Abstract
Background
Focal vasospasm (FV) of the occluded vessel can occur during the endovascular treatment of acute ischemic stroke (AIS). Nimodipine is commonly used to treat vasospasm and can play a role in distinguishing it from artery narrowing due to iatrogenic dissection or residual clot. However, nimodipine administration can result in arterial hypotension and subsequent enlargement of the ischemic core.
Objective
To assess the efficacy of preventive and continuous vasoactive amine infusion to counterbalance nimodipine-induced hypotension.
Methods
We reviewed data from a prospective registry of patients treated for AIS between January 2019 and January 2022 who were administered nimodipine. All patients were equipped with an arterial cannula for invasive blood pressure measurement and given vasoactive amines preemptively before general anesthesia and throughout the procedure. Data obtained from invasive monitoring of mean arterial blood pressure (MABP) in a time-lapse of 25 min before and after nimodipine administration were analyzed.
Results
MABP significantly decreased after nimodipine administration but remained within the recommended range (81.79 ± 0.49 mmHg). Nimodipine was effective in reducing FV caused by stent retriever passage in 76.3% of cases. Furthermore, it proved valuable in diagnosing iatrogenic dissection (9.2%), residual clot (10.5%), or intracranial stenosis (4%).
Conclusions
Infusion of vasoactive amines effectively counteracted the intraarterial nimodipine effect, thus avoiding frank arterial hypotension during endovascular treatment. Nimodipine has been useful in differentiating the diagnosis of FV resulting from mechanical thrombectomy and other potential causes, such as iatrogenic dissection or residual clot.
Introduction
A focal vasospasm (FV) of the target vessel can occur during mechanical thrombectomy (MTB) performed for the endovascular treatment of acute ischemic stroke (AIS) and is commonly related to the mechanical stress exerted by MTB devices over the vessel wall. Angiographically, FV appears as the narrowing of the vessel, which is visualized as a reduction of the diameter of the vessel lumen and a reduction of the anterograde blood flow. Nevertheless, such an appearance could be related to other causes such as iatrogenic dissection of the target vessel or the presence of residual clot. In clinical practice, nimodipine is commonly administered during the course of neurointerventions to treat FV.1–3 Nimodipine is a dihydropyridine drug, which blocks the voltage-gated calcium channels leading to the arterial smooth muscle relaxation and thus to vasodilation. 4 Hence, nimodipine can cause arterial hypotension and alterations in heart rate 4 and, consequently, a drop in blood pressure. Such a drop during AIS can result in increased neural loss with potentially negative consequences for the patient's outcome.5,6
The aim of the present study was to evaluate whether the preventive and continuous infusion of vasoactive amines could effectively counterbalance the hypotensive effect of intraarterial (ia) nimodipine in the treatment of FV triggered by the interaction between thrombectomy devices and the target artery.
Methods
We conducted a retrospective review of the clinical and radiological records of our institutional stroke prospective registry, which included all patients treated at our center for an AIS between January 2019 and January 2022 by MTB. To investigate the impact of ia nimodipine administration on mean arterial blood pressure (MABP) during MTB procedures for the treatment of AIS, we conducted an analysis of MABP values measured every minute through an arterial cannula during endovascular treatment and ia nimodipine infusion. We also investigated whether the preventive and continuous administration of amines could maintain MABP values within the recommended range.
MABP data collection consisted of measuring MABP once per minute for 10 min before nimodipine administration (T10–T0), for 5 min immediately after nimodipine administration (T1–T5), and for 10 more minutes after nimodipine administration (T6–T15). This resulted in a total of 25 measurements for each of the 75 patients included in the study. For each of these three intervals, the mean value and standard deviation of MABP were calculated.
We also investigated whether the administration of ia nimodipine played a role in the diagnosis of other pathological conditions, such as iatrogenic dissection or the presence of residual clot. Such conditions have been shown to be associated with a poor clinical outcome contributing to a reocclusion of a previously recanalized artery within 24 h. 7 Therefore, we analyzed the reasons for nimodipine administration, the site of the vascular stenosis where it was most frequently used, and the dosages administered. We then assessed the angiographic outcomes of each procedure according to the thrombolysis in cerebral infarction score (TICI). 8
To evaluate the clinical outcome, we assessed the National Institutes of Health Stroke Scale (NIHSS) score at 24 h and the modified Rankin Scale (mRS) score at 90 days. In addition, we investigated whether an enlargement of the ischemic core resulting from a potential decrease in MABP due to ia nimodipine administration had occurred. For this purpose, we compared the size of the ischemic core using pre- and posttreatment radiological imaging by evaluating the ASPECT score.
Endovascular procedures: Blood pressure management and measurement
During the procedure, all patients received general anesthesia and invasive and continuous monitoring of MABP was carried out using an arterial cannula. Our goal was to maintain a target MABP value of 90 mmHg 5 throughout the procedure until the target vessel was successfully recanalized. Noradrenaline dosage was adjusted as necessary, based on each patient's response to maintain the target MABP.
In cases where a focal narrowing of the artery was identified following a thrombectomy maneuver, ia nimodipine was administered at a dose of 2 mg. The nimodipine solution was prepared by diluting it 50% with a hydrosaline solution. Nimodipine administration occurred over a period of 3 min through the main catheter. However, if MABP decreased to 70 mmHg during the administration of nimodipine, the administration was immediately stopped. If the reduction in artery caliber was determined to be caused by iatrogenic dissection, an intracranial stent was considered in the acute phase to address the issue. Conversely, if residual clot was suspected to be present, additional thrombectomy maneuvers were performed to remove the clot and improve vessel patency. A cone beam CT was performed at the end of each procedure and the presence of intracranial hemorrhage was scored using the Heidelberg bleeding classification. 9
Statistical analysis
Descriptive statistics, including the mean, standard deviation, median, quartiles, and frequencies, were used to analyze the data. Statistical comparisons of MABP values before (T10–T0), immediately after (T1–T5), and 5 min postadministration of nimodipine (T6–T15) were analyzed by a parametric general linear model with repeated measures after ensuring that the data were normally distributed. Missing values were excluded. SPSS Version 22 was used for the analysis with a significance level of 0.05 (two-tailed).
Results
Between January 2019 and January 2022, a total of 554 patients were treated for AIS at our center. Of these, 90 received ia nimodipine. Fifteen patients were excluded due to noninvasive, blood pressure monitoring with a sphygmomanometer. Among the 75 included patients, one underwent treatment for simultaneous bilateral occlusions of the middle cerebral arteries, resulting in a total of 76 treated occlusions. Table 1 provides an overview of the baseline, procedural, and clinical variables of the included patients.
Clinical and procedural variables.
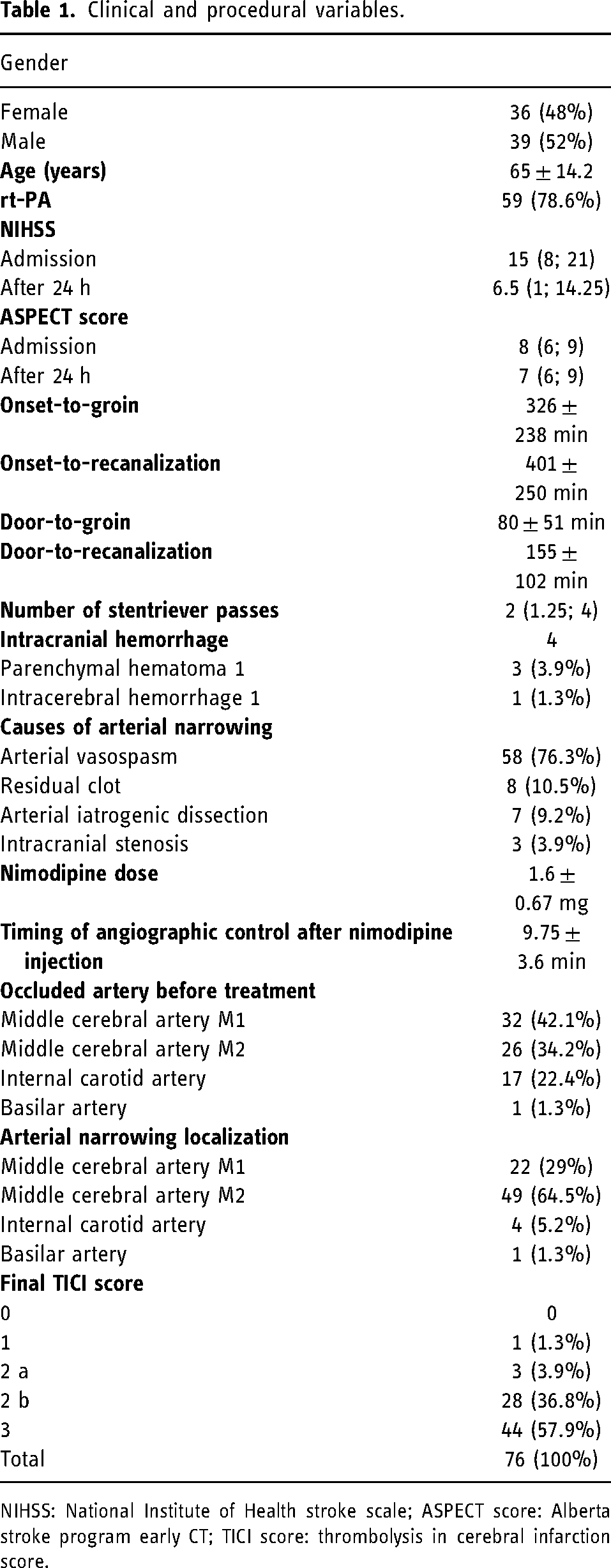
NIHSS: National Institute of Health stroke scale; ASPECT score: Alberta stroke program early CT; TICI score: thrombolysis in cerebral infarction score.
There were significant differences between all three time periods (p < 0.001), with nimodipine having a significant blood pressure-lowering effect despite the concomitant administration of amines. MABP values were 95.24 ± 0.47 mmHg before ia nimodipine administration (T10–T0), 91.84 ± 0.69 mmHg 5 min after nimodipine injection (T1–T5), and 81.79 ± 0.49 mmHg 15 min after nimodipine injection (T6–T15) (Figure 1). A comparison of the ischemic core volume before and 24 h after symptom onset was performed, based on the ASPECT score on pre- and postprocedural imaging. Ten patients were excluded due to the absence of either one of the two radiological examinations. In 53/65 (81.5%) cases, the volume of the ischemic core remained unchanged as assessed by the ASPECT score, while in 8/65 (12.3%) cases, the ischemic core expanded by 1 ASPECT score point and in 4/65 (6.1%) cases, it expanded by more than 1 ASPECT score point.
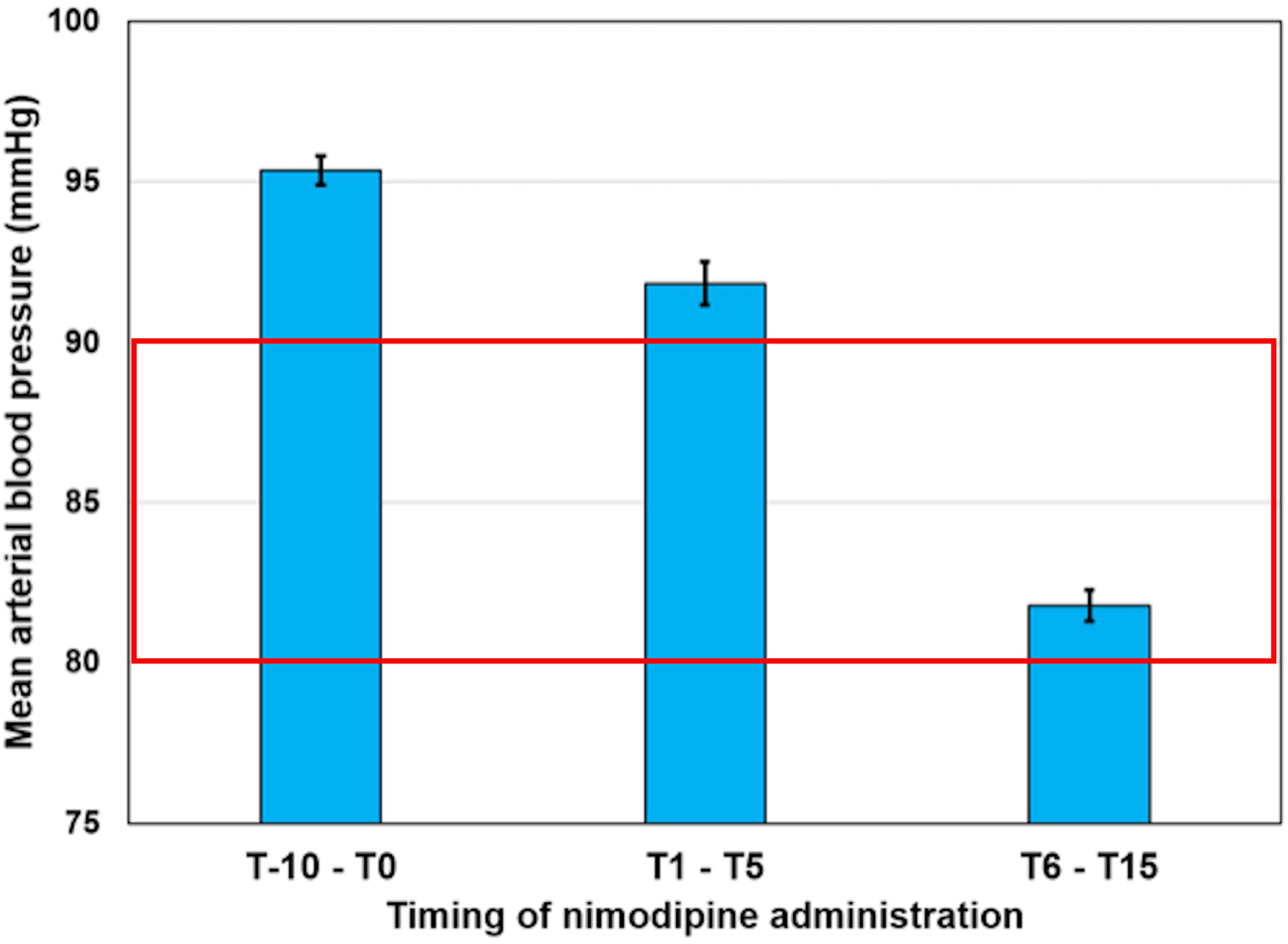
Mean arterial blood pressure before (T10–T0), immediately after (T1–T5), and post (T6–T15) administration of nimodipine (mean ± SE). Target values to be maintained during endovascular treatment for mean arterial blood pressure are included in the red rectangle.
On average, 1.6 ± 0.67 mg of nimodipine per artery was administered, and angiographic control was conducted at an average of 9.75 ± 3.6 min after nimodipine injection. In 58/76 (76.3%) cases, ia nimodipine revealed and resolved FV (Figure 2(A)–(D)). Additionally, it allowed the diagnosis of iatrogenic dissection in 9.2% of cases, residual clot in 10.5% of cases, and intracranial stenosis in 4% of cases. Among the cases of iatrogenic dissection (n = 7) and intracranial stenosis (n = 3), stent placement in the acute phase was performed in 5 and 1 case, respectively, resulting in a TICI score of ≥2b in 3/6 cases (Figure 2(E)–(G)). In the eight cases diagnosed with residual clot, additional thrombectomy maneuvers were conducted, achieving a TICI score of ≥2b in 7/8 cases (Figure 2(H)–(K)). Overall, a TICI score of ≥2b was obtained in 94.7% of cases at the end of the procedure, which aligns with the recanalization rate observed in a recent clinical trial. 10
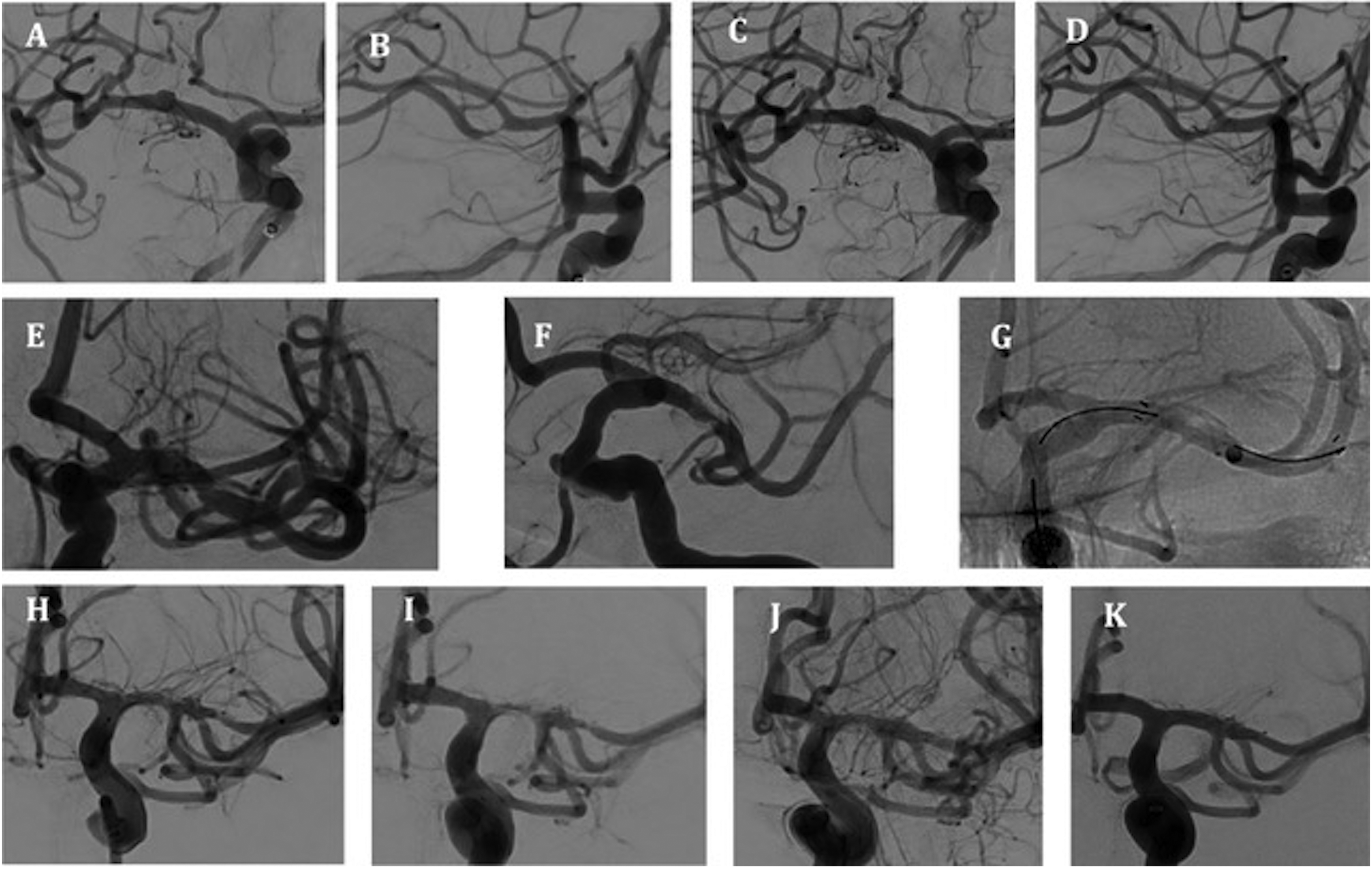
(A–D). Angiographic example of focal vasospasm following stent retriever thrombectomy. (A–B). Before nimodipine administration (anteroposterior and latero-lateral views, respectively). (C–D). Results at angiographic control at 10 min (anteroposterior and latero-lateral views, respectively). (E–G). Angiographic example of iatrogenic dissection. (E). Image after stent retriever thrombectomy; (F) image 10 min after nimodipine administration; (G) control after intracranial stent placement. (H–K). Angiographic example of artery narrowing due to residual clot. (H) Angiographic image after thrombectomy with a stent retriever; (I) angiographic image 10 min after administration of nimodipine; (J) angiographic image with open stent retriever and visualization of clot within the stent retriever; K, angiographic result after thrombectomy maneuver.
Cone beam CT performed at the end of the procedure showed intracranial hemorrhage in 4/75 (5.3%) patients (three cases with PH1 and one case with HI1). 9 Three-month follow-up was collected for 72/75 patients, revealing an mRS score ≤2 in 39 (54%) patients (Figure 3).
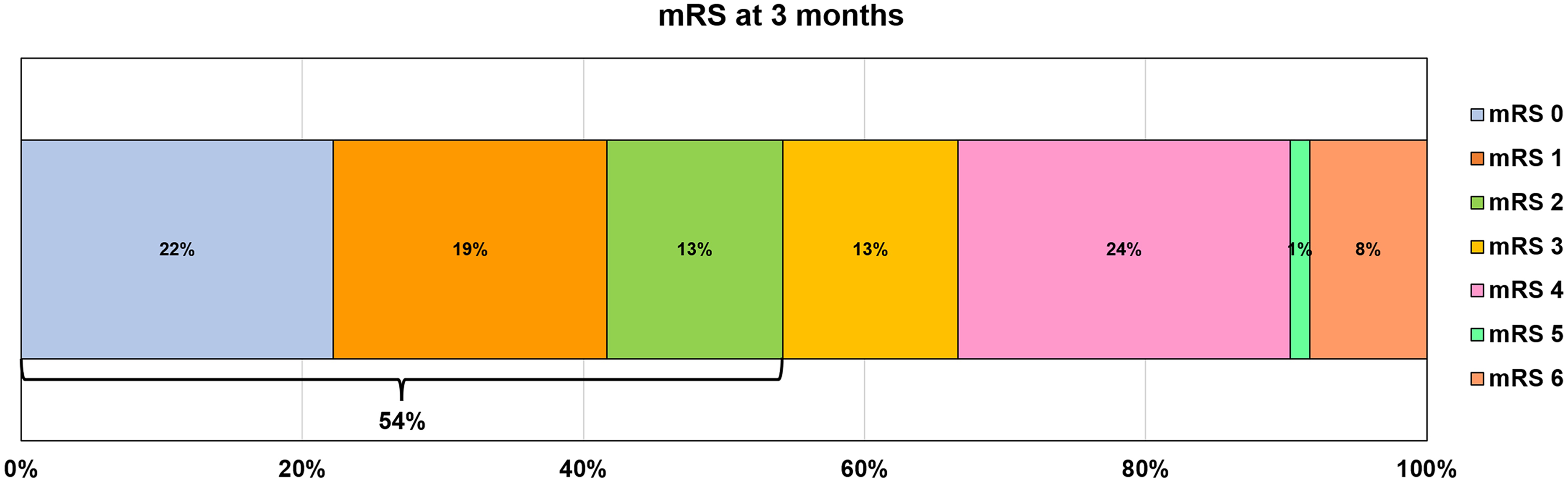
mRS distribution at three-month follow-up after endovascular treatment.
Discussion
We aimed to evaluate whether a prior infusion of vasoactive amines was able to reduce the hypotensive effect of nimodipine administered for the purpose of resolving FV or unmasking the presence of iatrogenic target artery dissection or residual clot. Our findings revealed that despite MABP significant reduction, the preventive administration of vasoactive amines played a crucial role in maintaining MABP at an acceptable level of 81.79 ± 0.49 mmHg, following ia nimodipine administration during endovascular treatment for AIS. Previous studies by Markus et al. 11 and Henry et al. 12 suggest that MABP is a more sensitive indicator than the systolic blood pressure of the brain parenchymal perfusion. Target MABP values recommended by Rasmussen et al. 5 for the optimal endovascular treatment of ischemic stroke fall within the range of 70–90 mmHg. Prolonged periods of MABP below 70 mmHg or above 90 mmHg have been associated with unfavorable clinical outcomes. MABP serves as a reliable indicator for assessing cerebral perfusion and its reduction can exacerbate the extent of ischemic damage. 13 Consequently, our MABP target during AIS endovascular procedures was set at 90 mmHg. However, it is important to acknowledge that patients may respond differently to various anesthesia drugs, emphasizing the necessity of strict MABP monitoring, particularly when patients undergo treatment under general anesthesia. 14
Jesser et al. 3 conducted a comparative analysis between two groups of patients who underwent endovascular treatment for AIS. One group received ia nimodipine based on the presence of FV, while the other did not. At 24 h after symptom onset, patients who received ia nimodipine during thrombectomy exhibited a lower ASPECT score and a higher NIHSS score compared to the control group. This suggests that in this specific patient population, the hypotension induced by ia nimodipine administration may have compromised cerebral perfusion, leading to an enlargement of the ischemic core. However, an enlargement of the ischemic core may also depend on late or incomplete reperfusion of the involved vascular territory.15,16 Our MABP management protocol aims to mitigate the hypotensive effects of ia nimodipine through the early administration of amines, thereby ensuring that MABP values remain above 70 mmHg. In our study, the comparison between ASPECT scores at patient admission and 24 h after symptom onset revealed no ischemic core enlargement of the ischemic core shift in 81.5% of patients. This finding indicates that the size of the ischemic core remained stable in most patients, thus confirming the adequate control of MABP and maintenance of cerebral perfusion. Furthermore, the three-month clinical follow-up showed an mRS ≤2 in 39/72 (54%) patients.
While FV induced by the passage of a stent retriever is often mild and transient, it is important to recognize that an apparent FV can mask other underlying conditions, such as iatrogenic dissection or residual clot. Endovascular treatment of a medium vessel occlusion increases the likelihood of acute arterial FV or iatrogenic artery dissection following the use of a stent retriever.1,2,17 Notably, our study revealed that arterial narrowing predominantly occurred in the M2 segment of the middle cerebral artery in 64.5% of cases in our patient cohort. In our study, ia nimodipine was effective in reducing FV caused by stent retriever passage in 76.3% of cases. Furthermore, when ia nimodipine administration did not resolve arterial narrowing, it proved valuable in diagnosing iatrogenic dissection (9.2%), residual clot (10.5%), or intracranial stenosis (4%).
Limitations
The study's main limitation is its retrospective and observational nature, conducted solely within a single center. All procedures were performed under general anesthesia with the assistance of a dedicated team of anesthesiologists, highlighting the need to consider the specific context and resources available when interpreting and applying these results.
Conclusions
Infusion of vasoactive amines effectively counteracted the intraarterial nimodipine effect, thus avoiding frank arterial hypotension during endovascular treatment. Nimodipine is useful in differentiating the diagnosis of FV resulting from MTB and other potential causes, such as iatrogenic dissection or residual clot.
Footnotes
Contributors’ statement
All authors have substantially contributed to the conception or design of the work; or the acquisition, analysis, or interpretation of data for the work; drafting the work; revising it critically for important intellectual content; final approval of the version to be published; and agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: PM is consultant for Stryker and Medtronic.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Schweizerischer Nationalfonds zur Förderung der Wissenschaftlichen Forschung (grant numbers 320030_188942 and 32003B_182382).
