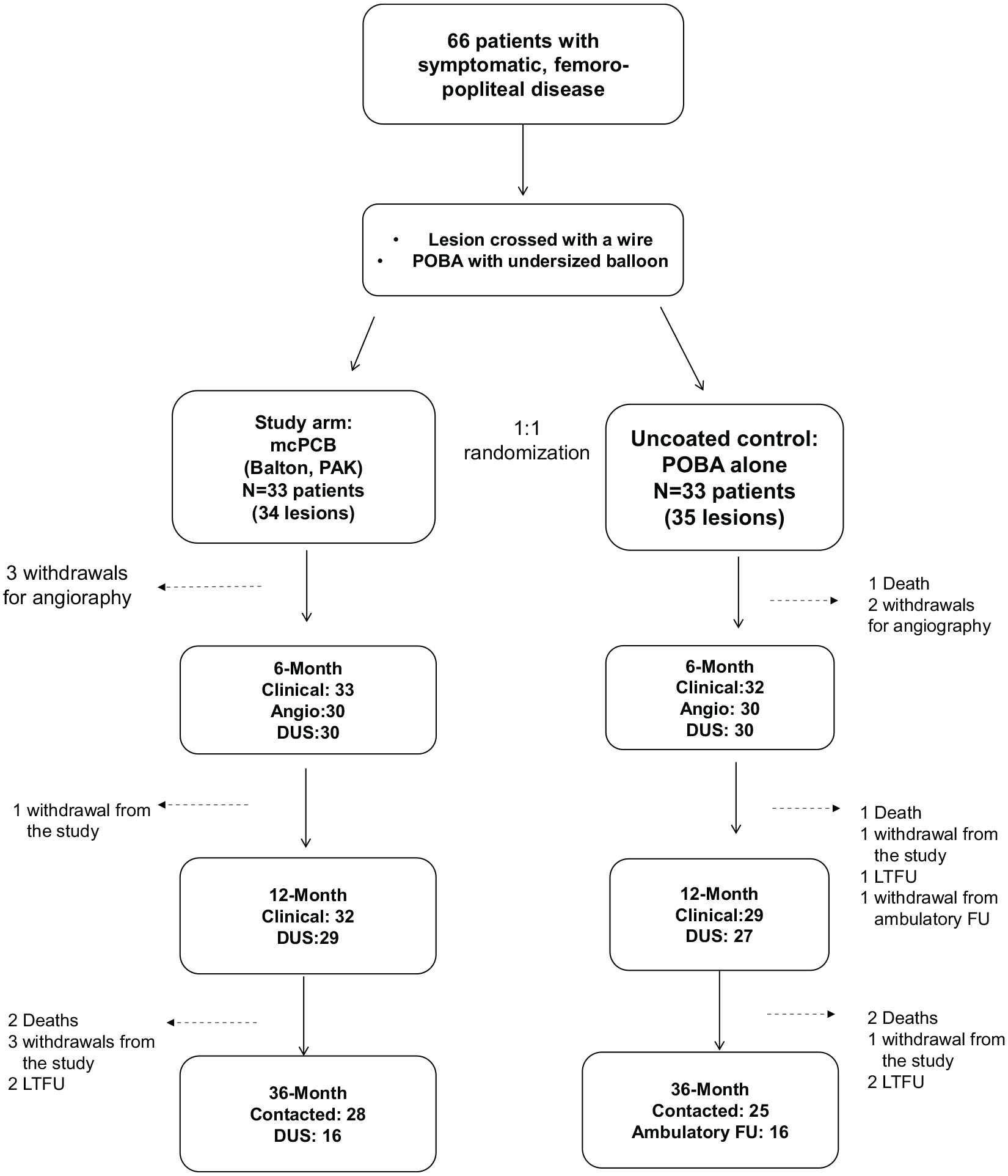
Study flowchart. Values reported at 6, 12, and 36 months are the number of patients.
Baseline characteristics
There were no significant differences in the patients’ baseline clinical and demographic characteristics except for the number of male participants, which was 24% higher in the study group compared to the uncoated control (Table 1). Additionally, there were more post-coronary artery bypass graft patients in the POBA group. Most of the study and control participants presented with coronary artery disease. The majority of the pathologies were de novo lesions in the superficial femoral artery. Intermediate- to high disease complexity was confirmed by a high proportion of chronic total occlusions, particularly in the mcPCB group. Mean lesion length was 60 mm in both groups; arterial diameters were also similar (Table 2).
Patient baseline clinical characteristics.
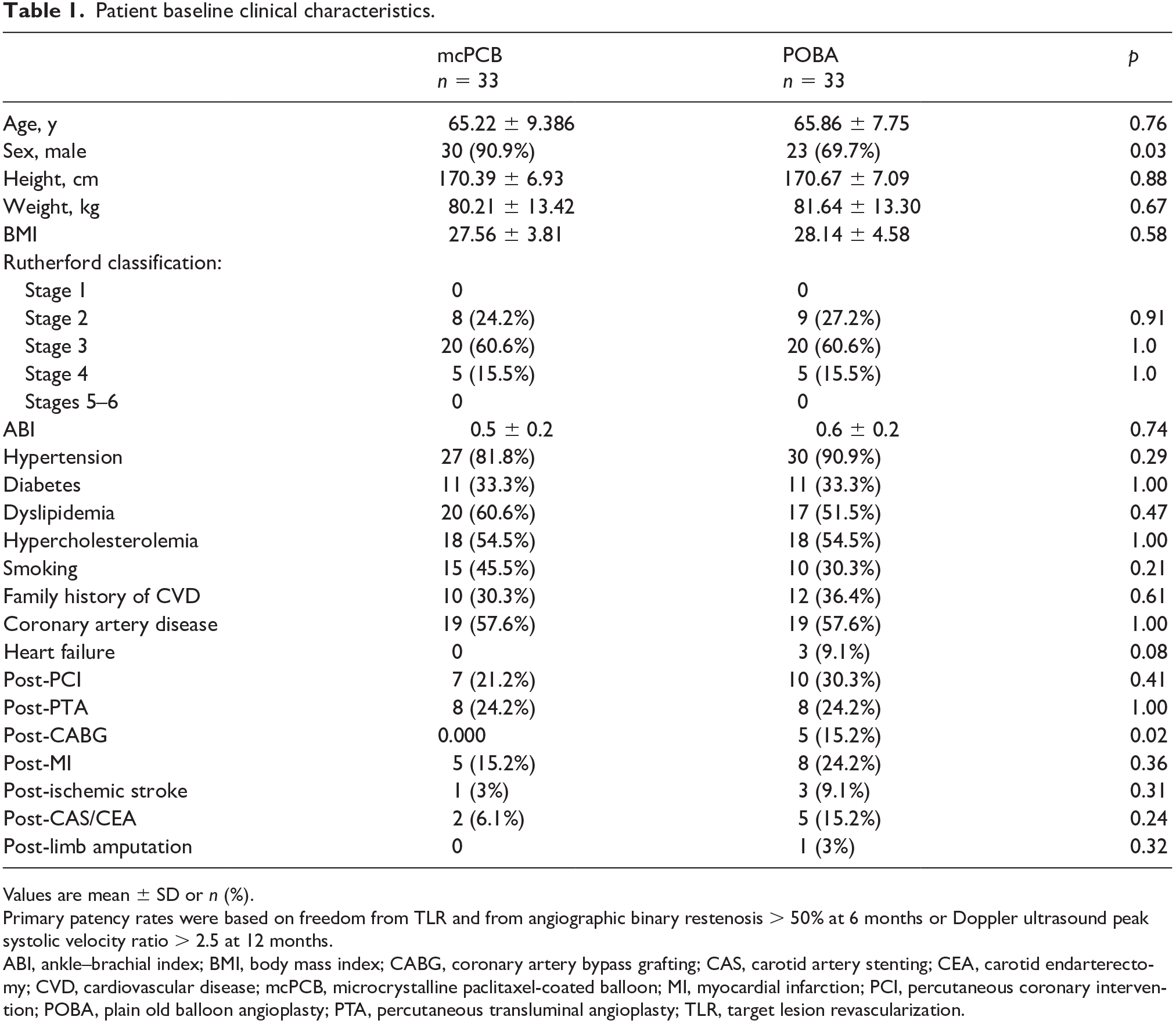
Values are mean ± SD or n (%).
Primary patency rates were based on freedom from TLR and from angiographic binary restenosis > 50% at 6 months or Doppler ultrasound peak systolic velocity ratio > 2.5 at 12 months.
ABI, ankle–brachial index; BMI, body mass index; CABG, coronary artery bypass grafting; CAS, carotid artery stenting; CEA, carotid endarterectomy; CVD, cardiovascular disease; mcPCB, microcrystalline paclitaxel-coated balloon; MI, myocardial infarction; PCI, percutaneous coronary intervention; POBA, plain old balloon angioplasty; PTA, percutaneous transluminal angioplasty; TLR, target lesion revascularization.
Procedural characteristics.
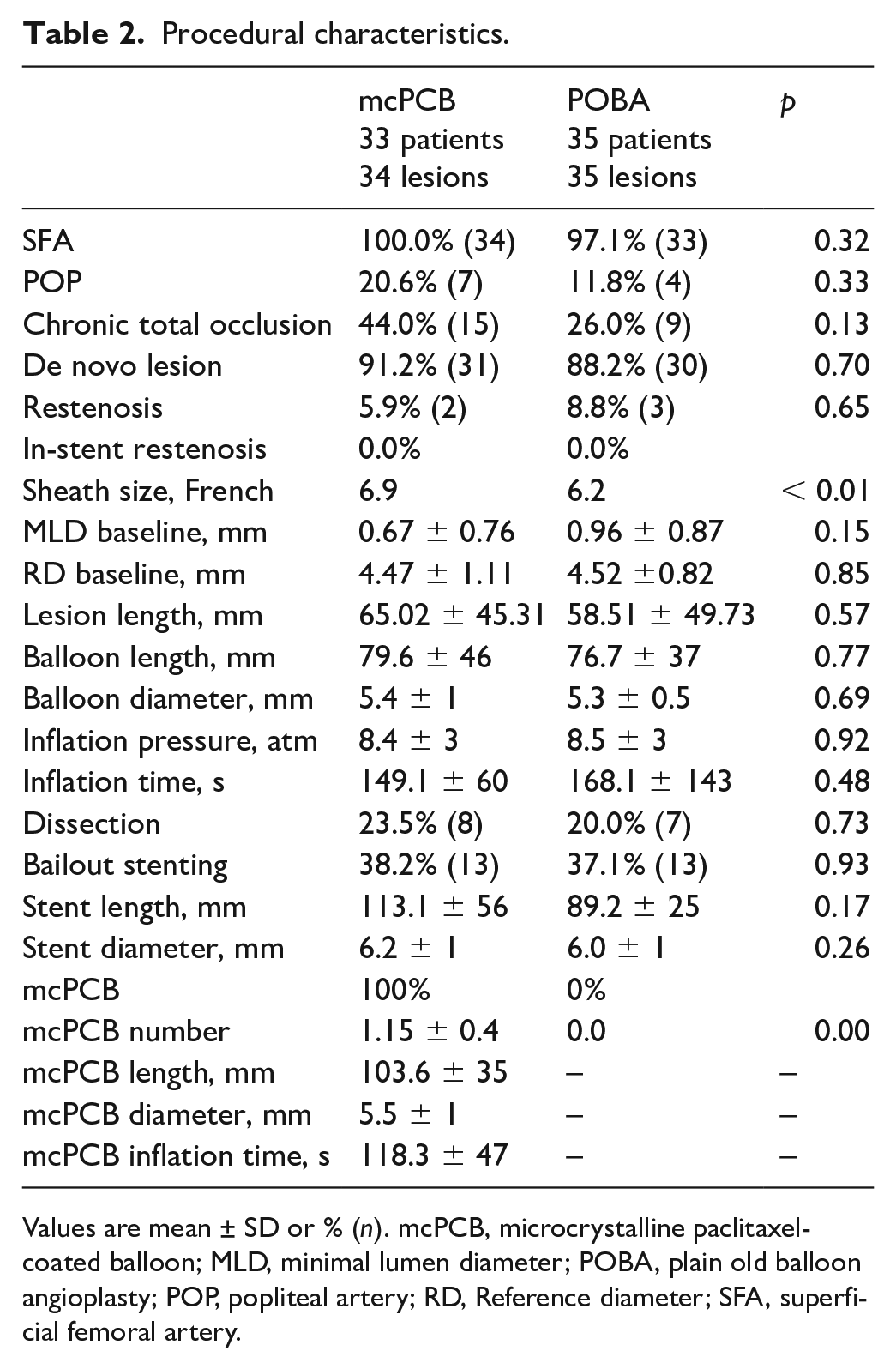
Values are mean ± SD or % (n). mcPCB, microcrystalline paclitaxel-coated balloon; MLD, minimal lumen diameter; POBA, plain old balloon angioplasty; POP, popliteal artery; RD, Reference diameter; SFA, superficial femoral artery.
There were no differences with regard to procedural details between the groups (Table 2). In most patients, the procedures were performed using antegrade femoral artery access; in two cases access was gained through a contralateral approach. The average sheath size was larger in the mcPCB group. In all cases, device delivery to target lesions was successful and followed by uneventful deployment, thus resulting in 100% procedural success. Bailout stenting was performed in over 30% of patients in both groups. The mcPCB catheters were 5–10 mm longer than regular POBA balloons used for predilation.
Angiographic outcome at 6 months
The primary endpoint was reported previously. 16 Briefly, LLL was significantly lower (by 63%) in the study group compared to the control (0.52 ± 1.2 mm vs 1.39 ± 1.1; absolute difference: 0.87, 95% CI for the difference: 0.29–1.44), thus confirming the superiority of mcPCB (p < 0.01). The binary restenosis was over twofold lower in the mcPCB group (odds ratio (OR): 0.27; 95% CI: 0.09–0.81; p = 0.02).
Clinical outcomes at 12 and 36 months of follow-up
At 12 months, the rate of SAE was significantly lower in the mcPCB group (outcomes were reported previously 13 ). The TLR (OR: 0.22; 95% CI: 0.07–0.69; p = 0.01) and TVR (OR: 0.31; 95% CI: 0.1–0.9; p = 0.03) rates were over twofold lower in the study group.
Clinical outcomes at 36 months
At 36 months, the rate of SAEs remained significantly lower in the mcPCB group, as presented in Figure 2A and in Table 3. The majority of SAEs were repeat revascularizations of the treated limb. After 1 year, there were two additional ischemia-driven revascularizations in the control group and one in the drug-coated balloon group. As a result, the TLR (hazard ratio (HR): 0.36; 95% CI: 0.17–0.81; class p = 0.01) and TVR (HR: 0.38; 95% CI: 0.17–0.85; p = 0.02) rates were over twofold higher in the control group. There was a significant difference in the mean Rutherford stage between the groups in favor of mcPCB (Table 4).
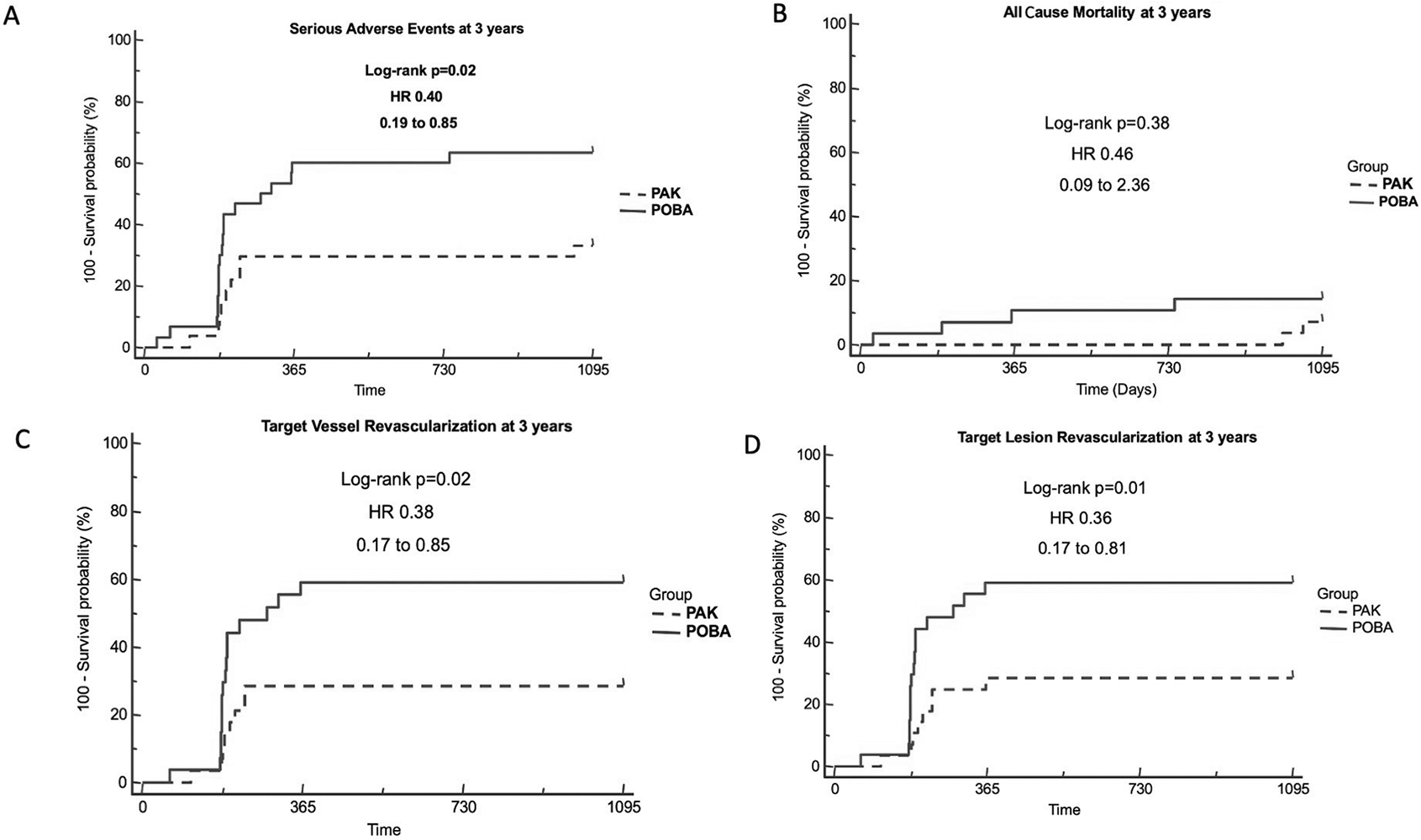
Outcomes of endovascular procedures in the BIOPAC trial. (A) Serious adverse events at 3 years; (B) all-cause mortality at 3 years; (C) target vessel revascularization at 3 years; (D) target lesion revascularization at 3 years.
Detailed analysis of cumulative SAEs at 36-month follow-up.
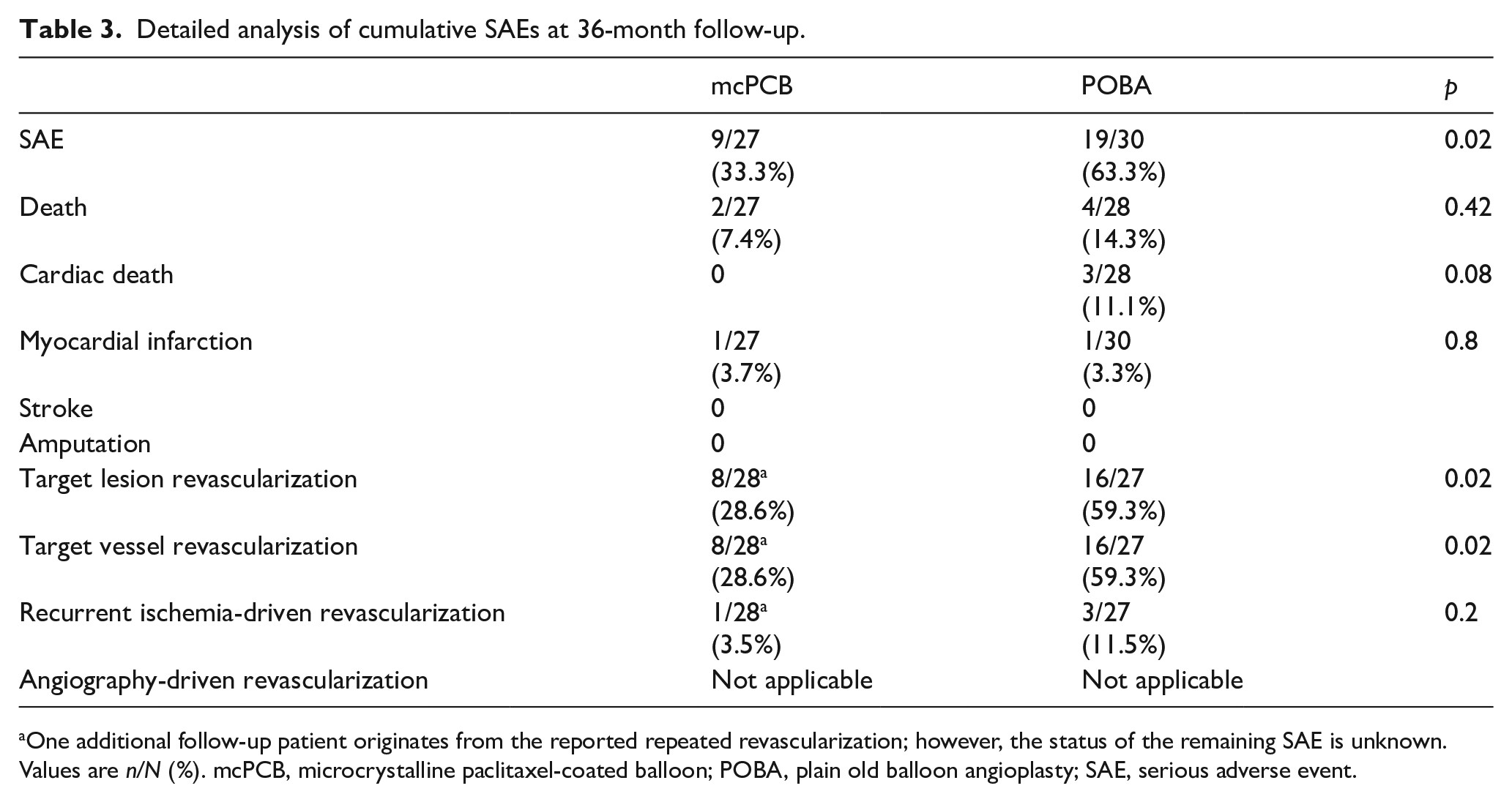
One additional follow-up patient originates from the reported repeated revascularization; however, the status of the remaining SAE is unknown.
Values are n/N (%). mcPCB, microcrystalline paclitaxel-coated balloon; POBA, plain old balloon angioplasty; SAE, serious adverse event.
Clinical and Doppler ultrasound assessment at 36-month follow-up.
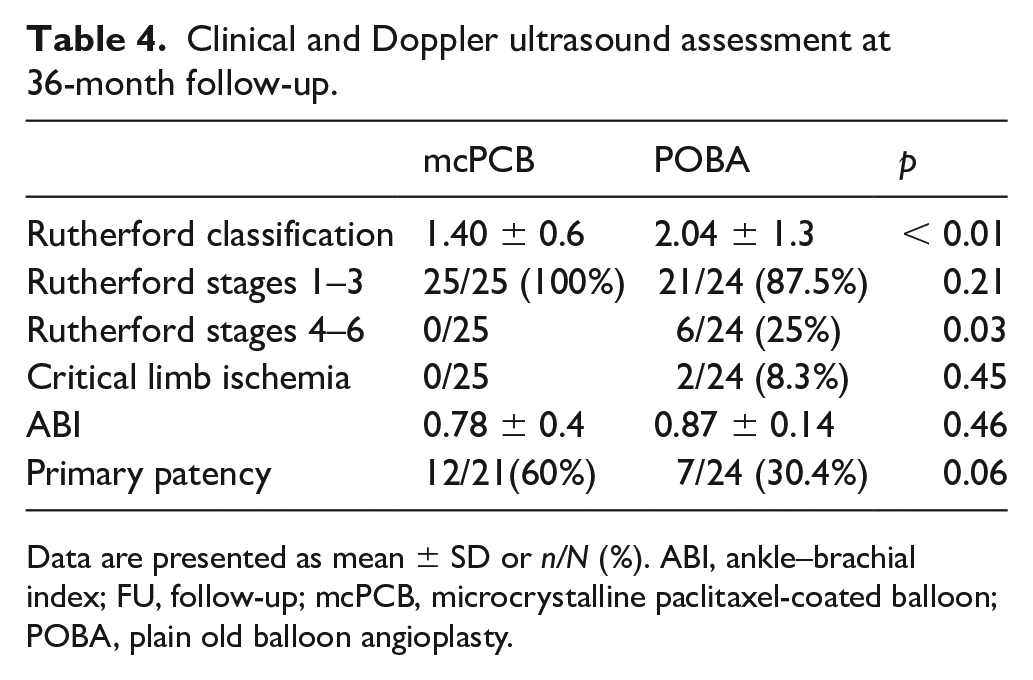
Data are presented as mean ± SD or n/N (%). ABI, ankle–brachial index; FU, follow-up; mcPCB, microcrystalline paclitaxel-coated balloon; POBA, plain old balloon angioplasty.
The study and control groups did not differ significantly with respect to total mortality (Figure 2B). There were two deaths beyond 1-year follow-up in the mcPCB group – both noncardiac. One patient died of acute pancreatitis and the other of unknown cause (most likely pulmonary embolism) (Table 3). There were four deaths in the uncoated balloon group – three patients died of heart failure within 1 year of follow-up and one beyond 1-year follow-up of unknown cause.
No amputations or cerebrovascular accidents were noted in the study and control groups during the 36-month follow-up.
There was one confirmed myocardial infarction (NSTEMI) in the study and control groups; the one in the study group occurred at 2 years and was treated with percutaneous coronary intervention.
Duplex Doppler and ischemia evaluation
Patients in the study group were less symptomatic when compared to controls, as shown by lower Rutherford stages (Table 4). All mcPCB patients remained within stages 1–3, whereas nearly a quarter of the POBA group exhibited stages 4–6 symptoms (p = 0.02). There were no differences in ABI and flow velocities within, proximally, and distally to the lesion. Primary patency at 6- and 12-month follow-ups was reported previously.
Secondary prevention measures
As shown in the online Supplemental Table 2, there were more smokers in the mcPCB group than in the POBA group at 3-year follow-up. There were no significant differences with regard to adherence to ambulatory follow-up. However, the mcPCB patients were less likely to attend ambulatory visits compared to those who had undergone POBA only. There were no significant differences with regard to mono or dual antiplatelet regimen; however, patients in the POBA group were more frequently on clopidogrel rather than low-dose aspirin. There were no differences with regard to hypolipemic treatment.
Discussion
First, second-generation PCBs have shown improved vessel healing and efficacy in the experimental setting, 14 although clinical studies to confirm this hypothesis are very limited. Second, warnings of increased mortality after PCB treatment in the long-term have recently been raised. 11 Therefore, our first-in-human, randomized, controlled study was aimed at testing the clinical safety and efficacy of restenosis inhibition by a novel biocompatible polymer and mcPCB technology in patients with occlusive femoropopliteal disease. Herein we report 3-year outcomes that seem to confirm long-term efficacy and safety as represented by a twofold reduction of SAE and necessity for repeat revascularization. Importantly, there were no differences in mortality between the study and control groups.
The novelty of the tested mcPCB has two aspects. First, the coating consists of microcrystalline paclitaxel particles characterized by decreased particle release (data on file at Balton). Second, the novel proprietary Poligrade polymer has been used as an excipient. In the pre-clinical setting, the tested mcPCB has shown 50% reduction of neointimal hyperplasia compared to POBA, whereas healing was complete. 2
As reported before, the primary endpoint (i.e. late lumen loss at 6 months) was reduced by 63%, thus meeting the pre-specified criteria for superiority when compared to the uncoated control. Binary restenosis decreased by over 50%. The composite endpoint of SAE at 6 and 12 months was approximately 2.5-fold lower compared to the control group, which was mainly caused by a reduction in repeat revascularization rates sustained throughout a 3-year follow-up. The difference was statistically significant. This was achieved despite slightly higher lesion complexity in the mcPCB group as evidenced by higher prevalence of Trans-Atlantic Inter-Societal Consensus (TASC) B, C, and D lesions compared to previously cited studies. This reduction in TLR is consistent with previous other reports in which 3 μg/mm2 paclitaxel concentration was used and proved superior to balloons with 2 μg/mm2. No differences were noted in repeat revascularization rates between the latter and uncoated devices.6,7,12 There were no thrombosis or amputations of any of the treated limbs, confirming the safety of the intervention. During follow-up, CLI was only observed in the control group.
During a 36-month observation period, there were two deaths in the study group and four in the control group. Deaths in the mcPCB group were caused by pancreatitis (one patient) and suspected pulmonary embolism; both occurred after 1 year of follow-up. Three control participants died of heart failure and one of unknown cause. Consequently, and contrary to other reports,10–12 patients in the mcPCB group showed lower mortality rates compared to POBA.
At 3-year follow-up, patients in the mcPCB group were less symptomatic and less frequently presented with severe claudication. However, this positive effect could have influenced the adherence to secondary prevention measures. Our analysis showed that mcPCB participants were more likely to smoke and less likely to attend ambulatory care appointments, especially beyond 1-year follow-up, when regular check-ups were no longer scheduled. As a result, fewer patients were on dual antiplatelet therapy. This is consistent with the observations of other authors, including patient-level meta-analysis. 13 It could be that lesser patient adherence to secondary prevention measures, caused by lack or very few symptoms, may lead to adverse events in the future.
Although a second-generation device was used in this study, clinical advantages of this technology remain unresolved. We therefore believe that a direct clinical comparison of first- and second-generation technologies (including sirolimus-coated balloons) is required and well-justified. Evaluation in below-the-knee arteries and CLI is also needed where reduction of paclitaxel particle size and improved vessel healing may play an important role.
Study limitations
The major study limitation is the small number of patients. Nevertheless, the sample size was sufficient to prove mcPCB superiority in terms of late lumen loss reduction at 6 months. The uneven proportion of male and female sex in the study group is accidental and caused by the relatively small number of patients. We excluded patients with chronic kidney disease.
Conclusions
In this trial, at 6 months, a novel mcPCB with polymer coating has shown superior efficacy regarding restenosis inhibition and LLL prevention after femoropopliteal artery revascularization when compared to POBA. At 36 months post-intervention, the rate of SAE remained significantly lower in the mcPCB group, mainly as a result of an approximately twofold decrease in repeat revascularization rates. The safety of the mcPCB with a Poligrade polymer technology seems comparable to that of uncoated balloons. Importantly, our study did not confirm higher mortality rates in patients treated with mcPCB. Further investigations in different anatomical regions, clinical scenarios, and comparison with first-generation technologies are required.
Supplemental Material
sj-docx-1-vmj-10.1177_1358863X20988360 – Supplemental material for Microcrystalline paclitaxel-coated balloon for revascularization of femoropopliteal artery disease: Three-year outcomes of the randomized BIOPAC trial
Supplemental material, sj-docx-1-vmj-10.1177_1358863X20988360 for Microcrystalline paclitaxel-coated balloon for revascularization of femoropopliteal artery disease: Three-year outcomes of the randomized BIOPAC trial by Przemysław Nowakowski, Wojciech Uchto, Eugeniusz Hrycek, Mateusz Kachel, Tomasz Ludyga, Filip Polczyk, Aleksander Żurakowski, Paweł Kaźmierczak, Juan F Granada, Iwona Nowakowska, Radosław S Kiesz, Krzysztof P Milewski, Paweł E Buszman and Piotr P Buszman in Vascular Medicine
Supplemental Material
sj-docx-2-vmj-10.1177_1358863X20988360 – Supplemental material for Microcrystalline paclitaxel-coated balloon for revascularization of femoropopliteal artery disease: Three-year outcomes of the randomized BIOPAC trial
Supplemental material, sj-docx-2-vmj-10.1177_1358863X20988360 for Microcrystalline paclitaxel-coated balloon for revascularization of femoropopliteal artery disease: Three-year outcomes of the randomized BIOPAC trial by Przemysław Nowakowski, Wojciech Uchto, Eugeniusz Hrycek, Mateusz Kachel, Tomasz Ludyga, Filip Polczyk, Aleksander Żurakowski, Paweł Kaźmierczak, Juan F Granada, Iwona Nowakowska, Radosław S Kiesz, Krzysztof P Milewski, Paweł E Buszman and Piotr P Buszman in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Piotr P Buszman and Przemysław Nowakowski received travel grants and lecture fees from the Balton company that were not directly related to the study. The other authors have nothing to declare.
Funding
This study was funded from the unrestricted grant provided by the Balton Company, Warsaw, Poland. Balton company did not have any role in the study execution, data analysis, or manuscript creation.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
