Abstract
VenaSealTM is composed of a cyanoacrylate adhesive compound often utilized for chronic venous stasis treatment. Rare case reports of hypersensitivity reactions to this compound exist. We present the first case of dermatographism and angioedema after utilization of VenaSealTM successfully treated via high dose antihistamines. We also present a case of type IV hypersensitivity to VenaSealTM, a cyanoacrylate, occurring in a patient with known meth(acrylate) allergy indicating a possible cross reactivity between these acrylate groups.
Introduction
VenaSealTM (Medtronic, Minneapolis, Minnesota) consists of a cyanoacrylate adhesive compound (n-butyl- 2-cyanoacrylate) that when injected endovenously, solidifies via polymerization and causes an inflammatory reaction with the venous wall. Since its FDA approval in 2015, VenaSealTM has been used as a therapy for pathologic saphenous venous incompetence. 1 In the initial clinical trials assessing the efficacy of VenaSealTM, there were several cases of delayed onset erythematous and pruritic dermal reactions. A follow up study to further characterize these hypersensitivity reactions following VenaSeal procedures led to identification of delayed onset pruritic rash in 6.3% patients. The rash presented 3–28 days after the procedure in the area of cyanoacrylate (CA) exposure and successfully treated via corticosteroids in majority of cases. 2 Subsequently, other supporting cases reports have noted type IV hypersensitivity reactions following VenaSealTM therapy.3,4 We present the first case of type I hypersensitivity following CA exposure, as well as a case of type IV hypersensitivity due to cross reactivity between cyanoacrylate and meth(acrylate).
Case report 1
A 66-year-old female presented with 3 weeks of dermatographic urticaria, pruritus and an episode of angioedema following great saphenous varicose vein closure with VenaSeal Closure Therapy. Symptoms began on post-operative day (POD) 1 and progressively worsened over 3 weeks. The patient had no known food or drug allergies and had not had similar symptoms prior to this episode.
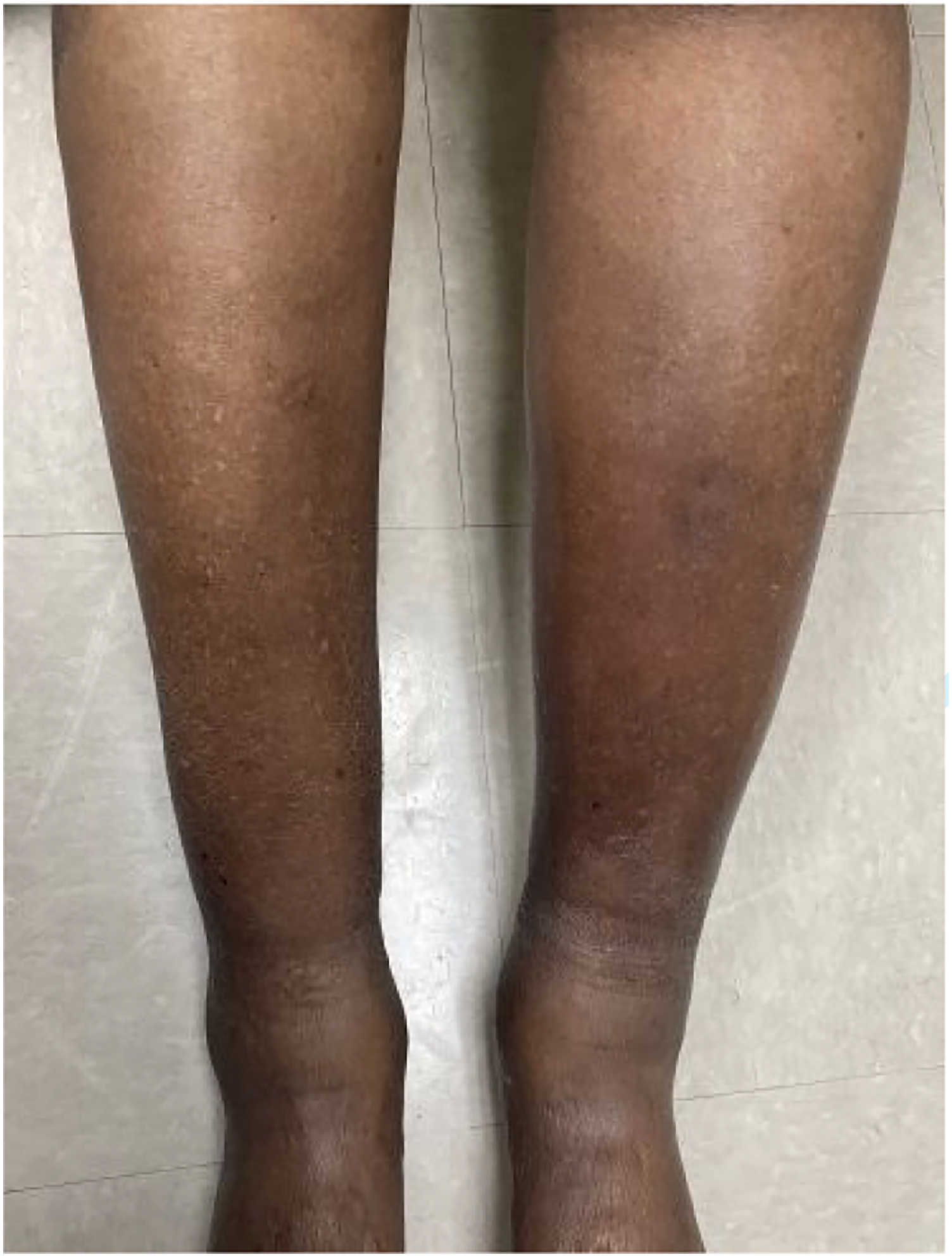
The patient’s ultrasonography demonstrated reflux of the left lower extremity great saphenous vein, without evidence of deep venous thrombosis. The patient subsequently underwent great saphenous vein closure for superficial venous reflux with pain. On POD 1, the patient noticed pruritus without a rash on the medial aspect of the treated left lower extremity that progressed to include the entire lower extremity over the following 2 weeks. On POD 12, she presented to the emergency department with tongue and lip swelling along with acute onset dyspnea. After a course of oral steroids and antihistamines, she had resolution of angioedema but progression of full body pruritus and development of dermatographism (Figure 1) with painful lower extremity erythema and edema (Figure 2). Elevated dosing of antihistamines improved her urticaria and pruritus over the following 2 months, with resolution of pruritis, urticaria, and erythema upon return visit on POD 169. Acute dermatographic urticaria on patient’s right upper extremity. Progressive, painful left lower extremity erythema and edema on POD 21.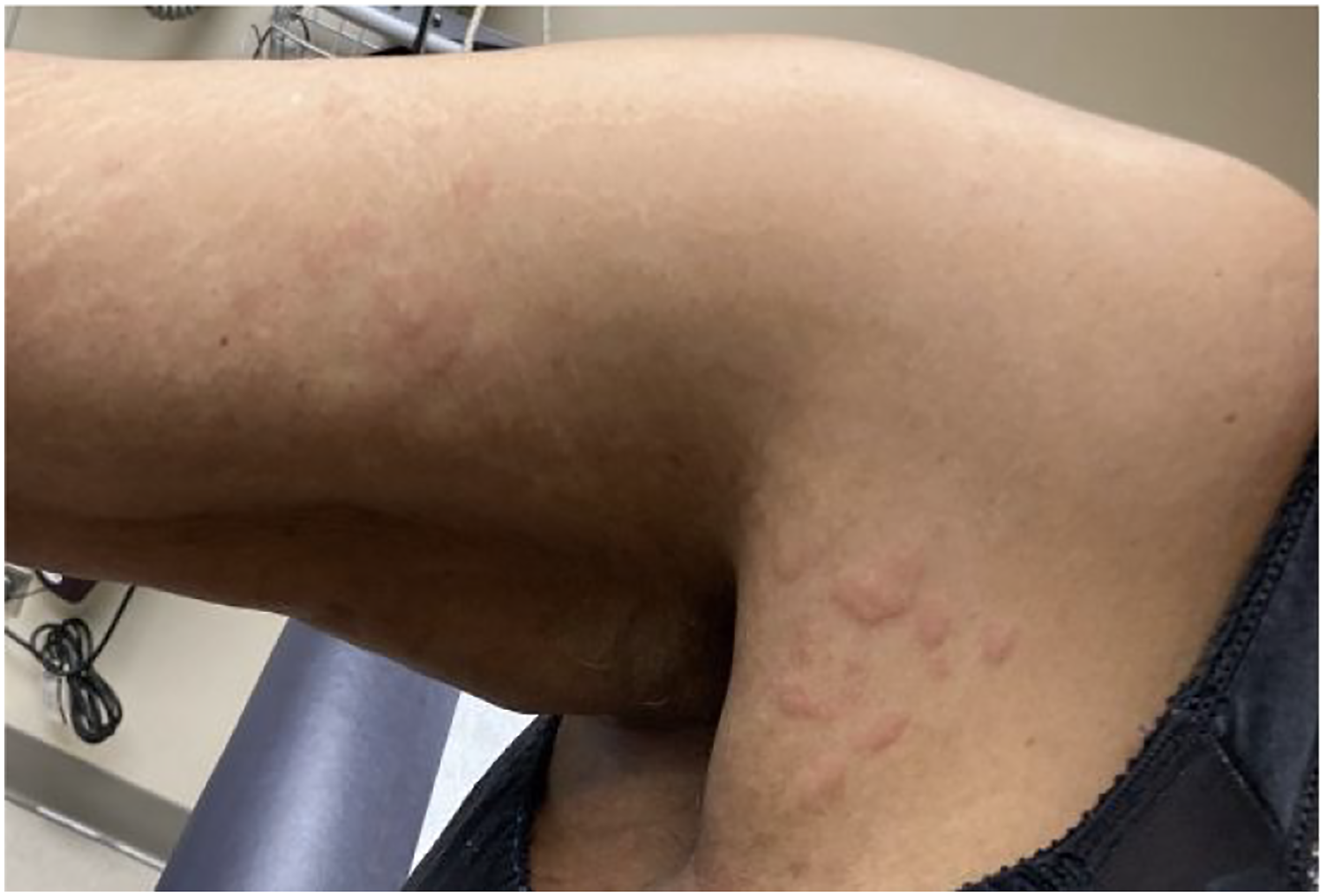
Case report 2
A 57-year-old female presented with new onset localized pruritic rash on POD 1 after vein closure with VenaSealTM Closure Therapy on left lower extremity. Medical attention was not sought by the patient until POD 6 when she underwent the same VenaSealTM Closure Therapy procedure for her right lower extremity and was given a course prednisone following the procedure to treat the initial rash. The following day (POD 7&1), approximately 24 hours after the second closure, the patient developed a similar pruritic rash overlying the right lower extremity despite starting the corticosteroid course. The rash progressed leading her to present to the allergy clinic on POD 13&6 where she was noted to have an erythematous-violaceous patch within the defined areas surrounding the puncture mark from catheter placement that appeared to spread superiorly, including left posterior distal lower extremity and right posterior proximal lower extremity. Review of her history indicated allergic contact dermatitis to various adhesives and chlorhexidine of which the latter was not used during either procedure. She was treated via a course of oral and topical corticosteroids which led to progressive improvement.
Discussion
VenaSealTM Closure Therapy utilizes cyanoacrylate (CA) adhesive compound (n-butyl- 2-cyanoacrylate) to treat pathologic saphenous venous incompetence. Rare cases of hypersensitivity reactions have been reported to date.
Our first case is the first report to our knowledge of new onset dermatographism and angioedema after implantation of CA. Our second case supports previous reports of allergic contact dermatitis presentation to CA exposure, however is the first to report cross reactivity between two different forms of acylate. These cases highlight the versatility in acylate induced hypersensitivity reactions (histaminergic and T-cell mediated, respectively) that may be seen due to VenaSealTM Closure Therapy.
Acrylates have been utilized in the generation of adhesives, glues, and plastic. Its presence can been found in beauty products (eyelash and artificial nail adhesives), medical products (wound dressings, denture adhesive), and industrial products (rubber and plastic generation). With increased exposure, the rate of hypersensitivity reactions has also increased leading to acrylate being the contact allergen of the year in 2012 and 2020. These reactions have tended to be a type IV hypersensitivity mechanism with rates of reaction based on patch testing to be 1.4% – 3.4% in 2015–2016 based on the various formulations of acrylate. 5 CA hypersensitivity reports have all indicated a delayed type IV (T-cell mediated) hypersensitivity as seen with other acrylate exposures.2,3,4 Our patient had the unique manifestation of dermatographism and angioedema, indicating a type I hypersensitivity (histaminergic), that was successfully treated with high dose antihistamines alone. This signifies an opportunity to educate patients regarding potential outcomes that should be self monitored within the first day of therapy. Furthermore, treatment does not have to include corticosteroids given the histaminergic mechanism; rather urticarial rashes often resolve with up to fourfold dosing of a second generation antihistamine.
Mechanism of type IV hypersensitivity (T-cell mediated) has been supported by positive patch testing results to VenaSealTM in patients with delayed pruritic rash. In these same patients, patch testing to other acrylates, such as (meth)acrylate and cyanoacrylate allergens (ethyl cyanoacrylate or 2-octyl cyanoacrylate), at the same time were reported to be negative indicating tolerance to other acrylate products.4,6 Contrary to this data, our second case identifies an allergic contact dermatitis (ACD) reaction to CA in a patient with a known of history of ACD to meth(acrylate), indicating cross reactivity between these two different forms of acrylate. Clinically, this suggests the potential benefit in inquiring about adhesive specific allergy in patients prior to VenaSealTM Closure Therapy. Screening positively on history would then be an indication for further evaluation such as patch testing prior to VenaSealTM therapy.
Conclusion
Utilization of acrylates in medical therapy is increasing. As a result, various forms of hypersensitivity will likely be seen. It is important to educate patients on the possibility of urticaria and angioedema development after VenaSealTM therapy. Furthermore, there is a potential of cross reactivity between patients reporting meth(acrylate) hypersensitivity and CA indicating a tool to screen higher risk patients prior to therapy. Both manifestations have clinical implications in patient education, as well as potentially reducing risk of new onset hypersensitivity reactions.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
