Abstract
Meeting the growing demand for sustainable energy is one of the major challenges of the 21st century. In the Kyoto conference on global climate change, numerous nations committed to large reductions in greenhouse gas emissions. Reduced greenhouse gas emissions are a major advantage of using biodiesel on diesel engines. The test uses three biodiesels (soybean oil, palm oil, and waste edible oil) in the engine, comparing engine performance and exhaust emissions with the baseline (pure petrodiesel) case. The results indicated that the use of biodiesel produces decreased smoke opacity but increased fuel consumption up to 7.38% compared with petrodiesel. Exhaust emissions, such as CO and HC, were reduced using biodiesel in fuels, primarily because of the effect of complete combustion of the oxygen contained in biodiesel.
Introduction
Rapid urbanization and economic growth of developing countries have produced air pollution, particularly in the road transport sector. The increasing demand for petroleum-based fuels and their combustion in internal combustion engines have had adverse effects on air quality, human health, and global warming. This has triggered awareness to find alternative sources of sustainable energy. Similar to petroleum diesel, biodiesel can be used in compression ignition engines with little or no modification. The primary advantages of using biodiesel are its renewability and superior quality of exhaust gas emissions. Biofuels are renewable, can supplement HC fuels, assist in their conservation, and mitigate their adverse effects on the environment. To fulfill emission regulations and contribute to the reduction of CO2 from transportation, one strategy is the use of cleaner fuels. Bioderived biodiesel fuels are viable candidates as a renewable alternative and for meeting bioenergy consumption targets in the transport sector.1–3
Many countries are seeking alternative fuels for diesel engines. So far, biodiesel is one of the alternative fuels being promoted. The search for alternative fuels is motivated by the goals of sustainable development, energy conservation, efficiency, and environmental preservation. Nearly all (97%) of the fossil fuel energy consumption in the transportation sector is from oil. However, the use of various fossil fuels, such as petroleum products and coal, leads to several environmental problems such as reduction in underground-based carbon energy sources and subsidence of the ground surface after the extraction of fuels and minerals. Among them, biodiesel seems a very promising resource.4–7
Monoalkyl esters can be used in modern diesel engines in their pure biodiesel forms or can be blended with petroleum diesel. In addition to being renewable, biodiesel is biodegradable, nontoxic, has a higher flash point, causes reduced exhaust emissions, and is more eco-friendly than petroleum diesel fuel. 5 The physical and chemical properties of biodiesel fuel have been shown to be similar and compatible with those of petrodiesel fuels. Vegetable oil–based diesel fuel substitutes have proven to be clean and effective, and they apparently increase overall lubricity when added to diesel fuel at nominally low levels. Biodiesel can be produced from vegetable oils, as well as edible and nonedible and animal fats. Edible oil resources, such as soybeans, palm oil, sunflower, safflower, rapeseed, coconut, and peanut, are considered the first generation of biodiesel feedstock because they were the first crops to be used for biodiesel production.7,8
Waste cooking oil is a promising alternative to vegetable oil because of its reduced raw material cost. Restaurant waste oils and rendered animal fats are less expensive than food grade soybean oil. Waste edible oil (WEO) cannot be discharged into drains or sewers because it causes blockages, odors, and vermin problems and may also pollute watercourses, causing problems for wildlife. Although WEO was once used as an ingredient in animal feed, it was banned by the European Union because of animal health hazards.9,10 The disposal of large amounts of WEO is problematic in numerous countries. Production of biodiesel from waste cooking oil is one of the less problematic methods of using it efficiently and economically. Looking at the requirement for diesel fuel and the availability of waste cooking oil, the biodiesel obtained from waste cooking oil could partially replace diesel fuel.11,12
Biodiesel fuels can be used in high-pressure combustion engines, such as common rail injection engines in which high injection pressures allow rapid atomization and combustion resulting in higher efficiencies and lower emissions. When burned, biodiesel produces pollutants with few detrimental human health effects. In addition, its lubricity is superior to that of diesel fuel.13–15 Biodiesel can be blended in any proportion with mineral diesel to create a biodiesel blend or can be used in its pure form.
Demirbas 16 reported that oxygen improves the efficiency of combustion, but it takes up space in the blend and thus slightly increases the apparent fuel consumption rate observed while operating an engine with biodiesel. Altiparmak et al. 17 tested various blends of biodiesel and pure petrodiesel in a single-cylinder direct injection diesel engine at full-load capacity. They observed that the engine torque and power output with biodiesel and petrodiesel fuel blends increased by up to 6.1% and 5.9%, respectively. Agarwal 18 evaluated the performance characteristics of biodiesel and various blends in a direct injection diesel engine. It was found that heating the biodiesel was adequate to reduce the viscosity and close range to diesel. Based on brake-specific fuel consumption (BSFC), thermal efficiency, and smoke opacity, 200 bar is the optimal fuel injection pressure for biodiesel oil and diesel. Suryawanshi and Deshpande 19 reported that at partial and full loads, HC emissions were significantly lower in biodiesel blends than in pure petrodiesel. The HC emissions of a fuel blend can be reduced further by increasing the biodiesel content. Unburned HC can be reduced by 53% in engines operated with biodiesel. Charles and Todd 20 concluded that complete combustion converts HC fuels to CO2 and water. Diesel fuel, represented by C16H34, releases 3.11 kg of CO2 per kilogram of fuel used in combustion. Biodiesel, represented by C22H43O2, releases 2.86 kg of CO2 per kilogram of fuel used in combustion. According to Pal et al., 21 a 30% biodiesel blend of petrodiesel showed a higher brake power, brake thermal efficiency, reduced BSFC, and smoke opacity compared with pure petrodiesel.
Karaosmanoglu et al. 22 studied the effects of biodiesel on engine behavior over a 50-h period and observed no remarkable changes. The results were considered promising for this fuel, although diesel engines are expected to last much longer than 50 h. In Karaosmanoglu et al., 22 an engine running on sunflower oil showed no significant decrease in power, fuel consumption, and balancing force. In Charles and Todd, 20 rapeseed methyl ester emission tests showed a reduction in unburned hydrocarbon (55.6%), carbon monoxide (50.6%), and nitrogen oxides (11.8%) and an increase in carbon dioxide (1.1%) and particulate matter (PM; 0.3%). The PM results did not significantly differ from those for diesel. 7 Helwani et al. 23 reported that combustion of neat biodiesel decreases carbon monoxide (CO) emissions by 46.7%, PM emissions by 66.7%, and unburned hydrocarbons by 45.2%. Pramanik 24 reported the performance of using biodiesel and diesel fuel blends in diesel engine. There are significant improvements in engine performance when compared to biodiesel. The specific fuel consumption was reduced due to decrease in the viscosity of the biodiesel.
Many studies of the effects of biodiesel on engine performance consistently show slightly decreased torque and power with increased specific fuel consumption.8–14 Although reported emission results for biofuels vary, most studies report lower CO, HC, and smoke emissions and higher NOx emissions. These reductions in exhaust emissions have caused changes in engine design for biofuel use and control of the fuel injector system. Therefore, in making an attempt to promote the use of biodiesel, it is important to evaluate the performance and engine exhaust emission characteristics of biofuel used in a diesel engine. The studies have shown that biodiesel produces substantially lower CO, CO2, PM, polyaromatic HC, nitrated polyaromatic HC, sulfur oxides (SOx), and HC emissions than pure petrodiesel. Although many studies have confirmed acceptable performance results for engines operating with biodiesel, very few have performed a long-term analysis of engine exhaust emissions. There is a need to study possible usage of other biodiesel at large scale and their impact on human health and environment. Other biodiesels need to be examined for engine performance and emission characteristics.
Experimental procedure
This study was a comparison of biodiesel and pure petrodiesel in terms of engine exhaust emitted by a diesel engine. The water-cooled diesel engine used in the engine test had a four-cylinder four-stroke engine with direct injection (Figure 1). The engine was operated at 3000 r/min at a constant 70% throttle position. The engine operation showed good stability at 70% rather than 100% throttle position. Hence, for safety reasons, the 70% throttle was chosen. The engine specifications are shown in Table 1. The essential fuel properties are given in Table 2. To examine the emission characteristics, a portable SINCRO exhaust gas analyzer (model EGA2001C) was used to measure the concentration of exhaust gases of the test engine such as HC and NOx (ppm); CO, CO2, O2, and smoke (%vol) and exhaust gas temperature (°C).

Experimental setup of the diesel engine testing system.
Test engine specifications.
DOC: diesel oxidation catalyst; PCV: positive crankcase ventilation; EGR: exhaust gas recirculation.
Base fuel properties.

WEO: waste edible oil.
Results and discussion
Various types of diesel fuel were used for the same testing procedures, which included pure petrodiesel, soybean oil, palm oil, and WEO. The type of testing engine used was a four-stroke diesel engine. Pollutants from diesel engines included CO, HC, CO2, NOx, and smoke. Most of the diesel engines run on exclusively lean mixtures, such as 14:1 times of stoichiometric air–fuel ratio. Therefore, the CO emission is typically lower than that of gasoline engines. 25 The CO emissions for various types of fuel at 3000 r/min are shown in Figure 2. The CO emission at 3000 r/min was 0.103%, 0.068%, 0.070%, and 0.077% for pure petrodiesel, soybean oil, palm oil, and WEO, respectively. Soybean oil has the lowest CO emission, followed by palm oil, and WEO. On average, soybean oil, palm oil, and WEO reduced CO emission by 33.9%, 32.0%, and 25.2%, respectively.

CO emissions for various types of fuel.
HC emissions were the result of incomplete fuel combustion from flame quenching in crevice regions in the cylinder wall. A comparison of the unburned HC emissions of various fuels at 3000 r/min is shown in Figure 3. The HC emission at 3000 r/min was 28.0, 22.8, 26.2, and 25.1 ppm for pure petrodiesel, soybean oil, palm oil, and WEO, respectively. It can be seen that the lowest level of HC was produced from soybean oil fuel, followed by WEO and palm oil. The soybean oil contained 82% fatty acid and produced complete combustion compared with palm oil and WEO fuels. The oxygenated compounds available in the biodiesels improve the fuel oxidation reducing HC emissions. 18 When the oxygen content of fuel is increased, it requires less oxygen for combustion. However, oxygen content of fuel is the main reason for more complete combustion and HC emission reduction. Another, higher cetane number of biodiesels reduces the combustion delay, and such a reduction has also been related to decreases in HC emissions.26–28
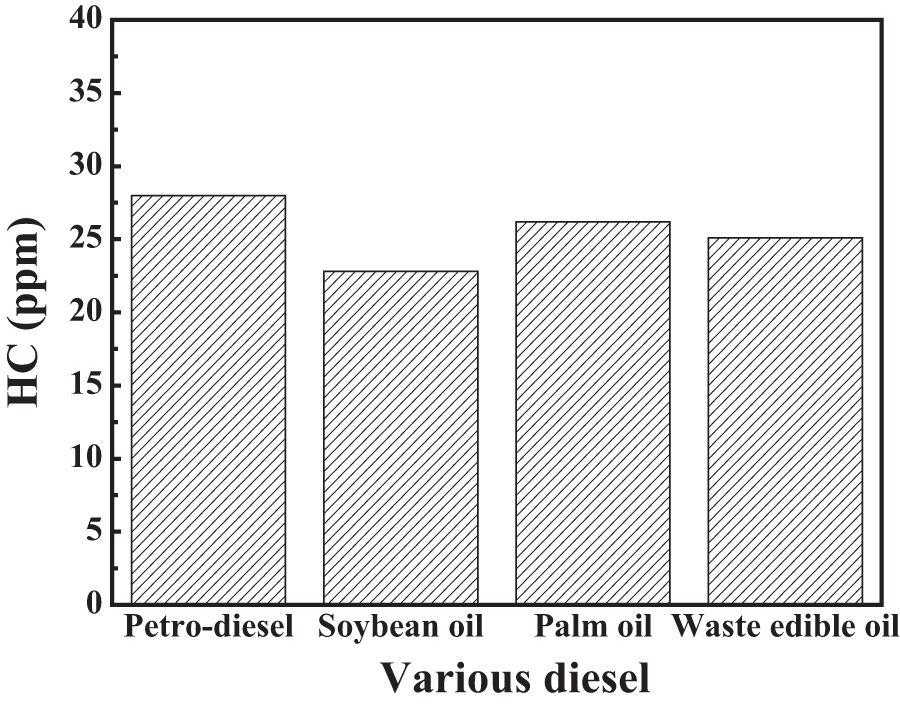
Hydrocarbon emissions for various types of fuel.
The CO2 is the key parameter that indicates the combustion performance of fuel. Increasing CO2 emission indicates increased combustion. A comparison of the CO2 emissions of various fuels at 3000 r/min is shown in Figure 4. The maximum CO2 emission was observed for biodiesel, resulting from increased combustion at the current throttle-speed engine operating condition. The CO2 emission for all types of fuel was found at 3000 r/min: 2.51%, 2.64%, 2.72%, and 2.87% for pure petrodiesel, soybean oil, palm oil, and WEO, respectively. WEO oil produced higher amounts of CO2 than palm oil and soybean oil fuels. This is mainly due to the effect of high unsaturated fatty acid in WEO. The WEO contains 81% fatty acid and produces complete combustion compared with palm oil and soybean oil fuels.

CO2 emissions for various types of fuel.
Compared with pure petrodiesel, soybean oil increases NOx emissions by 10.34%, palm oil reduces them by 22.4%, and WEO increases them by 31.0% (Figure 5). The difference was primarily due to the increase in combustion temperature caused by the biodiesel fuels, which can be attributed to biodiesel fuels producing a higher heat release rate at the premix combustion phase, which higher the peak combustion temperature and increase NOx emissions. Pradeep and Sharma 29 suggested that using hot exhaust gas recirculation to control NOx in diesel engine fueled with biodiesel can effectively reduce NOx emission, which lowers peak combustion temperature and thus reduces NOx emissions. In addition, the NOx emission is strongly related to lean fuel with high cylinder temperature or high peak combustion temperature. A fuel with high heat release rate at premix or rapid combustion phase and lower heat release rate at mixing controlled combustion phase will increase NOx.11,25
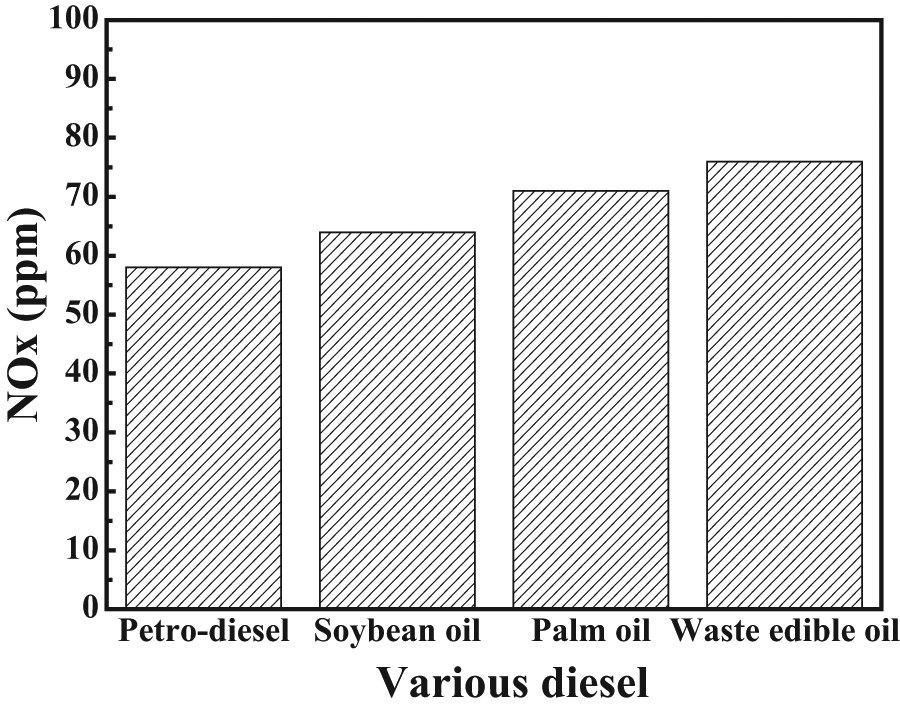
Nitrogen oxide emissions for various types of fuel.
Smoke is strongly related to the fuel cooling effect, as well as the air–fuel mixing problem that increases combustion delay. WEO oil showed the lowest smoke emission level compared with palm oil and soybean oil (Figure 6). The ignition delay of WEO oil was shorter than that for pure petrodiesel, which affected smoke reduction. The lowest smoke emission was observed for WEO oil, followed by soybean oil and palm oils. This implied that WEO oil produced a more complete combustion than palm oil and soybean oil. On average, the WEO oil and soybean oil fuels showed 18.5% and 20.3% less smoke opacity than pure petrodiesel fuel did. Because biodiesel is free from sulfur, less sulfate emissions and particulate reduction were reported in the exhaust.
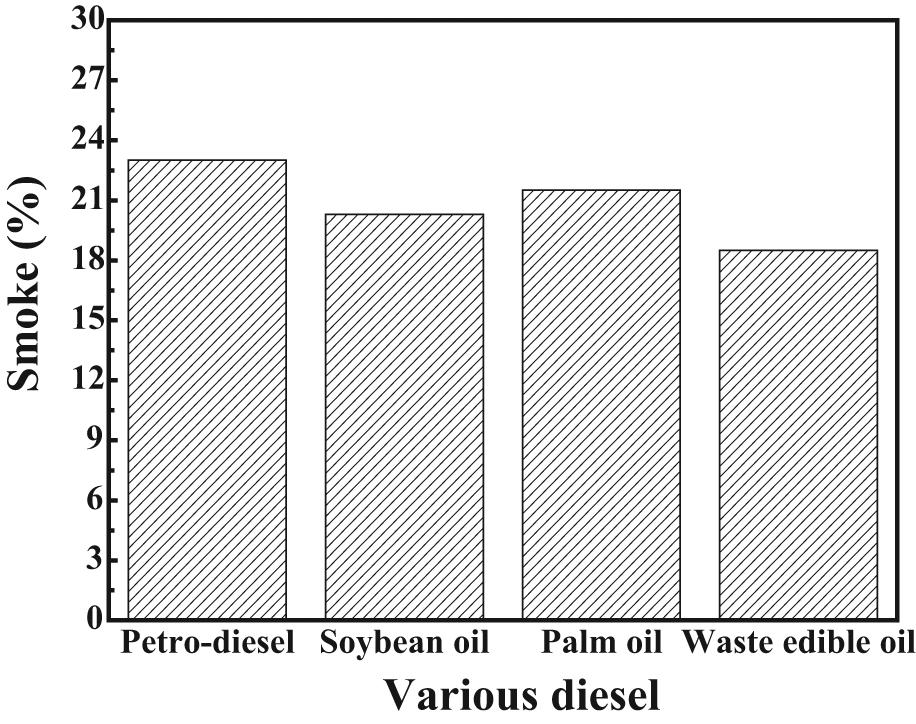
Smoke emissions for various types of fuel.
The O2 emission of various fuels from the diesel engine is shown in Figure 7. At 3000 r/min, the O2 concentration recorded was 16.62%, 17.85%, 17.47%, and 18.97% for pure petrodiesel, soybean oil, palm oil, and WEO, respectively. Soybean oil, palm oil, and WEO showed higher O2 concentrations because soybean oil, palm oil, and WEO contain higher amounts of O2 than pure petrodiesel.

Oxygen emissions for various types of fuel.
The variation in exhaust gas temperature for all tested fuels is shown in Figure 8. The lowest level of exhaust gas temperature was found in pure petrodiesel followed by WEO, palm oil, and soybean oil. Exhaust temperature for biodiesel fuels varied between 321°C and 344°C, as compared with 321°C for petrodiesel, indicating limited variation in exhaust temperature; this could be due to the same quantity of fuel being consumed per hour for diesel and biodiesel fuels. Both the WEO and palm oil contain fuel-inbound oxygen molecules, but the fatty acid in WEO is highly saturated (80%) compared with palm oil (50%). The highly saturated fatty acid (in WEO) does not increase combustion products compared with palm oil. The saturated fatty acid is sufficient to produce complete combustion, and it could represent a good diesel fuel to reduce exhaust emissions.

Exhaust gas temperature for various types of fuel.
The variation in fuel consumption for all tested fuels is shown in Figure 9. The lowest level of fuel consumption was found in petrodiesel, followed by soybean oil, WEO, and palm oil. The high fuel consumption obtained by soybean oil, palm oil, and WEO in comparison with petrodiesel is mainly owed to their respective lower heating values. However, higher oxygen content also led to lower calorific values, which may result in substantial power losses and the increase in specific types of fuel consumption.
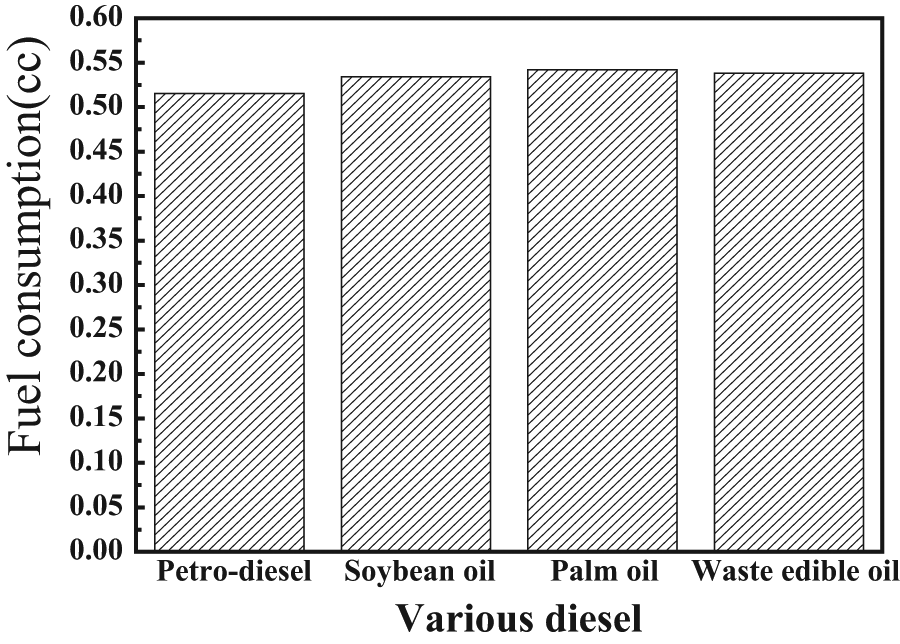
Fuel consumption for various types of fuel.
Compared with pure petrodiesel fuel, biodiesel has several superior combustion characteristics. For example, when biodiesel is used as an engine fuel, its approximately 10 wt% oxygen content may promote a more complete combustion and effectively reduce the production of unburned HC, CO, as well as suspended aerosol carbon granules (smoke); however, NOx emissions slightly increase. These increased fuel consumptions for biodiesels are because they contain oxygen content in the fuels, which result in the lower heating value. The lower heating values and higher densities of those biodiesels require larger mass fuel flows for the same energy output from the engine, leading to the increase in the fuel consumption to compensate the reduced chemical energy in the fuel. 28
Conclusion
The tests apply four diesel fuels on the engine performance, and exhaust emission results are compared to the baseline (pure petrodiesel) case. Results show that the fuel consumption is increased by 7.38% for the biodiesel case, the engine combustion temperature can be increased by 7.17% for the biodiesel case, the O2 emission can be increased by 14.14%, CO emissions can be reduced up to 25.24%, HC emissions can be reduced up to 18.57%, and smoke emissions can be reduced up to 19.6%. Thus, the biodiesel can be applied in the diesel fuel to upgrade the engine performance. The exhaust emissions of biodiesel are also lower than those of pure petrodiesel because of the presence of O2 in the molecular structure of the biodiesel. Biodiesel is increasingly used instead of pure petrodiesel because it is a renewable and sustainable alternative fuel for compression ignition engines.
Footnotes
Academic Editor: Ruey-Jen Yang
Declaration of conflicting interests
The author declares that there is no conflict of interest.
Funding
This work was supported by the National Science Council of the ROC with grant no. NSC 101-2221-E-539-003.
