Abstract
The Tigertriever is a novel, radially adjustable stentriever that addresses limitations in traditional mechanical thrombectomy devices by providing enhanced user control over clot integration. This provides the ability to adapt to patient-specific factors such as varying vessel sizes and clot compositions and may be particularly crucial for ensuring efficacy and safety in distal locations. This consensus paper synthesizes the clinical techniques from a consortium of experienced international operators. It outlines the current data on the Tigertriever, discusses the new operator-controlled capabilities, and provides a recommended approach for both proximal and distal mechanical thrombectomy, emphasizing the “FLEX” approach (Fast Controlled Expansion with Relaxation) for optimal integration and reduced clot disruption.
Introduction
The Tigertriever (Rapid Medical, Yokneam, Israel) is a novel, radially adjustable, fully visible stentriever that permits the operator to align radial expansion with target vessel diameters. It is designed to increase the integration of the thrombus, facilitating retrieval and reducing downstream embolization (Figure 1). Relaxing the device upon removal may also reduce the risk of vessel distortion, injury, and hemorrhagic complications, particularly in distal locations. These advancements offer greater user control regardless of anatomy or clot composition. This paper explains the Tiger's unique attributes and presents an international consensus on device operation during proximal and distal vessel thrombectomy.
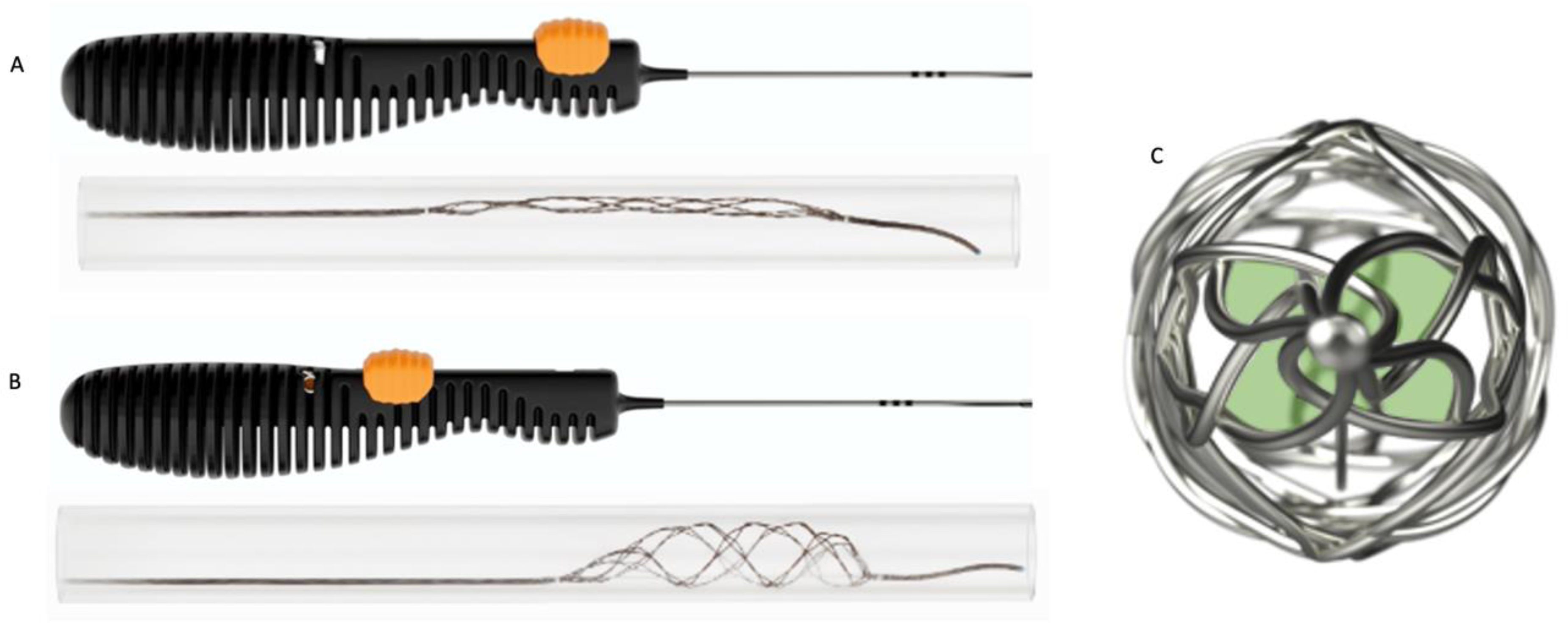
The Tigertriever device in the (A) closed configuration, (B) expanded configuration, and (C) the distal embolic net with the small filter-like pore sizes highlighted in green.
Tigertriever demonstrated promising efficacy and safety in the pivotal TIGER trial (Treatment with Intent to Generate Endovascular Reperfusion). 1 The study compared the device to performance goals derived from adjudicated outcomes in six completed prospective trials of two U.S. Food and Drug Administration (FDA)-cleared predicate stentrievers, Solitaire (Medtronic, Minneapolis, Minnesota, USA) and Trevo (Stryker, Kalamazoo, Michigan, USA). One hundred sixty patients were enrolled in the study and treated with the Tiger at 17 centers. Forty-three patients were lead-in, and 117 were in the main study. The primary efficacy endpoint, successful revascularization (modified treatment in cerebral infarction score (mTICI) 2b-3) after Tiger use, was achieved in 84.6% of the main study phase group compared with the 73.4% historical rate (p < 0.01). The primary safety composite endpoint rate of mortality and symptomatic Intracranial Hemorrhage (sICH) was 18.1%, comparable to the 20.4% historical rate. A very low sICH rate was observed (1.7%). Secondary effectiveness endpoints were also positive, achieving 58% good clinical outcome at 90 days, 57.8% first-pass effect, and 95.7% final successful reperfusion (mTICI 2b-3). The safety secondary endpoint, embolization to new territory (ENT), met the criteria of superiority: The 2.6% (95% CI 0.5%–7.4%) ENT rate was superior to the 7.4% in the pooled comparator trials (p = 0.0403). Finally, the median groin-to-revascularization time was 24 min, comparing favorably to the fastest times (25 min) reported in prospective studies, including the aspiration arm of the COMPASS study. 2
Following the completion of the TIGER trial, the Tiger 21 and 17 were cleared for marketing in the US by the FDA in April 2021—Tiger 13 clearance for large vessel occlusion treatment followed in July 2022. While the device remains under investigation for distal vessel occlusions (DVOs) in the US, it is indicated in Europe for this purpose. All TIGER models have CE marking. The devices’ performance has been demonstrated in 20 peer-reviewed publications covering over 600 patients. An estimated 10,000 patients have been treated with the Tiger device since European approval.
Considering the unique adjustable nature of Tiger and its increasing utility worldwide, questions about the most effective technique remain. A recent international consortium of experienced Tiger users developed a consensus on their standard operating steps. Due to proximal and distal artery differences, the consensus group agreed to separate the recommended techniques for these anatomies. Additionally, it was recognized that using Tiger 13 in more distal anatomies may benefit from specific operational steps.
Key distinctive features and general terminology
Three key technical features differentiate Tigertriever devices from self-expanding stentrievers: (1) The stent is expanded and relaxed on-demand in 19 increments or clicks (2) The stent is 3D-braided with radiopaque drawn-filled tubing (DFT) wires of nitinol and either platinum or tantalum, making it fully visible under fluoroscopy (3) Tiger 13 can be advanced through catheters with a minimum internal diameter of 0.013″, optimized for use with Headway Duo 167. To streamline communication, the consensus group chose “expansion” and “relaxation” to describe device opening and closing.
User-controlled expansion
Standard stentrievers are made from laser-cut nitinol tubes that are heat-set to a predetermined pattern. Although they vary in geometry, their mechanics remain the same. Once unsheathed, they expand to their original form—the structure and radial force profile cannot be altered. This classic deployment mechanism hinders their ability to pass through larger microcatheters, as uncontrolled expansion induces friction. Conversely, the ability to manually expand the Tiger allows it to pass through larger catheters while remaining fully collapsed. Standard stentrievers have limited clot integration methods as follows:
The length of the stentriever exposed (by partially unsheathing the stentriever). The time the stentriever is deployed. The physician can load the stentriever to increase the stent's expansion. A technique called “push and fluff”.
3
1. Amplitude 2. Frequency 3. Speed
Tiger maintains these options at the physician's discretion and offers additional capabilities. The device opens when a physician pulls a handheld slider, contracting a control wire fixated to the distal end of the stent. Contracting the wire shortens and expands the device, leading to an overall loss of length and gain in width. With the first eight clicks, the core wire retracts 1.4 mm per click. With the last 11, the core wire retracts 2.2 mm per click. This foreshortening from distal to proximal explains why it is essential to position the proximal marker at the proximal clot face. The user control of Tiger increases the operator's armamentarium with three new parameters:
The degree of mesh expansion modifies outward radial force, thus altering clot integration and conformation to the vessel wall. Expansion facilitates mesh integration into the clot, and relaxation reduces retrieval force and avoids thrombus shearing as it enters the aspiration catheter.
Real-world evidence shows that repeated expansion/relaxation of the device enhances the revascularization rate compared to a single deployment.4,5 The cyclical action (colloquially described as massage) impacts the work performed on the clot and integration forces.6–8 Whereas self-expanding stentrievers expand via chronic outward force until stress equilibrium, penetration with Tiger is an incremental process controlled by the number of cycles, or frequency, of deployment.
Speed of expansion/relaxation may also enhance clot integration. It is commonly known that faster insertion of a tool or needle causes less tissue deformation and easier penetration.7,8 Like slicing tomato or making surgical incisions, adding a sliding motion perpendicular to an advancing blade or wire decreases the force required for penetration, resulting in less trauma.6,7,9–11 Figure 2 shows the effect of the sliding speed on force reduction—even the slightest perpendicular motion significantly decreases the required compressive push forces.10–12 With Tiger, the device foreshortening creates this sliding motion, reducing the radial force necessary for clot penetration. The reduced force and thrombus strain may decrease the risk of clot fracture and secondary embolization
13
; this may have contributed to the Tiger study showing the lowest ENT of any clinical trial.
1
Also, Sreenivasan et al.
5
found no ENT in their patient series treated with the rapid repeated expansion/relaxation technique.
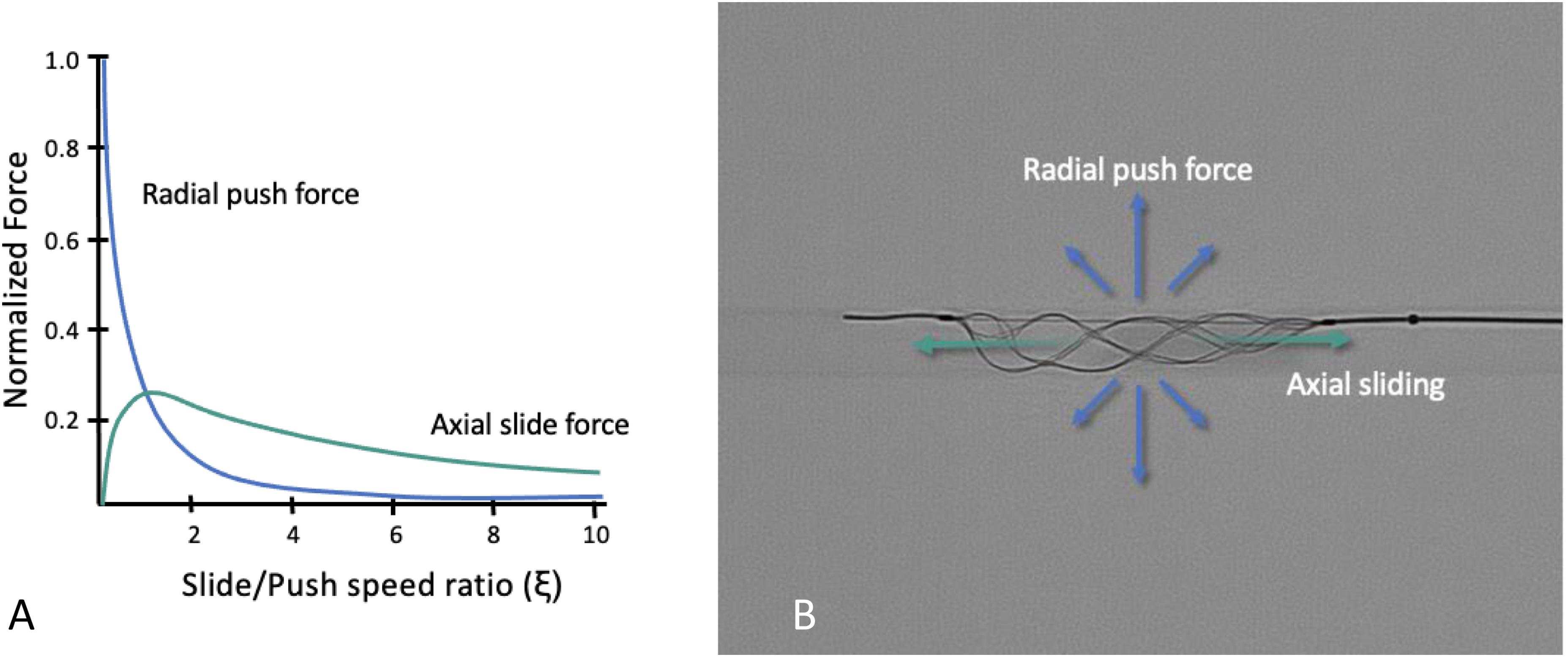
Demonstration of the slide-push physics (A) Graph of normalized forces in a frictionless environment performed on viscoelastic biomaterials.11,12 The radial force needed to penetrate the thrombus decreases when the device foreshortening creates perpendicular sliding motion (ξ > 0). (B) Illustration of the directional forces.
In-vitro testing examined the effect of actuation frequency and speed on clot integration. Fine-cut CT quantified the final clot volume incorporated into the stent for three different techniques: static (single expansion for 2 min), slow (six cycles of expansion and relaxation in 2 min), and fast (six cycles of expansion and relaxation in 30 s). The results showed that clot integration was significantly higher with the fast technique. 14 Time between cycles was constant, though it may be another variable for future study.
3D braided stent with radiopaque wires
The radiopacity of nitinol is not visible on standard fluoroscopic imaging. 15 Tiger solves this problem by adding a radiopaque core of either platinum or tantalum to nitinol DFT wires. 16 Each device wire is visible during the procedure, and radiopaque markers flank the proximal and distal ends of the stent for positioning ease. Device visibility is critical to the Tiger procedure, as incremental changes in size require this degree of visual feedback.
The end of the stent is closed, creating a distal embolic net, which may prevent distal emboli during retrieval. Illustratively, in a partially relaxed configuration, the Tiger 17 net openings have an average area of 90 μm2 (range 50–130 μm2) (Figure 1(c)). The distal coverage combined with the optimal balance of enhanced clot integration and reduced clot disruption may explain the lowest-ever reported ENT rate in the Tiger trial. 1 The device harbors a soft radiopaque tip with a 30-degree angle measuring 7 mm long and 0.015″ wide for Tiger 21 and 17 and 0.012″ for Tiger 13 (Figure 1(a) and (b)).
Consensus technique for proximal mechanical thrombectomy
Tiger 17 is compatible with a 0.0165″ microcatheter and indicated for use in 1.5–2.5 mm vessels with a working length of 19–23 mm between markers. Tiger 21 is compatible with a 0.021″ microcatheter and is indicated for use in 2.5–6 mm vessels with a working length of 21–30 mm. Tiger 17 is best suited for use in the M1, proximal M2, A1, distal basilar, and PCA due to its superior radial force. Tiger 21 is best suited for use in the ICA, proximal M1, vertebral, and proximal basilar due to its larger size and working length. While Tiger 13 is indicated for proximal thrombectomy, it is often used in smaller, more friable vessels, requiring a separate technique discussed in the next section.
The Tiger's additional functionality increases operators’ ability to address clinical situations in situ over self-expanding stentrievers. The technique is summarized by the acronym FLEX:
Recommended technique
Conscious sedation or general anesthesia should be employed at the discretion of the interventionalist.
A combined approach with Tiger and contact aspiration is recommended. Additional use of a balloon guide may be beneficial for recanalization rates and patient outcomes. 1
Position the microcatheter beyond the occlusion such that the proximal radiopaque marker is just proximal to the occlusion. Contrast injection through the guide or aspiration catheter may be performed to ensure optimal positioning of the Tiger. Proper positioning of the proximal marker relative to the face of the clot ensures ideal positioning of the working length since the device will shorten distal to proximal during expansion.
With the microcatheter and device in the optimal position, the aspiration catheter can be advanced just proximal to the occlusion site. If the aspiration catheter fails to advance, the Tiger can be used as an anchor once deployed to maximize distal access.
Unsheathe the device by pinning the pusher wire with one's right hand and retracting the microcatheter with the left (pin and pull technique) until there is a small gap between the microcatheter and the proximal device marker. Note: Ensure Tiger is fixed in place while unsheathing since it is only anchored once actively expanded!
Begin expansion of the device under live fluoroscopy until optimally opened for the vessel size. This is apparent when the wires at the distal end of the device approximate a 90-degree angle (Figure 3). Note: The device can be safely opened to the maximum diameter—the MAUDE database has no reported events of vessel rupture or dissection related to this. The stent does not overexpand; rather, the tip infolds and the radial force decreases as a safeguard. Infolding is another signal that maximum opening was achieved and surpassed; relax the device until it returns to its standard configuration.
Real-world evidence shows that repeated expansion and relaxation of the device enhances the revascularization rate.4,5 This cyclic actuation can be done rapidly and often to maximal expansion and relaxation. A maximum of five cycles is likely adequate over 30–60 s and performed under direct visualization.
Prior to removing the device, reduce tension on the system, or friction against the endothelium, by relaxing the Tiger until the device visibly lengthens slightly. Lengthening indicates that the device is ideally relaxed within the vessel. The device does not need to be closed entirely and can be relaxed further at any point during retrieval in response to resistance. Note: With conventional stentrievers, some operators prefer to remove the microcatheter before the thrombectomy system to maximize the aspiration force. However, fluid dynamics and in-vitro testing show that a microcatheter left in situ results in no appreciable loss of suction force as long as the microcatheter is retracted slightly from the catheter tip to avoid obstructing the suction surface area.18,19 This positioning is relevant for Tiger since the microcatheter cannot be removed due to the fixed handle.
Under concurrent aspiration, withdraw the aspiration catheter, microcatheter, and device as a unit. It is strongly recommended to visualize the device under live fluoroscopy during withdrawal into an access catheter, adjusting to the catheter diameter as needed to facilitate entry (Figure 4). Overly expanded devices can distend and develop a slight curve that can catch at the aspiration catheter tip.
As with any stentriever, repeated use may deform the device or hinder its ability to expand fully. Before reuse, the Tiger device should be manually inspected and actuated, particularly after employing the FLEX technique.
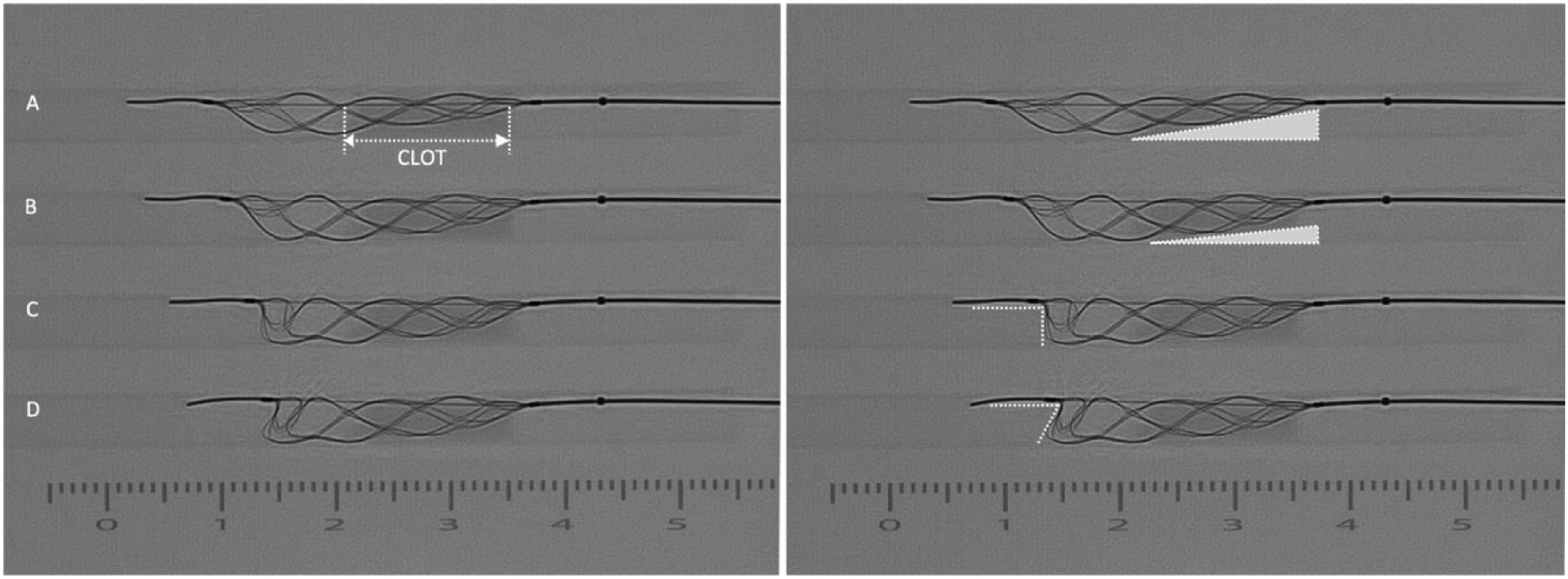
The visual cues associated with Tiger's stages of expansion. The images depict T21 in a 4.5-mm internal diameter (ID) straight tube with a wall thickness of 0.12 mm, similar to the ICA and MCA, 17 and a synthetic clot. The series on the left and right are identical, with the right adding annotations to highlight the progressive changes for each phase of expansion: (A) Initial wall apposition–seen in the distal section of the device. (B) Continued expansion opens the stent further and increases apposition across the proximal section. Once fully apposed, only the distal end moves inward, as noted by its change in angulation from image B to C. (C) Maximum expansion is indicated by a 90-degree angle at the device apex. (D) Overexpansion is displayed by an acute angle at the apex as the distal end folds inward.
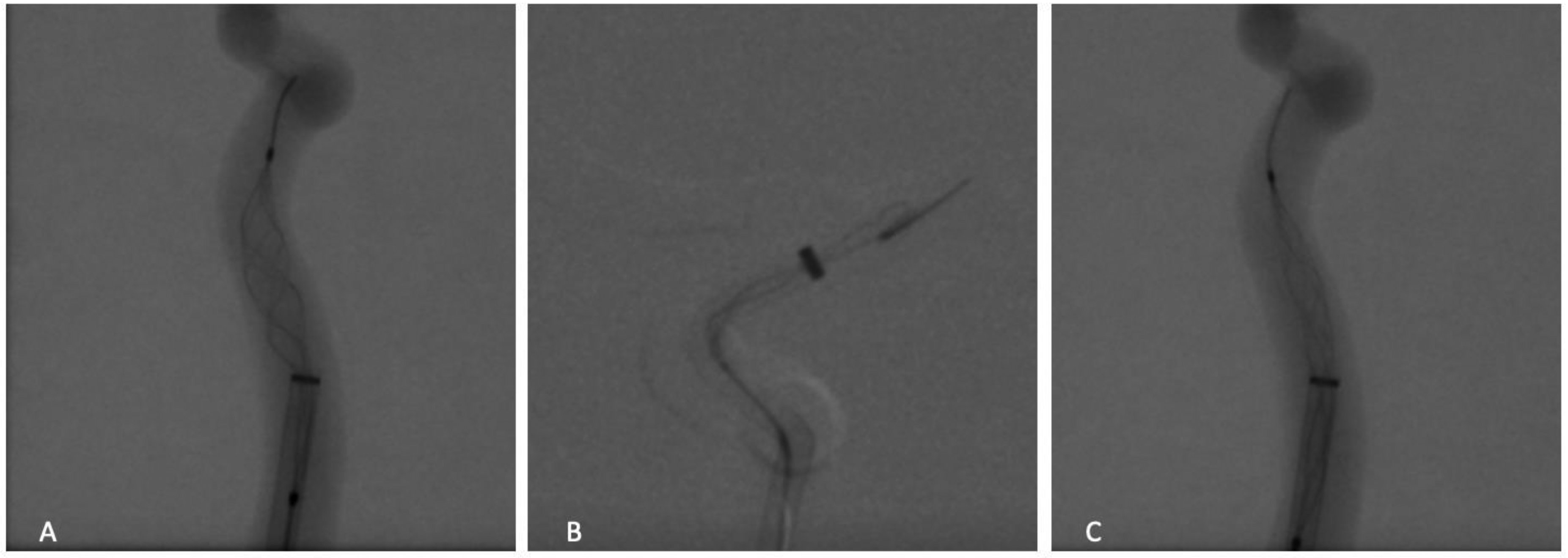
Relaxation facilitates entry into an access catheter. (A) Growing distention as the expanded device enters the guide catheter. (B) The resultant curved end when continuing to withdraw an overexpanded device into the catheter; the distal marker is now involuted and proximal to the tip of the distal embolic net. (C) The desired adjustment to the guide catheter diameter from (A) to correct distention and prevent hooking.
Consensus technique for distal mechanical thrombectomy
Tiger 13 is the lowest profile stentriever in the world and is compatible with a 0.013″ microcatheter. It expands from 0.5–2.5 mm with a working length of 12–19 mm between markers. The microcatheter profile constitutes a 24% reduction in distal outer diameter (OD) compared to the second lowest profile device (1.3F down from 1.7F), facilitating access in vessels previously thought too dangerous. A recent consensus statement proposed the consideration of thrombectomy down to a vessel diameter of 0.7 mm. 20 There is no current single optimal technique for treating DVO. As such, two distinct access methodologies will be discussed. Regardless, it is clear that the precise positioning of the Tiger 13 is essential. Ample time and patience should be allowed for perfect placement at the proximal end of the clot, especially considering the functional device length can range from 19.4 mm in a 1-mm diameter vessel to 11.9 mm in a 2.5-mm diameter vessel. Furthermore, with primary DVO, the presenting deficit is usually less severe; thus, the risk profile should be reduced. The most common complications include vessel perforation or sulcal SAH. While the lowest profile aspiration catheters can now track to the aforementioned distal locations, traumatic SAH is quite common.21,22 Using the smallest microcatheter in the smallest vessels likely provides the lowest risk. The ability to access these vessels with only a 1.3F OD microcatheter and to relax the stentriever before pulling potentially makes Tiger 13 the safest and most effective device for treating DVO.
Recommended technique
General anesthesia is favored to reduce patient movement during catheter navigation to the more distal and fragile target arteries.
While there is consensus that using contact aspiration with a distal aspiration catheter is the preferred technique for thrombectomy in proximal vessel occlusion, the working group could not define a single optimal technique for distal thrombectomy. However, two methods were clearly favored.
There were multiple concerns about advancing aspiration catheters into distal vessels. First, the added rigidity of the system could lead to vessel distortion and perforator avulsion. Second, aspiration through a distally placed intermediate catheter, particularly if sized to the vessel, could lead to vessel collapse and hinder clot adherence to the Tiger 13.23,24 To avoid the need for a distally placed aspiration catheter, one might consider simply using a classic combination of a proximal BGC and microcatheter to deploy the Tiger device. Another option is to maximize distal support with a triaxial system and use in-situ aspiration with a midsize catheter such as Zoom 45, Zoom 55, Phenom Plus, 5F Sofia, or 5F Esperance. This enhances 1:1 responsiveness with catheter and wire advancement and may avoid loss of clot by shortening the vessel length the Tiger and clot must traverse before entering a catheter. Considering the above-stated concern that a catheter “wedged” in a distal vessel could lead to vessel collapse, one may consider waiting until Tiger 13 is at the aspiration catheter tip to initiate pump aspiration rather than before the pull.
As stated above, the most important cause of failed distal vessel thrombectomy is failure to position the device so that the fully expanded Tiger encompasses the clot. While GA and road mapping are essential, a contrast injection in the vessel of interest as close to the thrombus as possible can optimize localization of the proximal clot face; this can be accomplished either through the distal access catheter or the more distal microcatheter. When uncertain about the clot location or after a failed thrombectomy, consider advancing through the clot, removing the microwire, and performing a contrast injection distal to the clot. To further enhance this technique, withdraw the catheter retrograde through the clot with continuous contrast puffs to fully localize the proximal and distal ends. While losing access may be frustrating due to the lack of perforators, gently advancing the catheter without a wire or using the lead wire of the Tiger 13 (0.012″ tip diameter) appears safe and can avoid the need to readvance the microwire. This technique may risk clot dislocation or fragmentation; however, in the group experience, this consequence appears less likely in distal vessels.
The positioning of Tiger 13 should optimize the working length, with the proximal marker as close as possible to the proximal thrombus since the device will shorten distal to proximal during expansion.
Unsheathe the device by pinning the pusher wire with one's right hand and retracting the microcatheter with the left until there is a small gap between the microcatheter and the proximal device marker. Note: Ensure Tiger is fixed in place while unsheathing since it is only anchored once actively expanded!
Begin slow expansion of the device under live fluoroscopy until optimally opened for the vessel size. This will be apparent when the wires at the distal end of the device approximate a 90-degree angle (Figure 3) or the vessel changes shape. Count the number of clicks to facilitate step #7.
Real-world evidence shows that repeated expansion/relaxation of Tiger enhances the revascularization rate.4,5 This cyclic actuation can be done rapidly and most often to maximal expansion in proximal vessels but should be performed slowly and usually to less than full expansion in distal vessels. Five cycles are likely adequate over 30–60 s and performed under direct visualization.
Reduce stress on the system when retrieving Tiger 13 by relaxing it until the device lengthens. If any tension is felt or visualized (by vessel stretch or distortion), continue relaxing the device until it moves easily, a notable safety feature of TIGER over static stentrievers. For DVOs, err toward complete relaxation to avoid hemorrhagic complications. If using a distal access catheter, pay attention to the distal tip location. During the pull, Tiger 13 may act as an anchor and cause advancement of the aspiration catheter, which is more prone to vessel perforation or injury in the distal vasculature. Again, erring toward complete relaxation of the device or initially backing up the aspiration catheter may be necessary.
Pay particular attention to the arterial angles in the Sylvian fissure. Consider further relaxation in response to resistance or vessel deflection while pulling through this segment, reducing the risk of vessel injury and SAH.
Conclusions
The Tiger devices represent a significant advancement in the field of intracranial thrombectomy. The design increases the parameters neurointerventionalists can modify to optimize success, thereby addressing patient variables in real-time. Since this changes the procedure from passive to active control, these recommendations mark the first step in consolidating the worldwide knowledge base to improve outcomes and reduce complications. Much more is likely to be learned from continued use and collaboration. Additional research and utilization of artificial intelligence could help enhance optimal technique.
Supplemental Material
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
