Abstract
Objectives
Periodontal disease and tooth loss were found to be associated with several peripheral vascular disorders. Nonetheless, an evaluation of the literature on the broader domains of oral health in individuals with peripheral vascular disorders is lacking. This systematic review aims to collate the current evidence on the oral health status of individuals with peripheral vascular disorders.
Methods
Five electronic databases were searched for studies assessing oral health parameters in individuals with peripheral vascular disorders. Outcome measures considered were periodontal health, dentition status, caries indices, oral prostheses, oral pathologies and oral hygiene behaviours. The Newcastle-Ottawa scale was used to appraise the quality of the studies.
Results
From 3025 records identified, 24 studies involving 1232 participants with peripheral vascular disorders were included in this review. In nine studies, periodontitis was significantly more prevalent in peripheral vascular disorders compared to non-peripheral vascular disorders participants. A further six studies reported individuals with peripheral vascular disorders also had significantly fewer teeth and increased rates of edentulism. Only one study reported a higher incidence of dental caries in peripheral vascular disorders participants. Other aspects of oral health such as oral prosthesis, oral pathology and oral hygiene behaviours were seldom assessed.
Conclusions
The scarcity of studies reporting on broader domains limited our ability to arrive at a conclusion regarding the oral health status of individuals with peripheral vascular disorders. Future studies ought to assess these domains in individuals with peripheral vascular disorders and controls to gain a more complete understanding of oral health and its potential association with peripheral vascular disorders.
Introduction
Emerging evidence illustrates an association between certain oral health conditions, such as periodontal disease and tooth loss, and several peripheral vascular disorders (PVD).1–6 The International Statistical Classification of Diseases and Related Health Problems (ICD) defines PVD as an umbrella term for any disorder affecting blood flow in arteries or veins outside the heart. 7 A recent meta-analysis 8 reported a significantly higher risk of periodontitis and tooth loss amongst individuals with peripheral arterial disease (PAD), a type of PVD, than healthy controls. Their review only considered periodontitis and no other essential aspects of oral health as per the World Health Organisation’s (WHO) manual for standardised oral health assessment. These include dentition status, dental erosions, oral mucosal lesions, dental caries, dentition and prosthesis status and oral hygiene behaviours. 9 More specifically, dental caries and poor oral hygiene had been investigated as markers of early initiation of atherosclerosis and were found to be associated with increased carotid intima–media wall thickness. 10 , 11 Poor oral hygiene also increases the risk of periodontitis 12 and can lead to increased abundance of potentially pathogenic bacteria colonising the teeth. 13 These bacteria may exacerbate atherogenesis via the oral infection–inflammation pathway. 2
Several measures have been taken by vascular surgeons regarding their patients’ dental health, including dental antibiotic prophylaxis to prevent vascular graft infections following dental sepsis. 14 , 15 However, the oral health status of individuals with PVD remains incompletely assessed clinically. This is due to a lack of consensus regarding the need for oral health assessment in this group and whether this would warrant an improvement in outcomes of PVD. The aim of this study was to review the literature relating to the oral health status of individuals diagnosed with PVD. Further, where studies are available with controls (individuals without PVD), a comparison of the oral health status between these two groups was undertaken.
Methods
Search strategy
We used the Preferred Reporting Items for Systematic Review and Meta-analysis guidelines (see supplemental material for the checklist) for this systematic review. The Population Intervention/Exposure Comparator Outcome criteria were used to form the review question – What are the oral health findings (O) in individuals with PVD (P) whom have undergone oral health assessment (I) compared to individuals without PVD (C). Electronic database and hand searches for articles in April 2020 were conducted in the following databases: MEDLINE, SCOPUS, EMBASE, Web of Science and Cochrane Central Register of Controlled Trials. The search strategy was formed using the Medical Subject Headings and relevant free-text terms and was applied to each database (Supplementary Table 1).
Inclusion and exclusion criteria
Inclusion criteria: Adults aged ≥18 years Individuals with any vascular disease of arteries and veins outside the heart as grouped under the PVD diagnosis-related groups by the ICD
16
Assessed any of the following oral health measures: dentition status, remaining/missing teeth, prevalence of dental disease including periodontal indices, dental caries indices, oral infections, oral pathology, presence/absence of oral prosthesis and oral hygiene behaviours Published in English
Exclusion criteria: Systematic or literature reviews Case reports Studies with duplicate/overlapping cohorts.
Data extraction and quality assessment
Study characteristics and data on participants’ oral health were extracted from each included study and compiled on data extraction tables (Table 1 and Supplementary Tables 2 and 3). Titles and abstracts of all studies were independently screened by two reviewers (SAA and MM). Full texts of the selected studies were critically reviewed based on the inclusion and exclusion criteria. Although there were no disagreements, an arbitrator (GC) was available for mediation. For quality assessment, both reviewers independently used the Newcastle–Ottawa Scale (NOS) for case–control studies 17 and a modified form for cross-sectional studies (Supplementary Table 4).
Characteristics of included studies.
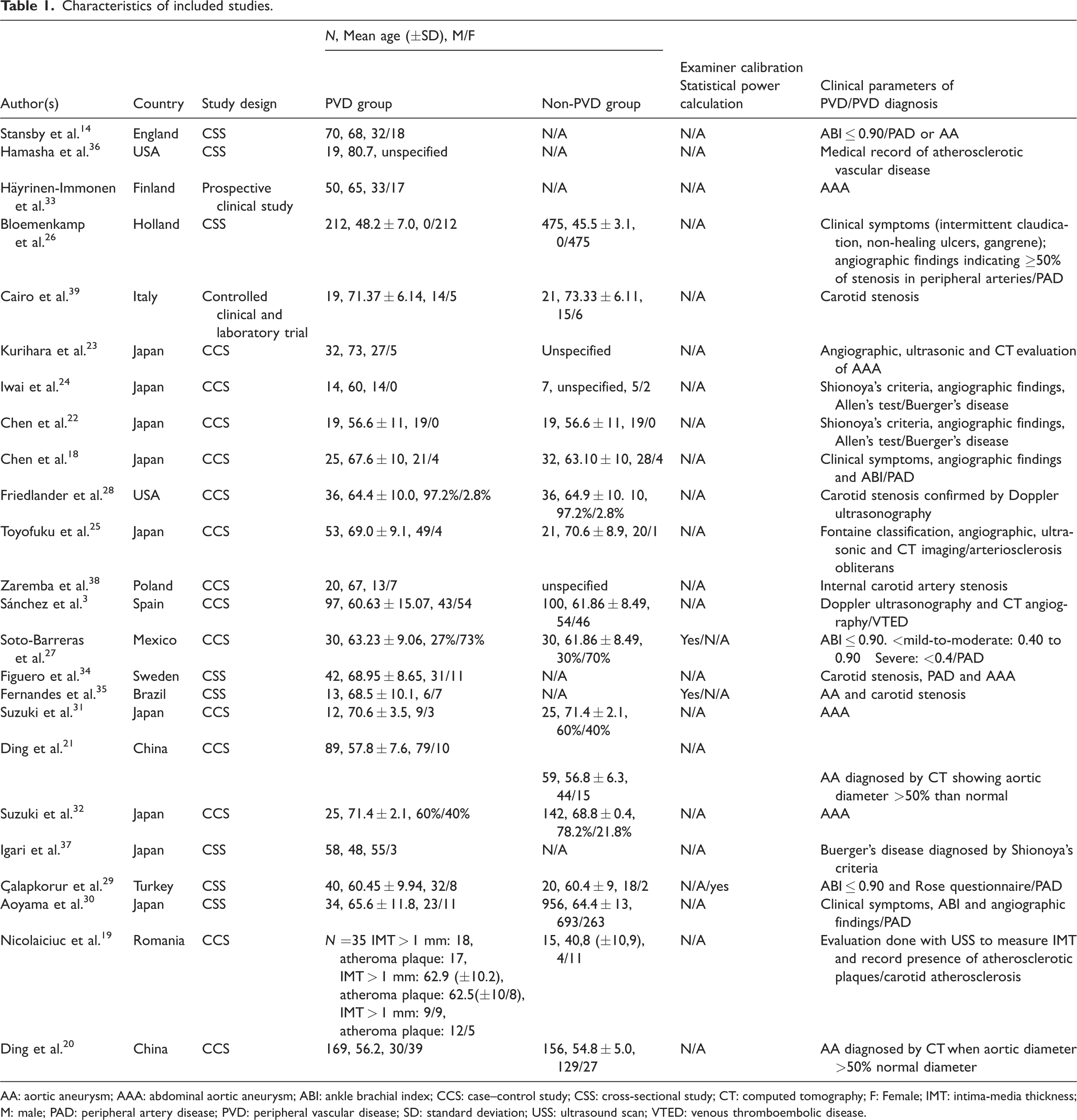
AA: aortic aneurysm; AAA: abdominal aortic aneurysm; ABI: ankle brachial index; CCS: case–control study; CSS: cross-sectional study; CT: computed tomography; F: Female; IMT: intima-media thickness; M: male; PAD: peripheral artery disease; PVD: peripheral vascular disease; SD: standard deviation; USS: ultrasound scan; VTED: venous thromboembolic disease.
Results
Search results
The search identified 3025 studies. RefWorks (ProQuest Refworks, 2020) was used to process the search results and to de-duplicate 80 studies. After application of inclusion and exclusion criteria, 58 studies were selected for full text screening, following which 24 studies comprising 1232 PVD participants were included in this systematic review (Figure 1).
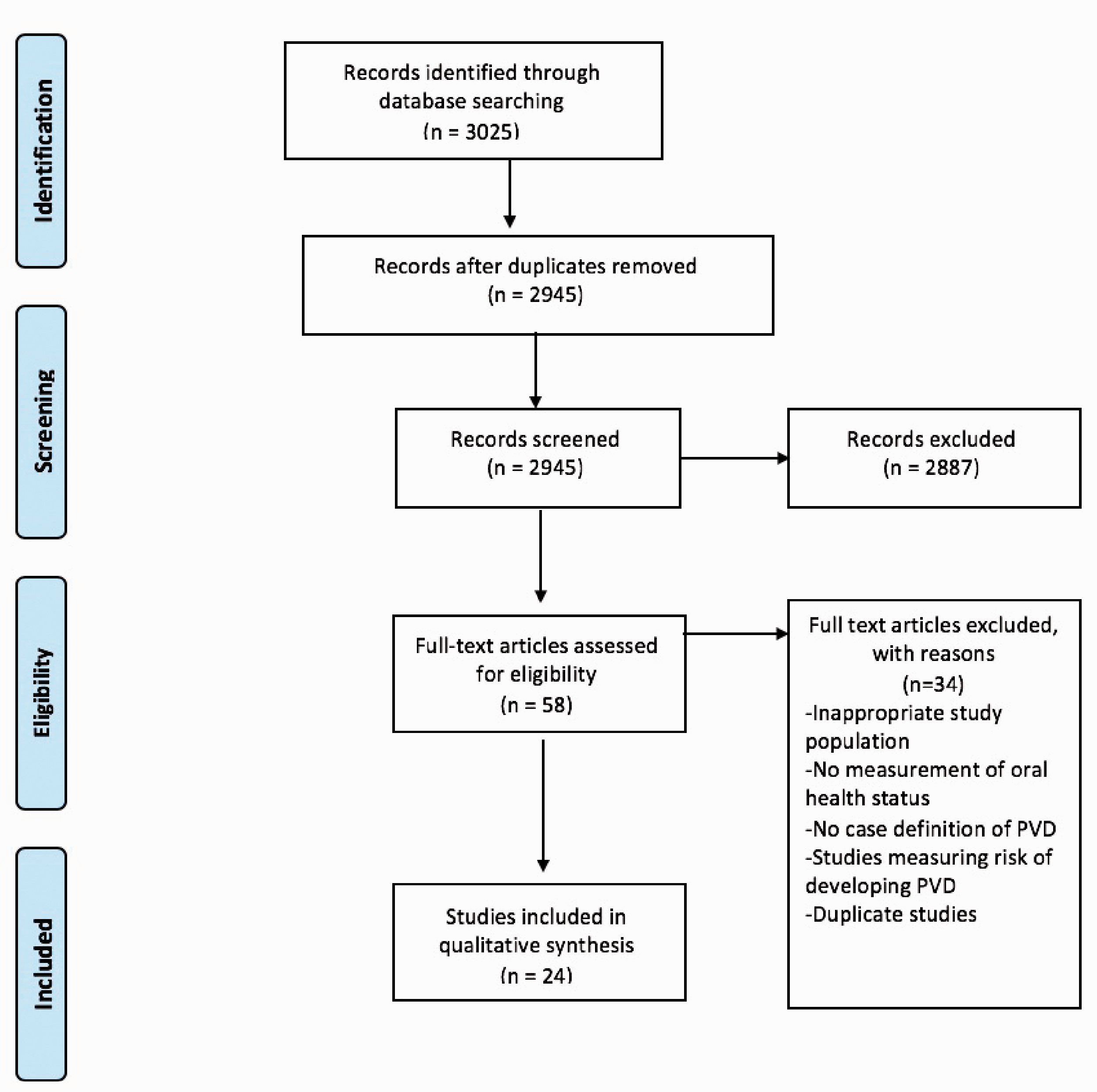
Preferred reporting items for systematic reviews and meta-analyses flow diagram of literature search and paper selection process.
Characteristics of studies
The characteristics of studies included in this systematic review are summarised in Table 1. These studies were published between 1994 and 2018 and were from 14 countries. Twenty-three studies were on arterial disorders, and one was on a venous disorder. Thirteen studies compared the oral health status between PVD participants and healthy controls, 3 ,18–29 whilst three compared the oral health of PVD participants to controls with cardiac diseases.30–32 Six studies 14 ,33–37 did not have controls. Another study included controls with cardiovascular disease; however, they did not measure oral health in the control group. 38 One study recruited edentulous PVD participants as controls; however, no oral health assessment was conducted in this group, therefore, a comparison was not done between cases and controls. 39
Examiner calibration and statistical power calculation
Of the 24 studies, three studies reported examiner calibration through intraclass correlation coefficient, 27 re-evaluation of random referred patients 29 and another through previously calibrated examiners using Kappa values (0.80–0.97). 35 Only one study provided details of statistical power calculation to determine sample size. 29
Case definition of PVD
The case definition of PVD varied amongst the studies (see Table 1). Seven studies reported a PVD diagnosis of case groups but with no description of the parameters used to diagnose PVD.31–35, 38 , 39 One study used previous medical records of vascular disease. 36 Four studies used clinical findings of PVD as diagnostic parameters such as ankle-brachial index, clinical symptoms and Rose questionnaire. 14 , 26 , 27 , 29 Stansby et al. 14 reported the parameters used for diagnosing PAD but had only reported a diagnosis of aortic aneurysm (AA). Six studies used imaging parameters only such as angiography, computed tomography and Doppler ultrasonography. 3 ,19–21, 23 , 28 Six studies used a combination of clinical findings and imaging parameters for diagnosis of PVD. 18 , 22 , 24 , 25 , 30 , 37
Oral health measures
Periodontal health
Periodontal health was assessed in 22 studies with results displayed in Supplementary Table 2. The case definition of periodontitis varied across each study (see Supplementary Table 2). Prevalence of periodontitis and moderate to severe periodontitis was reported to be significantly higher in participants with arteriosclerosis obliterans, 25 Buerger’s disease, 22 PAD, 18 , 26 , 27 carotid atherosclerosis, 19 AA 20 , 21 and venous thromboembolic disease (VTED) 3 compared to non-PVD participants. Two studies found no differences between groups. 28 , 29 In studies with no controls, a high percentage of the PVD participants had periodontitis 23 , 24 , 37 except for one study. 33 Gingivitis was more prevalent in non-PVD than in PVD participants in four studies. 20 , 21 , 26 , 29
Probing pocket depth (PPD) is a measurement of the distance from the gingival margin to the pocket base surrounding a tooth. This measurement is one of a range of clinical criteria used to diagnose and assess severity of periodontal diseases. 40 Aoyama et al. 30 and Çalapkorur et al. 29 found no difference between PVD and non-PVD participants in the mean PPD. Çalapkorur et al. 29 reported a significantly higher number of sites with PPD over 5 mm amongst PVD participants. Likewise, a significantly higher number of sites with PPD over 4 mm were seen in AA, 20 , 21 , 31 , 32 Buerger’s disease, 22 carotid atherosclerosis 19 and PAD participants 18 , 27 compared to non-PVD participants. It is important to note that Çalapkorur et al. 29 defined periodontitis as having at least five teeth with at least one site of PPD equal to or greater than 5 mm. This differed from the other studies which defined periodontitis as the presence of more than one site with PPD equal to or greater than 4 mm in each quadrant.
Clinical attachment loss (CAL) indicates the extent of periodontal tissue support loss around a tooth. A significantly higher percentage of sites with CAL greater than 4 mm was found amongst AA, 20 , 21 Buerger’s disease, 22 PAD 18 , 27 and VTED 3 participants compared to non-PVD participants. There was no significant difference in CAL greater than 4 mm between PVD and non-PVD participants in two studies. 29 , 30
Zaremba et al. 38 found 4 of 20 participants demonstrating presence of periodontal bacteria in their atherosclerotic plaques after carotid endarterectomy. These participants had significantly higher bleeding indices and PPD greater 4 mm compared to participants with no periodontal bacteria in the atherosclerotic plaque. Mean CAL was not significantly higher in the group with periodontal bacteria present in atherosclerotic plaques than those without.
Dentition status
Thirteen studies assessed the dentition status of PVD participants with results displayed in Supplementary Table 2. 3 , 14 , 18 , 20 , 23 , 28 ,30–36 Nine studies compared remaining or missing teeth in PVD to that of non-PVD participants. 3 , 18 , 20 , 28 ,30–32, 34 , 36 All but three 28 , 31 , 34 reported significantly less retained teeth, 3 , 18 , 30 , 32 more missing teeth 20 , 30 , 36 or higher edentulism rates 30 , 36 in PVD compared to non-PVD participants. Three studies reported lower percentages of PVD participants who were edentulous. 14 , 23 , 33 Fernandes et al. 35 found that 96.1% of teeth were missing amongst 13 PVD participants.
Dental caries
Five studies investigated caries in PVD participants with findings displayed in Supplementary Table 3.27–29,
33
,
35
The DMFT is the sum of the number of
Other oral diseases
Two studies commented on oral diseases other than caries and periodontal disease (see Supplementary Table 3). 28 , 33 Friedlander et al. 28 found no difference in the mean number of teeth with periapical lesions between PVD versus non-PVD participants. Immonen et al. 33 reported 80% of the PVD participants had an oral infection, with only 11% of them having ‘good oral health’. Candida infection was present in 17% of dentate and 47% of edentulous PVD participants. Fifty-four percent of dentate participants had intraosseous foci compared to 20% of edentulous participants. Periapical lesions and intraosseous foci are areas of localised chronic infection of dental origin that may influence chronic systemic diseases. 41
Dental prosthesis
Three studies assessed the presence of dental prosthesis in PVD participants (see Supplementary Table 3). Stansby et al. 14 reported 46% PVD participants had partial dentures. Another study reported that amongst a total of 56 dentures in 37 PVD participants, 45% of dentures were poor and had to be replaced. 33 A third study reported the mean number of implants but had no controls. 34
Oral hygiene behaviours
Only one study investigated oral hygiene behaviours. They used a self-reported assessment of oral hygiene behaviours amongst AA participants. 20 Compared to the non-AA group, more AA participants had inaccurate brushing methods, less brushing time and frequency, no flossing and less routine dental examinations (Supplementary Table 3).
Discussion
This review included broader oral health domains in the WHO’s manual to assess oral health in individuals. Participants with PVD were found to have compromised oral health across various measures such as poorer periodontal health, more missing teeth and a higher prevalence of edentulism in comparison to non-PVD participants. However, the findings on dental caries and presence of periapical lesions in PVD and non-PVD participants were conflicting. Although not reported in the studies, reasons for this may include differences in dental health-seeking behaviour in PVD participants, and in the diagnostic protocols in different countries. Other aspects such as oral prosthesis, oral hygiene behaviours and oral pathology were seldom assessed.
This review has also considered other subtypes of PVD such as PAD, carotid atherosclerosis, AA, Buerger’s disease and VTED. The assessment of oral health amongst individuals with a venous disorder was performed in only one of the included studies. 3 Association studies demonstrated a possible relationship with valvular incompetence in individuals with varicose veins, 42 the latter being a known risk factor for VTED. 43 This could be mediated by the role of certain bacteria in periodontal disease, which were found to be risk factors for vascular endothelial damage and pro-coagulation. 44 , 45 Despite Sánchez and colleagues’ 3 significant oral health findings in their cohort with a venous disorder, further studies are required to confirm the validity of these findings and to compare the oral health status between individuals with venous and arterial disorders. Two studies in Yang and colleagues’ 8 meta-analysis were excluded from our review, as they assessed the risk of developing PVD in participants with prior periodontitis. 1 , 46
Tooth loss in PVD participants
Several studies reported a significantly higher prevalence of tooth loss amongst PVD as opposed to non-PVD participants. 3 , 18 , 20 , 30 , 32 , 36 The putative mechanisms underpinning this finding could be varied. Firstly, tooth loss was shown to have a possible association with atherosclerosis, even with adjusting for shared risk factors including age, smoking, sex, diabetes mellitus and hypertension. 47 Secondly, an important mediator of incident tooth loss in PVD participants is antecedent periodontal disease. Periodontal infection via the oral infection–inflammation pathway may promote systemic inflammation and exacerbate atherogenesis. 2 Tooth loss in turn may lead to compromised masticatory ability, causing an altered diet which may predispose to PVD. 48 Finally, the attitude and approach to dental disease management amongst participants and their dental care providers in different countries may have resulted in increased tooth loss.
Dental prosthesis, other oral diseases and oral hygiene behaviours
The studies reporting on the presence of dental prostheses in PVD participants were non-controlled and descriptive in nature. 14 , 33 , 36 Therefore, no inference could be made on this oral health domain in PVD and non-PVD participants. The current literature is also insufficient to establish any difference between PVD and non-PVD participants regarding oral diseases such as Candida infections, periapical lesions and intraosseous foci. For a more complete assessment of these domains, further analytical studies are required.
Poor oral hygiene behaviours have been associated with an increased risk of cardiovascular events and elevated concentrations of inflammatory molecules such as C reactive protein. 49 Conversely, improvements in oral hygiene have been shown to reduce the risk of cardiovascular events. 50 Although an important determinant of oral health, oral hygiene behaviour was assessed in only one study. 20 That study only involved participants diagnosed with AA, who showed poor oral hygiene behaviours. Further analytical studies assessing oral hygiene behaviours in other PVD subsets are required.
Overall appraisal of included studies and recommendations for future research
Cross-sectional and case–control studies were the predominant study designs in this review. Twelve 14 , 19 , 21 ,23–25,33–35,37–39 studies did not adjust for shared risk factors that underpin both tooth loss and certain types of PVD such as age, diabetes mellitus, hypertension and smoking. Lack of adjustments in observational studies is a known limitation.
Key methodological limitations such as the lack of control groups, 14 ,33–35, 38 the use of controls with various cardiac30–32 or vascular 24 co-morbidities, and unmatched controls have undermined the comparability of results between PVD and non-PVD participants. Furthermore, high heterogeneity of outcome measures and case definitions precluded a quantitative analysis. The case definitions of PVD and the clinical parameters employed for diagnosis were varied between the studies included. Several studies stated the diagnosis of PVD and its subtypes.31–35, 38 , 39 They did not, however, describe the parameters used to make the diagnosis. Some studies used imaging modalities to diagnose PVD. 3 , 20 , 21 , 23 , 28 These studies would have further benefited from categorising different PVD severities according to imaging findings, as this may allow correlation with associated oral health findings. This was evident in one study in which greater severities of periodontitis were seen with increased intima–media thicknesses on imaging. 19 Other studies utilised a combination of clinical and imaging tools to diagnose PVD which would also allow correlation of PVD severity with the oral health findings. 18 , 22 , 24 , 25 , 30 , 37 However, none of these studies had performed such correlation, which could have established whether or not increasing PVD severities are associated with poorer oral health findings.
Similarly, differences in case definitions of periodontitis and value ranges for categorising different severities of periodontitis were observed. In addition, variations in the periodontal indices used to assess clinical parameters of periodontal health were apparent. Such variations may cause inaccuracies in the prevalence of periodontitis amongst PVD participants. In general, studies would benefit from using a standardised case definition of periodontitis as recommended by Eke et al. 51 which is aimed for use in population-based research. Standardisation of clinical assessment parameters is also essential in future studies. 52
Moreover, Bloemenkamp et al. 26 assessed periodontal disease using patient self-reporting. This method was shown to have acceptable validity for large-scale epidemiologic studies surveying periodontal disease. 53 Nevertheless, another review indicated that employing a combination of self-reporting alongside other clinical indicators, such as CAL and PPD, of periodontal disease may be most beneficial; 54 this approach was utilised in two studies. 20 , 21 Therefore, future studies utilising self-reporting methods amongst PVD participants may benefit by combining them with other parameters of assessing periodontal disease. One study performed periodontal examination one to two months postoperatively on PVD participants who had undergone vascular surgery. 34 There was no mention of whether these participants had received dental care during that period, when their oral health may have worsened.
The strengths of this review relate to the assessing a spectrum of essential oral health domains, other than periodontitis, in participants with various subtypes of PVD as per WHO guidance. The principle limitation was the inability to undertake a quantitative analysis due to high heterogeneity of the included studies. Additionally, only 4 of the 24 studies were considered of high quality (scores greater than six) according to the NOS scale. Further, only 3 of the 16 studies that included controls were graded as high quality. Therefore, the scarcity of high-quality studies compromises the ability to establish a clear description of the oral health status amongst PVD participants. Nor does it permit a meaningful comparison to be made between PVD and non-PVD participants. Several recommendations for future research on this topic are listed in Table 2 based on the format recommended by Brown et al. 55 These recommendations include but are not limited to:
Research recommendations (based on format from Brown et al., 2006). 55

PVD: peripheral vascular disorders.
Future studies to assess the magnitude of the relationship between PVD and oral health parameters other than periodontal disease and tooth loss
Using standardised case definitions for PVD and periodontitis
Using standardised parameters for PVD diagnosis and oral health assessment according to WHO guidance and customised from Oral Health Assessment and Review Dental Clinical Guidance of the Scottish Dental Clinical Effectiveness Programme. 56
Assessing the effect of oral health treatment on PVD.
Conclusion
Due to the paucity of the high-quality studies addressing oral health domains other than periodontal disease and tooth loss, a definitive conclusion regarding oral health status/conditions in individuals with PVD could not be made. Therefore, it is not yet possible to make an evidence-based recommendation regarding the value of routine oral health assessment in individuals with PVD. However, on considering the evidence regarding the link between oral and systemic health, it would be good practice to advise on oral health assessment and maintenance in individuals with PVD.
Supplemental Material
sj-pdf-1-vas-10.1177_1708538120963914 - Supplemental material for The oral health status of patients with peripheral vascular disorders: A systematic review
Supplemental material, sj-pdf-1-vas-10.1177_1708538120963914 for The oral health status of patients with peripheral vascular disorders: A systematic review by Sayed Abdulmotaleb Almoosawy, Mhairi McGowan Karolin Hijazi, Rona Patey, Paul Bachoo and George Cherukara in Vascular
Supplemental Material
sj-pdf-2-vas-10.1177_1708538120963914 - Supplemental material for The oral health status of patients with peripheral vascular disorders: A systematic review
Supplemental material, sj-pdf-2-vas-10.1177_1708538120963914 for The oral health status of patients with peripheral vascular disorders: A systematic review by Sayed Abdulmotaleb Almoosawy, Mhairi McGowan Karolin Hijazi Karolin Hijazi, Rona Patey, Paul Bachoo and George Cherukara in Vascular
Supplemental Material
sj-pdf-3-vas-10.1177_1708538120963914 - Supplemental material for The oral health status of patients with peripheral vascular disorders: A systematic review
Supplemental material, sj-pdf-3-vas-10.1177_1708538120963914 for The oral health status of patients with peripheral vascular disorders: A systematic review by Sayed Abdulmotaleb Almoosawy, Mhairi McGowan Karolin Hijazi Karolin Hijazi, Rona Patey, Paul Bachoo and George Cherukara in Vascular
Supplemental Material
sj-pdf-4-vas-10.1177_1708538120963914 - Supplemental material for The oral health status of patients with peripheral vascular disorders: A systematic review
Supplemental material, sj-pdf-4-vas-10.1177_1708538120963914 for The oral health status of patients with peripheral vascular disorders: A systematic review by Sayed Abdulmotaleb Almoosawy, Mhairi McGowan Karolin Hijazi Karolin Hijazi, Rona Patey, Paul Bachoo and George Cherukara in Vascular
Supplemental Material
sj-pdf-5-vas-10.1177_1708538120963914 - Supplemental material for The oral health status of patients with peripheral vascular disorders: A systematic review
Supplemental material, sj-pdf-5-vas-10.1177_1708538120963914 for The oral health status of patients with peripheral vascular disorders: A systematic review by Sayed Abdulmotaleb Almoosawy, Mhairi McGowan Karolin Hijazi Karolin Hijazi, Rona Patey, Paul Bachoo and George Cherukara in Vascular
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: SAA was funded by the NHS Grampian Vascular Surgery Department, and MM was funded by the University of Aberdeen Institute of Dentistry through the Aberdeen Summer Research Scholarship as part of the Aberdeen Clinical Academic Training (ACAT).
Ethical statement
No application to the Research Ethics Services was required for this study.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
