Abstract
Purpose
The aim of this study is to assess the impact of bridging intravenous thrombolysis (IVT), infarct core growth rate (ICGR) and their interaction on neurological outcomes in patients undergoing endovascular thrombectomy (EVT) in acute ischemic stroke (AIS) with anterior large vessel occlusion (LVO).
Methods
Patients undergoing EVT due to anterior LVO (ICA to M2 branches) between 2018 and 2022 in a tertiary care center were included. Patient's baseline characteristics, peri-procedural factors of EVT and neurological outcomes were retrospectively analyzed. ICGR was determined by infarct core volume on perfusion CT divided by the time from stroke onset to imaging. Endpoints included early neurological improvement (ENI), defined as NIHSS reduction of ≥8 points after 24 h or NIHSS < 2 at discharge, and intracranial hemorrhage (ICH) on CT at 24 h.
Results
Of 205 patients, 128 (62.4%) received bridging IVT. Neither bridging IVT, ICGR, nor their interaction was significantly associated with ENI or ICH occurrence. Factors significantly associated with ENI included short groin puncture to reperfusion time (OR = 0.98, CI 0.97–0.99, p < 0.001), low pre-stroke mRS (OR = 0.76, CI 0.65–0.89, p = 0.001), and high baseline CT ASPECTS (OR = 1.15, CI 1–1.31, p = 0.049). Factors associated with reduced odds of ICH were short symptom onset to admission time (OR = 1, CI 0.998–1, p = 0.021), short groin puncture to reperfusion time (OR = 1.01, CI 1–1.02, p = 0.039), and complete reperfusion (OR 0.89, CI 0.81–0.97, p = 0.009).
Conclusion
Neither bridging intravenous thrombolysis, infarct core growth rate, nor their interaction significantly impacted early neurological improvement or intracranial hemorrhage rate.
Introduction
In the last decade, numerous studies have demonstrated the efficacy of combining endovascular thrombectomy (EVT) with best medical treatment for improving neurological outcomes in patients with acute ischemic stroke due to anterior large vessel occlusion (LVO), compared to best medical treatment alone.1,2 Moreover, both national guidelines and those from the European Stroke Organisation (ESO) recommend the utilization of bridging intravenous thrombolysis (IVT) with EVT.3,4 Some earlier studies have suggested that direct EVT (DEVT) as a standalone procedure may be non-inferior to the combination of bridging IVT and EVT.5–9 However, recent investigations highlight the potential therapeutic benefits of bridging IVT, showing improved neurological outcomes and reduced incidence of intracranial hemorrhage (ICH).10–12 Additionally, bridging IVT appears to positively influence procedural aspects of EVT, including shorter procedure times and increased odds of successful reperfusion.13,14 Nevertheless, decision for bridging thrombolysis requires careful prior consideration of contraindications (including extensive acute brain infarction and previous active bleeding events, etc.) to mitigate the risk of hemorrhagic complications. 4
Previous studies suggest that the impact of bridging IVT on patients’ neurological improvement may depend on the velocity of infarct core progression, but the published data were controversial.15,16 For instance, the study by Lin et al. showed that patients with fast infarct core growth rate (>15 ml/h) might benefit from the bridging IVT with a better functional outcome at 90d. Contrary to this, Marnat et al. found no significant correlation between the infarct speed and the odds of favorable outcome independent of bridging IVT or DEVT alone. However, in their study, prior IVT was associated with significantly reduced occurrence of any ICH among slow progressors, whereas this was increased in fast progressors (>1.2 points ASPECTS/h). Beside these controversial findings in the literature, there was also a discrepancy regarding the thresholds used to define fast and slow infarct progression across the studies, in addition to different imaging modalities chosen for infarct core estimation.
Therefore, our study aimed to address this gap by assessing the impact of bridging IVT, infarct core growth rate (ICGR), and in particular their interaction, on neurological outcomes and post-EVT bleeding occurrence, without specifically categorizing patients into fast and slow infarct progression groups. Additionally, we aimed to evaluate the potential influence of prior IVT on peri-procedural factors of EVT.
Methods
This study was approved by the local review board and conducted in accordance with the Declaration of Helsinki. Informed consent was waived due to the retrospective nature of the study.
All data were extracted from the stroke registry of a tertiary care center, which prospectively recorded all stroke relevant information of incoming patients. The study encompassed all individuals diagnosed with acute ischemic stroke between 2018 and 2022 (selection process in Figure 1). The inclusion criteria included:
Patient age ≥ 18 years. Acute anterior LVO is defined by the occlusion of the internal carotid artery (ICA) and/or the M1/M2 branch of the middle cerebral artery. EVT is the primary treatment with best medical treatment, either with or without bridging IVT. Availability of baseline CT perfusion imaging (CTP).
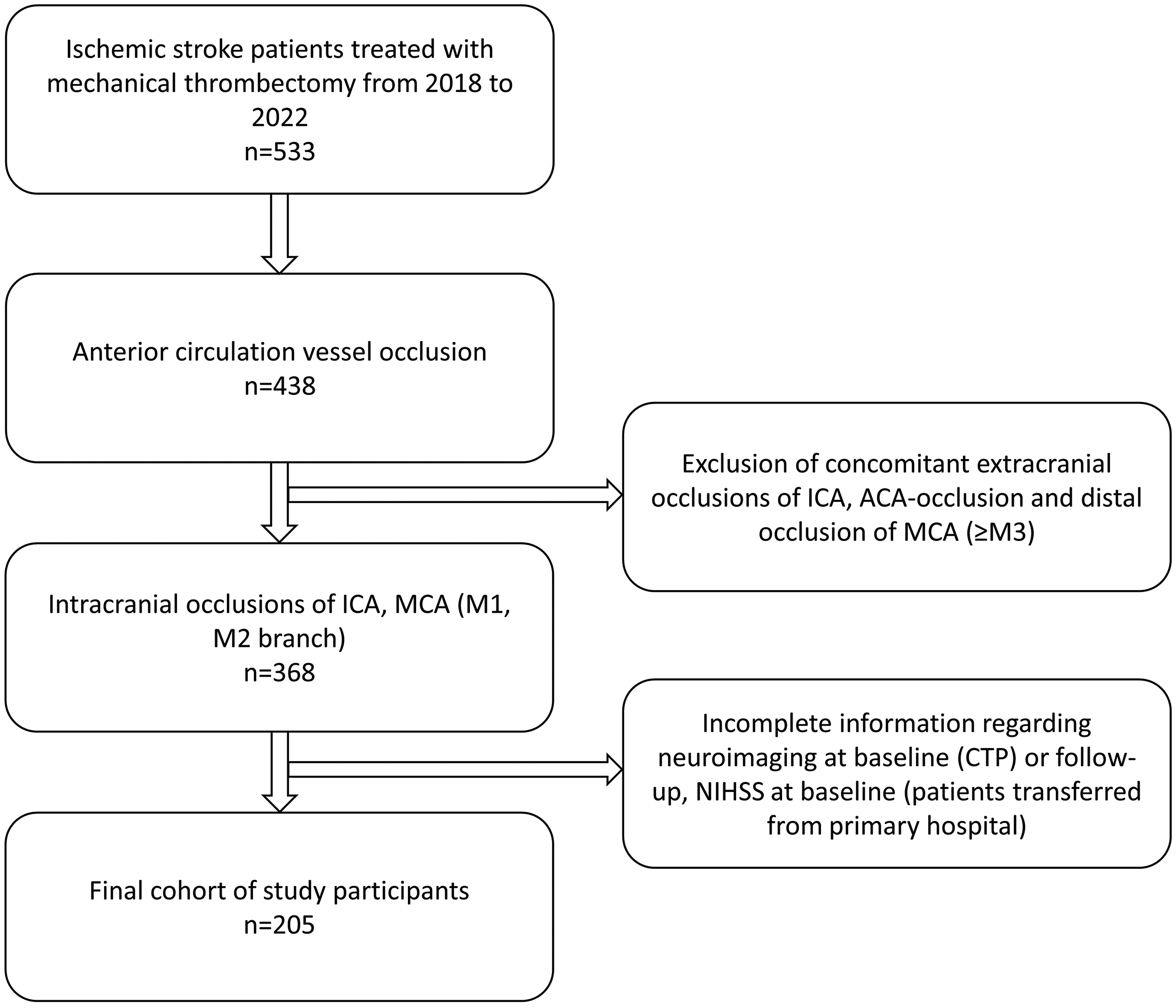
Flow chart of selection process.
Exclusion criteria were acute stenting of the carotid artery and/or intracranial vessels, as well as intra-arterial administration of thrombolytic agents.
Clinical and neurological assessment
The time points of symptom onset, hospital admission, groin puncture, and reperfusion were recorded. In case of unwitnessed strokes, the time of the last known well was regarded as the symptom onset time. Patient characteristics, including age, gender, pre-stroke modified Rankin Scale (mRS), and a medical history of diabetes mellitus and atrial fibrillation, were evaluated. The neurological status of patients was continuously assessed using National Institutes of Health Stroke Scale (NIHSS) scores at admission, as well as at 24 h, 48 h, 72 h, and discharge.
Stroke workflow and neuroimaging analysis
Upon arrival, all patients with symptoms of acute ischemic stroke underwent a standardized stroke workflow. The imaging protocol comprised an initial non-contrast CT scan followed by head-neck CT angiography and CTP. The Alberta Stroke Program Early CT Score (ASPECTS) was used to quantify the extent of early ischemic changes. Hypoperfusion and ischemic core volume were estimated using a postprocessing software (VEObrain GmbH, Freiburg, Germany). The volume of hypoperfused tissue was defined as regions exceeding a Tmax > 6 s, while the ischemic core volume was determined as cerebral blood flow <30% in comparison to the unaffected hemisphere. The penumbra was calculated as the difference between hypoperfused volume and infarct core volume.
The decision for bridging IVT was at the discretion of the attending neurologist according to the national and ESO guidelines.4 Post-EVT reperfusion grade was assessed using the Thrombolysis in Cerebral Infarction (TICI) Scale, with the first-pass effect defined as complete reperfusion (TICI 3) after the first pass.
At 24 h, all post-EVT patients underwent a dual-energy CT scan to evaluate the extent of infarction and differentiate potential intracranial hemorrhage from contrast agent extravasation. Hemorrhages were categorized using the Heidelberg Bleeding Classification. The ICGR was defined as the infarct core volume divided by the time interval from stroke onset to imaging (per 10 min). Neuroimaging analysis was conducted independently by two board-certified neuroradiologists (YL, CD), who were blinded to the clinical and interventional patient information, with any discrepancies resolved through consensus meetings.
Definition of study endpoints
Early neurological improvement (ENI), defined as a reduction of ≥8 NIHSS points after 24 h or NIHSS <2 at discharge,17,18 was set as the primary outcome. The secondary outcome was occurrence of any intracranial hemorrhage at 24 h. Primary and secondary outcome were tested for their association with bridging IVT, ICGR, and in particular their interaction.
Statistical analysis
Statistical analyses were performed using SPSS (version 29.0.2.0, IBM Corp., Armonk, NY, USA), R-software (version 4.3.2; R Foundation for Statistical Computing, Vienna, Austria) and the “Induced Smoothed lasso” package. 19 A significance level of p < 0.05 was considered statistically significant. Patient demographics, clinical characteristics, and procedural details were summarized using descriptive statistics. Categorical variables were presented as frequencies and percentages, while continuous variables were expressed as medians with interquartile ranges (IQR). Numeric variables were compared using Mann-Whitney U test and nominal variables were assessed using the χ2-test.
ENI and ICH at 24 h were tested separately for their association with bridging IVT, ICGR, and their interaction (included as a product term, ICGR × IVT), as well as other relevant factors, using induced smoothed ridge regression. The interaction term was included to assess whether the combined effect of ICGR and IVT on the outcomes differed from the sum of their individual effects, thereby allowing us to investigate if the relationship between ICGR and the outcomes varies depending on the administration of IV thrombolysis. Potential multicollinearity among variables was assessed through variance inflation factors, with values >5 suggesting significant overlap. Odds ratios (OR) and their corresponding 95% confidence intervals (CI) were calculated to quantify the strength of association for each variable with ENI or ICH.
Results
Baseline characteristics and peri-procedural factors
A total of 205 patients were included. The median age was 77 years and 81 patients were male (39.5%). 55.7% of the patients had a history of atrial fibrillation and 25.9% had diabetes mellitus (Table 1). The median NIHSS score was 14 points at patients’ admission. 128 (62.4%) patients received bridging IVT. The DEVT group had significantly longer time from symptom onset to admission (234 min [IQR 93–594] vs. 155 min [IQR 51.75–427.75] for the bridging IVT group, p = 0.015) and lower median baseline ASPECTS (9 [IQR 8–10] vs. 10 [9–10], p < 0.001). The DEVT group also had a significantly lower rate of pre-stroke functional independence (median mRS 2 [IQR 0–3] vs. 1 [0–2], p < 0.001). The ICGR was significantly lower in the DEVT group (median 0.55 ml/10 min [IQR 0.08–2.94] vs. 1.31 ml/10 min [IQR 0.19–5.42], p = 0.033).
Baseline characteristics and peri-procedural factors of reperfusion therapy.
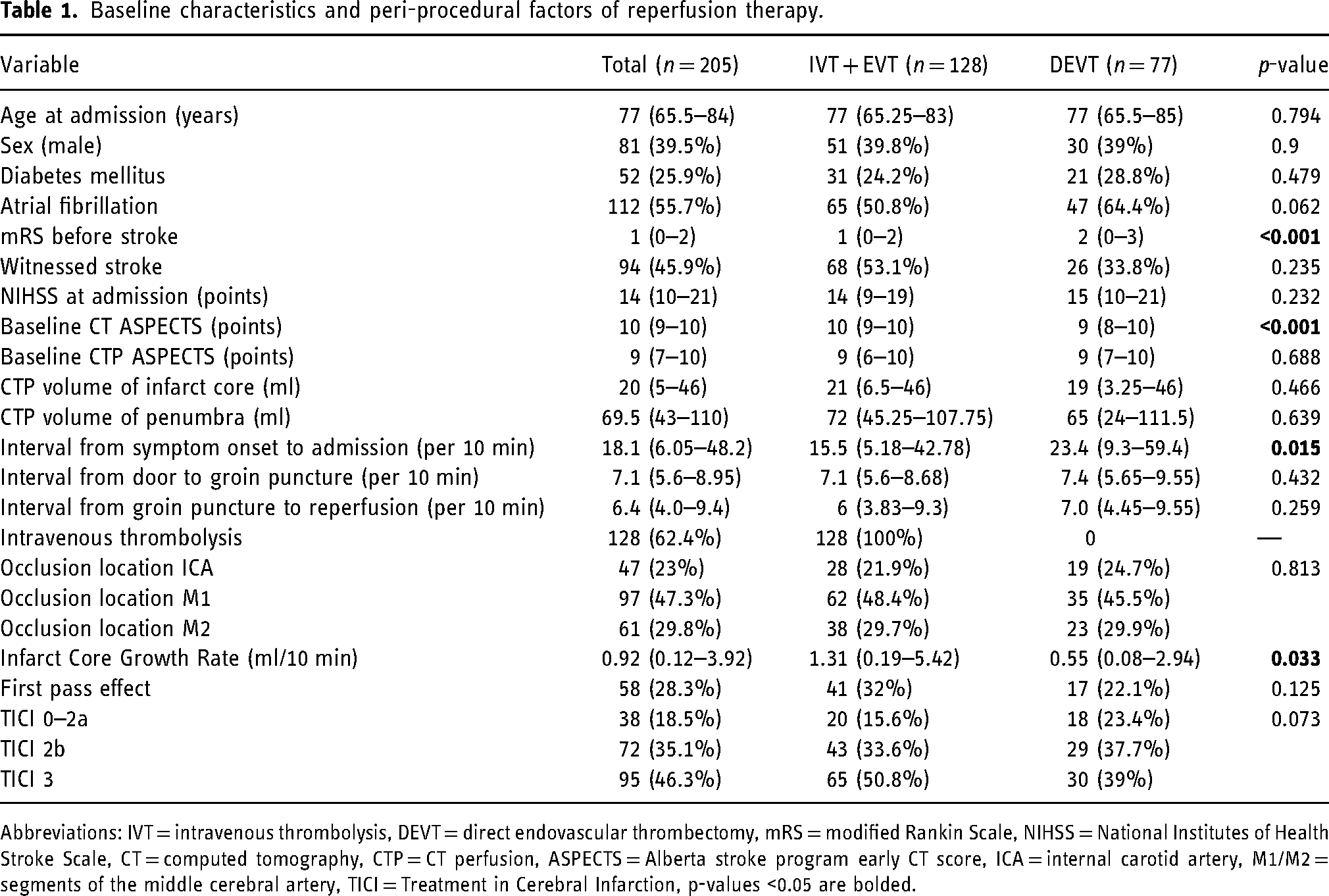
Abbreviations: IVT = intravenous thrombolysis, DEVT = direct endovascular thrombectomy, mRS = modified Rankin Scale, NIHSS = National Institutes of Health Stroke Scale, CT = computed tomography, CTP = CT perfusion, ASPECTS = Alberta stroke program early CT score, ICA = internal carotid artery, M1/M2 = segments of the middle cerebral artery, TICI = Treatment in Cerebral Infarction, p-values <0.05 are bolded.
In both groups, the most frequently observed reperfusion grade was TICI 3 (50.8% in bridging IVT vs. 39% in DEVT), followed by TICI 2b (33.6% in bridging IVT vs. 37.7% in DEVT). First pass effect was achieved in 32% of bridging IVT group and in 22.1% of EVT patients (Table 1). Procedure time from groin puncture to reperfusion was not significantly different in both groups and took an average of 64 min (Table 1).
Neurological endpoints and predictors of ENI and ICH
ENI was observed in 57 patients, with the majority (47/57) experiencing NIHSS reduction of ≥8 points at 24 h. The remaining 10 patients had a relatively low median baseline NIHSS of 7 (IQR 5–9) and were discharged with a NIHSS score of <2 after a median hospital stay of 7 days (IQR 4–14). In the bridging IVT group, ENI was observed in 30.5%, while in the DEVT group, it was documented in 23.4% of patients. Any ICH at 24 h after thrombectomy occurred in 54 cases (26.3%), with only 11 cases classified as 1a/1b according to the Heidelberg Bleeding Classification. The distribution of ICH was similar in cases with and without bridging IVT (25.8% vs. 27.3%). The differences regarding ENI and ICH at 24 h between the two groups were not statistically significant (Table 2).
Study endpoints and bridging IVT vs. DEVT.

Abbreviations: IVT = intravenous thrombolysis, DEVT = direct endovascular thrombectomy, ENI = early neurological improvement.
In the smoothed ridge regression analysis, neither bridging IVT (OR = 1.0, CI 0.79–1.26, p = 1), nor ICGR (OR = 0.99, CI 0.94–1.04, p = 0.676), nor their interaction (OR = 0.97, CI 0.92–1.03, p = 0.310) was significantly associated with the occurrence of ENI (Figure 2, supplementary material Table S1). Neither did bridging IVT (OR = 1.0, CI 0.92–1.11, p = 0.836), nor ICGR (OR = 0.99, CI 0.96–1.02, p = 0.374), nor their interaction (OR = 0.99, CI 0.96–1.03, p = 0.60) have a relevant impact on ICH occurrence (Figure 3, supplementary material Table S2).
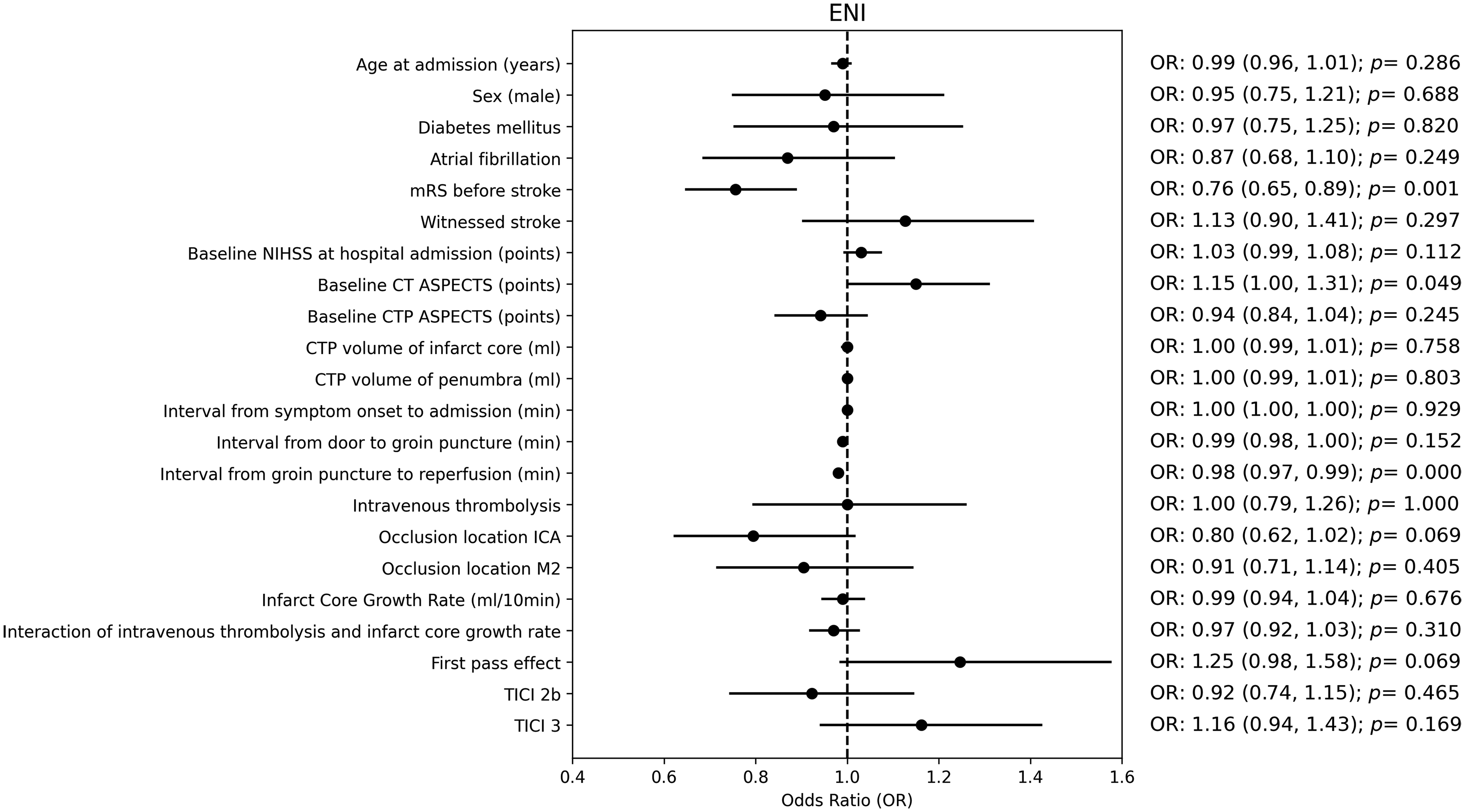
Forest plot of different variables in predicting early neurological improvement.
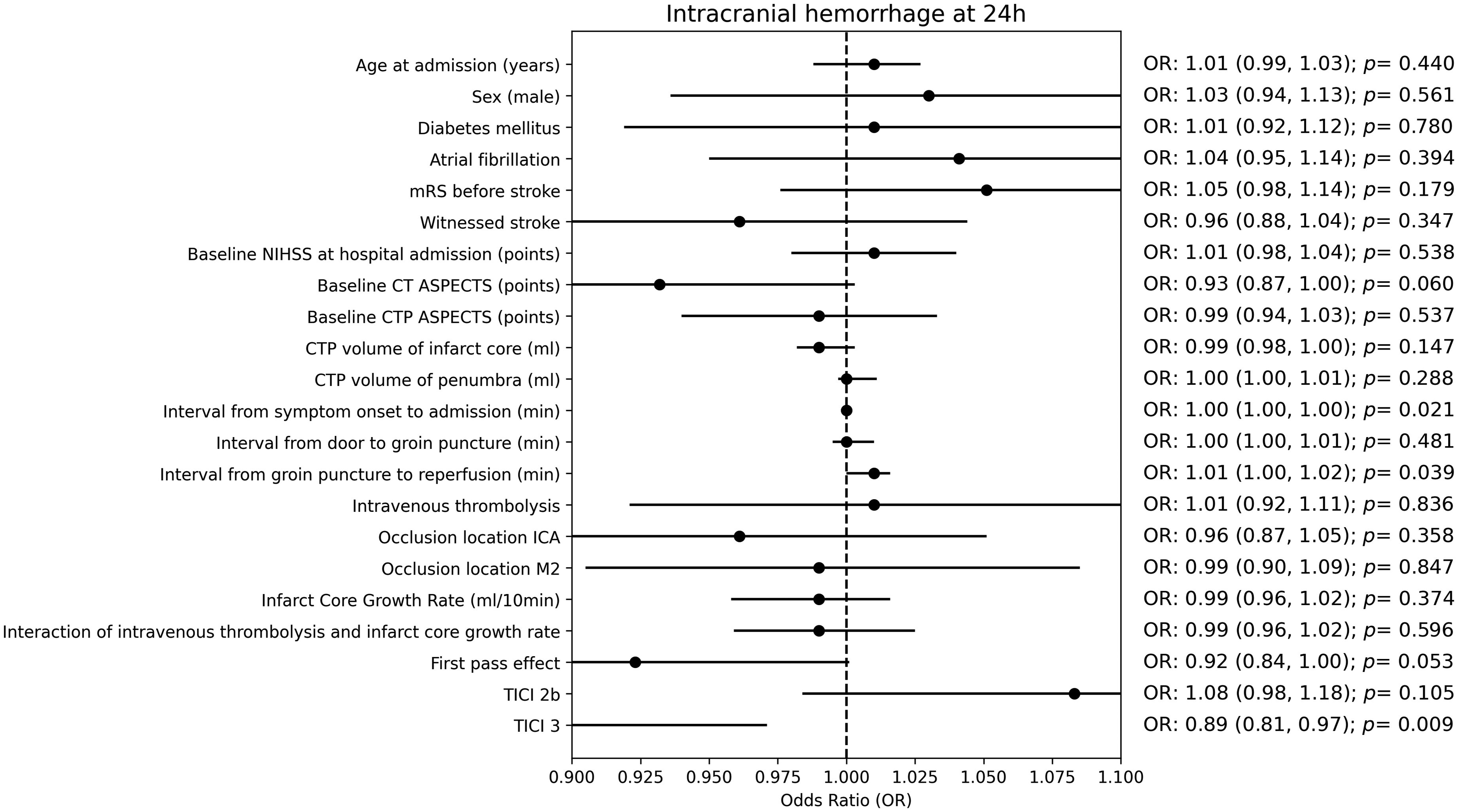
Forest plot of different variables in predicting intracranial hemorrhage.
A short groin puncture to reperfusion time (OR = 0.98, CI 0.97–0.99, p < 0.001), low mRS before symptom onset (OR = 0.76, CI 0.65–0.89, p = 0.001), and high baseline CT ASPECTS (OR = 1.15, CI 1–1.31, p = 0.049) were significantly associated with ENI. A short symptom onset to admission time (OR = 1, CI 0.998–1, p = 0.021), short groin puncture to reperfusion time (OR = 1.01, CI 1–1.02, p = 0.039), and TICI 3 (OR 0.89, CI 0.81–0.97, p = 0.009) were significantly associated with reduced odds of ICH.
Association between IVT and peri-procedural factors
A shorter procedure time (median 60 min vs. 70 min in DEVT, p = 0.259, Table 1), a higher rate of complete reperfusion and a lower incidence of unsuccessful reperfusion were observed in the IVT group (TICI 3 of 50.8% vs. 39%, TICI 0-2a of 15.6% vs. 23.4% in DEVT, p = 0.073). Moreover, the first pass effect occurred more frequently in the bridging IVT group (32% vs. 22.1% in DEVT, p = 0.125). The differences in procedure time, reperfusion grade, and first pass effect between the two groups remained statistically non-significant (Table 1).
Discussion
In this retrospective study, we investigated the impact of bridging IVT, infarct core growth rate, and their interaction on ENI and occurrence of post-EVT bleeding complications in the context of ischemic stroke due to anterior LVO. Neither bridging IVT, nor ICGR, nor their interaction showed significant association with ENI or occurrence of post-EVT ICH. Despite a shorter procedure time and a higher rate of both complete reperfusion grade and first pass effect being found in the bridging IVT group, there was no statistically significant difference compared to the DEVT group. Shorter procedure time, however, as well as lower pre-stroke mRS, and higher baseline CT ASPECTS, were significantly associated with ENI. On the other hand, a shorter symptom-onset-to-admission time, a shorter procedure time, and complete reperfusion were significantly associated with reduced odds of ICH.
In the literature, studies about the influence of infarct growth rate before reperfusion therapy, the bridging IVT, and particularly the interaction of both on patients’ neurological outcomes were limited and the results were conflicting. One key issue was the heterogeneous definition of ICGR, especially the infarct core volume, which was determined by different imaging modalities such as diffusion-weighted imaging (DWI), 20 non-contrast CT 16 and CTP parameters.15,21 In our study, infarct core volume was defined by CBF < 30% on CTP due to its robust determination, showing higher interrater agreement and reproducibility compared to other imaging modalities. 22 Furthermore, CTP provides more precise detection of early infarct core compared to non-contrast CT. 23 Despite varying definitions of ICGR, findings about the neurological impact of ICGR and bridging IVT remained controversial. Some studies have suggested a significant impact of ICGR on neurological outcomes. Sarraj et al. reported that patients with a slow ICGR (<10 ml/h) were more likely to attain functional independence at 90d after EVT compared to fast progressors (mRS ≤ 2 in 60.5% vs. 35.3% in fast progressors, p < 0.001), particularly in cases beyond a 6 h time window. 24 This finding could be explained by the presence of better collateral flow resulting in slower stroke progression, since favorable collateral status has been proven as a valid predictor of better functional outcome in case of successful reperfusion. 25 In comparison to our study, the overall median ICGR was lower (median 3.6 ml/h [IQR 0–12.5] vs. 5.5 ml/h [IQR 0.74–23.5]), which might have contributed to better functional outcomes at 90d (mRS ≤ 2) in their study. A study by Lin et al. showed that only in patients with fast ICGR (>15 ml/h), the additional prior IVT might have a favorable therapeutic effect regarding patients’ functional outcomes at 90d. 15 Accordingly, in fast progressors with bridging IVT, the final post-EVT infarction volume was reported to be lower than in the DEVT group (median 96.3 ml vs. 153 ml in DEVT, p = 0.021). In line with our study, they also observed shorter procedure times in the bridging IVT group. Nonetheless, our study did not show any significant association of bridging IVT, ICGR, and their interaction with ENI. This aligns with findings from the latest investigation by Marnat et al., which similarly found no evidence for a significant interaction of the ICGR and IVT on the neurological outcome. 16 However, it is important to note that Marnat et al. only included patients within the early time window of <4.5 h after symptom onset, and absolute infarct core volumes were unavailable because the infarct core was defined with CT ASPECTS in their study.
In our study, bridging IVT appeared to be safe without increased post-EVT bleeding complications, regardless of infarct speed (25.8% vs. 27.3% in DEVT, p = 0.814). Previously, Marnat et al. found a significantly higher ICH rate in fast progressors (>1.2 points ASPECTS/h) after bridging IVT (49.4% vs 26.8%, p-value for interaction <0.001). 16 A possible explanation for this may be the different definition of ICGR and the larger extent of early ischemic changes at baseline CT in their study (median ASPECTS 8 vs.10 in our study). As reported in earlier studies, the bleeding rate was higher after IVT in patients with initial low ASPECT scores. 26 In contrast, Lin et al. found no significant interaction between ICGR and IVT in predicting parenchymal hematoma (interaction OR 0.65 [0.15–2.82], p = 0.566) or sICH (interaction OR 1.22 [0.12–12.7], p = 0.867). 15
These controversial results in the literature and our study again reflect the complexity of the interaction between infarct progression speed before reperfusion and the subsequent change in neurological function. This interplay is influenced by numerous factors besides IVT that could affect the efficacy of leptomeningeal collateral blood flow to endure ischemic conditions. 27 The hypothesized mechanisms include anatomical collateral status and many other physiological parameters, including peri-procedural blood pressure, head position, CO2 levels, temperature, and blood glucose etc. 28 Furthermore, in our study the ENI was more present in the bridging group, which might be partially due to the generally shorter time from symptom onset to admission (median 155 min vs. 234 min, p = 0.015). Additionally, within the bridging group, shorter procedure times, a higher incidence of complete reperfusion and first-pass effect were observed, which ultimately led to a reduction in the total ischemic time and higher reperfusion grade. In the literature, the time from symptom onset to admission and the total ischemic time has been proven to be valid predictors for favorable functional outcome. 29
In summary, our study results revealed that the decision regarding bridging IVT should not be solely based on ICGR, and patients with higher ICGR should not necessarily be excluded from bridging IVT. Additionally, prior IVT tends to have a favorable effect on the EVT procedure. Furthermore, ICGR could be important in decision-making, particularly for fast progressors, when the anticipated patient transfer time from a primary center to a comprehensive stroke center is prolonged. It may help guide the decision on whether to proceed directly to the angio suite or perform repeat imaging to assess infarct core progression.30,31
The main strength of our study was the utilization of CTP to assess the ICGR. Another strength was the investigation of the impact of the interaction between IVT and ICGR on study endpoints, since we did not divide patients into any slow or fast infarct speed group. There are several limitations of our study. First, due to the retrospective nature of this study selection bias was present, since IVT was administered mostly in patients within early time window <4.5 h, potentially influencing the neurological outcome. Second, in cases of unwitnessed stroke, which constituted up to 54.1% of cases, the time of last known well was used for ICGR calculation. This likely diluted the impact of ICGR on neurological outcomes and could significantly compromise the validity of our findings, although it mirrors the real-world experience in acute stroke care. Finally, patient information regarding functional independence at 90 days was unavailable. Nonetheless, previous studies have identified ENI as a robust predictor for functional independence (mRS ≤ 2). 32
Conclusion
Neither bridging intravenous thrombolysis, infarct core growth rate, nor their interaction had a significant impact on early neurological improvement or intracranial hemorrhage rate. Hence, patients with fast infarct progression should not be necessarily excluded from bridging thrombolysis prior to thrombectomy.
Supplemental Material
sj-docx-1-ine-10.1177_15910199251336889 - Supplemental material for Impact of bridging intravenous thrombolysis and infarct core growth rate on early neurological improvement in patients with acute anterior circulation ischemic stroke and mechanical thrombectomy
Supplemental material, sj-docx-1-ine-10.1177_15910199251336889 for Impact of bridging intravenous thrombolysis and infarct core growth rate on early neurological improvement in patients with acute anterior circulation ischemic stroke and mechanical thrombectomy by Natalie van Landeghem, Christoph Ziegenfuß, Aydin Demircioglu, Benedikt Frank, Martin Köhrmann, Benjamin Stolte, Ramazan Jabbarli, Philipp Dammann, Johannes Haubold, Michael Forsting, Isabel Wanke, Cornelius Deuschl and Yan Li in Interventional Neuroradiology
Footnotes
Acknowledgements
We appreciate the support by the UMEA Junior Clinician Scientist Scholarship of the Medical Faculty of the University of Duisburg-Essen.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This study was approved by the Ethics Committee of the University of Duisburg-Essen (number 22-10821-BO) and conducted in accordance with the Declaration of Helsinki. Informed consent was waived due to the retrospective nature of the study.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
