Abstract
Fuel lubricity prevents wear between metallic parts in relative motion inside the injection system of combustion engines. Among diesel fuels, paraffinic (gas-to-liquid or hydrotreated oils) and biodiesel (methyl esters) fuels are emerging since some of them are renewable and, in the case of paraffinic fuels, present excellent properties that can be exploited in compression ignition engines. However, the lubricant properties of raw paraffinic fuels are poor. This work explores the potential of individual methyl esters, found in different biodiesel fuels derived from a wide variety of sources, as lubricity additives for paraffinic fuels. Blends at 1% and 2% ester content in a surrogate of paraffinic fuel were tested under the standardized high-frequency reciprocating rig test for lubricity determination. Results confirm the extremely poor lubricity of the surrogate and that the wear scar diameter measured (the higher this, the lower the fuel lubricity) can be significantly reduced with any of the tested esters just at 1% concentration. Higher ester concentration (2%) does not always improve the lubricity further. The number of double bonds in the ester was revealed very significant, but to boost the lubricity of the blend and fulfill the limits set in fuel quality standards, two or more polyunsaturated esters are necessary.
Introduction
Fuel lubricity is defined as the ability of a fuel to reduce friction between two surfaces in relative motion, 1 therefore reducing wear and scuffing. 2 Diesel fuel pumps (single-cylinder, inline, and rotary types) and fuel injectors rely entirely on the fuel for ensuring correct lubrication. This is more critical in rotary pumps 3 and in high-injection pressure systems, such as common-rail systems, because of the finer mechanical tolerances allowed, and in highly loaded engines (such as those built under the “downsizing” concept) and engines with several injection events per cycle. In current gasoline engines, which have evolved toward direct injection (gasoline direct injection (GDI)) and higher injection pressures than before, the fuel lubricity is becoming essential as well. Apart from the automotive sector, these lubricity features may be extended to marine and aviation engines, as reported in Anastopoulos et al.4,5
Automotive engine damages and injection pump failures, all related to insufficient lubricity, were massive in the 1990s, coinciding with the removal of sulfur in fuels to avoid poisoning after-treatment catalysts. This unexpected effect of sulfur-removal was manifested earlier in the 1960s in aviation turbines. 6 Hydro-treating, the chemical process used in refineries for this task, also eliminates much of the naturally contained high-lubricant hydrocarbons present in crude oil diesel cuts, such as nitrogen and oxygen polyaromatic hydrocarbons (PAHs). The lubricity loss must be compensated by the addition of lubricating additives but ensuring the proper lubricant properties is increasingly complex due to the number of fuels in the market. This is because the pool of fuels and fuel components available or shortly expected in the market is spreading to accomplish the ambitious targets set for penetration of renewable sources in transport (see the proposal for a Directive on the promotion and use of renewable energy, 7 in Europe, or the Renewable Fuel Standard Program, 8 in the United States).
As a result, these and other regulations have encouraged the use of biodiesel fuels for compression ignition (CI) engines all over the world. Other less-known fuels, such as gas-to-liquid (GTL), a highly paraffinic hydrocarbon mixture produced from natural gas (then non-renewable) through a Fischer–Tropsch process, possess some inherent high-quality properties, mainly a high cetane number, that may be exploited in existing engine calibrations to achieve cleaner combustion modes. Current GTL prices are not competitive, but investments and technology improvements are dropping the costs quickly. 9
On the properties side, the main handicap for GTL is its poor lubricity compared to fossil diesel due to the null aromaticity, and some authors have focused their research to find a solution via additives.10,11 Also, the GTL production reactions can be adjusted to yield longer alkanes, which are characterized by a better lubricant capacity, 12 but at the expense of worsening the cold flow properties. Contrarily to GTL, esters (the components of biodiesel fuels), as other fatty acid compounds, are magnificent lubricants since it possesses a polar end in their molecule which is adsorbed on surfaces and a long non-polar chain that provides a thin liquid film that prevents wear. Biodiesel lubricity has been investigated in many publications, either neat 13 or blended14,15 with fossil diesel fuel but not with paraffinic fuels as it is evaluated in the present work. In biodiesel–diesel blends, the results confirm that biodiesel contents lower than 2% improve lubricity sharply, while the remaining contents do not contribute significantly. 16 Lubricity improvement was higher when no neat methyl esters but actual biodiesel fuels were added into diesel, which may be interpreted as minor biodiesel constituents (free fatty acids, glycerin, glycerides) having an additional benefit in lubricity. 17
Under this context, GTL fuels additivated with biodiesel emerge as an option to keep the outstanding diesel properties of paraffinic fuels while the biodiesel content enhances the lubricity of the final blend. This work examines the lubricity of a surrogate of paraffinic fuel, designed to mimic the properties of a commercial GTL, and blends of this with pure methyl esters at low content. Among the esters, those present in the ester profile of biodiesel fuels from waste sources or sources not adequate for food markets have been included, as these feedstocks, which are more uncommon in literature, are being encouraged by authorities for biodiesel production in detriment of conventional ones. GTL blending with pure esters rather than biodiesel fuels allows to isolate the effect of each ester (therefore inferring the effect of key molecule characteristics such as the chain length and the unsaturation level of the ester) and to avoid the effect of minor contaminants typically existing in commercial biodiesel fuels (glycerides, glycerol, acids). This approach will confirm whether a sufficient lubricant capacity may be achieved with a balance of saturated, monounsaturated and polyunsaturated methyl esters, as some authors have suggested. 14
Experimental and fuel samples
High-frequency reciprocating rig (HFRR) tests were performed to evaluate the lubricity of the fuel samples, following the European Standard EN ISO 12156-1:2016. HFRR method is used worldwide and preferred over other methods (scuffing load ball-on-cylinder lubricity evaluator (SLBOCLE)) as it can discriminate better between two samples with different lubricity values. 3 Compared to the former version (EN ISO 12156-1:2007), the new one reduces the ambient temperature and humidity range allowed to perform the test, which in practice makes recommendable the use of a climatic chamber. Figure 1 illustrates the range for ambient conditions permitted in the past and the present version of the Standard, along with the average values attained during the tests carried out in this work.
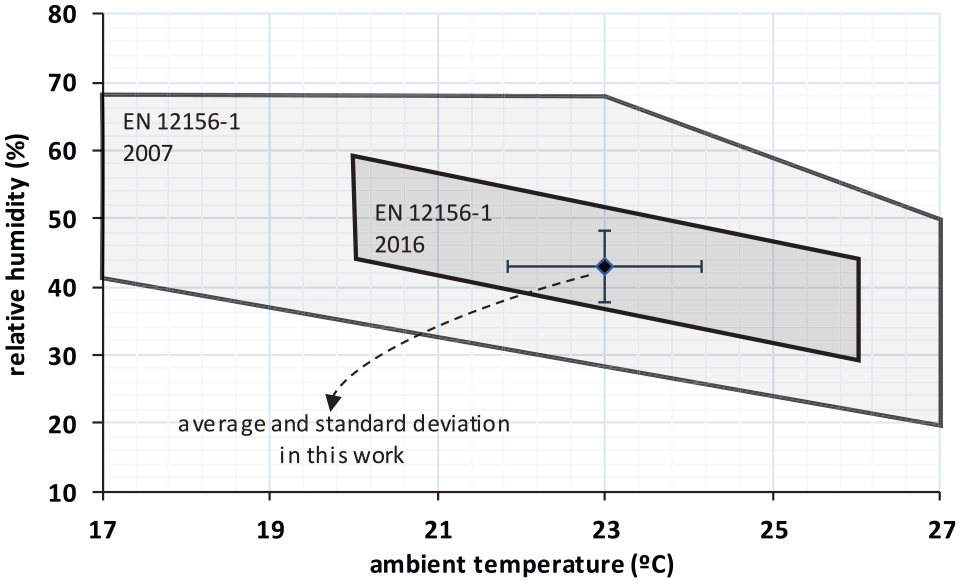
Ambient conditions (temperature, humidity) allowed in EN 12156-1 and attained during this work.
To perform a test, the fuel sample is heated up to 60°C and creates a film between a steel ball and a steel disk. As illustrated in Figure 2, a vibrator arm loaded with a 200 g weight maintains the contact between ball and disk during the test (75-min duration) and causes a wear scar on the ball. This scar is later measured in a stereomicroscope Optika SZR1 equipped with 100× magnification and the WSD (wear scar diameter) is calculated as the arithmetic average of the scar length in the axis parallel and perpendicular to the displacement of the ball.

HFRR equipment.
The HFRR equipment was located inside a climatic chamber from PCS Instruments, where temperature was kept at 22°C–24°C. A supersaturated dilution of potassium carbonate in deionized water was employed in the chamber to maintain the humidity at around 43%. 18 As shown in Figure 1, the ambient temperature–humidity variation during these tests was narrower than permitted in the Standard, which eliminates the uncertainty that different ambient conditions would add to the results (it has been confirmed 12 the effect of ambient humidity on the WSD).
Samples of the pure methyl esters (100 mg each) that are listed in Table 1 were purchased from Sigma-Aldrich, all having a purity higher than 99%. Some of the esters (myristic, palmitic, stearic, and oleic) are present in significant concentrations in conventional vegetable oils (soybean, palm, rapeseed) and animal fats that have been used as biodiesel sources for long. Other esters are only at minor concentrations in these sources, but they are present in non-conventional sources (jatropha oil, fish oil, algae, some wastes) that will benefit from current enhancing politics that pursue the use of sustainable sources that do not interfere with the food market. For example, fish oil is rich in palmitoleic (15%–20%) and docosahexanoic (5%–25%) acids,19–24 while fish fat presents erucic and eicosenoic acids as well.19,25 Algae may show higher concentrations of palmitic and linolenic acids than traditional biodiesel sources, and significant amounts of highly polyunsaturated acids, such as docosahexanoic acid.26–29 Biodiesel from larvae of black soldier fly, a fat-containing insect, presents 15%–35% of palmitoleic ester.30–32 Finally, non-traditional vegetable oils also possess some of these esters, such as lunaria oil (erucic and nervonic), 33 jojoba oil (eicosenoic), 34 sabah yellow durian (linolelaidic and palmitoleic) 35 or Umbelliferae, Araliaceae, and Garryaceae species (petroselinic). 36
Methyl esters used in the work.

GTL fuels are liquid fuels produced from natural gas through a Fischer–Tropsch process, followed by an up-grading that is mainly by isomerization. 37 As a result, the final product is composed of linear and branched paraffins, which are responsible for their very high cetane number. Thus, a purely paraffinic surrogate blend containing 87% (v/v) n-hexadecane (linear paraffin) and 13% (v/v) iso-cetane (branched paraffin) was used as the base fuel (BF) in this study. These two components at such concentrations mimic the properties and the composition of a commercial GTL fuel that has been used in other works of this research group 38 (see Table 2). This fuel could not be used in the present work since lubricity enhancers were added in origin for commercial purposes, as it is inferred from the WSD, 211 μm, shown in Table 2.
Properties of a commercial GTL fuel 38 and the surrogate used as base fuel in this work.

GTL: gas-to-liquid; WSD: wear scar diameter.
All blends described next were prepared using micro-syringes to accurately dose the esters in the BF and a scale (1 mg resolution) to read the mass. Micro-syringes were disposed after the first use to avoid contamination of the samples. Between tests, all the components were submerged for 10 min into ultrasonic-baths using toluene and a final cleaning bath with acetone.
Test design
Three packages of experimental tests were designed by adding methyl esters into the surrogate GTL fuel. The goals were (1) to determine the effect of the sample volume, (2) to explore the effect of the main parameters of the tested esters (i.e. the chain length and the unsaturation level, and (3) to investigate whether the double bond characteristics (i.e. location and configuration) alter the lubricity.
The first package was intended to check the repeatability of the procedure and the equipment and to determine the minimum sample volume needed to perform a lubricity test satisfactorily. For this, two fuels (the BF and a 1% (m/m) oleic methyl ester blended in the BF) and three volumes (2, 1.5, and 1 mL) were selected. A volume of 2 mL is required in the lubricity European Standard, while 1.5 and 1 mL were chosen to check if it were possible to reduce the sample volume without affecting the results. Three repetitions of each test were executed.
The second package aims to evaluate the effect of the ester chain length and the saturation level on the lubricant properties of the esters when blended at 1% (m/m) in the BF. Based on the conclusions of the first package (see “Results” section), sample volume was fixed at 1 mL and three repetitions were planned. For some esters, blends at 2% ester content were also tested to explore the effect of the concentration. Larger ester contents were discarded as they are not typical of fuel additives.
In the last package, four different isomers of C18:1 were blended at 1% in the BF to study the effect of the location and configuration of the double bond. These methyl esters were (1) oleic (C18:1ω9), which is the most typical in most vegetable oils and fat sources from all C18:1 and presents its double bond in the center of the molecule; (2) petroselinic (C18:1ω12) and (3) vaccenic (C18:1ω7), where the double bond is moved toward one of the two ends of the molecule; and (4) elaidic (C18:1ω9t), with the double bond at the same position than oleic by the trans-configuration. Both location and configuration of double bonds are parameters that may change from one biodiesel fuel to another and that could affect the characteristics of the liquid film that prevents wear in the metallic surfaces. The first (location) depends on the source for biodiesel production, as already described, while the second (configuration) changes with some chemical and physical processes applied to biodiesel to improve its fuel properties, such as hydrogenation. 39
Results
The results of the first package of tests are represented in Figure 3, where the error bars correspond to the 95% confidence interval. For both fuels tested, it is observed that the sample volume does not affect the value of the WSD or the comparison between fuels (within the volume range evaluated). Therefore, a volume of 1 mL was selected for the following test packages, as the available quantity of esters was very limited and higher volumes could compromise the number of tests if additional repetitions were needed.
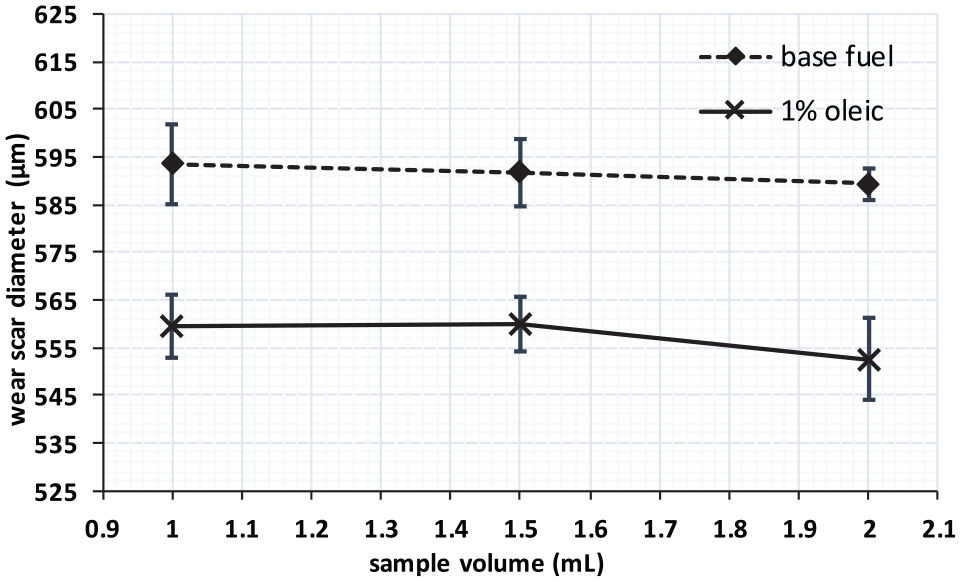
Effect of the sample volume.
The largest error bar in Figure 3 was lower than 10 µm, well below the maximum deviation admitted in the test standard (up to 50 µm is admitted in one in 20 cases). In terms of test repeatability, this result confirms that three repetitions are enough to discriminate results. These conclusions are not expected to change if other samples from Table 1, different from the BF and 1% oleic methyl ester, had been selected for this test. However, for more volatile fuels such as alcohols and alcohol-containing fuels (e-diesel), higher volumes may be imperative since sample temperature is raised up to 60°C for the test.
The effect of the ester chain length, evaluated in the second package of tests, is shown in Figure 4 for blends of saturated methyl esters in the BF, at 1% and 2% ester content. For a reference, a line indicating the lubricity of the BF in the absence of additives has been drawn in the figure. All blends showed a noticeable lubricity improvement compared to the BF, despite the low ester contents tested. In general, there is a decrease of the WSD when the chain length of the ester increases. Results with neat esters and esters blended in regular diesel fuels 40 showed similar trends; therefore, this effect of the chain length is now validated for ester-additivated paraffinic fuels.

Effect of the chain length and ester content for saturated methyl esters blended in base fuel.
Nevertheless, the lubricity gain with longer ester chains was not uniform along the whole range evaluated. While lubricity of the blends improved with longer ester chain lengths up to that of palmitic ester (C16:0), there was no further improvement thereinafter. This result suggests that better lubricity is expected if GTL fuels are additivated with palmitic-rich biodiesel (such as palm oil-derived biodiesel) rather than with other saturated biodiesel rich in shorter esters, such as coconut-derived biodiesel (rich in lauric ester, C12:0). But, compared to palm oil biodiesel, no additional benefit is anticipated if saturated sources rich in stearic (and longer) ester (C18:0), such as animal fat biodiesel, are used as lubricity additives.
For the palmitic ester (C16:0) at 1% content, Figure 4 evidences a sharp increase of the lubricant capacity compared to the immediate shorter ester, C14:0. Though the reasons for this sudden improvement at low content are not documented, other authors have found a marked good lubricity for C16:0 compared to esters quite similar in carbon chain. 40
In general, lubricity results were better for an ester content of 2% compared to 1% (Figure 4). The benefit of increasing the ester concentration is more evident for shorter esters (C6:0 and C14:0). Moreover, in the range of C16:0 to C20:0, the error bars of the two ester content lines overlap; therefore, there is no evidence for these esters that adding ester in more than 1% will improve the lubricant capacity of the blend. This behavior is typical in those properties that change in a strongly non-linear manner, as it is the case of lubricity. In such properties, once a threshold of concentration is reached, the value of the property remains constant or the change is only minor. The results in Figure 4 indicate that the content threshold for lubricity depends on the type of ester, being around 2% for C16:0 to C20:0, but higher than 2% for shorter esters.
The effect of the unsaturation (i.e. the number to double bonds) level of the esters is presented in Figure 5 for a fixed ester content of 1% in the BF. For all five chain lengths represented in the figure, the presence of a first double bond in the ester molecule did not contribute to the lubricity capacity of the blend (for 14 and 16 carbon atoms), or even reduced it (for 18–22 carbon atoms). Data published by Knothe et al. 41 and Lapuerta et al. 42 for pure methyl and ethyl esters, respectively, revealed that the first double bond did increase slightly the lubricity of the ester, but the results in the present work prove that this trend is not maintained when the esters are additivated into paraffinic fuels. This finding has serious consequences for practical blends of GTL with biodiesel fuels: oleic ester (C18:1), a major component of virtually all conventional biodiesel sources, and erucic ester (C22:1), the main unsaturated ester present in same rapeseed species, are detrimental for lubricity of the blend.

Effect of the unsaturation level of methyl esters blended in base fuel at 1%.
For higher numbers of double bonds in the ester (i.e. polyunsaturated esters), Figure 5 shows a strong improvement of the lubricity of the blend, which is now in agreement with the most reported trend for pure methyl esters. 41 Even more, just 1% of any of the three polyunsaturated esters in the BF was enough to fulfill the limit of 460 μm maximum set in EN-590 (quality European standard for diesel) and EN-15940 (for paraffinic synthetic fuels). This result highlights the potential of non-conventional biodiesel sources as additive improvers, such as fish oil (rich in C22:6) and, especially, algae (rich in both C20:4 and C22:6). Biodiesel from algae is receiving attention recently since the European authorities specially encouraged the use of algae cultivated in bioreactors as a source of advanced biodiesel. 43 Meanwhile, the production of algae has been proved to be economically and ecologically sustainable. 44
Finally, the third package of test pointed to the effect of the location of the double bond and its cis-/trans-configuration (Figure 6) for a fixed carbon chain length and unsaturation level (C18:1) of the ester blended at 1% in BF. The mean values indicate that this effect is the weakest of all the effects evaluated in this work. The lubricity of the blend is only slightly worse (higher wear diameter) when the double bond of the ester is at the center in the molecule and improves when the bond moves toward either end. Nevertheless, the differences observed fell into the confidence bars; thus, for this specific combination of ester chain length, unsaturation level, and content in the blend, it is not possible to conclude that this effect is significant. The same has been reported elsewhere 45 for several C18:2 esters. From a chemical basis, the location of the double bond modifies the symmetry and the polarity of the molecule, which is more pronounced for shorter molecules. Therefore, the position of the double bond/s in shorter esters could possibly exert some influence on the lubricity.
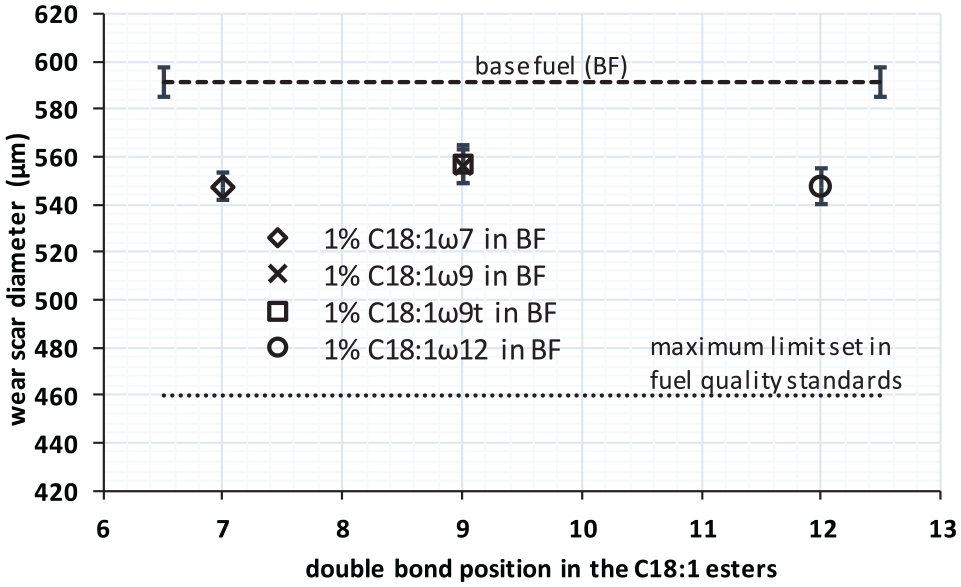
Effect of the double bond position in C18:1 methyl esters blended in base fuel at 1%.
Regarding the cis- or trans-configuration, the results in Figure 6 show that there is no influence at all on the lubricity of the blends. Again, it is not possible to discard some influence if shorter ester molecules were used instead.
Conclusion
The work presented has proved the potential of combining biodiesel and paraffinic fuels for use as blend in diesel vehicles. The poor lubricant properties of raw paraffinic formulations can be satisfactorily compensated with the addition of methyl esters (biodiesel) at very low concentrations. Based on the results on specific methyl esters, some recommendations are drafted on how to select the biodiesel sources and blend content:
Among all the tested esters, the highly unsaturated ones showed the most promising results. When 1% of these esters were added into the base paraffinic fuel, the WSD was reduced by 30% compared to the BF case (from 595 to 430 μm, approximately), a reduction that is enough to meet the current limit set in European standards for quality of both automotive and paraffinic fuels.
Based on the former, biodiesel sources such as fish oil and algae are suggested here as there is significant presence of unsaturated esters in their ester profiles. Since these sources have no use in food markets, they are pointed out by regulations for production of advanced biofuels, which are being encouraged recently.
Regarding the biodiesel content, 1% was adequate in almost all tests to achieve, or at least get close to, the limit set in the standards. Real biodiesel fuels are expected to behave even better than the pure methyl esters tested here, because the presence of minor concentrations of other compounds (glycerol, glycerides), inherent to all biodiesel fuels, has positive effects on lubrication capacity. Just in the case of shorter esters (lower than 16C atoms), higher biodiesel content, probably at or around 2%, is recommended.
Footnotes
Handling Editor: James Baldwin
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study has been carried out under the framework of project ENE2016-79641-R, financed by the Spanish Ministry of Economy, Industry and Competitiveness.
