Abstract
Objectives
Abdominal aortic aneurysms (AAAs) demonstrating hostile neck anatomy (HNA) are associated with increased perioperative risk and mortality. A number of these patients are not suitable for standard endovascular aneurysm repair (EVAR) and are high risk for open surgery. We present our experience with the first implantations in Scotland of a novel conformable aortic stent-graft designed to overcome some of the challenges of HNAs.
Methods
From May 2018 to March 2022, 24 consecutive patients with non-ruptured AAAs demonstrating HNAs (neck length < 15 mm, or angulation > 60°) were treated with GORE Excluder Conformable AAA endoprosthesis (CLEVAR) (CEXC Device, W.L. Gore and Associates, Flagstaff, AZ, USA) at a Scottish vascular centre. We assessed clinical outcomes and technical success of CLEVAR during deployment, primary admission and the post-operative period at 3- and 12-month clinical follow-up alongside CT angiography.
Results
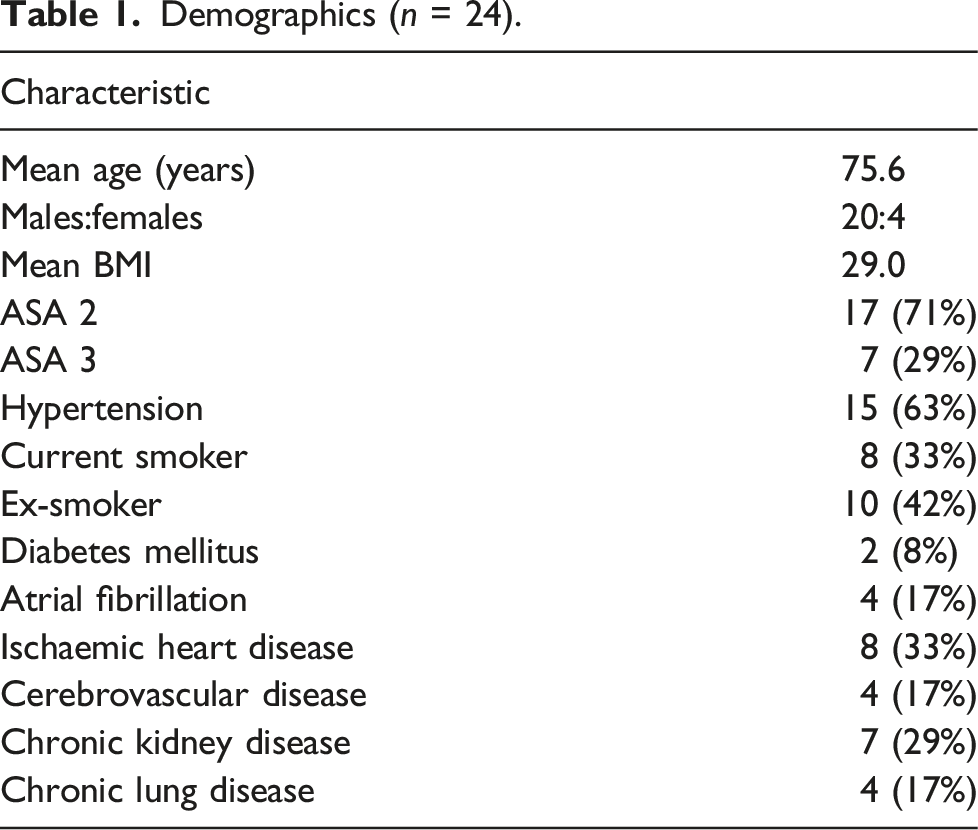
Twenty-four patients (20 males, mean age 75.6) were included. Primary technical success of proximal seal zones and CLEVAR deployment (no type 1/3 endoleaks, no conversion to open repair, AAA excluded and patient leaving theatre alive) was achieved in 100% of patients. All patients were alive and clinically stable at 3- and 12-month follow-up. There were five patients requiring re-intervention; at the 3-month follow-up, one patient (4.2%) developed a type 1b endoleak requiring graft limb extension, one patient developed a right common femoral artery dissection requiring open repair and one patient required a limb extension of the right iliac limb due to risk of developing a type 1b endoleak. At the 12-month follow-up, two patients required embolization of type 2 endoleaks and no patients demonstrated type 1 or type 3 endoleaks.
Keywords
Introduction
The introduction of endovascular aneurysm repair (EVAR) over the last 2 decades has enabled patients with abdominal aortic aneurysms (AAA) to undergo aneurysm repair with a very favourable morbidity and mortality profile.1,2 Unfortunately, a number of patients are unsuitable for standard infrarenal endografting and must undergo open repair or an endograft with an adjunctive procedure such as fenestration or suprarenal fixation. Such patients are known to have higher perioperative risk and lower survival rates.3,4
In many cases, the main complicating factor for endografting is aortic neck morphology. The phenomenon of hostile neck anatomy (HNA) has been well described. Standard endografting requires a healthy 15 mm aortic neck, at less than 60° of angulation. Indeed many validated scoring systems exist to predict outcome. 5
We present our experience with a novel conformable aortic stent-graft designed to overcome some of the challenges of HNAs. The GORE Excluder Conformable AAA endoprosthesis (CLEVAR) (CEXC Device, W.L. Gore and Associates, Flagstaff, AZ, USA) is designed to conform to short and tortuous AAA neck anatomy. In this prospective evaluation study, we assess 3- and 12-month survival data and follow-up clinical and angiographic outcomes of CLEVAR implantations, including the first implant inserted in Scotland.
Methods
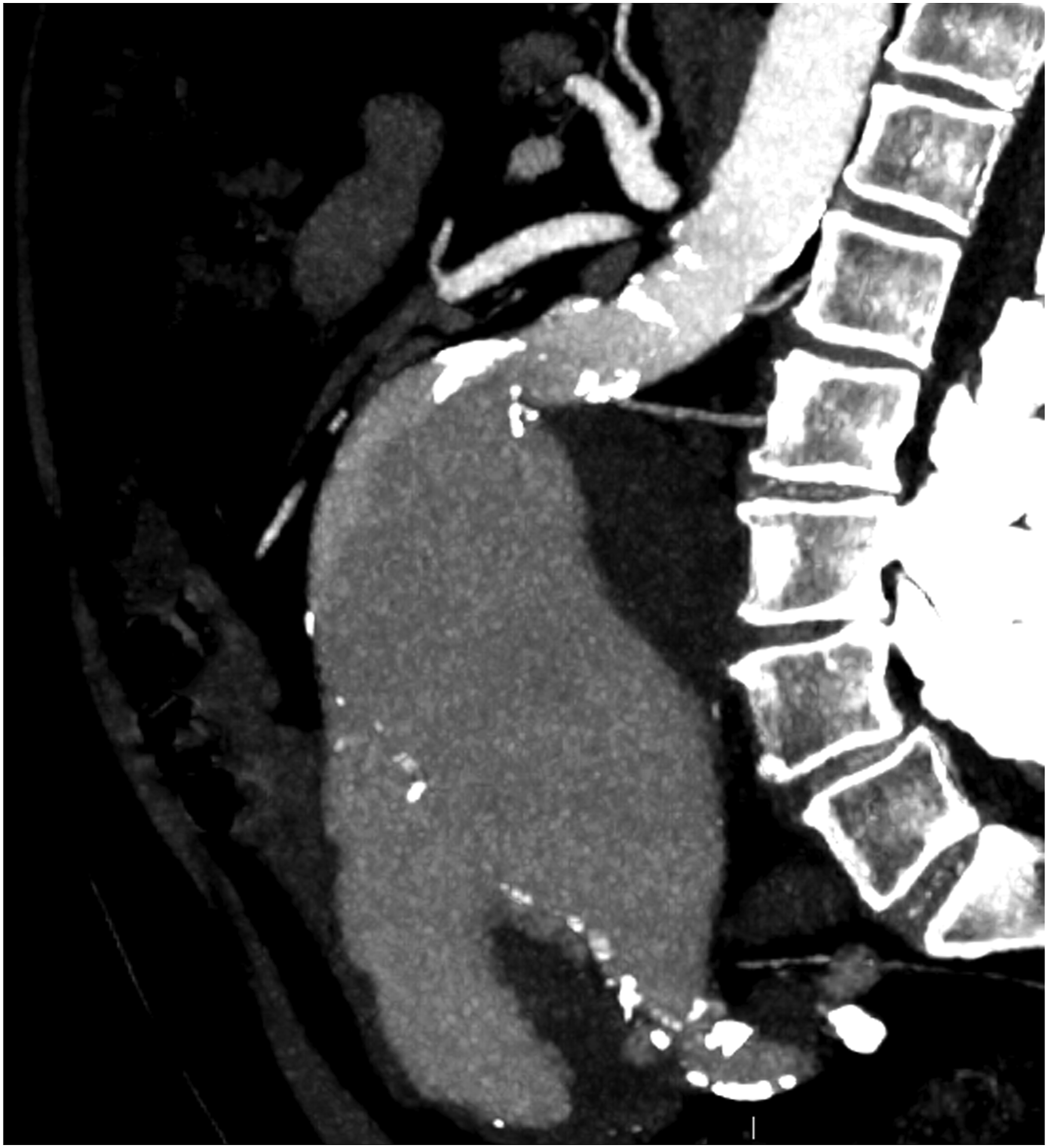
Twenty-four consecutive patients undergoing CLEVAR implantations from May 2018 to March 2022 at a single vascular centre (tertiary centre teaching hospital) were prospectively recruited. Hostile neck was defined as the presence of at least one of the following: neck length < 15 mm, or neck angulation > 60° as shown in Figures 1 and 2. Our primary outcomes were proximal zone seal, endoprosthesis migration, and type 1 endoleak assessed with fluoroscopy during deployment and with computed tomography (CT) angiography post-operatively at 3- and 12-month clinical follow-up. Our secondary outcomes included in-hospital and long-term aneurysm-related re-intervention, morbidity and mortality at 3- and 12-month follow-up. Baseline patient characteristics and comorbidities were also recorded. Pre-operative ultra-serpiginous iliac access vessels with highly angulated aortic neck. Pre-operative sagittal CT angiography demonstrating HNA.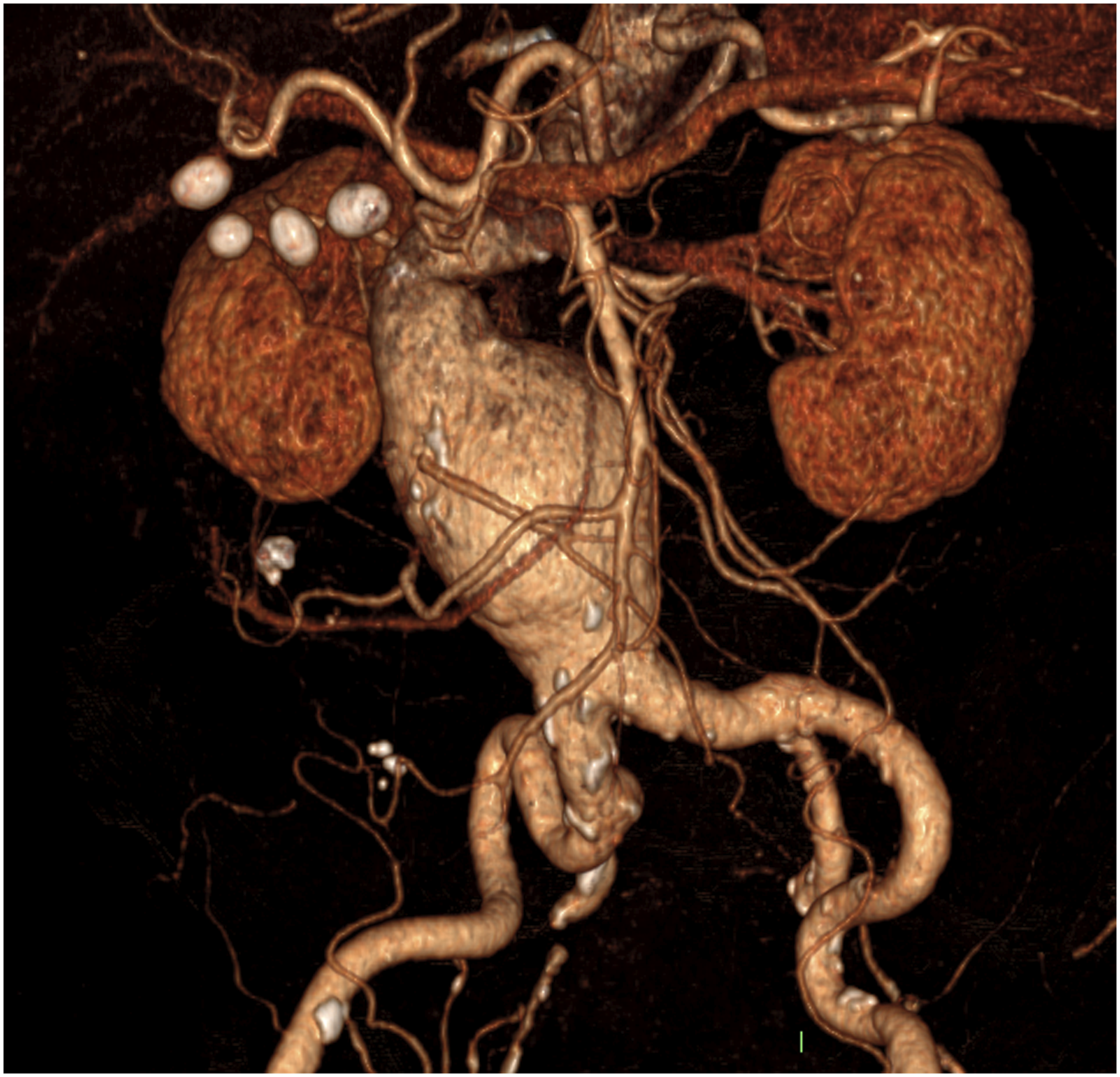
Inclusion criteria are as follows: patients aged > 55 years with non-ruptured infrarenal AAAs whose anatomy was deemed suitable for CLEVAR and warranting surgical intervention. All patients were discussed at vascular multidisciplinary meetings involving multiple consultant vascular surgeons deeming the patients unsuitable for conventional EVAR repair and at high risk for open repair. Patients with active infection, history of previous aortic surgeries, penetrating aortic ulcers, dissections, intramural haematomas or any clinically significant comorbidities which in the opinion of the treating vascular surgeon would contraindicate against a CLEVAR were excluded. This accounted for medical conditions reducing life expectancy to < 2 years (including but not limited to: end stage renal failure, severe heart failure [NYHA grade III/IV] metastatic malignancy, dementia and end stage COPD). Although not absolute contraindicating factors in themselves, frailty (i.e. history of numerous falls, mild cognitive impairment and frequent hospital admissions) and social and psychiatric factors predisposing the patient to poor follow-up were taken into account as exclusion criteria by the treating surgeon. Therefore, participants were also considered on an individual patient basis in the context of a novel therapy. Written informed patient consent was obtained for the publication of this study and the accompanying images.
Data availability statement
All data not published in this article will be made available to qualified investigators on request to the corresponding author.
Results
Demographics (n = 24).
Intraoperative characteristics (n = 24).

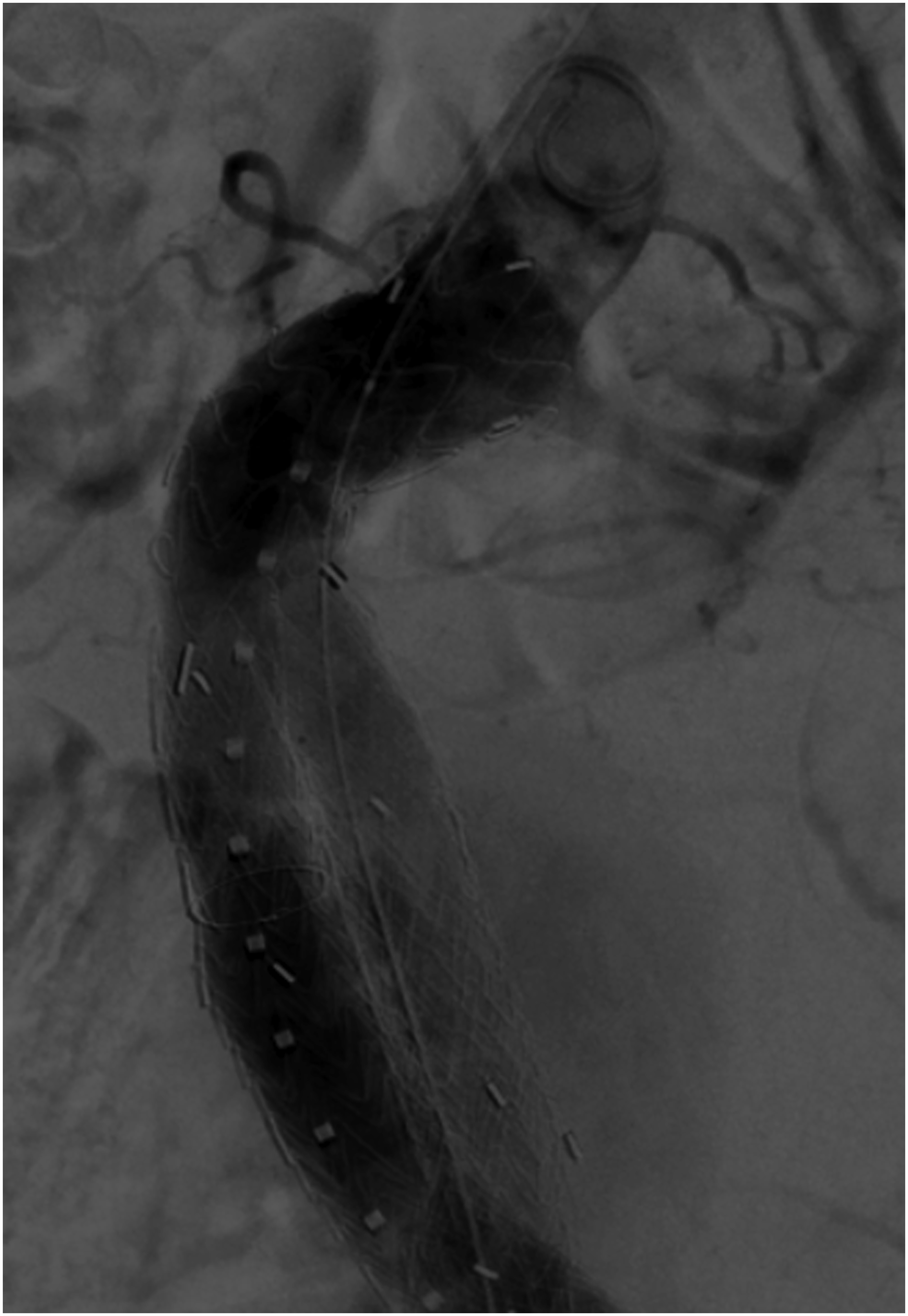
Intraoperative digital subtraction angiography of CLEVAR in-situ.
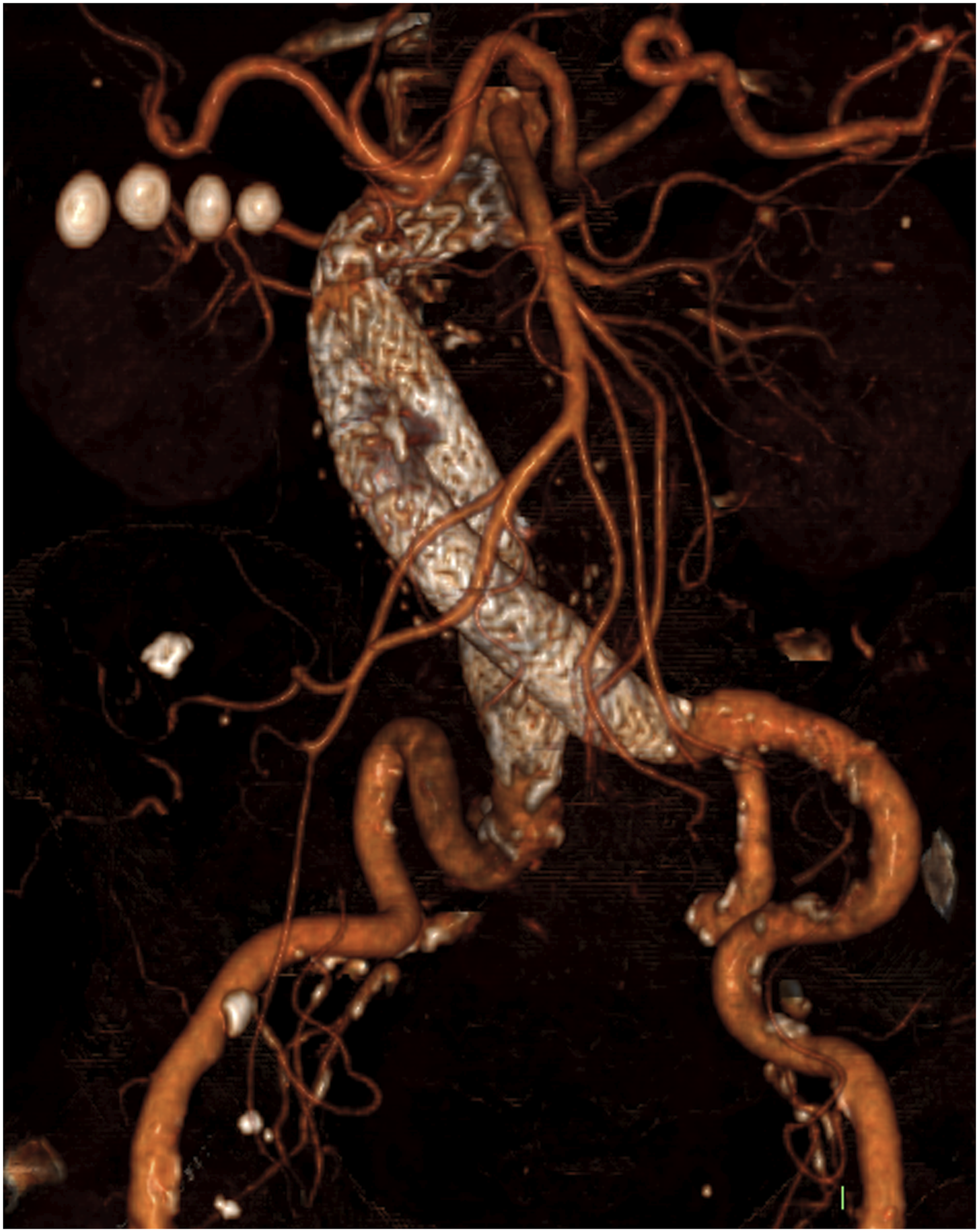
Volume rendering image of post-operative CT scan demonstrating endograft conforming to tortuous infrarenal neck and iliac access vessels.
Follow-up characteristics. Notes: CFA: common femoral artery.
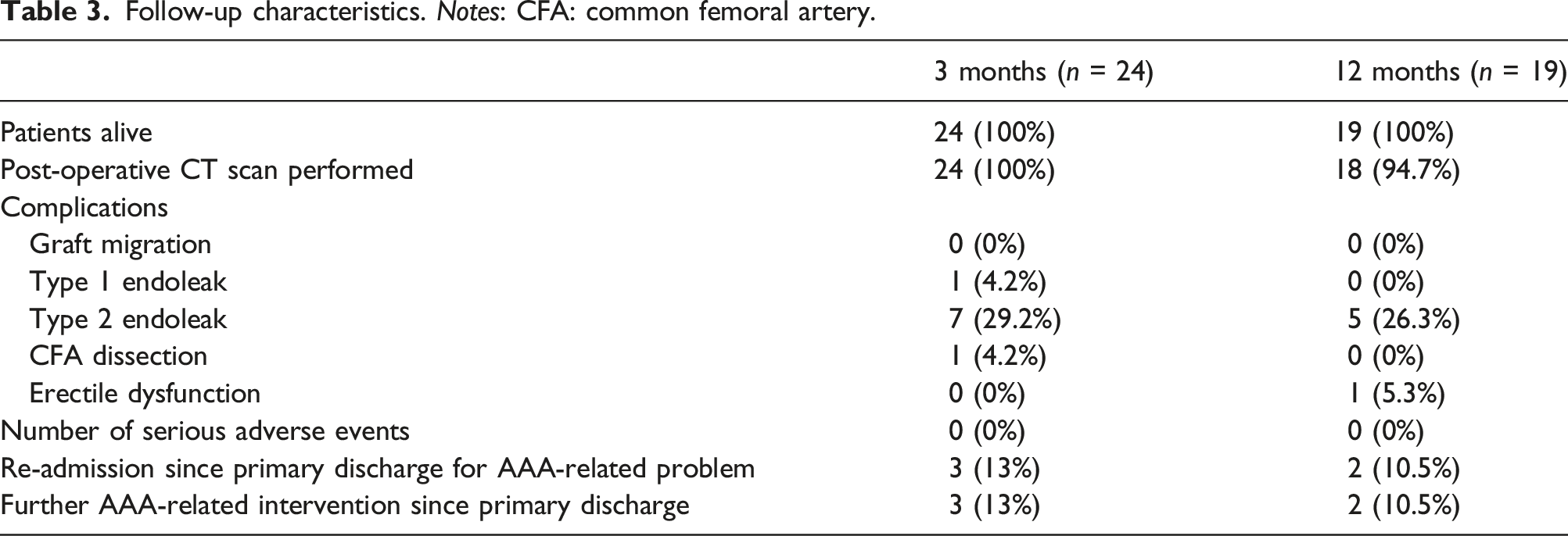
Nineteen patients underwent surgical intervention more than 12 months ago to be followed up at the 12-month clinical appointment with CT imaging performed. All 19 patients were alive. Five patients (26%) demonstrated type 2 endoleaks requiring no intervention and one patient (5%) developed erectile dysfunction. Two of these patients (11%) were readmitted and underwent successful embolization for type 2 endoleaks. There were no type 1 endoleaks in patients at the 12-month follow-up clinic, with all implants remaining in optimal position with good proximal zone seals and no significant distal migration (<2 mm since implantation).
Discussion
This prospective study demonstrates that GORE Excluder Conformable AAA endoprostheses (CLEVAR) are feasible in select patients, providing a treatment option for unruptured aneurysms with hostile neck anatomy (HNA). The re-constraining and angulation features of the device performed as intended, achieving 100% technical success in patients (n = 24) on deployment. Moreover, all patients were alive and clinically stable in the immediate post-operative period and at 3- and 12-month clinical follow-up. The low incidence of secondary intervention in this study further reinforces its feasibility.
Robust operative planning and careful patient inclusion, alongside a multidisciplinary team environment with specialist experience in managing AAAs has been key to favourable outcomes reported in our study. Although CLEVARs are yet to be adopted as standard treatments by regulatory bodies (British Society of Endovascular Therapy and NICE in the United Kingdom), the technique is still evolving and early data proves encouraging. Peer-reviewed case-series across multiple centres have also described the feasibility and favourable early outcomes of CLEVARs.6–8 The IDEAL framework sets out phases describing innovation for novel surgical interventions and this study fits within phase 2a of the framework. 9
Treating patients outside of instructions for use (IFU) is one of the commonest causes for graft dysfunction. Despite this, up to 58% of AAAs are treated outside of endograft IFU due to complex aortic neck morphology. 10 Current therapies provide suboptimal exclusion; pooled data demonstrate that HNAs treated with EVARs confer an increased risk of 30-day mortality and late type 1 endoleaks. 11 Moreover, meta-analyses evidence no statistically significant differences in outcomes between open repair, EVAR, and fenestrated EVAR in treating HNAs with respect to primary technical success rate, 30-day mortality and late AAA-related mortality. 12
Endoleaks are significant risks associated with HNAs. Infrarenal neck angles play a critical role in stent fixation as extreme curvature reduces seal zone length. The active angulation system with CLEVARs allowed pre-alignment of the proximal endograft segment, with individual stent rows allowing conformability with tortuous anatomy. With type 1 endoleak rates of 8.3% at 3 months and 0% at 12 months post-procedure, and no incidences of type 3 endoleaks, our data adds to the body of evidence that CLEVARs widen the indication for endovascular aortic repairs in treating HNAs.
Antoniou et al. reviewed 21 studies involving 102 patients with HNAs treated with chimney or snorkel EVAR grafts, reporting a pooled 91% technical success rate, 5% perioperative mortality rate and 13% early type 1 endoleak rate. 4 This systematic review concluded that these grafts provide a complementary technique in select high risk patients. Other interventions for HNAs aim to extend the proximal seal zone using branched or fenestrated grafts. In a review by Cross et al. involving 660 fenestrated procedures, target vessel perfusion rates ranged from 90.5% to 100%, 30-day mortality rate was 2% and type 1 endoleak rates at deployment, early post-operative and late post-operative periods were 2.9%, 1.2% and 2.6%, respectively. 13 Our immediate post-operative, 3- and 12-month follow-up data highlights the safety and efficacy of CLEVARs in context of existing therapies used outside their IFU.
Other studies have also reported endografts developed specifically for HNAs. Weale et al. studied the outcomes of 30 patients treated with the Aorfix endovascular graft (Lombard, Oxfordshire, UK), reporting intraoperative type 1 endoleak rates of 10% and a 30-day technical success rate of 96.7% with no incidences of 30-day type 1 or 2 endoleaks and no mortality. 14 A retrospective multi-centre study by Goncalves et al. involving 45 patients treated with the Endurant endograft (Medtronic, Minneapolis, Minnesota), reported a technical success rate of 100% during primary deployment, type 1 endoleak rates of 0% at completion angiography and 30-day post-operatively and early survival rates of 96.9%. 15 Our early clinical outcomes of CLEVAR perform well in context with other endovascular therapies designed for HNAs.
A further important factor to note regarding this study was the impact of Covid-19. As has been well documented, including in the COVER Study, mortality from vascular interventions during the first wave of the pandemic were unexpectedly high, be that due to the vascular population’s high rate of comorbidities or due to decisions to delay surgical intervention. 16 In this study, 88% of procedures were performed during the Covid-19 pandemic. With the option of performing CLEVAR in suitable populations, that is, those with HNA, it avoided the need of performing higher risk procedures such as open repair or FEVAR, and the potential delays associated with the logistics of device planning and manufacturing.
Limitations of this single-arm prospective study include selection and sampling bias. Patients were recruited by the consultant vascular surgeon who deemed the patient suitable for CLEVAR, although these biases were mitigated through consecutive recruitment of patients satisfying objective CT imaging HNA criteria. Moreover, all patients were discussed at multidisciplinary meetings involving multiple consultant vascular surgeons and radiologists. We did not compare CLEVARs with standard EVARs, open repair or other endovascular devices/adjuncts designed for HNAs. Further studies involving heterogeneous patient populations and larger sample sizes are warranted to investigate for the possibility of late complications. Head-to-head trials comparing CLEVARs, open repair and other endovascular devices/adjuncts designed for HNAs will also prove useful.
Conclusion
GORE Excluder Conformable AAA endoprosthesis demonstrates feasibility as a treatment option for clinically stable patients with unruptured AAAs demonstrating HNA. Further research involving heterogeneous and larger sample sizes is warranted to determine long-term clinical outcomes of CLEVAR.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: B.R is the BSET-CLEVAR trial local principal investigator for the North of Scotland. S.H.L, R.M, S.K, L.B and A.W report no conflicts of interest or disclaimers relevant to the manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
