Abstract
For tribologists, continuing challenges in engine design include reduced emissions and increased operating temperatures. Regulating the sulfur content in diesel fuel is expected to reduce the lubricity of these fuels, which may result in increased wear and damage of fuel injection systems in diesel engines. However, the use of biodiesel improves lubricity and cetane number requirements, particularly in ultra-low sulfur petro-diesel. In this study, the effect of temperature on the tribological properties of palm biodiesel was investigated using a wear machine. Palm biodiesel was used as a lubricating additive to petro-diesel at six concentrations of 2%, 5%, 10%, 20%, 50%, and 100%, and these biodiesel blends were named as B2, B5, B10, B20, B50, and B100, respectively. The tribological experiments were performed using the ball-on-ring method. Tests were conducted at 30°C, 60°C, 90°C, and 120°C, under a normal load of 50 N for 2 h and at a speed of 500 r/min. The wear scar diameter and the worn surface of tribopairs were studied and analyzed. According to the wear experimental results, the addition of a small amount of palm biodiesel to petroleum diesel can bring about a considerable reduction in the wear and friction under boundary lubrication. Results show that friction and wear increase with increasing temperature.
Introduction
Increasing industrialization, modernization, and development has led to a high demand for petroleum worldwide. In the Kyoto conference on global climate change, nations worldwide have committed to reducing greenhouse gas emissions. The use of various fossil fuels, such as petroleum products and coal, leads to several environmental problems such as reduction in underground-based carbon energy sources and subsidence of the ground surface after the extraction of fuels and minerals. Because of the environmental impacts and fluctuating prices of petroleum products, biodiesel has drawn considerable attention in recent years as a clean and renewable fuel. 1 The world is presently confronted with the twin crises of fossil fuel depletion and environmental degradation. Recently, because of increase in crude oil prices, limited resources of fossil oil and environmental concerns, there has been a renewed focus on vegetable oils and animal fats to make biodiesel. These reductions in exhaust emissions have been caused by engine design such used biofuel and control of the fuel injection. Therefore, an attempt has been made on promotion and application for biofuel that is important to evaluate the performance and lubricity characteristics using biodiesel fuel blends on a diesel engine.2,3
Increasingly strict regulations of sulfur content in commercial petroleum diesel fuels decrease their lubricity. This reduced lubricity can cause damage to the engine and fuel injection systems that use these fuels. This will lead to drastic reduction in sulfur content and increase in cetane number, which in turn will adversely affect the lubricity characteristics of the diesel fuel. 4 However, the use of biodiesel improves lubricity and cetane number requirements, particularly in ultra-low sulfur diesel. Biofuels are renewable, can supplement hydrocarbon fuels, assist in their conservation, as well as mitigate their adverse effect on the climate. The advantages of biofuel include increased employment and improved economic conditions, particularly in rural areas; enhanced energy security owing to reduced dependence on oil imports; foreign exchange savings; reduced vehicular pollution; and only a negligible contribution to global warming. 5
Interest in diesel fuel lubricity has increased since the early 1990s. It was at this time that ultra-low sulfur diesel fuel (ULSDF < 50 ppm) was commercialized, and coincident with this, there was an increased incidence of drivability problems and pump failures. The ULSDFs showed that their inherent lubricity was unacceptable. Therefore, to promote the use of biofuel, the performance and lubricity characteristics of biofuel blends used in a diesel engine must be evaluated.6,7 Biodiesel derived from these sources can generally be classified as mono-alkyl esters of long chain fatty acids. In addition to being a renewable fuel source, biodiesel has several attractive features. Biodiesel is biodegradable, non-toxic, has higher flash point, causes reduced exhaust emission, and eco-friendly with petroleum diesel fuel.8,9 Engine design and tribology engineers are constantly challenged to develop advanced method and products with low emission, and higher operating temperatures. The resulting engine systems must also meet more demanding emissions and fuel economy targets. The main factor in the durability and tribological behaviors of engine components and lubricants is fuel characteristics.
A diesel engine can perform satisfactorily on biodiesel blends without any engine hardware modifications. Long-term endurance test using biodiesel proved that biodiesel can be used for substituting mineral diesel in long run. Smoke emissions decrease appreciably when biodiesel is used in the engine. Esterification has proven effective for preventing long-term problems associated with utilization of vegetable oils such as fuel filter plugging, injector coking, formation of carbon deposits in combustion chamber, ring sticking, and contamination of lubricating oils.10–12 Other advantages of biodiesel are low engine wear, low cost, and easy availability. When burned, biodiesel produces pollutants with few detrimental human health effects. Since biodiesel is free from sulfur, less sulfate emissions and particulate reduction are resulted in the exhaust. Palm oil has great potential to meet future demand because of its high production rate and its high oil content. Palm oil (5950 L per hectare) has the highest oil productivity, which is approximately 13 times higher than that of soybean oil and followed by Calophyllum inophyllum oil.13,14 Since palm biodiesel is an oxygenated fuel, its more complete combustion compared to conventional fuels results in lower CO emissions in the exhaust.
Demirbas 15 stated that biodiesel provides significant lubricity improvement over petro-diesel fuel. Lubricity results of biodiesel and petro-diesel using industry test methods indicate that there is a marked improvement in lubricity when biodiesel is added to conventional diesel fuel. Bettis et al. 16 studied that use of biodiesel in diesel engine results in a slight reduction in brake power and a slight increase in fuel consumption. The exhaust emissions of biodiesel are also lower than those of diesel due to the presence of oxygen in the molecular structure of the biodiesel. Moreover, biodiesel fuel is environmentally friendly because it does not produce SOx and does not contribute to global CO2 emissions. Fazal et al. 17 investigated the effect of speed and biodiesel concentration on wear scar diameter and friction coefficient by four-ball wear machine. They found that wear scar diameter and friction coefficient had an almost linear relationship with engine speed. However, both wear scar diameter and friction coefficient were negatively associated with the concentration of biodiesel in the blend.
One of the main factors in the durability and tribological behavior of engine components and lubricants is fuel characteristics. New engines tend to require improved diesel fuel lubricity to avoid excessive wear of the fuel injection system. Lubricity is a critical property of diesel fuel because it affects engine performance. Current and future regulations are expected to reduce the sulfur content of diesel fuel. Low lubricity (i.e. low sulfur content) increases diesel engine wear and damages the fuel injection system. Alternative types of fuel have reduced diesel engine exhaust emissions and improved the friction and wear properties of engine parts. Biodiesel reduced long-term engine wear in diesel engines, and it was an effective lubricant, demonstrating a superior performance compared with petro-diesel. The possible uses of other types of biodiesel and their effects on human health and the environment warrant large-scale studies.
Non-sulfur polar compounds were also eliminated from petro-diesel during the desulfurization process and it was likely the loss of these compounds and not the sulfur-containing materials that caused the loss of lubricity. Poor lubricity is linked with the failure of engine parts. The biodiesel exhibits improved load capacity wear resistance. Analysis of lubricity characteristics of the biodiesel fuel blends showed a nonlinear relationship between biodiesel concentration and wear loads. However, the lubricant properties of biodiesel were more effective than those of petro-diesel and increased engine life. The use of biofuels as diesel engine fuels can play a vital role in helping the developed and developing countries to reduce the environmental impact of fossil fuels.
Although many studies have confirmed the acceptable performance results of engines operating with biodiesel, few studies have analyzed wear and maintenance at different temperatures. The use of diesel engines has increased in recent years because of their high fuel efficiency and low emissions. However, the tribological behavior and durability of the fuel injection component of an engine that runs on biodiesel are unclear. In this study, the tribological measurements included motion resistance and wear. The tribological properties of biodiesel used as an additive to pure petro-diesel were examined using a ball-on-ring wear tester to determine the optimal concentration; the mechanism of the reduction of wear and friction was investigated using optical microscopy (OM).
Experimental procedure
This study investigated the effect of temperature on the lubricity of different palm biodiesel blends. The essential fuel properties are given in Table 1. The objective was to determine the correlation between wear quantities being measured and percentage of biodiesel content in fuel oil. All test fuels were mixed for 30 min at 3000 r/min by a mechanical homogenizer machine. The blends contained no water molecules and showed no phase separation. A biodiesel additive was added to ULSDF (less than 50 ppm) at weight percentages of 0 wt%, 2 wt%, 5 wt%, 10 wt%, 20 wt%, 50 wt%, and 100 wt% (labeled as B0, B2, B5, B10, B20, B50, and B100). The resulting engine systems must also meet the strict emissions and fuel economy targets that have been recently established. Cold starts require diesel engine components to work at room temperature (28°C–31°C). Some of the diesel engine components work at high temperatures. Hence, wear tests were conducted at four temperatures, 30°C, 60°C, 90°C, and 120°C, to evaluate the tribological behaviors of the diesel under normal diesel engine operating temperatures. The tribological experiments were performed using the ball-on-ring method (Figures 1 and 2). The wear and friction characteristics of biodiesel blends were investigated at four temperatures from 30°C to 120°C under a normal load of 50 N for 2 h at a speed of 500 r/min.
Base fuel properties.


Ball-on-ring motion.

TE-53 wear test rig.
The ring was made of bearing steel (AISI 52100 steel with HRC of 60–62, of composition: 0.95–1.05% C, 0.15–0.35% Si, 0.2–0.4% Mn, <0.027% P, <0.020% S, 1.3–1.65% Cr, <0.3% Ni, and <0.25% Cu) with 70 mm diameter and thickness of 17 mm. The ball used in this study was made of bearing steel GCr15 with hardness of HRC 59–61 and diameter of 12.7 mm. Both surfaces were polished to roughness Ra = 0.05 µm for ball and Ra = 0.20 µm for ring. The test conditions were as defined by the ISO standard defining air conditions for testing diesel fuel lubricity. The relative humidity was kept between 50% and 55% while the mean ambient temperature in the laboratory was maintained at an approximate temperature of 28°C–31°C. The mean diameter of wear scar on the ball was then measured from five identical tests using a digital-reading microscope with accuracy of 0.01 mm. The friction coefficients were calculated from the applied load and measured strain on the ball-on-ring tester. The steel balls were rinsed in ethanol immediately and then the topography of the scar surfaces after the wear test was observed by OM. After each test was competed, all components in contact with the tested fuels were cleaned with toluene and acetone.
Results and discussion
Fuel blend stability tests were conducted to evaluate whether phase separation of the fuel blends occurred. The test was performed using 0%, 2%, 5%, 10%, 20%, and 50% biodiesel by weight in petro-diesel fuel. The blended fuels were maintained at 30°C, and the stability was tested every 2 h for the first 24 h and every day thereafter for 2 months. During this time, no precipitation or opacity was observed to determine any differences in the stability and miscibility between the six fuels (Figure 3).

The sedimentation of biodiesel in petro-diesel of pure petro-diesel (left), 2% biodiesel, 5% biodiesel, 10% biodiesel, 20% biodiesel, and 50% biodiesel (right).
Figure 4 shows the wear scar diameter and friction coefficient for different additive concentrations of biodiesel in petroleum diesel obtained by tribological test at room temperature (30°C), which confirmed that adding biodiesel to petroleum diesel can reduce the wear scar diameter of steel balls in comparison with pure petroleum diesel. A wear scar was clearly largest (diameter, 1.39 mm) in the case of pure petro-diesel. The optimal concentration of biodiesel in terms of minimizing wear scar diameter and friction coefficient is 10 wt%.
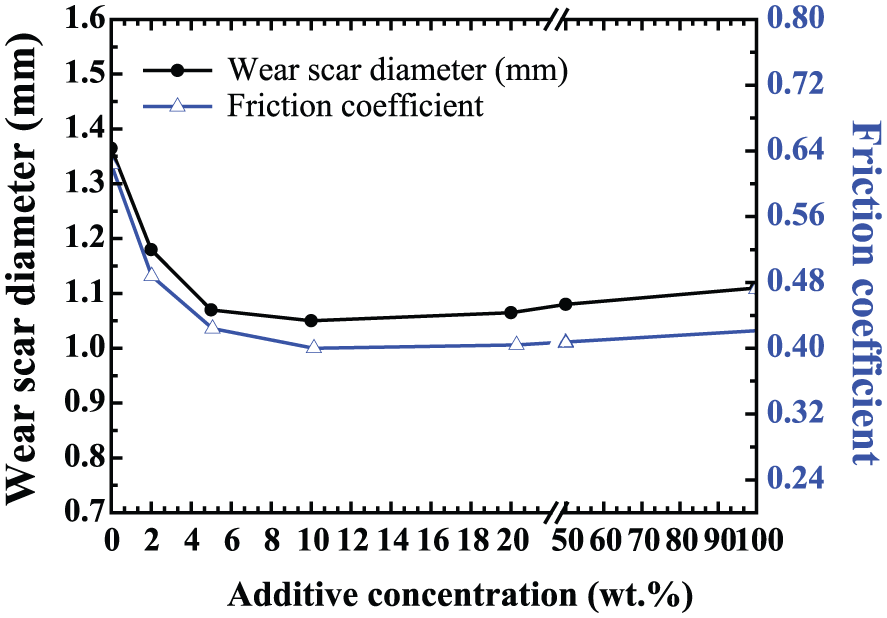
Wear scar diameter and friction coefficient related to the additive concentration of biodiesel in pure petroleum diesel at room temperature (ball-on-ring, 500 r/min, Ra: 0.20 µm, 50 N, 120 min).
Apparently, 2–5 wt% content of the biodiesel in petro-diesel oil considerably reduces the diameter of a wear scar compared to that of petro-diesel. A pronounced minimum can be observed in the concentration range of 10%–20%. It is found that the lubricity in terms of friction and wear increases with increasing concentration of biodiesel. This difference was also attributable to the presence of aliphatic fatty acids in biodiesel, such as stearic acid, which could enhance lubrication properties by controlling friction and wear between contact surfaces by developing lubrication films. The oxygen-containing compounds and the mixture of several fatty acids in methyl esters which were adsorbed onto the rubbing surface reduce friction and improve the film thickness of the boundary lubrication regime. This trend is also seen in the incremental viscosity as the chain length increases. However, at biodiesel concentrations above 10%, the wear scar diameter slightly increased, and small amounts of metal merged. This may be attributable to oxidation or chemical corrosive wear during the wear process.
The percentage share of the lubricant film covering the surface under friction conditions increases as concentration increases. The protective films can reduce thermal energy in sliding contact and thereby improve lubricity. These lubricity-enhancing films are most likely based on carboxylic acid moiety. Another possible explanation is trace components in the biodiesel fuel, such as free fatty acids, monoglycerides, and diglycerides, which reportedly improve the lubricity of biodiesel. Working temperature plays a crucial role in lubricant wear performance. Hence, wear tests were conducted at different temperatures, such as room temperature (30°C), 60°C, 90°C, and 120°C, to evaluate the tribological behaviors of the biodiesel blends at normal diesel engine operating temperatures. At room temperature, Figure 5 shows that B10 exhibits the highest antiwear performance, followed by B5 and B2. All biodiesel blends yield a smaller wear scar diameter compared with pure petro-diesel. The wear scar diameter decreases drastically for B2 and B5 compared with pure petro-diesel.

Variation of wear scar diameter with temperature for different concentration of biodiesel (ball-on-ring, 500 r/min, Ra: 0.20 µm, 50 N, 120 min).
Some diesel engine components work at high temperatures, which may induce dissimilar wear mechanisms. To investigate the effect of biodiesel on tribological behavior, the blended fuels were tested at 30°C–120°C. Figure 5 shows that the wear scar diameter increased slightly with increasing temperature. At 120°C, a superior antiwear performance was observed for B5 and B10 because of the promoting effect of biodiesel with the effective formation of physical adsorption films. At high temperatures, the effect of physical adsorption is considerably weak, suggesting that the tribological behavior is dominated by chemical adsorption and chemical reaction films.18,19
Figure 6 shows that the friction coefficient for different biodiesel blends increases somewhat with increasing temperature. The biodiesel blends have a lower friction coefficient than that of pure petro-diesel, and higher blends tend to have a lower friction coefficient. The magnitude of the friction coefficient confirms that the lubrication mode is located in the boundary lubrication regime, and the properties of the boundary films dominate the wear behavior. Figure 7(a)–(d) shows the typical frictional behaviors of the ball-on-ring wear tests for pure petro-diesel (B0), B2, B5, and B10, indicating that the coefficient of frictions tested for B5 and B10 are the most stable at room temperature (30°C). In addition, variations in the friction coefficients for B5 and B10 confirm a short run-in process, which can reduce the amount of wear. Figure 7(e)–(h) shows the typical frictional behaviors tested for the biodiesel blends at 120°C. Studies have indicated that increasing the oxygen content improves the friction and wear properties of pure petro-diesel. The lowest friction coefficient was observed when pure petro-diesel was contaminated with 10% biodiesel (B10). The analysis of fuel properties reveals that biodiesel has the advantages of a high cetane number, extremely low sulfur and aromatic contents, and favorable lubricity. Biodiesel is primarily composed of fatty acid methyl ester compounds, and it has a higher dynamic viscosity than that of petro-diesel and is similar to surface active agents that have a high affinity toward metals. In addition, biodiesel can be used as an oil additive for enhancing lubrication and reducing wear.20–22 The laboratory wear test data for biodiesel as compared to that of diesel fuel have been summarized in Table 2. 17

Variation of friction coefficient over last 2 h for each test with temperature for different concentration of biodiesel.

Typical friction behavior of ball-on-ring tested in the various oils at 30°C and 120°C. (ball-on-ring, 500 r/min, Ra: 0.20 µm, 50 N): (a) B0 (30°C), (b) B2 (30°C), (c) B5 (30°C), (d) B10 (30°C), (e) B0 (120°C), (f) B2 (120°C), (g) B5 (120°C), and (h) B10 (120°C).
Wear data from laboratory wear test: wear in biodiesel as compared to diesel. 17

L = less wear, S = similar wear, H = higher wear, HFRR = High-Frequency Reciprocating Rig, and RME = Rapeseed Methyl Ester.
Figure 8(a)–(d) shows the worn surfaces of the ball specimens tested at room temperature (30°C) for the biodiesel blends. Figure 8(a) shows the microcutting and abrasive wear caused by hard particles penetrating the brittle solid surface. As shown in Figure 8(d), B10 demonstrates the highest antiwear performance. Comparing Figure 8(b)–(d) shows that the worn surfaces of B10 are smoother than those of B2 and B5. The percentage share of the lubricant film covering the surface under friction conditions increases as the biodiesel concentration increases. Protective films can reduce thermal energy on sliding contact, thereby improving lubricity.23–25 These lubricity-enhancing films are likely based on carboxylic acid moieties. Figure 8(e)–(h) shows the worn surfaces of the ball specimens tested at 120°C for the biodiesel blends. The worn surfaces tested at 120°C are comparatively darker than those tested at 30°C. The wear mechanisms of the worn surface tested for B2 and B5 are similar to those for B10 (Figure 8(f)–(h)). Figure 8(f)–(h) shows that the ball specimens tested for B2, B5, and B10 are characterized by abrasive wear. The wear scar profiles perpendicular to sliding direction on worn surfaces were analyzed using a Talysurf-120 profilometer. Figure 9 shows the steel ring surface roughness analysis results. The surface exposed to pure petro-diesel after the lubricity test revealed a residue at the wear scar periphery and had a surface roughness of 0.565 µm. Average roughness (Ra) determined through a surface profiler decreased from 0.531 to 0.304 µm after biodiesel usage, which indicated improved lubrication. The surface roughness analysis also showed good results. A possible reason is that the additional lubricity properties of the biodiesel reduced friction.

Optical microscopy micrographs of worn surfaces (ball-on-ring, 500 r/min, Ra: 0.20 µm, 50 N, 120 min): (a) B0 (30°C), (b) B2 (30°C), (c) B5 (30°C), (d) B10 (30°C), (e) B0 (120°C), (f) B2 (120°C), (g) B5 (120°C), and (h) B10 (120°C).
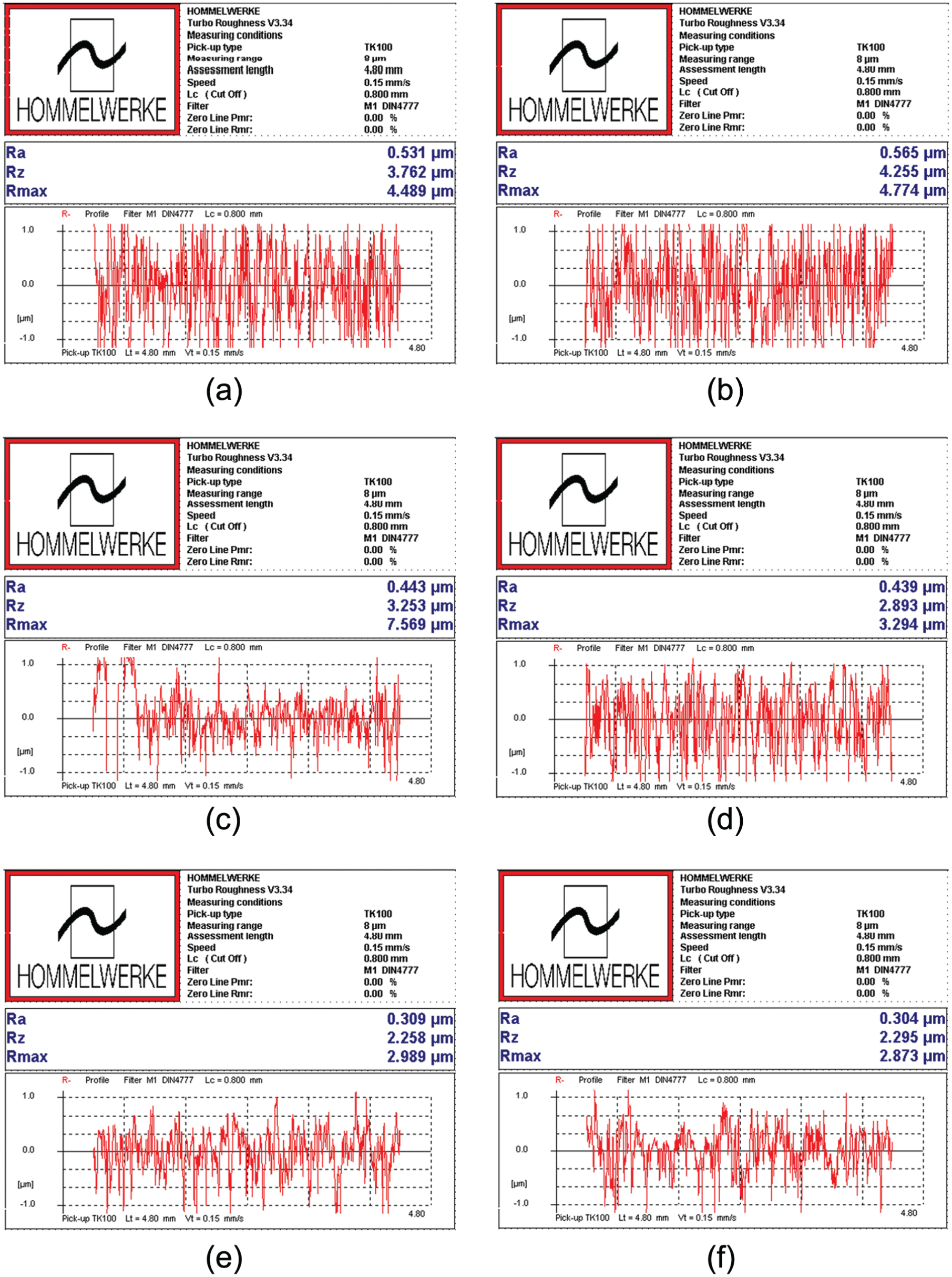
Roughness of the steel ring surface and rubbed surface after wear test (500 r/min, 50 N, 120 min): (a) profiles across the new steel ring, (b) rubbed surface lubricated by pure petro-diesel, (c) petro-diesel added by 10 wt% biodiesel, (d) petro-diesel added by 20 wt% biodiesel, (e) petro-diesel added by 50 wt% biodiesel, and (f) petro-diesel added by 100 wt% biodiesel.
Conclusion
This study analyzes the tribological effect of pure petro-diesel blended with 2%, 5%, 10%, 20%, 50%, and 100% biodiesel fuels. The experimental results are attributable to biodiesel contamination acting as a friction-reducing additive in pure petro-diesel. Regarding antifriction and antiwear properties, the lubricity of palm biodiesel increases as the biodiesel concentration increases. The low wear rate in biodiesel may result from fatty compounds. Biodiesel components, such as fatty acid methyl esters, free fatty acids, and monoglycerides, reportedly improve the lubricity of biodiesel, thereby substantially reducing wear tendencies. However, the lubricity of biodiesel at higher temperatures is relatively decreased.
Footnotes
Academic Editor: Mustafa Canakci
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the National Science Council of the R. O. C. with Grant no. NSC 101-2221-E-539-003.
