Abstract
Background
Accurate estimation of ischemic core on baseline imaging has treatment implications in patients with acute ischemic stroke (AIS). Machine learning (ML) algorithms have shown promising results in estimating ischemic core using routine noncontrast computed tomography (NCCT).
Objective
We used an ML-trained algorithm to quantify ischemic core volume on NCCT in a comparative analysis to pretreatment magnetic resonance imaging (MRI) diffusion-weighted imaging (DWI) in patients with AIS.
Methods
Patients with AIS who had both pretreatment NCCT and MRI were enrolled. An automatic segmentation ML approach was applied using Brainomix software (Oxford, UK) to segment the ischemic voxels and calculate ischemic core volume on NCCT. Ischemic core volume was also calculated on baseline MRI DWI. Comparative analysis was performed using Bland–Altman plots and Pearson correlation.
Results
A total of 72 patients were included. The time-to-stroke onset time was 134.2/89.5 minutes (mean/median). The time difference between NCCT and MRI was 64.8/44.5 minutes (mean/median). In patients who presented within 1 hour from stroke onset, the ischemic core volumes were significantly (p = 0.005) underestimated by ML-NCCT. In patients presented beyond 1 hour, the ML-NCCT estimated ischemic core volumes approximated those obtained by MRI-DWI and with significant correlation (r = 0.56, p < 0.001).
Conclusion
The ischemic core volumes calculated by the described ML approach on NCCT approximate those obtained by MRI in patients with AIS who present beyond 1 hour from stroke onset.
Introduction
Accurate and timely estimation of ischemic core on baseline imaging has treatment implications in patients with acute ischemic stroke (AIS). 1 While noncontrast computed tomography (NCCT) is the most commonly used and widely available imaging modality in acute stroke setting, it has proven challenging for accurate assessment of ischemic core. Early ischemic changes are depicted as hypoattenuation on NCCT can be often subtle and difficult to definitively measure resulting in variability and inconsistent results. 2
The Alberta Stroke Program Early Computed Tomography Score (ASPECTS) was developed in an effort to provide a standardized scale for definition of the extent of early ischemic change on NCCT. 3 After being validated by several randomized clinical trials for endovascular treatment decision making, ASPECTS ≥6 on NCCT is now considered sufficient for treatment decision making according to the current the American Heart Association and the American Stroke Association (AHA-ASA) guidelines, in patients with anterior circulation large vessel occlusion within the first 6 hours from the symptom-onset. 1 However, ASPECTS only offer a semi-quantitative approach that can vary considerably depending on which regions of the brain are involved. In addition, modest interobserver agreement and reproducibility especially in the absence of adequate expertise4–6 can further be limiting. Therefore, a more quantitative assessment of ischemic core remains an attractive alternative.
Thus far volumetric analysis of ischemic core has been reserved largely for more advanced imaging techniques such as CT perfusion (CTP) or magnetic resonance imaging (MRI) that is commonly performed for patients presenting beyond 6 hours from the stroke onset. 1 However, there is precedent for the use of volumetric ischemic core as an eligibility criterion for endovascular therapy (EVT) decision making even in early treatment window based on randomized clinical trials. In fact, core volume obtained through CTP or MRI was used to determine EVT candidacy in a subset of patients in the SWIFTPRIME (< 50 mL) 7 and the EXTEND-IA (<70 mL) trials. 8 Although cross trial analysis and comparison is difficult, use of quantitative imaging to assess ischemic core volumes in these two trials was associated with slight treatment benefits and improved outcome compared to trials using ASPECTS only.
Machine learning (ML) algorithms have shown promising results in estimating ischemic core on NCCT by providing automated quantitation and reducing variability as compared with human observers.9–12 This has opened a window of opportunity to quantify ischemic core volume on commonly used NCCT and obtaining quantitative results that are currently reserved for more advanced imaging techniques such as CTP or MRI.
In this study, by adopting an ML-trained algorithm (Brainomix), we aimed to quantify ischemic core volume on baseline NCCT and to compare the results to pretreatment MRI-diffusion-weighted imaging (DWI) as the reference standard in patients with AIS.
Methods
Study design
This single institutional retrospective study was approved by a local institution review board. Patients with anterior circulation large vessel occlusion who presented to our hospital between 1/2010 and 12/2019 were reviewed and included if they had both baseline pretreatment NCCT and MRI-DWI within 6 hours. Demographic and baseline clinical data including age, sex, stroke-onset time, time of CT and MRI, affected side, and baseline National Institute Health Stroke Scale (NIHSS) scores were collected from patient electronic medical records and our stroke registry.
Image acquisition
NCCT was performed with a multislice CT scanner (Sensation 64, Siemens, Erlangen, Germany) with the following parameters: 120 kVp, Collimation: 24 × 1.2 mm, and 5 mm slice thickness.
MRI was performed on either a 1.5 T MR scanner (Avanto, Siemens, Erlangen, Germany) or a 3 T MR scanner (Trio, Siemens, Erlangen, Germany) within our hospital. DWI was acquired using a single-shot spin-echo echo planar imaging sequence (repetition time/echo time, 4900/98 ms [1.5 T] or 4100/95 ms [3 T]; field of view, 220 × 220 mm; matrix, 128 × 128 mm; slices, 30 × 5 mm). Diffusion gradients were applied along 3 orthogonal directions with b = 0 and 1000 s/mm2.
Image analysis
Ischemic lesion volume was calculated on MRI-DWI using Food and Drug Administration-approved postprocessing software (Olea Sphere (SP21); Olea Medical Solutions, La Ciotat, France). Volume of infarction was calculated by applying a volume of interest on DWI, using a voxel-based signal intensity method subsuming the entire region of DWI hyperintensity. 13
Ischemic core volume was calculated automatically using a commercially available software (Brainomix, Oxford, UK). The image processing algorithm uses an artificial intelligence approach, with a combination of traditional three-dimensional graphics and statistical methods, and ML classification techniques. The training dataset included a large number of (> 10000 CT images) real-world CT scans from stroke patients and negative controls, with ground-truth data from MRI acquired within 1 to 2 hours of the CT scans. In short, the input Digital Imaging and Communications in Medicine data is first resampled to correct any gantry tilt and standardize the input resolution. Then, a fast proprietary registration approach is applied to realign the data, removing any tilt and rotation. The algorithm then uses a trained ML classifier to identify both early and non-acute signs of ischemia in order to derive a voxel-wise probability map. The voxels identified as acute ischemia will then be segmented and volume of ischemic core will be calculated. The output of this ML-trained algorithm was recorded as the volume of ischemic core without any adjustment or human interactions.
In addition, ASPECTS were evaluated by two board certified neuroradiologists (xx with 10 and yy with 8 years of experience) independently. The discrepancies were resolved by consensus.
Statistical analysis
Demographic characteristics and neuroimaging variables were presented as mean (standard deviation, SD) for continuous data and as median and interquartile range (IQR) for categorical data.
The estimated ischemic core volumes from NCCT were compared against MRI-DWI using a Wilcoxon–Mann–Whitney rank test. Mean (SD) and median (IQR) of differences between NCCT-estimated ischemic core volume and MRI were calculated for each patient. Bland–Altman plots were generated to compare the NCCT and MRI-derived ischemic core volumes, and their correlation was tested by Pearson correlation. The data was further dichotomized based on time from stroke onset, either <1 hour, between 1 and 3 hours, and between 3 and 6 hours, and time-based analysis was performed to compare the mean difference of estimated ischemic core between MRI and ML-segmentation on NCCT. Interobserver agreement between two neuroradiologists for ASPECTS was performed using a weighted kappa test with calculation of 95% confidence interval (CI) for both the raw/original scores and dichotomized ASPECTS ( ≥6 and <6). The significance level was defined as p < 0.05. Statistical analyses were performed with MedCalc® statistical software (version 20.008, MedCalc Software Ltd, Ostend, Belgium).
Results
A total of 79 patients with anterior circulation large vessel occlusion who had both CT and MRI before treatment were reviewed. Seven patients were excluded due to unavailable or nondiagnostic imaging resulting in a total of 72 patients for analysis. Demographic information, baseline clinical variables, and mean volumes of ischemic core are listed in Table 1.
Clinical and radiological data of patients with AIS (total of 72 patients).
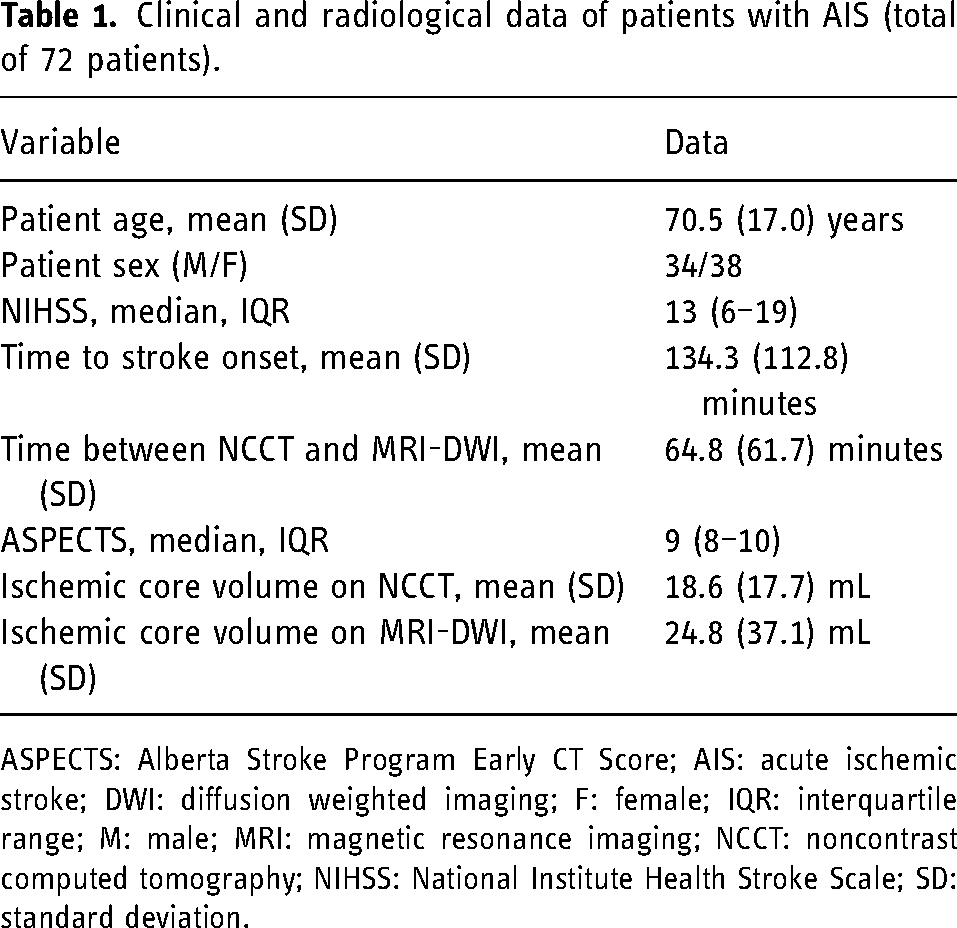
ASPECTS: Alberta Stroke Program Early CT Score; AIS: acute ischemic stroke; DWI: diffusion weighted imaging; F: female; IQR: interquartile range; M: male; MRI: magnetic resonance imaging; NCCT: noncontrast computed tomography; NIHSS: National Institute Health Stroke Scale; SD: standard deviation.
The majority (94.5%) of our cohort presented within 6 hours from symptom onset (mean/median: 134.2/89.5 minutes), with only 4 patients (5.5%) presented after 6 hours from the stroke onset. The mean/median time difference between NCCT and MRI was 64.8/44.5 minutes, with a total of 44 patients (61%) with NCCT and MRI performed within 1 hour and 62 patients (86%) within 2 hours. There were 61 patients who had NCCT performed first and 11 patients who underwent MRI first.
ASPECTS analysis
The median (IQR) for ASPECTS was 9 (7–10) for reader 1 and 9 (8–10) for reader 2. The interobserver agreement was moderate with k = 0.43, 95% CI 0.29 to 0.57. For the dichotomized ASPECTS (ASPECTS ≥6 or <6), the interobserver agreement was improved with k = 0.55, 95% CI 0.34 to 0.70. For the final consensus read, the median (IQR) for ASPECTS was 9 (8–10). In the dichotomized consensus read, a total of 67 patients had ASPECTS ≥6, and 5 patients had ASPECTS <6.
Ischemic core volume analysis
The volume of ischemic core (mean ± SD) was 24.8 ± 37.1 on MRI-DWI and 18.6 ± 17.7 mL via ML-segmentation on NCCT (mean difference: 20.9 mL, median; IQR: 8.2; 2.6–29.3, p = 0.9).
In a time-based analysis, the mean difference of estimated ischemic core between MRI and ML-segmentation on NCCT were analyzed based on time from stroke onset and time difference between CT and MRI (Table 2). ML-segmentation algorithm demonstrated a significant difference in the ischemic core volume within the first hour of presentation (p = 0.005, Table 2). In particular, in five patients with large hemispheric strokes who presented within 1 hour from onset, ML algorithm showed a significant difference in the ischemic core volume with mean difference of 100.0 mL in comparison to MRI (Figure 1). The mean time difference between CT and MRI in this group was 39.5 minutes (all 6 performed within 1 hour).
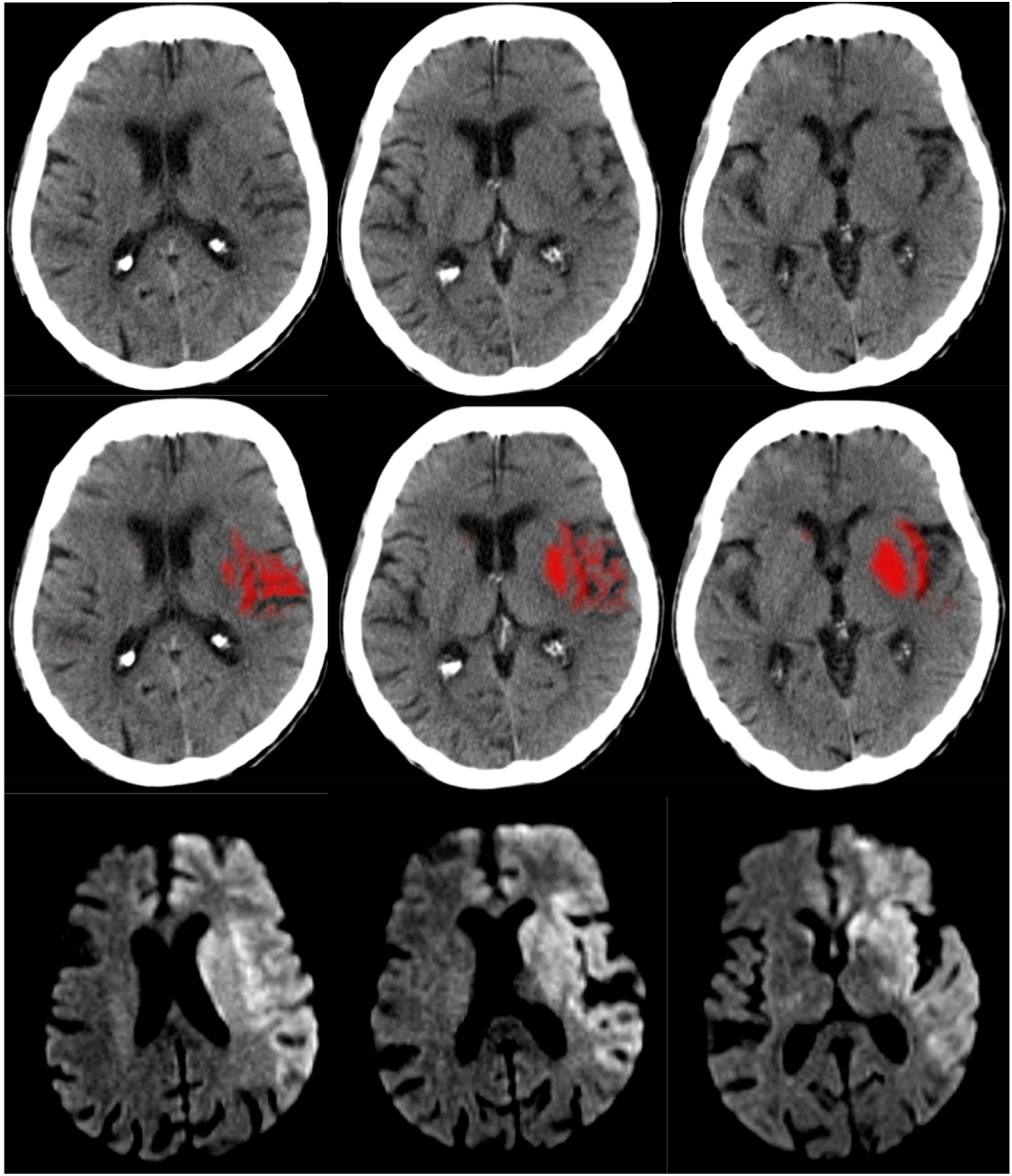
An adult patient with left MCA M1 occlusion (not shown), baseline NIHSS: 33. CT was performed in 45 minutes from the stroke onset followed by MRI 38 minutes later. Ischemic core volume on NCCT is estimated at 27.4 mL automatically via an ML-segmentation algorithm employed by Brainomix software (Oxford, UK) (top row). Consensus ASPECTS was 7. MRI-DWI shows a much larger area of ischemia (bottom row) with calculated ischemic core volume of 126.2 mL. ASPECTS: Alberta Stroke Program Early Computed Tomography Score; CT: computed tomography; DWI: diffusion-weighted imaging; ML: machine learning; MRI: magnetic resonance imaging; NCCT: noncontrast computed tomography; NIHSS: National Institute Health Stroke Scale; MCA: middle cerebral artery.
Differences of estimated ischemic core between MRI and CT categorized based on time from stroke-onset and time difference between CT and MRI.

CT: computed tomography; CI: confidence interval; IQR: interquartile range; MRI: magnetic resonance imaging; r: correlation coefficient; SD: standard deviation; SE: standard error.
Beyond 1 hour from stroke onset, there was no significant difference in estimated ischemic core volume between MRI and ML-NCCT (Table 2). Similarly, no significant difference was identified in estimated core volume based on time difference between CT and MRI (Table 2).
The ischemic core volume in patients presented between 1 and 3 hours from stroke onset (n = 46) was 7.8 mL, 4.3 to 23.7 (median, IQR) on MRI-DWI and 13.1 mL, 5.4 to 28.8 (median, IQR) via ML-segmentation on NCCT (mean difference: 18.5 mL, median 8.15 mL, p = 0.336). The ischemic core volume in patients presented between 3 and 6 hours from stroke onset (n = 11) was 4.1 mL, 1.4 to 13.4 (median, IQR) on MRI-DWI and 13.5 mL, 7.9 to 25.8 (median, IQR) via ML-segmentation on NCCT (mean difference: 14.8 mL, median 8.3 mL, p = 0.21).
There was a significant correlation between estimated core volume on MRI and ML-segmentation on NCCT (r = 0.56, p < 0.001) in patients presenting beyond 1 hour. Figure 2 shows an example of comparable ischemic core estimation on MRI and ML-NCCT.
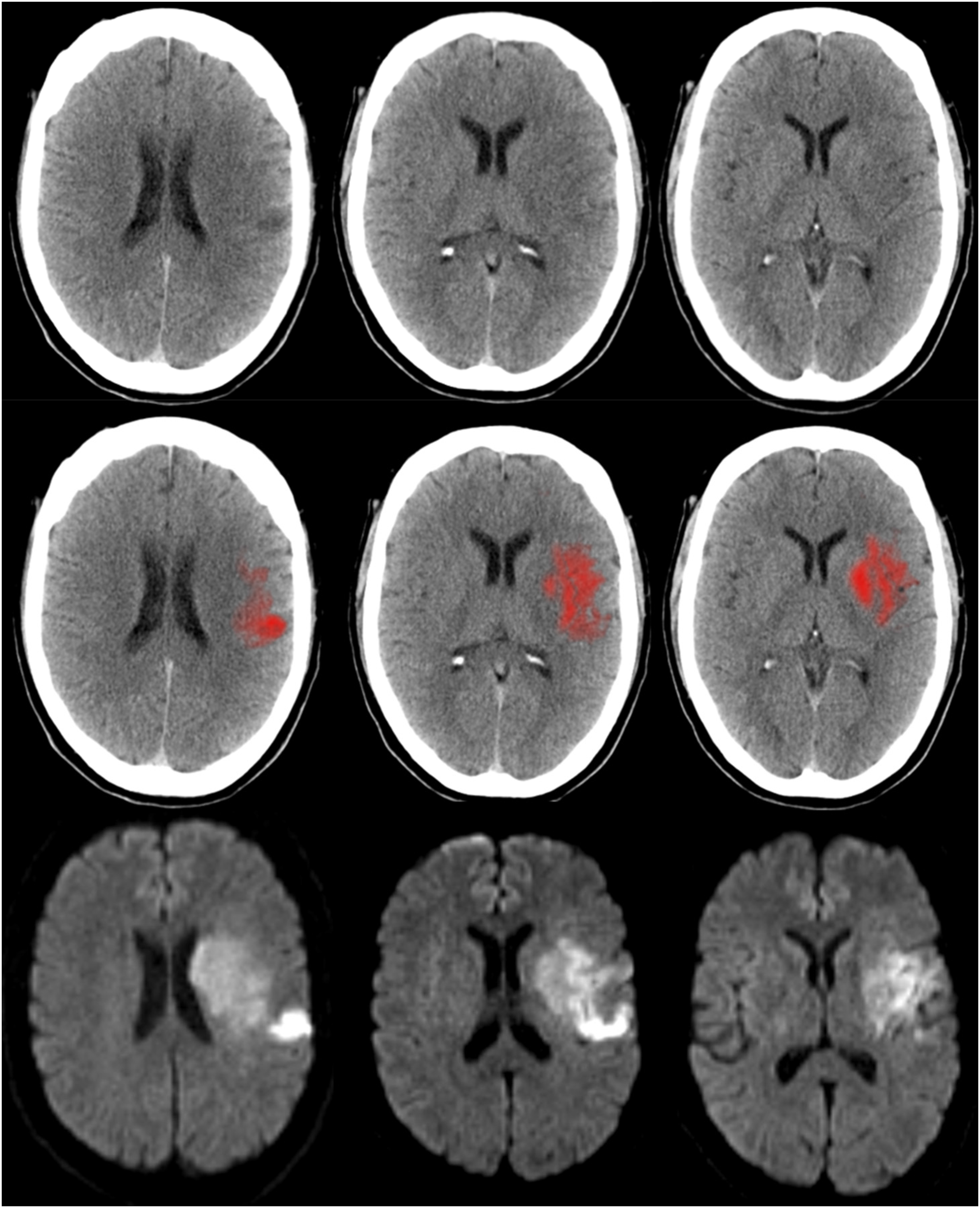
An adult patient with left MCA M1 occlusion (not shown), baseline NIHSS: 15. CT was performed in 76 minutes from the stroke onset followed by MRI 66 minutes later. Ischemic core volume on NCCT is estimated at 29.2 mL automatically via an ML-segmentation algorithm employed by Brainomix software (Oxford, UK) (top row). Consensus ASPECTS was 5. MRI-DWI (bottom row) shows a comparable volume of ischemic core calculated at 34 mL. ASPECTS: Alberta Stroke Program Early Computed Tomography Score; CT: computed tomography; DWI: diffusion-weighted imaging; ML: machine learning; MRI: magnetic resonance imaging; NCCT: noncontrast computed tomography; NIHSS: National Institute Health Stroke Scale; MCA: middle cerebral artery.
Among 4 patients who presented beyond 6 hours from the stroke onset, the estimated ischemic core volume on MRI and ML-segmentation on NCCT were nearly similar (MRI-vol/CT-vol/: 14.6/13.5 mL; 1.8/2.3 mL; 12.9/14.6 mL; 22.5/23.3 mL).
Bland–Altman plots for differences in estimated ischemic core volume in all patients, in patients who presented less than 1 hour from stroke onset, and in patients who presented beyond 1 hour from stroke onset are shown in Figure 3.
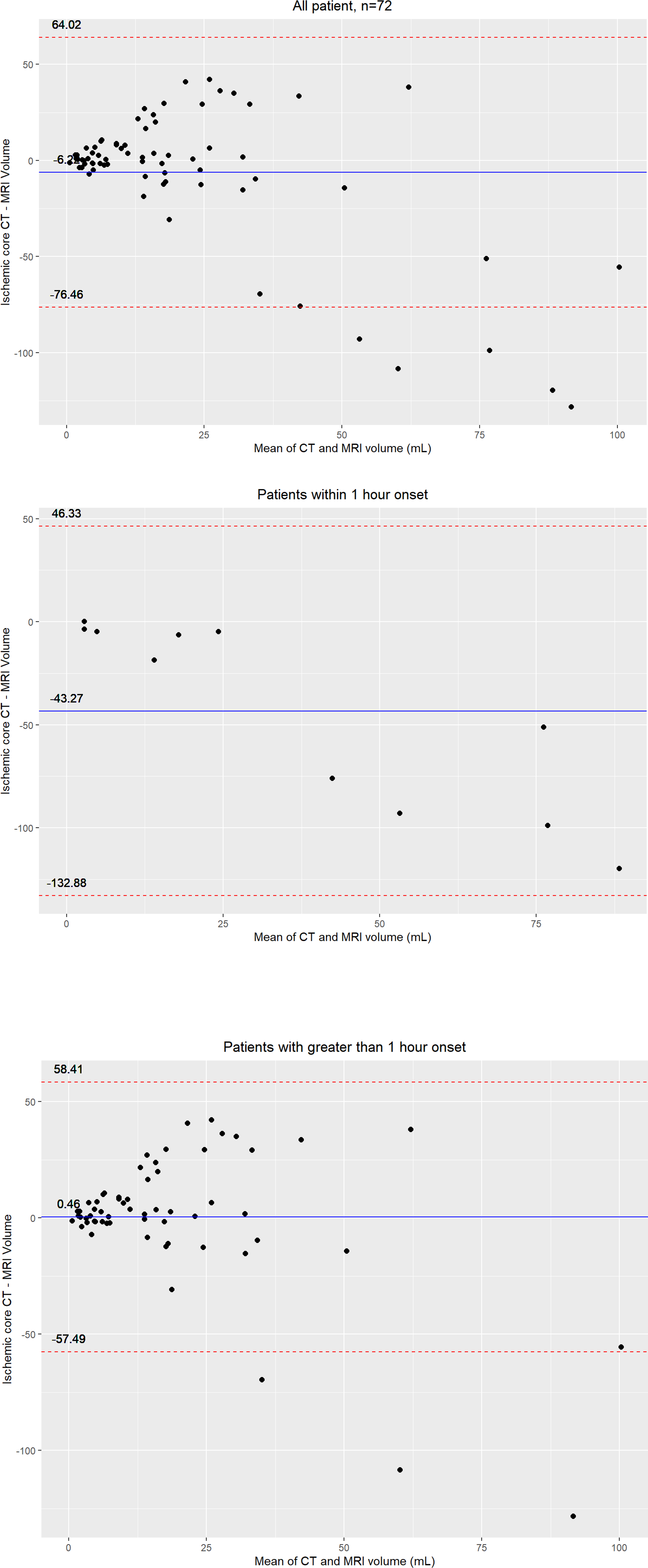
Bland–Altman analysis comparing ML-segmented ischemic core on NCCT and MRI-DWI in all patients (n = 72) (A), patients presenting < 1 hour from stroke onset (n = 10) (B), and patients presenting beyond 1 hour from stroke onset (n = 62) (C). DWI: diffusion-weighted imaging; ML: machine learning; MRI: magnetic resonance imaging; NCCT: noncontrast computed tomography.
Difference of estimated ischemic core volume by time-to-stroke onset is demonstrated in Figure 4.
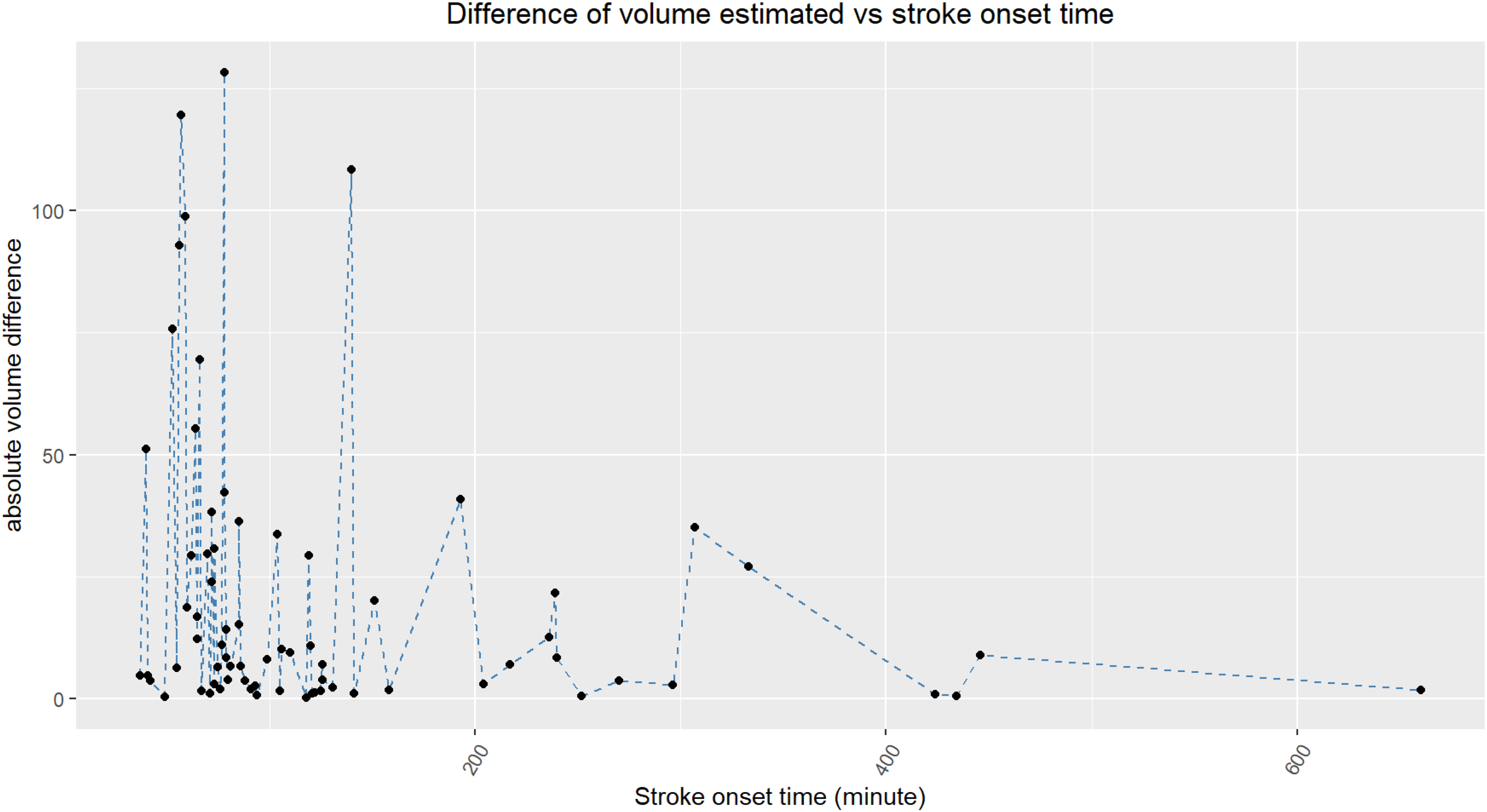
Difference of estimated ischemic core volume plotted by time-to-stroke onset.
Discussion
Our results showed that automated estimated ischemic core volumes obtained by the described ML-approach on NCCT approximate those obtained by MRI-DWI in patients with AIS beyond 1 hour from stroke onset. Although the measurement of ischemic core volume on NCCT can be challenging and time consuming, introduction of ML-approach algorithms provides an opportunity for automating ischemic core volume quantification as shown by our results and others.9–12
The volumetric quantification of ischemic core volume has been reserved mainly for advanced imaging such as CTP or MRI as NCCT tends to be less sensitive in detecting ischemic changes, particularly in early presentation. Introduction of ML techniques has presented a promise to improve detection of early ischemic changes by minimizing the variability factor related to human interaction.9,14,15 In fact, current automated software programs trained based on ML provide automated ASPECTS with promising results in comparison to human interpretation.16–18 On the other hand, quantitative and volumetric assessment of ischemic core on NCCT as shown by our results is now plausible as an attractive alternative approach that can not only mitigate the variability issue related to human interpretation but also provide the incremental value obtained by CTP or MRI. In a recent study by Bouslama et al., 19 using same ML-approach technique (Brainomix e-stroke suite), a promising result was shown in obtaining NCCT-based ischemic core volumes in comparison to CTP. Our results further support the potential for this algorithm in comparison to MRI.
We also showed that ML-estimated ischemic core volume on NCCT is significantly different in comparison to MRI in patients with hyperacute stroke when they present within 1 hour of stroke onset. This may highlight the significant challenges of delineation of early ischemic changes on NCCT in the hyperacute stage of ischemia even for machines. 20 This difference in ischemic core volume in patients with hyperacute strokes may have clinical implications for management, including the consideration for thrombectomy. Some of these challenges are related to conventional image-based segmentation techniques, suboptimal thresholding, noise, partial volume effects, and modest variation in density and texture difference between normal and ischemic brain. 21 Not surprisingly in 4 patients who presented after 6 hours from the stroke onset, ML-approach estimated ischemic core volume was essentially identical to that from MRI, indicating a time-dependent improvement in performance in delineation of early ischemic changes as supported by the literature.17,22
Further work is needed to improve the performance of current ML-based algorithms for more accurate estimation of ischemic core by reducing the impact of confounding factors such as noise and partial volume effect, extraction of more imaging features and improving deep-learning algorithms to increase robustness in delineating subtle ischemic changes. In addition, improvement in image acquisition and reconstruction algorithms has shown promise in increasing accuracy and reliability in detecting early parenchymal ischemic changes.23,24 Finally using dual-energy CT and calculation of ischemic maps may hold promise to further improve core delineation and quantification using NCCT. 25
Despite increased use of ASPECTS in clinical practice, a major limitation remains due to limited interobserver agreement and reproducibility, which is well known in the literature and redemonstrated in our study with only fair interobserver agreement (k = 0.43), even for experienced neuroradiologists.4–6 Another limitation of ASPECTS is that although it can be used as a surrogate for the extent of acute ischemic changes, it does not constitute an accurate representation of ischemic core volume. A recent analysis of the Endovascular Therapy Following Imaging Evaluation for Ischemic Stroke 3 (DEFUSE 3) trial showed a very poor correlation between ischemic core volume and ASPECTS in the middle range (ASPECTS 3–8), suggesting inconsistency in classifying treatment eligibility. 26 In our cohort, we had a total of 5 patients with ASPECTS < 6 who would have been ineligible for EVT according to the current AHA-ASA guidelines. In a hypothetical scenario, if we were to use volume of ischemic core < 70 mL for EVT eligibility (according to what was used in SWIFTPRIME), 3 of these patients would be correctly reclassified to EVT eligible by ML-based volumetric approach as having volume < 70 mL, concordant with MRI (Figure 2). On the other hand, if MRI-estimated ischemic core was used for treatment decision making, 8 patients would have been excluded from EVT based on MRI (ischemic core volume >70 mL). Among these, 2 patients would have been correctly classified as EVT-ineligible by CT (CT-vol >70 mL), while 6 patients would have been falsely classified as EVT eligible due to underestimation of ischemic core (CT-vol <70 mL). Five of these 6 patients were presented within 1 hour from the stroke onset, a described limitation of our algorithm.
Our study has several limitations. The retrospective nature introduces unknown bias, and the single-center experience limits generalizability. Our sample size is small. Since the majority of stroke patients undergo CT or MRI before treatment, the requirement for having both pretreatment NCCT and MRI was a major limiting factor resulting in only 79 patients being available for review despite extending the chart review to a 10-year span. The discussion about treatment eligibility based on CT and MRI ischemic core volume is hypothetical since our study was not designed to address treatment implications based on ischemic core volume due to its retrospective nature. A future prospective study is needed to address the treatment implications of ischemic core volume estimation on NCCT. The comparison of ischemic core volume on NCCT (parenchymal density change) and MRI-DWI (cytotoxic edema) is not a parallel comparison. However, MRI-DWI is still the best available imaging modality and considered the gold standard for defining early ischemia. It is possible that by depicting cytotoxic edema in an acutely stunned brain, DWI may overestimate the extent of ischemia in the hyperacute stage that can exaggerate the differences observed between NCCT and MRI in patients presented within 1 hour from stroke onset. 27 Due to retrospective nature, we only had access to reconstructed 5 mm slices on NCCT. It is plausible that using thinner cuts CT images e.g. 1.25 mm) may improve the performance of ML techniques to delineate early ischemic changes. 28
In summary, the described ML method provided automated ischemic core volumes from baseline NCCT that approximated those obtained by MRI-DWI in patients with AIS presenting beyond 1 hour from stroke onset. If its potential is realized in a broader setting, this ML method may provide an opportunity for more objective quantitative assessment of ischemic core similar to what is obtained by CTP or MRI while affording the simplicity of NCCT.
Footnotes
Abbreviations
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institutes of Health (grant number R56 EB031993) for Dr. Kambiz Nael and Dr. William Hsu.
