Abstract
The pursuit of radically improving the strength and stiffness of materials while maintaining satisfactory level of ductility for formability has been on for decades. A patent has laid in 2005 the foundation for alloying a metallic matrix, such as steel matrix, with nano-meter-sized elements while exploiting amorphous cross-linking of the matrix. This patented innovation is expected to trigger the third industrial revolution based on mass production of the alloying of a metallic matrix, such as steel matrix, with nano-meter-sized elements in order to produce bulk metallic glasses. The first industrial revolution had been triggered by the mass production of steel in about 1840 and the second industrial revolution was triggered by the mass production of the synthetic polymeric materials and composites in about 1960. This article presents an approach of computer-aided manufacturing based on this patent for the mass production of an E-like-shaped spring made of the alloying of heterogeneous composite ferromagnetic metallic glass matrix with nano-meter-sized elements. The proposed heterogeneous nanocomposite metallic glass alloy exhibits features that are promising in applications such as vehicle suspension springs.
Introduction
Examining processes that affect key products such as automobiles becomes increasingly important nowadays. Although many improvements have been made regarding steel manufacturing processes, unsound castings are still produced under normal production conditions. Minimizing cost and risk, computer-aided manufacturing (CAM) offers improvement in each of the key four areas of manufacturing: information flow, process coordination, efficient utilization of resources and flexibility leading to a shorter product cycle.1,2 Adopting cooling curve experiments in CAM has the potential to evaluate the size of the alloying elements and the degree of modification achieved by these modifiers simultaneously. 3
Originally, the formation of a nanostructured meta-stable non-equilibrium-based material synthesized by solid-state powder processing was reported by Thompson and Politis 4 in 1987. A patented invention on a hybrid micro/nanocomposite material-based technology has laid in 2005 the foundation for alloying a metallic matrix, such as steel matrix, with nano-meter-sized elements in equilibrium/non-equilibrium synthesis through other ways of processing while exploiting amorphous cross-linking of the matrix.5,6 This patented innovation is expected to trigger the third industrial revolution based on mass production of the alloying of a metallic matrix, such as steel matrix, with nano-meter-sized elements in order to produce bulk metallic glasses (BMGs). The first industrial revolution had been triggered by the mass production of steel in about 1840 and the second industrial revolution was triggered by the mass production of the synthetic polymeric materials and composites in about 1960. A key advantage of bulk amorphous alloys is that they can soften and flow upon heating like glasses. This allows for easy processing, such as by injection moulding, similar to polymers. Quenching amorphous metallic structure results in avoiding crystallization during solidification, preserving the amorphous arrangement of the atoms that existed in the metal’s liquid phase. The amorphous structure of glasses results in a lack of slip planes so that metal glasses exhibit much greater resistance to deformation than crystalline metals.
Because amorphous metal has no anisotropic properties, which originated from a crystalline structure, and there are no crystalline grain boundaries, it can exhibit excellent properties. For instance, grain boundaries are a weak point for corrosion as they provide more surface area for the required chemical reactions to occur. The lack of grain boundaries in BMGs reduces their tendency to corrode. The alloying elements of boron and silicon with magnetic metals, such as iron, cobalt and nickel, have high magnetic susceptibility with low coercivity and high electrical resistance which together exhibit high permeability and high-saturation magnetic flux density. The high resistance leads to low losses by eddy currents when subjected to alternating magnetic fields. This property is useful in applications such as transformer magnetic cores. In addition, the BMGs exhibit high elasticity and ability to store mechanical energy. Yet, metal glasses are limited to low-temperature applications because of their characteristically low glass formation temperatures. 7 However, this is suitable for vehicle suspension system applications, such as the structural material of springs. Metal glasses also exhibit high hardness due to the lack of grain structure. The high hardness implies wear resistance which is preferable in vehicle suspension springs. In addition, high elastic energy storage per unit volume and mass, and the low damping, give metallic glasses (MGs) potential as vehicle suspension springs. Other possible applications of MG springs in devices include high-speed relays. BMG is expected to exhibit higher dynamic toughness (MPa m1/2) in structural vibration applications under loading rate ranging from 10−3 to 100 MPa m1/2/s. 8 Moreover, die casting metal glass parts exhibit almost no shrinkage due to no phase change as well as lower melting temperature that requires less cooling so that they do not require surface finish unlike die casting materials such as Iron. Yet, MGs at room temperature are not ductile and tend to fail in a brittle manner suddenly when loaded in tension, which limits the material applicability in reliability-critical applications. Thus, MGs usually have poor ductility in terms of low elastic strain limit, and hence exhibit poor tensile strength. Therefore, there is considerable interest in producing metal matrix composite materials consisting of an MG matrix containing dendritic particles or fibres of a ductile crystalline metal. In addition, due to the overall complex nature of the toughening mechanism in MG composites, there is a lack of understanding the toughening mechanism in MG composites. 9
Recently, research has been done proposing crystalline metal matrix nanocomposite, such as Wang. 10 The properties of crystalline metal matrix nanocomposite strongly depend on the amount, distribution and dispersion of nanoparticles inside the metal matrix. Yet, the nanoparticles in the crystalline metal matrix are rarely observed in the bands between intermetallic phase-rich regions in the metal matrix. 11 The depletion of nanoparticles in the band between intermetallic phase-rich regions indicates that the nanoparticles are pushed to the last solidified region during solidification. 12 Thus, the dispersion of nanoparticle clusters in the crystalline metal matrix can have negative effect on strength and elasticity of the nanocomposite. Even the improvement in Young’s modulus gained from the crystalline metal matrix nanocomposite is very limited because Young’s modulus is determined by atomic bonding.11,13
Other techniques have been proposed to develop steel structures such as surface mechanical attrition treatment (SMAT). The SMAT causes a deleterious influence on the corrosion resistance of steel. 14 The SMAT also causes increase in surface roughness, strain-induced martensite and dislocations nullifying the beneficial influence of surface nanocrystallization. 14 Further techniques have been proposed to develop steel structures such as twinning-induced plasticity (TWIP). The tensile strength of Fe–22% Mn–0.52% TWIP steel has reached the value of 1160 MPa. 15 The ultimate tensile strength of TWIP-based ultra-high-strength steels (UHSS) reaches values up to 1500 MPa. 15 Yet, the yielding strength of TWIP steels is as low as 300 MPa. 16
This article investigates the CAM for mass production of non-conventional shapes made of the alloying of heterogeneous composite ferromagnetic MG matrix with nano-meter-sized elements. It starts with investigating the alloying of a metallic matrix with nano-meter-sized elements. Strengthening and toughening mechanisms in the proposed structure follow. Next, the investigation of the mass production of non-conventional shapes made of the alloying of an MG matrix with nano-meter-sized elements is presented. Finally, a CAM-based method is proposed.
Alloying of an MG matrix with nano-meter-sized elements
The selection and design of modern high-performance structural engineering materials is driven by optimizing combinations of mechanical properties such as strength, ductility and fracture toughness. A key advantage of nanoparticles is their inherently large surface-to-volume ratio relative to that of larger particles. These nanocrystalline materials have comparative advantages over their microcrystalline counterparts in hardness, fracture toughness and ductility. 17 The origin of such microstructure depends on kinetic and thermodynamic contributions which are difficult to separate. As a top–down approach to the synthesis of nano-sized materials, the alloying process is hence adopted in this article and the adopted alloy development strategy aims at exhibiting improved fracture toughness and tensile ductility while retaining high strength of the parent bulk MG matrix. The way adopted in this study to produce BMGs is rapid cooling of solid solution. A heterogeneous nanocomposite ferromagnetic MG alloy is proposed in this study and is expected to be about three times stronger than titanium with elastic modulus that can nearly match bones. This alloy is expected to have a high wear resistance without producing abrasion powder. It does not undergo shrinkage on solidification. The adopted alloy composition is shown in Table 1.
Adopted alloy composition.

The ductile crystalline metals, such as nickel, titanium and copper, are particularly added to the composite structure as nano-meter-sized alloying elements. These reinforcing nanoparticles have an average diameter of 50 nm and expectedly have good wettability with Fe melt. An additional beneficial effect of incorporating copper in the is to improve the mechanical strength of the proposed material. The rest of modifying additives in this study are of the size of mostly less than 1 µm. The Cr content not only increases corrosion resistance but also reduces the stacking fault energy (SFE) of the alloy which helps in reducing free volume resulting in improved mechanical properties. 18 Elements such as Si can provide improved properties with minimum contents at a lower and less volatile price. In this sense, low melting point alloys are especially interesting since they provide the formation of a liquid phase that promotes the homogeneous distribution of alloying elements. 19
At higher molecular weight of nanoparticles in composites, the matrix does not wet the particles and the glass transition temperature decreases. 20 For particles of atomic number less than 42, the matrix wets nanoparticles efficiently. The elements of the proposed composite structure are thus of atomic number less than 42. Low molar mass systems tend to fill the structural pores completely, especially at temperatures about 50 K above the melting temperature or glass transition temperature. The majority of the elements of the proposed composite structure are thus of low molar mass.
A key advantage of the proposed heterogeneous nanocomposite ferromagnetic MG alloy is that the volume fraction of glass can be calculated beforehand using the rule of mixtures average of the glass matrix and the dendrites without even the need for differential scanning calorimetry (DSC) scans. In addition, since tensile ductility substantially varies with dendrite size, the proposed composite structure expectedly leads to a highly toughened composite. The microstructure with isolated dendrites in a BMG matrix expectedly stabilizes the glass against the catastrophic failure associated with unlimited extension of a shear band and results in enhanced global plasticity and more ductile failure. The homogeneity of solute nanoparticles, Hm, can be modelled as a logarithmic function of time, t, as follows

where ψ and
This is analogous to the proved homogeneity formula presented in Benjamin and Volin. 21 This paves the way to investigate the strengthening and toughening mechanisms in the proposed alloy.
Strengthening and toughening mechanisms in the proposed structure
Metals of vastly different size will have difficulty bonding with each other so that an alloy containing them would take longer to crystallize due to the ‘confusion effect’. The many elements of different sizes in an amorphous metal alloy lead to low free volume in molten state and hence higher viscosity than other monolithic metals in molten state. The viscosity prevents the atoms moving enough to form an ordered lattice. Thus, upon cooling at sufficiently fast rates, the constituent atoms simply cannot coordinate themselves into the equilibrium crystalline state before their mobility is stopped. In this way, the random disordered state of the atoms is locked in and the constituents become cross-linked. It takes much more energy to permanently shift these atoms around. Thus, activation energy and activation volume need to be in place for shear transformation zone (STZ) operations.
The formation of BMGs of several millimetres thick can occur at critical cooling rates of 10–100 K/s. Alloys containing an array of elements such as nickel and copper can reach critical cooling rates of as low as 10 K/s. The formation of the amorphous state is the ability for the cooling curve to miss the crystalline nose. Hence, lowering the melting temperature and raising or maintaining the glass formation temperature would yield a cooling curve with a lesser slope, and lower therefore cooling rate, that misses the crystalline nose.
Strengthening by grain size reduction is adopted in this study. Because of the very small size of the precipitates, a large fraction of the atoms in these materials would require lower interfacial energy during mechanical stir mixing for alloying; thus, high diffusion rates and consequently reduced manufacturing times are achieved. Also, virtually rounded precipitates result in reduced induced strain at interfaces and thus increased toughness.
Solid-solution strengthening is adopted as well in this study and is based on the fact that adding another element that goes into interstitial or substitutional positions in a solution increases strength. There are key factors other than size and concentration of solute atoms that influence solid solution strengthening: shear modulus of the solute atoms, valency of solute atoms for ionic materials and the symmetry of the solute stress field. The magnitude of strengthening is higher for non-symmetric stress fields because these solutes can interact with shear bands in amorphous structures immobilizing crack propagation. Generating an extremely large number of these bands could make the material tougher. If the number of bands is significantly increased, they start to interlace and form networks. When new cracks evolve in the glassy metal, these networks thus cluster around the crack tips, blocking them from growing. Non-symmetric stress fields cause not only volume change but also shape change. 22 The random distribution of the alloying nano-meter-sized elements in a metallic matrix results in non-symmetric stress fields which lead to higher magnitude of strengthening.
Most monolithic MGs in bulk dimensions do not exhibit rising fracture toughness, i.e. they fail catastrophically soon after yielding by unstable crack extension. Upon yielding, shear strain in MGs is highly localized into narrow, 10-nm-wide bands. The development of such shear bands in MGs does not necessarily result in fracture since multiple small-sized shear band formation without crack formation has been reported. 9 A heterogeneous bulk glassy alloy can demonstrate an unusual capacity for shielding an opening crack accommodated by an extensive shear band sliding process, which promotes fracture toughness comparable to those of the toughest materials known.
Before failure, a large number of shear bands form in the spacing within the second phase throughout the entire part section. 9 These shear bands carry the plastic deformation, and their large number results in observed global tensile ductility. As deformation proceeds, the complex stress field redirects shear bands towards neighbouring space in the second phase. Fracture occurs typically along the shortest distance between spacing in the second phase. 9 In addition to the spacing within the second phase, the magnitude of the stress concentrations depends on the shear modulus of the second phase. A second phase with significantly different shear modulus than the matrix causes high stress concentrations. The lowest stress concentrations are present for a second phase with a modulus similar to the matrix. However, in this case, the high values of elastic strain cannot be achieved. Thus, there is compromise between the lowest stress concentrations and high values of elastic strain. Hence, optimization suggests a second phase with a modulus closely smaller than that of the matrix.
The effectiveness of MG heterostructures also depends on the size (cross-sectional width perpendicular to the loading direction) of the sample,
In the proposed microstructural architecture for MG heterostructure, a critical design aspect is a soft second phase which is most effective when spacing within the second phase assumes the critical crack length of the MG. This spacing should coincide with the second phase’s size. The highest performance in terms of flow stress and flow strain is expected to be achieved in the proposed heterogeneous nanocomposite ferromagnetic MG alloy with spacing within the second phase close to the critical crack length. These toughening strategies are only effective in samples that are large compared with the spacing of the second phase, such as the adopted glassy E-like shapes of 5–19 mm in thickness.
The basic behaviours of amorphous metals are as follows: (1) high-temperature homogeneous flow and (2) low-temperature strain localization into shear bands. 23 The two microscopic mechanisms that lead to deformation in MGs are as follows: (i) STZ where several dozens of atoms deform in-elastically in response to an applied shear stress and (ii) redistribution of free volume where a single atom jumps from an area of low free volume to an area of higher free volume. 23 Having seen the strengthening and toughening mechanisms in the proposed structure, let us investigate the mass production of non-conventional shapes made of such matrix.
Mass production of non-conventional shapes made of the alloying of an MG matrix with nano-meter-sized elements
The high computational ability available at low cost of CAM systems meets the need for quality manufacturing and short lead times. The CAM systems provide (1) computer monitoring and control of the manufacturing process and (2) manufacturing support applications during the preparation for actual manufacturing and post-manufacture. 24 When the central computer schedules a part for manufacturing, the processing instruction and processing sequence for the part are determined automatically by the central computer. Operating data from the machine and quality data from the part are sent back to the central computer. 25 The number of parts tested is determined with the help of statistical methods which take into account the vendor’s historical quality performance record.
In order to identify a material with adequate performance to meet a property requirement at minimum cost, experiments should be supplemented by CAM systems that contain a database (DB) of related alloy systems relating properties to microstructure. The material engineer should then be able to use more quantitative approaches. 26 Cooling curves are helpful in relating the cooling curve parameters and the metallurgical characteristics of the alloy. Several DBs have been developed that help in predicting inoculation efficacy and the mechanical properties during the solidification process. 27 Semi-automated computer-aided continuous hot injection mould design system similar to semi-continuous die casting is adopted in this study integrating part load and time–temperature transformation (TTT) DB. 28 These DBs are utilized in the proposed CAM-based method in this study as illustrated in Figure 1.

Function flow chart for mass production of non-conventional shapes made of the alloying of a metallic glass matrix with nano-meter sized elements.
On-machine inspection (OMI) enables measurement taking, data collection as well as data feedback and process adjustment fully automated and integrated. With OMIs, a part can be measured at the machine and corrected there. Small samples can be made and then checked immediately in order to identify problems at an early stage. The STEP-NC (ISO 14649-10, 2004) provides the control structures for the sequence of programme execution, mainly the sequence of working steps and associated machine functions 25 . The ‘machining_schema’ entity contains the definition of data types which are generally relevant for different technologies (e.g. casting) (ISO 14649-10, 2004). 25 Probing working step is also defined in this part for inspection operations. However, the probing operations are specified in (ISO 14649-16, 2004). 25 This proposed CAM-based method is further presented in the next section.
Proposed CAM-based method
A strategy combining micro-alloying with thermo-mechanical processing is adopted in this article. A two-step fabrication method is adopted: fabrication of a mould and replication of this mould with a heterogeneous nanocomposite ferromagnetic MG alloy. Computer-aided design (CAD) drawing is used to fabricate the mould that comprises the negative image of the proposed heterostructure. The data can be extracted from the CAD model using the STEP data standard. The mould is replicated through thermoplastic forming-based compression moulding, and the fully amorphous state of the resulting heterostructure can be subsequently confirmed by DSC. Figure 2 shows a proposed computer-aided system for thermal characterization.

Computer-aided system for thermal characterization.
A correct thermal characterization requires the following conditions to be fulfilled with respect to Figure 2:
The weight of samples should be as constant as possible.
The structural material of the cups should not react with the melt.
In an endeavour to mix the nanoparticles into the matrix, a two-step strategy is adopted:
The first mechanical mixing step is designed to address two challenges for incorporating nanoparticles into molten metal: (i) the burning of non-oxide nanoparticles due to the large surface-to-volume ratio of nanoparticles and (ii) the floating of nanoparticle clusters without incorporation into molten metal due to the poor wettability of nanoparticles with the molten metal. In order to slash the burning of non-oxide nanoparticles, the nanoparticles are fed at semi-solid state of the metal by mechanical mixing at low feeding temperature. In order to slash the floating of nanoparticle clusters without incorporation into the matrix, the high viscosity at the semi-solid state prevents the floating nanoparticle clusters.
The second mechanical mixing step is designed to improve wetting and break nanoparticles' clusters. Since the wettability of nanoparticles with liquid metal and fluidity of liquid metal increase as temperature increases, this step is conducted at a temperature higher than the liquidus temperature, about 150 °C higher than the liquidus temperature. The enhanced fluidity at higher temperature allows liquid metal to penetrate into nanoparticle clusters better, which significantly reduces the force needed to break nanoparticle clusters. The shear force generated by the rotating impeller shown in Figure 2 will further break nanoparticles' clusters into smaller ones. The stirring blade should be hardened and coated in order to prevent contamination from the blade. Uniform distribution and dispersion of nano-meter-sized particles in molten BMG matrix of nanocomposite alloy can be achieved by ultrasonic-assisted fabrication. In ultrasonic processing of nano-meter-sized particles, a source of ultrasonic waves is used to distribute and disperse the nano-meter-sized particles in the molten BMG matrix.
The wettability in this composite structure is determined based on the interaction of the segregated particles with the matrix. During the dispersion of nanoparticles into liquid, wetting is about reducing surface tension differences between matrix and nanoparticles. High-intensity acoustic energy released due to ultrasonic vibration can help in dispersing nanoparticles into the melt and controlling dendritic structure. The ultrasonic energy can help in stabilizing the nanoparticles through charge stabilization. Mechanical stir mixing is hence combined with ultrasonic dispersion in this work. Acoustic cavitation is thus used. When the matrix is subjected to high-intensity ultrasonic vibrations, the alternating pressure above the cavitation threshold due to the cyclic nature of ultrasonic wave of compression and expansion brings about several tiny bubbles in the liquid metal. Acoustic cavitation involves the formation, growth, pulsation and collapse of tiny discontinuities or bubbles in liquid. The acoustic streaming results in bubble growth and then bubble collapse leads to achieving good dispersion and wetting. The high-pressure shock wave can break the agglomeration of nanoparticles.
The adopted ultrasonic system for the treatment of the proposed material consists of an ultrasonic power supply unit, a high-power ultrasonic converter, an acoustic waveguide and an acoustic resonator, as indicated in Figure 2. The waveform generated by the ultrasonic power supply unit is converted in repetitive multi-frequency mechanical vibrations by the ultrasonic converter, which are driven to the acoustic resonator through the waveguide, which also works as a booster. Since the central mechanism of ultrasonic processing of liquid alloys is cavitation, those factors that influence cavitation levels are of importance to the operation. Given that ultrasound loses its oscillation energy and sound intensity along the path in a melt or in a solid medium, if this loss is significant, the intensity rapidly falls to the cavitation threshold and cavitation ceases. This effect of sound attenuation depends on several parameters such as melt viscosity, resonator and waveguide temperature which needs to be under control. Surfactants can be used as wetting agents. The wettability of the nanoparticles is assessed by measuring the contact angles (CAs) of distilled water on composite matrix. The CA of water is used for calibration purposes, for instance, the static CA of pure water is measured to characterize wettability of a sample. A good interaction requires a small wetting angle. When the wetting angle is between 0° and 20°, the wetting quality becomes very good resulting in minimal CA hysteresis. Wetting behaviour characterized by the water CA is dependent on both surface chemistry, in terms of surface energy, and surface topography, in terms of physical roughness. 29 At equilibrium, the interfacial free energy of the interface should be minimum. This surface energy is a function of the radius of the particle, the interfacial tension, the area of the interfaces and the CA. The enthalpy of wetting of the nanoparticles by the matrix can be measured as an indication on the interaction energy. 30
Alloys are prepared by inductively melting the elements with purity better than 99.99% in quartz tubes under inert atmosphere. The ingot is sealed in a quartz tube under vacuum, which is then heated above its liquidus temperature. Alloy ingots are fluxed at a temperature above the crystalline C-curve of the corresponding cooling curve, for example, about 1200 K, for about 1000 s. Glass formation is achieved by melting fluxed ingots in quartz tubes with 0.5-mm-thick walls. Melting can be done by arc-melter. 31
The melt is then instantaneously directed to flow into the mould and to be compressed in the quartz mould which has quartz walls at the edges to seal the compression chamber and then to be rapidly water quenched. In this study, compression-based hot forming is adopted with very low applied strains, i.e., equivalent strain of about 1%. Melting the amorphous metal at vacuum pressure and casting it at an increased pressure of about 50 MPa can increase the efficiency of the production process. This is for a couple of reasons: (1) during solidification, the high pressure causes the liquid metal to maintain contact with the mould wall that provides the cooling to the material and (2) the glass forming temperature of metals increases at higher pressures due to the fact that during the propagation of shear bands, the internal friction between atoms results in a rise in temperature of several hundred degrees producing thus more flow serration. The hot compression is used to disperse nanoparticles further and eliminate solidification defects. The hot compression is conducted at a temperature less than 1200 K with a ram speed of about 60 mm/min. The average pressure required to carry out the formation of a part is expressed by the Hagen–Poiseuille equation. 32 Hot pressing of materials consumes about one-seventh of the excess energy apart from equilibrium in comparison with other techniques such as condensation from vapour, indicating less energy consumption. 33 By introducing compression stress into the structure, the tensile strength of the material can be increased. This can be done via compression moulding of MGs.
Quenching water or liquid nitrogen flows through the upper and lower parts of the mould as well as around the sealing quartz walls at the edges of the mould. The quartz tube water quenching method is expectedly more efficient in terms of glass formation than other techniques such as copper mould casting. Water quenching in the molten state in quartz moulds can be capable of forming glassy E-like shapes of 5–19 mm in thickness. The DSC (e.g. Perkin-Elmer Diamond DSC) can confirm the amorphous nature of the MG.
At the scale of nano-meter-sized alloying elements, based on Griffith-like criterion, the elastic energy release does no longer offset the energy barrier to form a shear band. Hence, on such a small scale, a deformation mechanism not relying on shear bands can result in tensile ductility. This nano-scale cyclic strengthening occurs when an MG is cycled at loads well below the load typically required to initiate a shear band, i.e. occurs in the regime of the elastic material response. This nano-scale strengthening contrasts the macroscopic behaviour of an MG where the elastic–visco-plastic nature of MGs usually results in softening of a glass under a load. As a matter of fact, shear bands form along a trajectory where the entire shear band path exceeds the macroscopic yield stress. 23 Therefore, this area of high stress during the cycling could induce structural rearrangements, which accumulate over time through to-and-fro STZ activity resulting in strengthening of a glass subtly. Saturation in this strengthening occurs beyond which further cycling does not provide additional strengthening. 23 This study proposes to perform this nano-scale cyclic strengthening after cooling down of the formed spring.
A part of the alloy is taken out for 60 min to be kept at 100°C for the purpose of conducting tests. 9 The mechanical properties are studied by uni-axial tension (STM-100KN; United Calibration Corporation, USA) and compression (Sinteh 10/GL; MTS, USA) tests. Tensile test samples with diameter of 6.3 mm and gauge length of 31.7 mm are machined from the alloy by turning. In this tension test, single edge-notched specimens under monotonic tensile loading are adopted in order to develop a local plastic zone at the crack tip. Under plane stress conditions, such a plastic zone is dominated by shear bands elongated in the direction of crack extension. 34 Young’s modulus is calculated from the elastic deformation part of the stress–strain curve under tension stress. The tensile strain is measured by extensometer for accurate measurement. The discs for the compression test with diameter of 10 mm and height of 10 mm are cut from the alloy by a low-speed diamond saw. The strain is calculated from the platen displacement, which includes the deformation of testing machine. The yield strength and plasticity of the samples are calculated from the compressive stress–strain curves due to the expectedly limited plastic deformation under tensile stress. The distribution of the nanoparticles can be investigated by scanning electron microscopy (SEM; LEO 1530 Gemini; Zeiss). The geometrical effect caused by the E-like shape of the formed spring increases the elasticity and as a result enhances the deformation of the MG heterostructure, as can be gathered from the following comparison.
Numerical analyses and results
Three diversified configurations of steel springs, multi-leaf spring, E-like-shaped spring and C-like-shaped spring, are compared using COSMOS/M finite element package. The material type in these three finite element models is AISI 9255 alloy steel. These models are subjected to the same utmost level of dynamical full-load condition of 545 kg based on the quarter car model of a light commercial vehicle. The element group for the steel semi-elliptic multi-leaf spring model is taken as a SHELL4T four-node thick shell element. The element group for the other two models of the steel E-spring and the steel C-spring is taken as a TETRA4R four-node tetrahedral with trans/rot degree of freedom (DOF). Figures 3–5 present the maximum vertical displacement Y and the deformed shape of the steel semi-elliptic multi-leaf spring, steel E-spring and steel C-spring, respectively. The summary of these numerical analyses is shown in Table 2.

Maximum vertical displacement and deformed shape of steel multi-leaf spring.
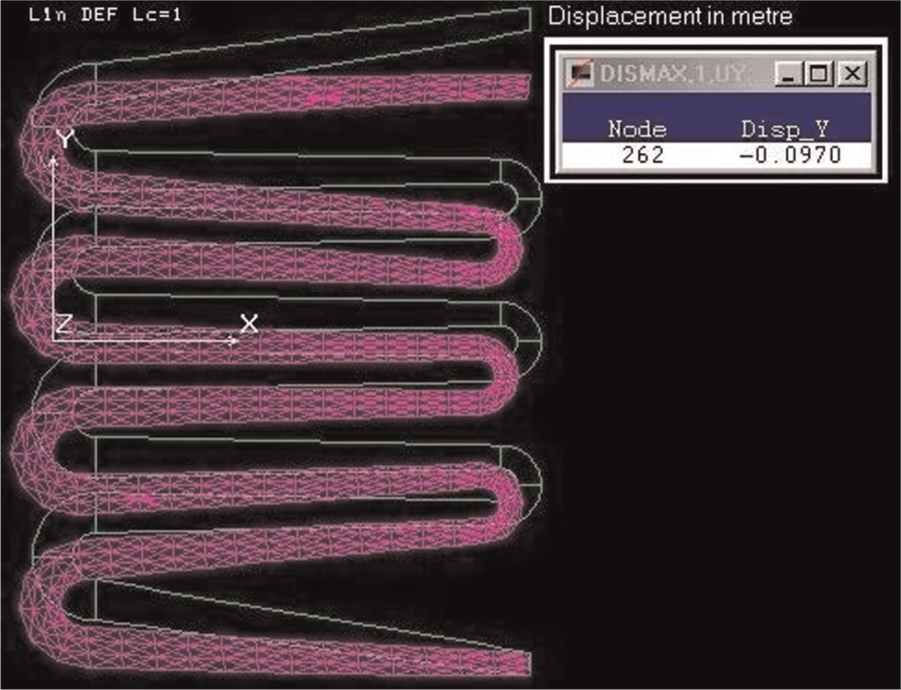
Maximum vertical displacement and deformed shape of steel E-spring.

Maximum vertical displacement and deformed shape of steel C-spring.
Summary of the numerical analyses of the geometrical effect on elasticity.
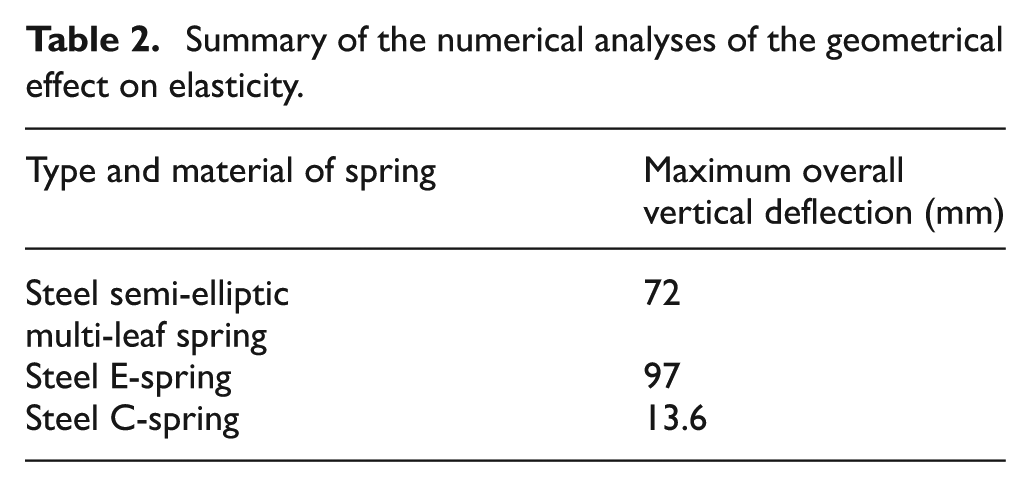
As can be gathered from Table 2, the E-like-shaped spring demonstrates highest maximum overall vertical deflection in comparison with the other geometries which proves the geometrical effect on elasticity.
Conclusion
Just like today’s mass produced materials such as steel, producing BMGs in large quantities would likely offset much of the overhead costs per unit of product associated with small-scale production. The combination of fracture toughness, strength and ductility that can be achieved by alloying an MG matrix, such as steel matrix, with nano-meter-sized elements while exploiting amorphous cross-linking of the matrix extends beyond the benchmark ranges established by the toughest and strongest materials known. This combination thereby pushes the envelope of damage tolerance accessible to a structural metal. The proposed heterogeneous nanocomposite ferromagnetic MG alloy exhibits features that are promising in applications such as springs. The combination of properties of the proposed alloy is promising as well in other applications such as transformer magnetic cores and motor/generator parts in vehicle powertrains. Given the superior properties of the proposed alloy, it is expected to be more cost-effective than widely used alternatives such as low-carbon Fe–Mn martensite steel that needs to be subjected to ageing heat treatment. Environment wise the study advantageously utilizes recyclable material of superior characteristics to manufacture vehicle suspension springs so that reducing long-term impact on the environment.
Footnotes
Acknowledgements
The author would like to thank Professor Sayed M. Metwalli and Professor Badr S.N. Azzam from Cairo University, Egypt; Professor Hasan H. Dadoura from Helwan University, Egypt and Professor Nicola Pugno from the University of Trento, Italy.
Academic Editor: Yunn-Lin Hwang
Declaration of conflicting interests
The author declares that there is no conflict of interest.
Funding
Cairo University, Egypt, is thanked for the support provided.
