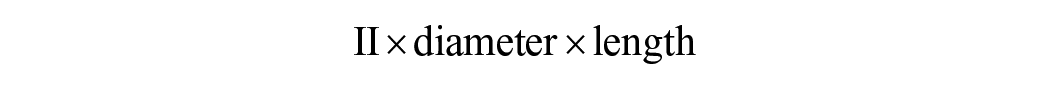Abstract
Objective:
Primarily to evaluate the area of neck coverage and secondarily, technical and clinical outcomes of the GORE® EXCLUDER® Conformable AAA Endoprosthesis (EXCC), designed to treat patients with significant angulation of the aortic neck, in routine clinical treatment of patients with abdominal aortic aneurysm (AAA).
Design:
A prospective, multicentre, observational cohort study.
Materials and Methods:
Patients aged >55 years with a nonruptured infrarenal AAA requiring treatment, were enrolled into the registry from June 2019 to December 2021. Baseline clinical, procedural, and follow-up data were collected pre-, peri-, and postoperatively at 3 and 12-months. CT images were assessed using a strict protocol at a core laboratory. The primary outcome was the estimated percentage of the total aortic neck that remained uncovered by the graft assessed on the first follow-up CT scan.
Results:
One hundred five patients with AAA underwent endovascular aneurysm repair (EVAR) with the EXCC device in 13 UK study centers (mean age 77.5 years, 14/105 female, 7/105 symptomatic). One hundred two participants with available CT scans were placed into group A-beta angulation <60° and aortic neck >10 mm (51/102); group B-beta angulation <90° and >60° and aortic neck >15 mm (37/102); and group C-those outside of instructions for use (14/102). The primary outcome of median percentage of neck that remained uncovered after EVAR was 5.1% [IQR 1.9–12.4]; 4.8% in group A, 6.0% in group B, and 3.8% in group C. Overall technical success was 96% (97.9%, 100%, and 84.6% in group A, B, and C, respectively). One-year overall and aneurysm related mortality was 9.1% and 0%. Four patients required re-intervention for proximal neck complications (including 2/14 patients in group C).
Conclusion:
The EXCC device can be accurately placed even in patients with significant angulation of the aortic neck. Clinical outcomes are acceptable to 1 year; however, physicians should exercise caution when treating patients where the graft is placed outside the instructions for use.
Clinical Impact
This prospective registry assessed the performance of a novel device with greater conformability, and angulation control of the delivery system to treat abdominal aortic aneurysm (AAA) with up to 90° neck angulation within instructions for use (IFU). In 105 patients, the primary outcome of estimated median percentage of uncovered aortic neck was 5.1% [IQR 1.9–12.4]. In patients with anatomy conforming to IFU, there was no difference in the percentage of neck that was uncovered in angulated (>60°) versus nonangulated patients (0–60°) and no early type 1a endoleak. This suggests excellent apposition of the graft to the aortic wall despite significant angulation of the aortic neck. Clinical outcomes were acceptable to 1 year.
Introduction
Aortic neck morphology is regarded as the primary anatomical feature determining the success of endovascular aneurysm repair (EVAR) in patients with abdominal aortic aneurysm (AAA). It has been known for many years that standard EVAR is not well suited to challenging aortic neck anatomy and, in patients with “hostile necks” leads to significant short- and long-term issues.1,2 Meta-analysis of over 11,000 patients 3 highlights that those with hostile necks require adjuncts more frequently and have early issues with migration and type 1 endoleak. Separately, and importantly, there are long-term issues, with endograft failure, necessitating further treatment. A recent study from the United Kingdom, the UK COMPlex AneurySm Study (UK-COMPASS) study, 4 specifically evaluated off label use of EVAR, compared to fenestrated and open repair in a cohort comparison with propensity stratification and adjustment. The late mortality and re-intervention rate were significantly greater after EVAR and FEVAR than open repair.
The angulated neck is a specific concern, as there may be issues with conformability of the proximal end of the graft leading to early type 1 endoleak, due to failure of accurate deployment. Equally, there may be late loss of seal zone secondary to neck tortuosity that increases the risk of long-term failure. There is a 10% excess of type 1 endoleak at a median follow-up of 7.4 years when angulated and nonangulated matched cohorts with a neck >15 mm are compared (freedom from type 1a endoleak, 86.1% vs 96.6%). 5
The effect of reduced neck coverage after EVAR appears to be an important determinant in longer-term failure of EVAR. In 32 patients with type 1a endoleak, matched with a population that did not, a clear relationship between short apposition length of the graft at the level of the sealing zone and subsequent endoleak can be demonstrated. 6
Recently, the GORE® EXCLUDER® Conformable AAA Endoprosthesis with ACTIVE CONTROL System (EXCC; W. L. Gore & Associates) has been developed for AAA patients with angulated neck, designed to be more effective in patients with an AAA with angulated necks. One may demonstrate endograft apposition at the aortic neck by examining CT angiograms after EVAR in a structured way.7,8
The CLEVAR Registry was set up to evaluate the device-specific performance outcomes of the EXCC device in routine clinical treatment of patients with AAA. The primary aim of the study was to evaluate aortic neck area coverage in patients with angulated and nonangulated AAA neck anatomy. Secondary aims were to evaluate technical success and clinical outcomes to 1 year.
Materials and Methods
Overview
The CLEVAR Registry is a prospective, multicentre, observational cohort study of patients with nonruptured AAA who have been treated with the EXCC device. This study was conducted in 13 centers in the United Kingdom. The study was registered in ClinicalTrials.gov (Identifier: NCT03966521). The study received Health Research Authority (HRA) approval and favorable ethical opinion by an independent National Research Ethics Committee (South East Scotland Research Ethics Committee 01, Ref: 19/SS/0067). This report follows the Strengthening the Reporting of Observational Studies in Epidemiology guidelines. 9
The GORE EXCLUDER Conformable AAA Endoprosthesis with ACTIVE CONTROL System device (Figure 1a and b) is more conformable at the proximal end with a longer body to mold to the angulated neck anatomy. There is modification of the delivery device to allow active control of the proximal end of the graft either in the collapsed or partially deployed form. The stiff wire is removed and replaced. The graft may then be angulated to run parallel to the centreline of the aorta at the level of the neck (Figure 2). The graft is repositionable. These modifications allow the operator to place the graft accurately in the angulated AAA neck, to ensure better apposition and coverage of the entire neck of the aneurysm, which may translate to better short- and long-term outcomes.
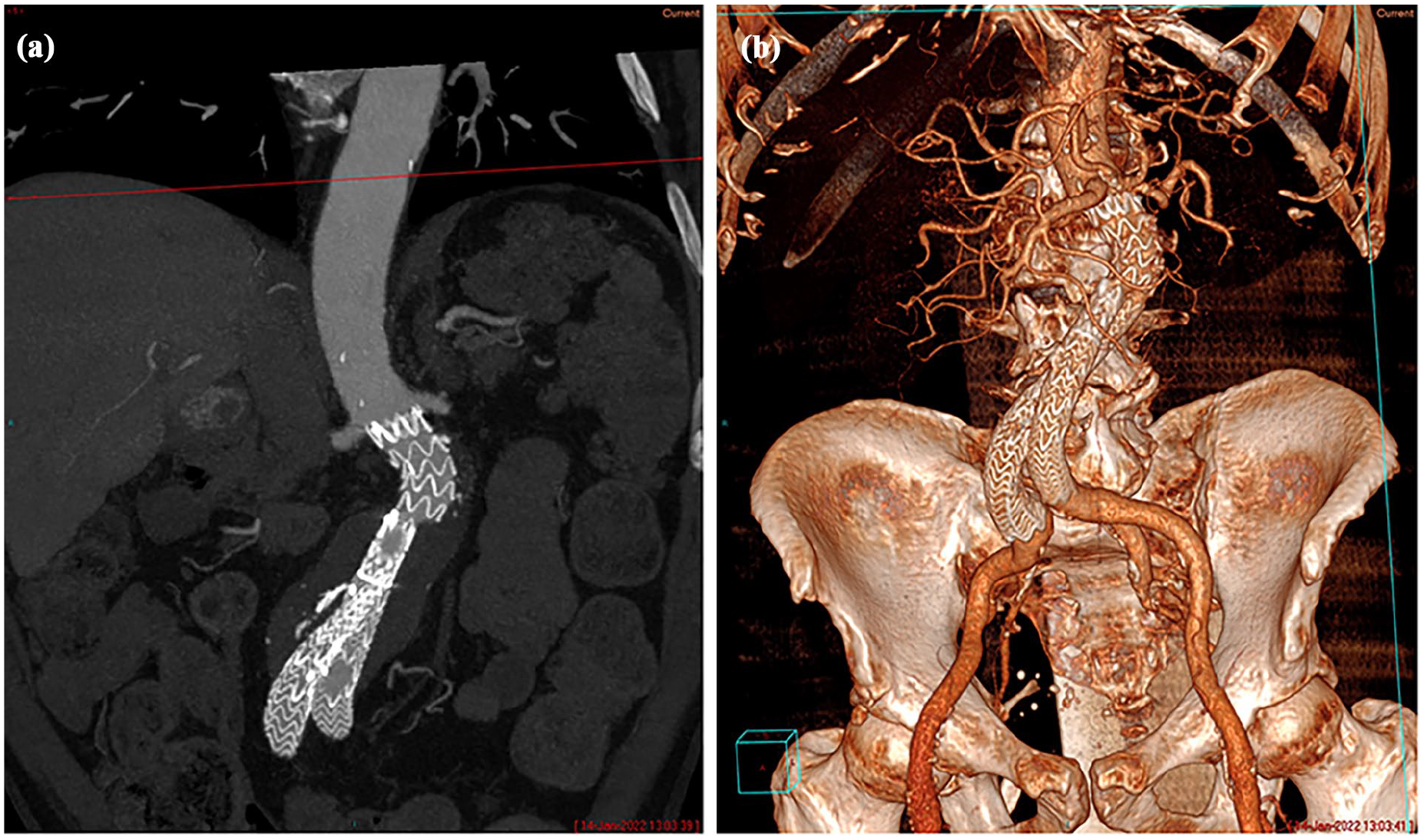
(a) A multiplanar and (b) a 3D reconstruction of the aorta of a patient with an abdominal aortic aneurysm, with an angulated neck, treated with the GORE EXCLUDER® Conformable AAA Endoprosthesis with ACTIVE CONTROL System (EXCC; W.L. Gore & Associates, DE, USA).
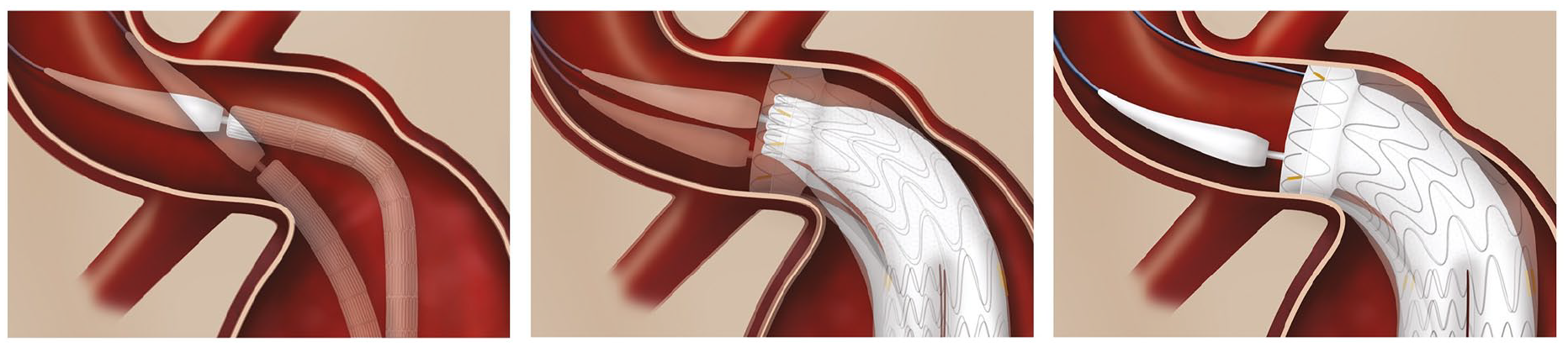
The GORE® EXCLUDER® Conformable AAA Endoprosthesis deployment stages.
Inclusion and Exclusion Criteria
Patients aged over 55 years with a nonruptured infra-renal AAA that required treatment, and, in the opinion of the treating vascular/interventional radiology physician at the study hospital, whose anatomy was adequate to receive the EXCC device, who could comply with protocol requirements, including follow-up, were eligible for inclusion.
Patients were excluded if they had a previous infrarenal aneurysm repair; been a participant in another aortic or thoracic medical device study within 1-year of study enrolment; if they had active infection; penetrating aortic ulceration, dissection, or intramural haematoma in the treated segment; or any clinically significant medical condition, which may interfere with the study follow-up requirements or reduce life expectancy to less than 2 years.
Enrolment
Between June 2019 and December 2021, 326 patients with AAA underwent EVAR in 13 centers. One hundred ninety-two patients did not meet the inclusion criteria: 182 were not included based on the decision of the local clinician (due to another stent-graft being more suitable to patient’s aortic anatomy or a co-morbidity/mental status making the patient unsuitable for enrolment), 5 had previous infra-renal aortic aneurysm repair, 2 had been treated in another thoracic medical device study within a year, 1 had an active infection, 1 had a penetrating aortic ulcer, and 1 was under 55 years old. A further 29 patients could not be enrolled in the study due to trained study personnel not being available to obtain consent during the COVID-19 pandemic (n=24) or the patient declining participation (n=5). Therefore, 105 patients were enrolled in the study. A flow diagram of enrolment is shown in Figure 3.
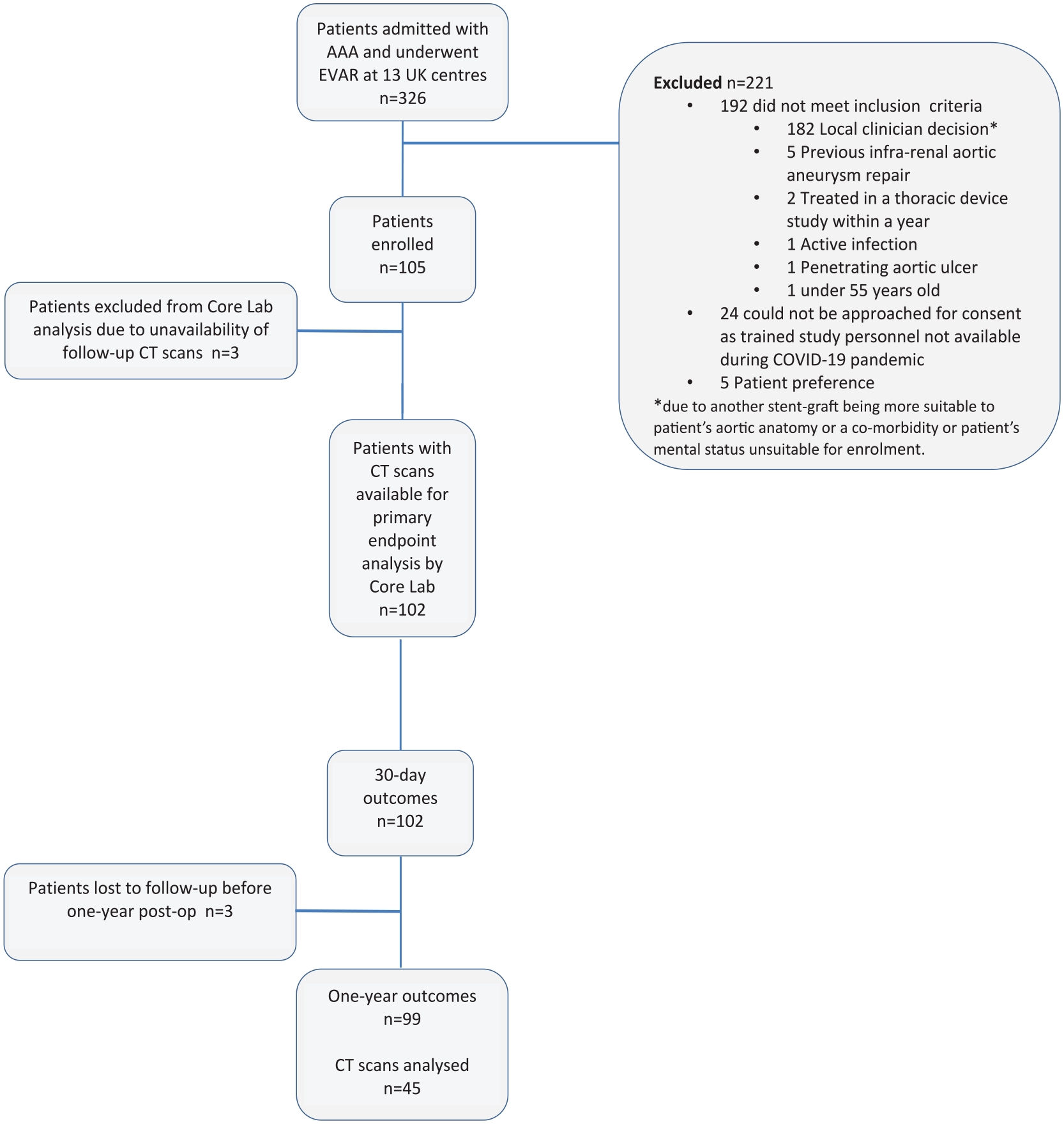
Enrolment of patients during the study period.
Data Collection
Suitable participants that met the inclusion criteria were approached and given information on the registry. After consent, demographics, medical history, and examination findings from the participant were recorded. During the perioperative period details of the operative procedure, adjunct procedures and clinical/technical success was recorded. Patients underwent follow-up according to each study center’s standard of care, with CT evaluation and follow-up within 3-months and further imaging (on ultrasound or CT scan), with clinical evaluation at 1 year. Details regarding serious adverse events and re-interventions were recorded. Anonymized data were transferred on electronic case report forms centrally for analysis.
Core Lab Analysis
Core Lab services for this study were provided the Department of Vascular Surgery, University of Liverpool, UK. Pre- and postoperative CT scans were transferred using a secure image transfer system. Pre- and postoperative CT evaluation was performed by a single trained investigator. A defined protocol was used to assess scans from each participant. The Corelab assessment of preoperative aneurysm neck morphology was undertaken using validated methodology from the UK COMPASS study. 10
The core lab measured: AAA diameter, neck length, supra-renal, and infrarenal angulation, neck diameter at 5 mm intervals along the neck, on preoperative scans. The length between the lowermost renal artery and the device (giving minimum and maximum measurements where the graft was not placed exactly parallel to the centreline of the aorta at the level of the neck), and the presence and type of endoleak was measured on postoperative scans.
The instructions for use (IFU) threshold for the EXCC device were defined according to the European definition as infrarenal aortic neck treatment diameter range of 16 to 32 mm, a minimum aortic neck length of 10 mm when proximal aortic neck angulation was ≤60° and a minimum aortic neck length of 15 mm when proximal aortic neck angulation was >60° and ≤90°. Subjects were divided into 3 groups by aortic morphology: Group A: Proximal aortic neck angulation ≤60° and minimum aortic neck length 10 mm; Group B: Proximal aortic neck angulation 61° to 90° and min. aortic neck length 15 mm; Group C-off IFU: Aortic neck angulation above 90° and/or less than required aortic neck length.
Analysis
The primary outcome was evaluation of the positioning accuracy and aortic neck coverage expressed as percentage of the total aortic neck that was uncovered by the graft assessed on the first follow-up CT scan.
To calculate the area of neck that remained uncovered after graft placement, the area of the neck was estimated using the generic formula for calculation of a cylinder:
The uncovered neck was calculated using the formula (Figure 4):
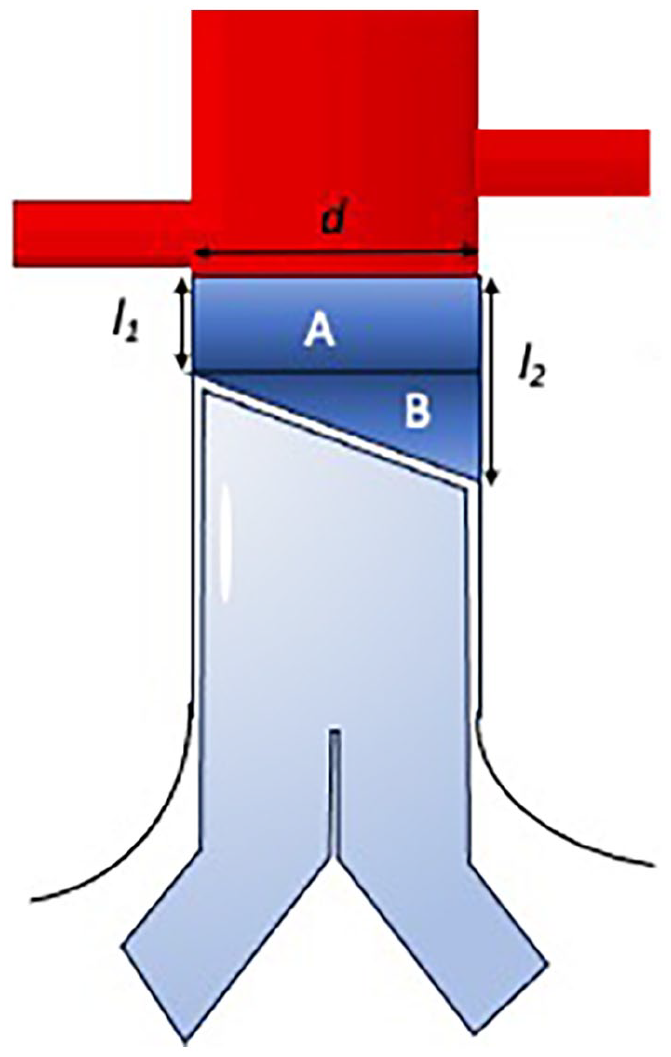
How the primary outcome was evaluated. To calculate the area of neck that remained uncovered after graft placement, the area of the neck was estimated by calculating the area of the cylinder A which is calculated using the formula
Secondary outcomes include the technical success (definition: no type I/type III endoleak and successful deployment), in-hospital mortality, adjuncts to treat type I endoleak at primary procedure, 12-month aneurysm-related re-intervention rates, and 12-month aneurysm-related mortality.
Small numbers in each subgroup precluded statistical comparison.
Results
Baseline Characteristics
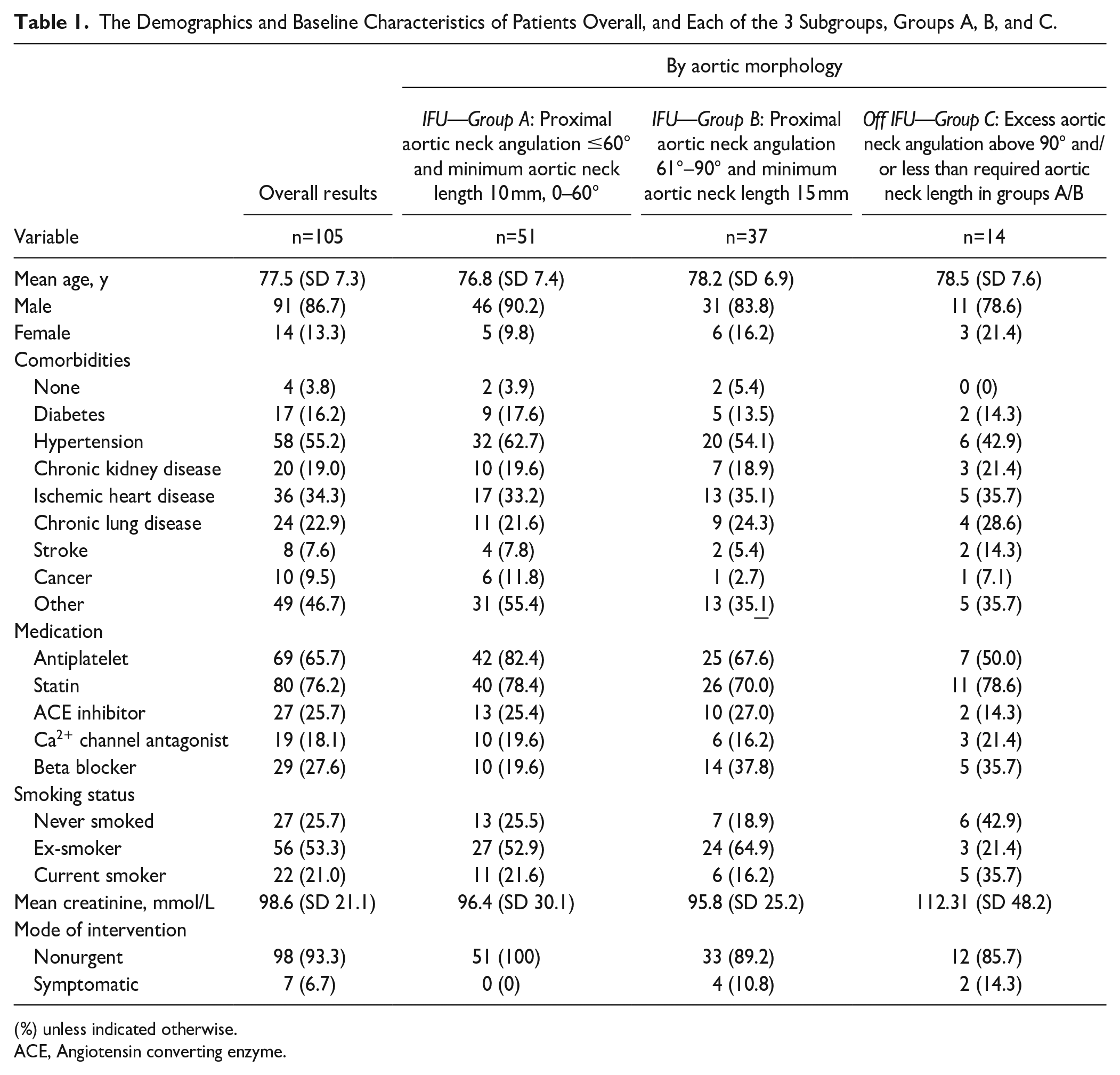
One hundred two of 105 CT scans were available for analysis by the core lab. Unfortunately, 3 CT scans could not be transferred by the end of the study deadline for analysis. The demographics of the participants in groups A, B, and C are shown separately. The vast majority of patients had significant co-morbidity, similar in groups A, B, and C.
Fifty-one patients had mild angulation of the neck and an aortic neck length of 10 mm or more. Fifty-one patients had necks that were angulated greater than 60°; 14 of these patients did not conform to IFU.
The demographics of the participants are shown in Table 1. Eleven of 14 female patients had angulated, shorter necks and were placed into groups B and C. Seven cases were urgent and symptomatic, all in groups B and C.
The Demographics and Baseline Characteristics of Patients Overall, and Each of the 3 Subgroups, Groups A, B, and C.
(%) unless indicated otherwise.
ACE, Angiotensin converting enzyme.
Procedural Outcomes
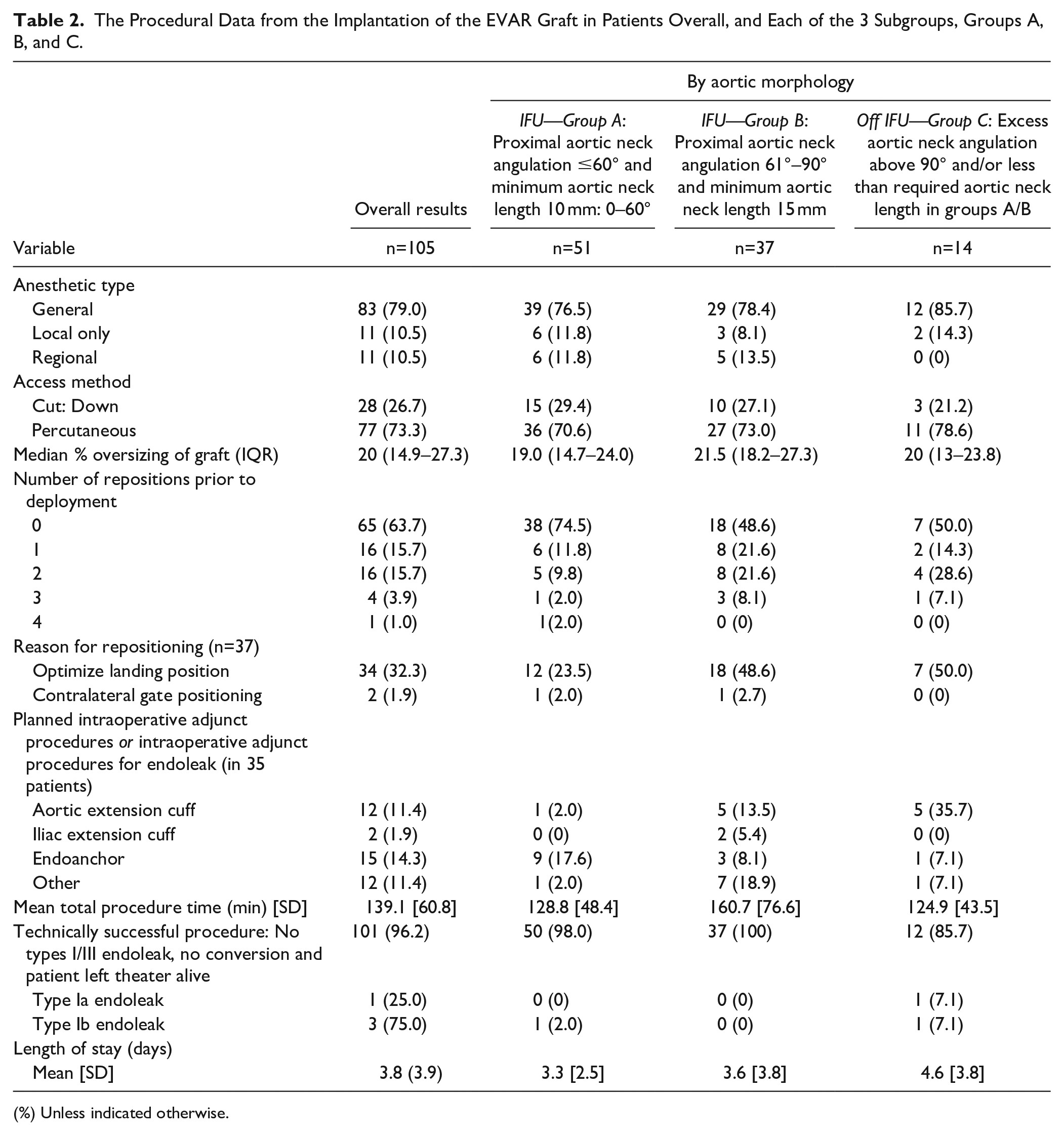
The procedural and in-hospital data are listed in Table 2. In this cohort, 37 patients (36.3%) had the graft re-positioned one or more times after placement with constriction of the proximal sealing ring, largely to optimize the landing position. Patients in group A had the graft repositioned 25.5% of the time. That is group B had repositioning 51.4% of the time, and those patients where the graft was placed outside IFU in group C had repositioning in 50% of procedures.
The Procedural Data from the Implantation of the EVAR Graft in Patients Overall, and Each of the 3 Subgroups, Groups A, B, and C.
(%) Unless indicated otherwise.
Thirty-five patients received adjunct procedures during the placement of the graft. This includes planned and unplanned adjuncts including aortic and iliac extension cuffs as well as endoanchors. A total of 35.7% of patients where the graft was placed outside IFU had an aortic cuff added.
A total of 101/105 cases were judged as technically successful. Four cases that were technically unsuccessful were secondary to 1 type 1a endoleak and 3 type 1b endoleaks on completion imaging. Two out of three patients, including the patient with a type 1a endoleak, who had CT scans suitable for core lab analysis, and technically unsuccessful procedures were in group C.
Aortic Morphology Assessed by the Core Lab
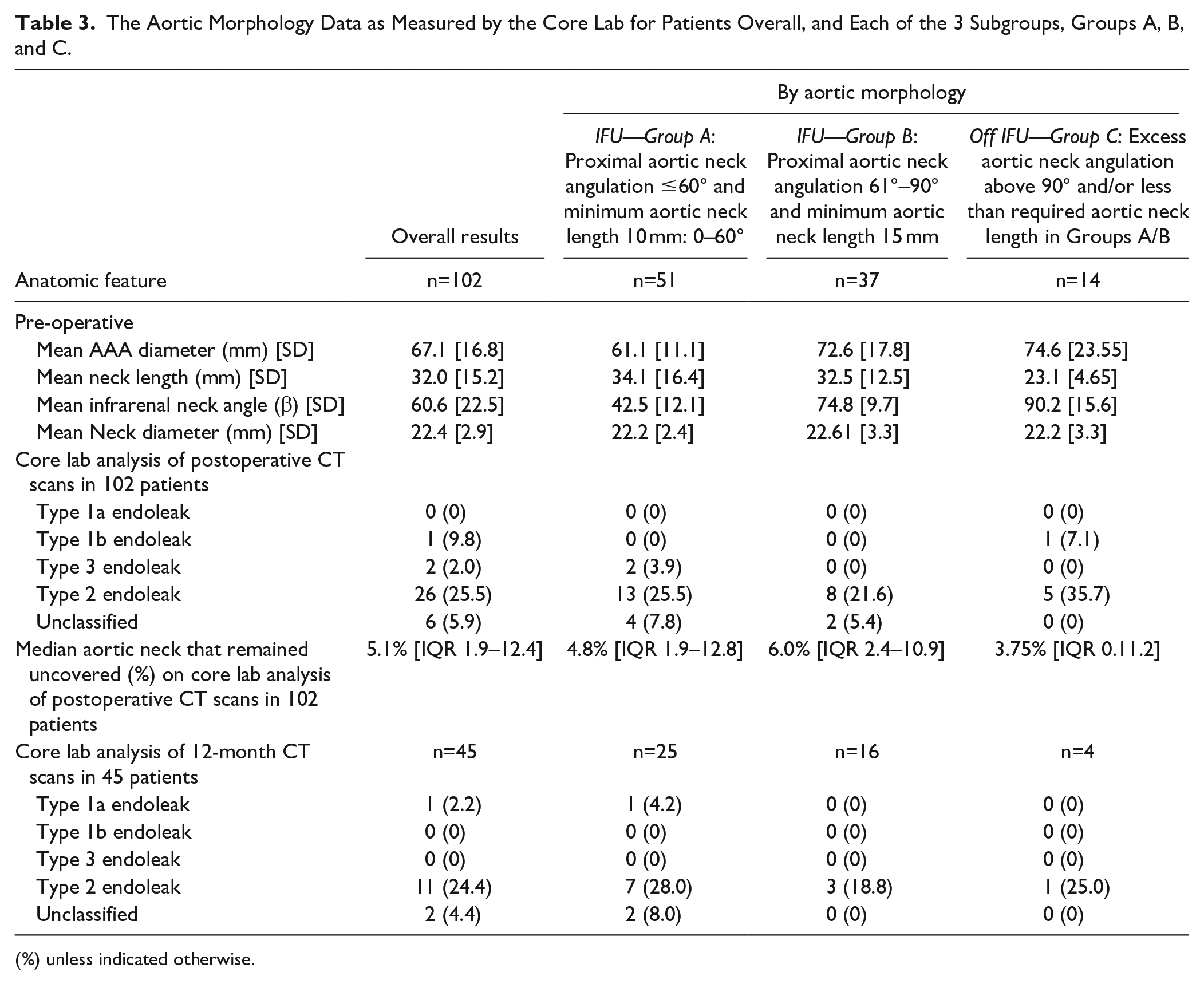
Table 3 shows the aortic morphology data assessed by the core laboratory. Each of the groups had broadly similar AAA characteristics, but the AAA size was larger, and the aortic neck was shorter in those patients where the graft was placed outside IFU.
The Aortic Morphology Data as Measured by the Core Lab for Patients Overall, and Each of the 3 Subgroups, Groups A, B, and C.
(%) unless indicated otherwise.
The primary outcome was measured in 102 of the 105 patients in the study. The median percentage of neck that remained uncovered after EVAR with the EXCC device and was 5.1% [IQR 1.9–12.4]. The median percentage of neck that remained uncovered was 4.8% [IQR 1.9–12.8] in group A; 6.0% [IQR 2.4–10.9] in group B; and 3.8% [IQR 0–11.2] in group C.
Early Outcomes
Core lab analysis of early CT scans took place in 102 patients. No type 1a endoleaks were detected. There was 1 type 1b endoleak, 2 type 3 endoleaks, and 26 type 2 endoleaks. A further 6 endoleaks were detected which were not able to be definitively classified according to the core lab protocols.
The 30-day clinical results are shown in Table 4. Thirty-day mortality was 0%. Eighteen patients had 1 or more complications. There was not an excess of complications in patients in groups B and C; however, 1 patient in group C required conversion to open repair. The site reported that the graft was placed appropriately, immediately below the level of the renal arteries and on the first CT scan at or around 30-days, the graft was found to have migrated proximally. This patient was readmitted for graft explantation due to a proximal position of the graft fabric, at the level of the superior mesenteric artery, compromising the renal arteries. Explantation resulted in renal ischemia on 1 side. The patient remains alive at 12-months and is not requiring renal support.
30-Day Outcomes for the Cohort, in Patients Overall, and Each of the 3 Subgroups, Groups A, B, and C.
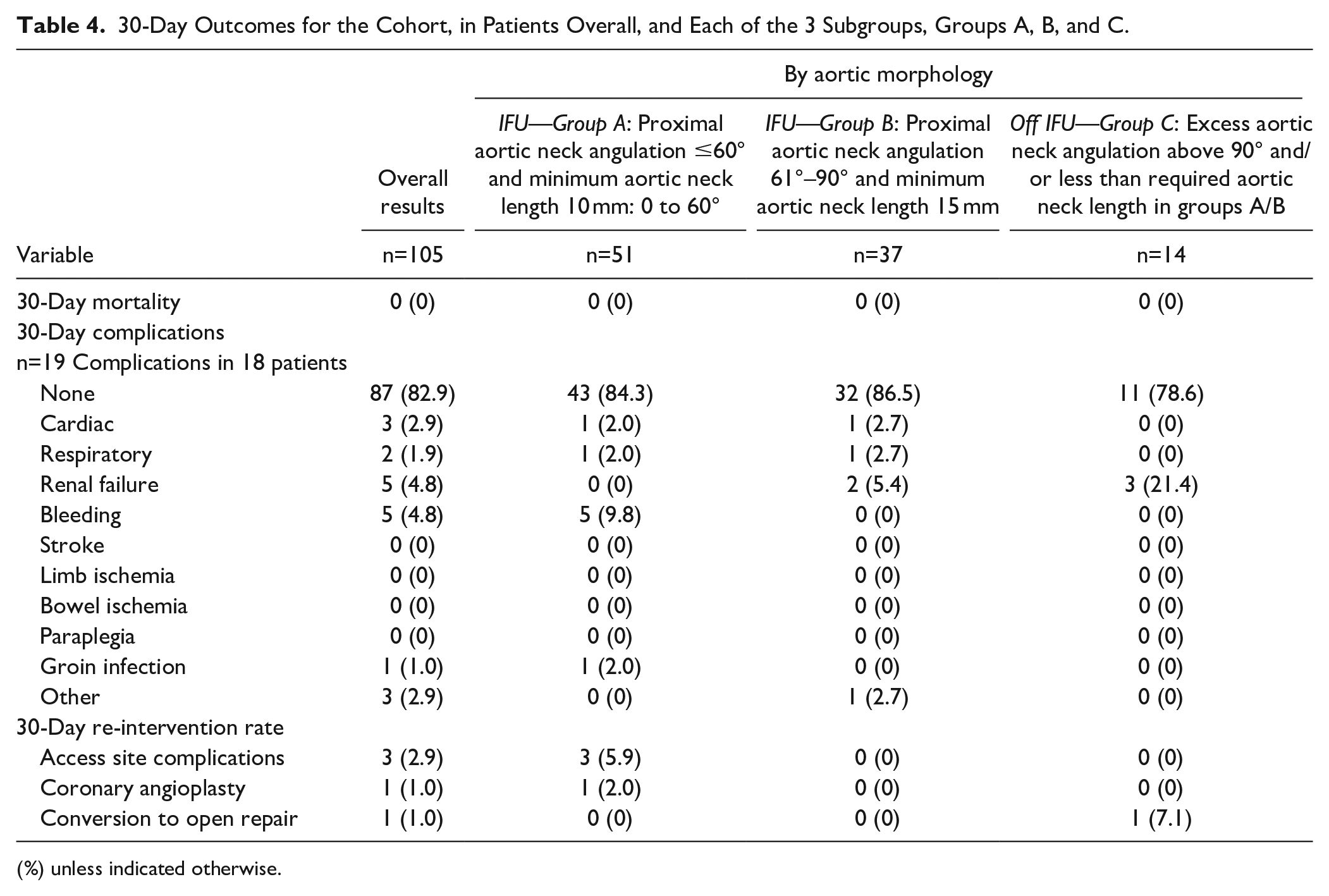
(%) unless indicated otherwise.
One-Year Outcomes
Twelve-month follow-up data were available for 99 (94.3%) of patients. Between 30 days and 12 months (Table 5), there was no recorded aortic-related mortality. Seven patients in the cohort died from causes unrelated to the aneurysm or aneurysm repair, and 2 patients died out of hospital, from unknown cause. Three of the patients that died (23.1% of this group) were in group C.
The 30-Day to 1-Year Outcomes in Patients Overall, and Each of the 3 Subgroups, Groups A, B, and C.
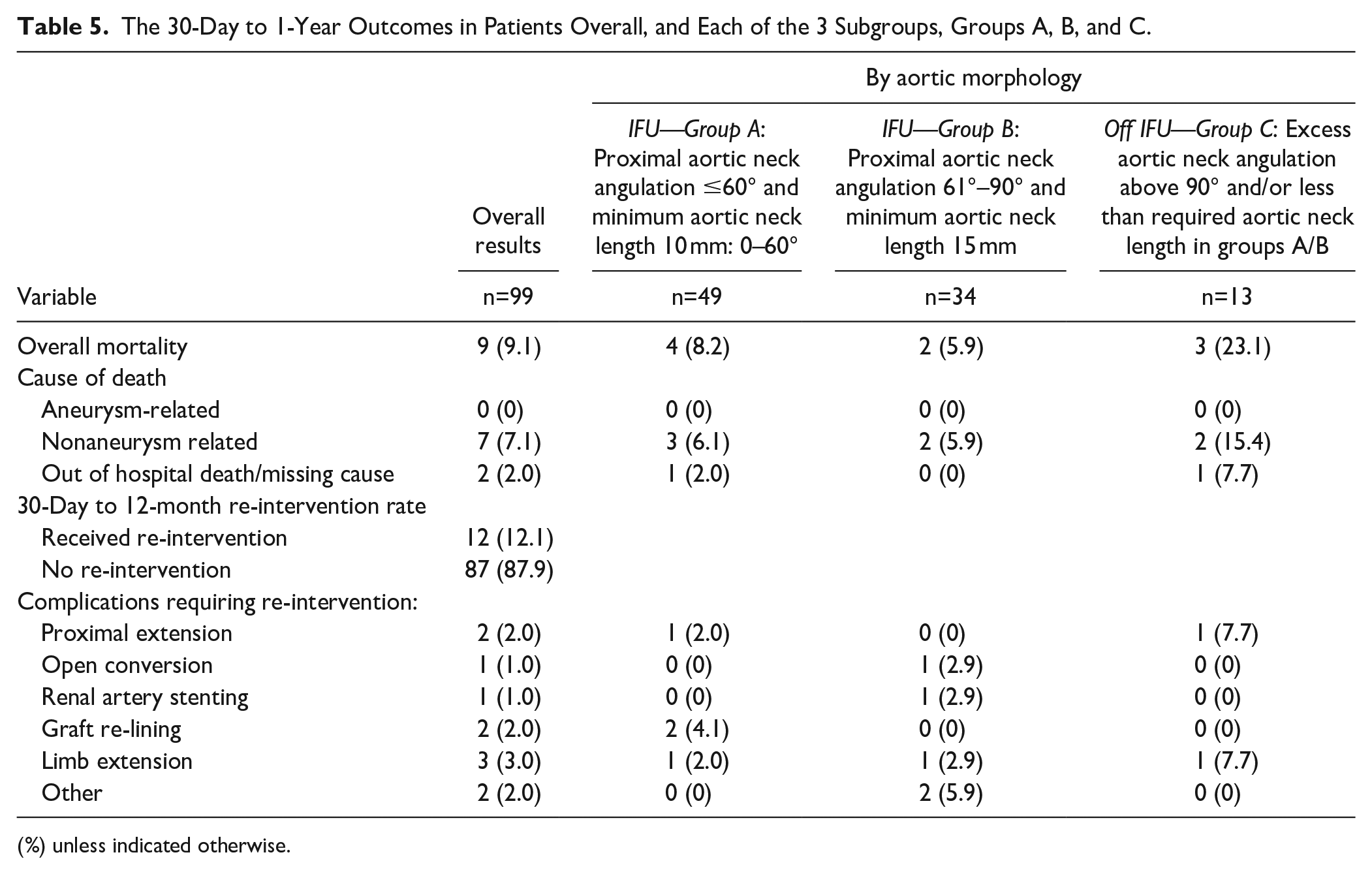
(%) unless indicated otherwise.
Eleven patients underwent re-intervention, including 2 cases of proximal extension. One proximal extension case was in group C and was a technical failure noted at the time of endograft placement. The other proximal extension was a chimney graft placement for rupture secondary to type 1a endoleak.
There was 1 open conversion, following rupture thought secondary to type 2 endoleak for a patient in group B.
Other re-interventions included 2 cases of stent relining for the cases of type 3 endoleak, 3 limb extensions for type 1 b endoleak (one of which required subsequent reconstruction of the common femoral artery and external iliac artery stenting), a case of thrombectomy for iliac limb thrombosis, psoas abscess drainage with stent graft infection, and 1 case of renal artery stenting as the graft was shuttering the renal artery origin.
Core lab analysis of late CT scans took place in 45 patients (as the majority underwent ultrasound evaluation). One further type 1a endoleak was found in a patient in group A with significant neck dilatation. There were 11 type 2 endoleaks and 2 unclassified endoleaks detected at this time point. There was no migration of the proximal portion of the graft greater than 5 mm in any of the 45 patients. No limb occlusions were noted.
Discussion
This study describes the imaging and clinical outcomes of patients with infrarenal AAA treated with the EXCC device in routine clinical treatment of AAA. The estimated aortic neck area coverage was chosen as the primary outcome as a clinical end point with a small sample size and short-term follow-up was not considered adequate to evaluate whether there is adequate, ongoing seal and fixation at the level of the neck of the aneurysm.
The estimated aortic neck area that remained uncovered was 5.1% overall. The percentage coverage of the neck that remained uncovered did not differ between groups, implying that the graft that is conformable and is able to be deflected to allow accurate placement, is effective with the grafts placed close to the renal artery origin.
The calculated graft apposition area appears to be significantly greater than in previous publications. In one study by Schuurmann et al, where apposition of the endograft was calculated from CT images, the endograft apposition area was approximately 75%. 8 In our article, we have estimated the aortic neck coverage from core lab measurements of average neck diameter, neck length, and distances from the graft to renal arteries. In other publications, the estimation of aortic neck coverage is calculated in different ways, with different assumptions.
The authors tentatively propose that the excellent utilization of the neck of patients in our study may well translate into improved 1-year outcomes. This is based on the fact that a shorter coverage of the aortic neck appears to predispose to a greater rate of type 1a endoleak. 6
The recently published European guidelines for treatment of AAA 11 warn against treatment of patients outside of IFU for the device. The number of patients that could be considered for this device is increased due to the fact that the graft is suitable for patients with AAA and a neck of up to 90° angulation. In these patients, outcomes were acceptable for a real-world cohort of patients, with challenging anatomy.
In group C, there was a greater need for repositioning and adjunct procedures and a lower technical success rate. These patients also suffered major complications requiring explantation and proximal extension for type 1a endoleak. The number of patients in this group is small, but the results cause some concern.
Specifically, regarding the greater need for adjunct procedures, this includes the addition of an aortic cuff. Although we presume this was necessary to treat in some cases and planned in other cases, it does seem to have been effective in covering the aortic neck shown by the absence of difference in the estimated area of uncovered neck between all 3 groups.
It is interesting to note that this group also had larger AAA maximum diameter size perhaps indicating that the operator chose a higher threshold for AAA repair in patients with difficult anatomy who were unfit for open surgery.
There are previous studies describing the early outcomes of patients with AAAs with angulated necks treated with the EXCC device. Rhee et al published the 1-year results of the investigational device exemption clinical trial at 31 US sites in 80 patients. 12 There was 100% technical success and no complications, aneurysm-related deaths or type I or III endoleaks. However, in this study, the neck angle was less than 60° with a mean of 35.7 (SD±4.55) degrees and a mean neck length of 23.9 (SD ± 13.07) mm.
A single-center series published by Mascoli et al 13 documented the results in 25 patients all with severe infrarenal neck angulation (beta angle ≥60°). Eleven (44%) of those had neck beta angle ≥90°. This series demonstrated 100% technical success rate with the need for only 1 aortic cuff to be placed. There were no type I endoleaks, and there was no aortic-related mortality. Approximately 10% of patients in our series of patients had a proximal aortic cuff placed. This is higher than in the series by Mascoli, but this may have been planned in many cases as those patients with a double bend in the aorta may benefit from graft placement to accommodate the first angulated segment and then an extension cuff around the second angled segment to sit just under the renal artery origins.
Finotello et al present an elegant study of patients. 14 In this series of patients demonstrating the initial clinical experience in 12 patients was favorable. Like our study, they examined the coverage of the aortic neck in patients with a median of 75° beta angulation and showed that the postoperative median apposition surface coverage was 79% of the available apposition surface. This is a slightly lower value than our estimation, but the method of evaluation was not the same. This publication used an automated method to calculate coverage. The assumptions made in the calculations differ from those in our article. This article also demonstrated that the CT evaluated neck curvature pre- and postoperatively does not appreciably change, and so, the authors concluded the conformability of the graft was adequate for the angulated neck aorta.
In another series by Zuidema et al, 15 treating 46 patients again the infrarenal and suprarenal aortic curvature did not significantly change during follow-up. There were 2 cases of migration in this series but no type I endoleaks at a median of 10 months.
Our series has a number of advantages in that it studies an unselected group of patients from 13 centers in the United Kingdom. The patients are representative of the typical patients seen in vascular clinics, and it is likely that the results of this study are generalizable to real-world practice.
The limitations are that this is an observational registry study and so suffers from the biases of all similar types of study, including selection bias in the enrolment stage. We were able to provide short-term outcomes for patients in this study, which are important as they demonstrated a higher rate of complications in those outside IFU, but assessment at 12-months is too short to make a robust assessment of the graft effectiveness and longer-term studies are of course needed. In addition, the numbers of patients in each of our 3 groups defined by anatomical boundaries are small and statistical analysis is not valid. Therefore, conclusions from the article regarding the subgroup analysis must be guarded. A further consideration is that in cases with a long proximal neck, there is an argument for not covering the entire aortic neck. However, it is true that aneurysms with angulated necks treated with EVAR do have an excess rate of type 1a endoleak irrespective of neck length and, therefore, it is reasonable to assume that best practice would be to cover the entire neck. In addition, the patients undergoing additional adjunct operative procedures include those that were planned before the procedure. Unfortunately, given the data collected, it was impossible to divide into planned and unplanned procedures. Many patients had planned endoanchors placed from one particular site. In addition, some had an aortic cuff placed which was planned to deal with a double “bend” in the aortic neck. In retrospect, given the different strategies employed by physicians in using this graft, we would advocate that further studies collect details on planning in the future. Lastly, our neck angulation is calculated from robust core lab data but does assume a cylindrical shape to the aortic neck and therefore, may differ from other estimations of aortic neck surface area coverage.
Conclusion
The CLEVAR Registry has provided real-world data for the use of the EXCC device up to 12 months, in a group of patients who presented electively and urgently, with significant angulation of the neck, demonstrating the estimated median percentage of neck that remained uncovered was approximately 5%. In patients with anatomy conforming to IFU, there was no difference in the percentage of neck that was covered in angulated and nonangulated patients and no type 1a endoleak at the end of the procedure, implying that the graft can be accurately placed even in patients with significant angulation of the aortic neck.
Clinical outcomes were acceptable to 1 year, with no aortic-related mortality; however, physicians should use caution when treating patients where the graft was placed outside the IFU.
Footnotes
Authors’ Note
BSET-CLEVAR Registry Investigators were responsible for recruitment, endograft implantation, data entry and management of the study at each site: Investigators include the following regional principal investigators and co-ordinators in order of site start date from earliest to most recent (numbers in parentheses indicate the number of patients entered into the study); and the members of the BSET council who have advised on the study: Members of the BSET council—Donald Adam, Ray Ashleigh, Paul Bachoo, Rachel Bell, Paul Bevis, Colin Bicknell, Dave Bosanquet, Patrick Chong, Martin Claridge, Patrick Coughlin, Nikesh Dattani, Shiva Dindyal, Murray Flett, Rick Gibbs, John Hardman, Seamus Harrison, Paul Hayes, Pete Holt, Mike Jenkins, David Lewis, Katharine Lewis, James McCaslin, Bijan Modarai, Paul Moxey, Hosaam Nasr, Simon Neequaye, Ian Nordon, Becky Sandford, Athanasios Saratzis, Nadeem Shaida, Kaji Sritharan, Chris Twine, Rao Vallabhaneni, Andy Wigham, Rob Williams, Emma Wilton, Hany Zayed. Imperial College Healthcare NHS Trust (20)—Colin Bicknell, Esther Negbenose, Jose Maria “Lloyd” Nunag, Lawrence Tarusan, Tuong Vi Le-Magowan, Amanda Henry, Rebeca Toledano, Natasha Grewal. Frimley Health NHS Foundation Trust (6)—Patrick Chong and Andrea Croucher. Guy’s and St Thomas’ NHS Foundation Trust (5)—Michael Dialynas, Lily Benton, and Eleanor Davis. Leeds Teaching Hospitals NHS Trust (3)—Rosie Darwood and Joanne Fletcher. Hull & East Yorkshire Hospitals NHS Trust (6)—Ian Chetter, Anna Firth, Josie Hatfield, Louise Mclean, and Tracey Roe. Bedford Hospital (15)—Arindam Chaudhuri, Gail Curran, and Rachel Lorusso. Gloucestershire Hospitals NHS Foundation Trust (8)—Sachin Kulkarni and Sarah Smith. NHS Grampian (19)—Bryce Renwick and Stacey Kerr. University Hospitals Dorset NHS Foundation Trust (8)—Sara Baker and Luke Vamplew. Cambridge University Hospitals NHS Foundation Trust (3)—Jon Boyle and Rico Boyce. Liverpool University Hospitals NHS Foundation Trust (5)—Robert Fisher, Simon Neequaye, Sophie Holder, and Lois O’Brien. Norfolk and Norwich University Hospitals NHS Foundation Trust (4)—Michael Delbridge and Mandy Burrows. NHS Tayside (3)—Murray Flett and Damian Smith.
A complete list of the BSET-CLEVAR Registry Investigators is provided in the Authors’ Note.
Declaration of Conflicting Interests
The author(s) disclosed the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Colin Bicknell has a consultancy agreement with GORE Medical and Medtronic outside of the scope of the submitted work and has received grant funding from GORE Medical and Medtronic for further unrelated studies. Jonathan Boyle was the Local PI for the GREAT Registry, funded by Gore. The British Society of Endovascular Therapy receives sponsorship for the annual meeting from GORE Medical and other entities. All other authors declare no potential conflict of interest with regards the submitted work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The British Society of Endovascular Therapy (BSET) initiated this prospective registry through its Council Members, which consists of Vascular Surgeons, and received funds from endograft manufacturer (W.L. Gore & Associates) to carry out the study. The Study Coordination Centre was based at Imperial College London (the Sponsor). BSET and the Sponsor had full responsibility for the conduct of this independent registry. W.L. Gore & Associates did not participate in design, analysis, or writing of manuscript.