Abstract
Background
A number of clinical trials have compared tenecteplase (TNK) and alteplase for the management of acute ischemic stroke (AIS) and the results are inconsistent.
Purpose
Present systematic review and meta-analysis is undertaken to analyse the efficacy and safety of TNK in AIS compared to alteplase.
Summary
A thorough literature search was performed through the databases Embase, Cochrane Library, PubMed, and clinicaltrials.gov, for a period from inception to September 2022, with the keywords i.e., “tenecteplase” and “alteplase” and “acute ischemic stroke.” Clinical trials published in English that compared the efficacy and safety of TNK to alteplase in AIS were included. The major outcomes of this meta-analysis were proportion of patients free from disability and functional independence at 90 days, early neurological improvement at 24 hours, all-cause mortality at 90 days, patients with intra cranial hemorrhage (ICH), and patients with severe disability at 90 days. A total of nine studies with 3,573 patients were included in the analysis. The proportion of patients with freedom from disability was comparable in both groups (relative risk [RR] = 1.04, 95 per cent CI = 0.92–1.17; p = .53). Similarly, proportion of patients with functional independence was comparable (RR = 1.12, 95 per cent CI = 0.96–1.31; p = .14). TNK group had a higher rate of early neurological recovery (RR = 1.56, 95 per cent CI = 0.96–2.54; p = .07). All-cause mortality at 90 days was comparable in both groups (RR = 0.97; 95 per cent CI = 0.72–1.29; p = .82). The proportion of patients with ICH was higher in TNK group (RR = 1.14, 95 per cent CI = 0.77–1.68; p = .52). The proportion of patients with severe disability was less in TNK group (RR =0.84, 95 per cent CI = 0.53–1.32; p = .44).
Key Message
TNK was similar to alteplase in terms of efficacy and safety. The patients in TNK group showed early neurological improvement but were simultaneously at higher risk of ICH. The TNK can be an alternative to alteplase if the benefits outweigh the risks.
Introduction
A stroke or cerebrovascular accident is characterized by impaired blood flow to the brain and is broadly divided into the ischemic and hemorrhagic stroke. Ischemic stroke is the most common type of stroke. 1 Sexual predilection varies with age; at a young age, females are at more risk, while in older adults, males are at slightly higher risk. 1 Worldwide, stroke is the second most common cause of death and ranks third among causes of death and disability combined. 2 Furthermore, approximately 10 per cent of acute ischemic stroke (AIS) patients die within a year, and 20 to 25 per cent suffer from severe disability. 3
The primary goal of AIS treatment is to restore brain tissue circulation as early as possible by achieving cerebral blood vessel recanalization. 4 To reduce the mortality and disability rate of patients suffering from ischemic stroke, prompt management with intravenous thrombolytics to recanalize blocked blood vessels is essential. 5 Various guidelines have proposed intravascular thrombolytics like recombinant tissue-plasminogen activator (RT-PA) within a suitable time window as an effective treatment method for AIS. 6
Alteplase (rtPA) leads to the conversion of plasminogen into plasmin to achieve thrombolysis. The United States Food and Drug Administration (USFDA) has approved it for AIS at a recommended dose of 0.9 mg/kg (total dose not to exceed 90 mg) infused intravenously over 60 minutes, with 10 per cent of the total dose administered as an initial bolus over one minute. It should be given at the earliest but no later than three hours after the onset of symptoms following a stroke episode. 7
Tenecteplase (TNK) is a rtPA variant with a longer half-life and 14-fold more fibrin specificity. A longer half-life (18 minutes) allows its single IV bolus administration, which makes it easier to administer. 8 It has not been approved for the treatment of AIS by the USFDA. TNK in the dose of 0.1–0.4 mg/kg was found to be safe for ischemic stroke in a non-randomized dose-increasing safety study. 9 Even so, the intravenous administration of TNK to treat acute stroke is still regarded as off-label. A growing number of patients are being treated for AIS using intravenous TNK, especially in nations where TNK is more affordable than rtPA.10–12 Although several recent international guidelines have endorsed its use in AIS, alteplase remains the only intravenous thrombolytic drug that has received regulatory approval for treating AIS.13–15 The USFDA has approved alteplase for treating pulmonary embolism, myocardial infarction with ST-segment elevation, ischemic stroke when administered within three hours of the onset of symptoms, and re-establishment of patency in occluded intravenous catheters. 16 TNK, on the other hand, has been approved for use in the reduction of mortality associated with acute myocardial infarction. 17 TNK, currently, lacks a labeled indication for AIS. Several randomized controlled trials have evaluated the use of TNK in AIS patients, but the results have been inconsistent, and the level of evidence needs to be increased. Furthermore, it is still being determined from the trials whether the pharmacokinetic advantage of TNK of a single bolus dose is worth using in clinical practice. As a result, this review will systematically evaluate whether rtPA or TNK is a better option for intravenous thrombolysis in AIS patients in terms of efficacy and safety in order to provide a reliable reference basis for clinical application.
Methods
The review authors performed the systematic review and meta-analysis according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 18
Search Strategy
A rigorous systematic search was performed of numerous databases, i.e., Embase, PubMed, Clinical Trial Registry
Study Selection Process.
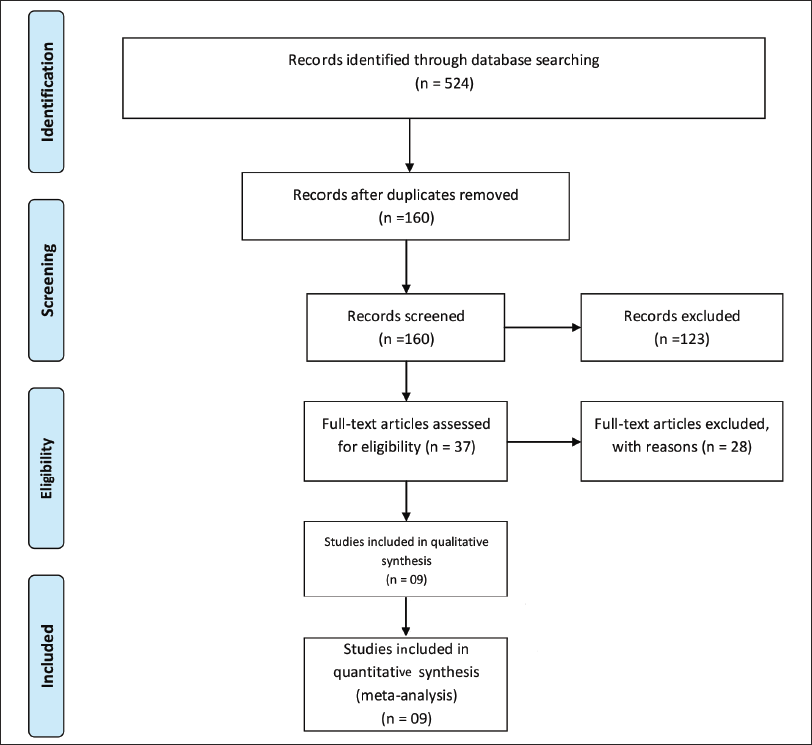
Study Selection
The randomized clinical trials involving patients of an AIS aged >18 years and compared TNK with Alteplase (ALT) in different phases of clinical trials were included.
Data Extraction
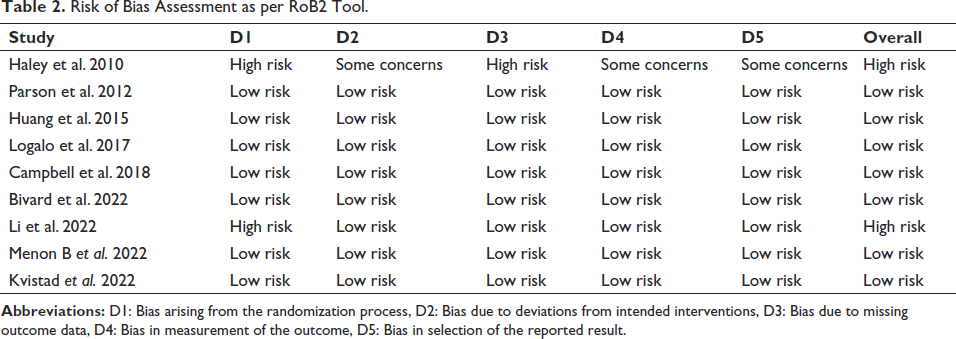
Two review authors (AS and MPS) performed the data extraction using Microsoft Excel 2016. Data extraction included demographic information, inclusion and exclusion criteria, treatment schedule, study design, and all outcomes. Any missing information was obtained from the protocol, statistical analysis plan, and other published analyses. Subsequently, all the relevant data was analysed in Review Manager 5.4 for Windows. The risk of bias (RoB) for the individual study was assessed using both, RoB and RoB2 assessment tools. The previous RoB tool was utilized for preparing the summary of findings (SoF); however, the RoB is presented in results using the RoB2 tool.19, 20 The biases assessed for each included selection bias, performance bias, detection bias, attrition bias, and reporting bias, as per previous RoB tool. The RoB2 tool was used to evaluate numerous biases, that is, bias arising from the randomization process, bias due to deviations from intended interventions, bias due to missing outcome data, bias in the measurement of the outcome, and bias in the selection of the reported result. Publication bias was checked by using a funnel plot for each outcome. The strength of evidence was judged with the GRADE approach considering the RoB, inconsistency, indirectness, imprecision, and publication bias. 21
Outcomes
The efficacy and safety endpoints are included below:
The proportion of patients with freedom from disability at 90 days [Modified Rankin Scale (mRS): 0–1]. The proportion of patients with functional independence at 90 days [mRS: 0–2]. The proportion of patients with early neurological improvement at 24 hours (NIHSS score decreased by ≥8 points or an NIHSS score of 0 or 1 at 24 hours posttreatment). All-cause mortality at 90 days. The proportion of patients with intracranial hemorrhage (ICH) with or without symptoms. The proportion of patients with severe disability at 90 days [mRS: 5]. Incidence of treatment-emergent adverse events (TEAEs). The proportion of patients with deterioration in mRS at least by one point at 90 days.
Statistical Analysis
The relative risks (RRs) with 95 per cent confidence intervals (CIs) were used for dichotomous data. The true heterogeneity among the included studies was assessed with I 2 statistics, and a p value of .10 was considered significant for the same. 22 The authors used the Random-effects model to conduct a meta-analysis. Sensitivity analysis was performed by removing one study at a time and using different models to assess the robustness of the results.
Results
Baseline Characteristics
The study includes 3,573 patients from nine randomized clinical trials.23–31 Of nine trials, two contributed 74.6 per cent of participants.26, 31 The mean age of patients was 71 years, and 57.1 per cent were male. The significant reasons for excluding studies were conference paper abstracts and posters with inappropriate details and others being narrative and systematic reviews. The study selection process is shown in Figure 1. The baseline information of included studies is listed in Table 1. Overall, majority of patients were white. The diagnosis of AIS was based on a CT scan, and hemorrhagic stroke was ruled out in all trials. All clinical trials used alteplase in a dose of 0.9 mg/kg given as an infusion and TNK in the dose range of 0.1–0.4 mg/kg as a bolus. Review authors included TNK in a dose of 0.25–0.4 mg/kg for comparison with Alteplase. The RoB is presented in Figure 2. Out of nine clinical trials, one trial was at high RoB as per the RoB2 tool. 23 The RoB in different domains for individual trials is presented in Table 2.
Baseline Characteristics of Included Studies.
Risk of Bias Summary.

Risk of Bias Assessment as per RoB2 Tool.
Efficacy Endpoints
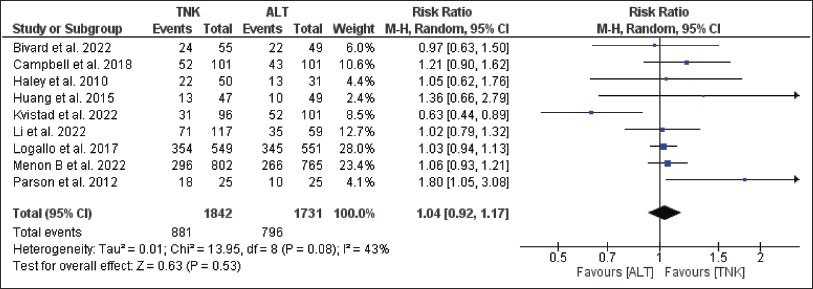
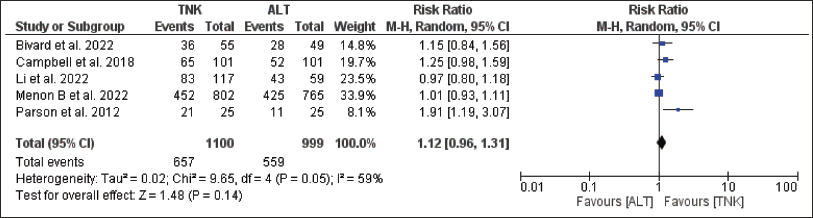
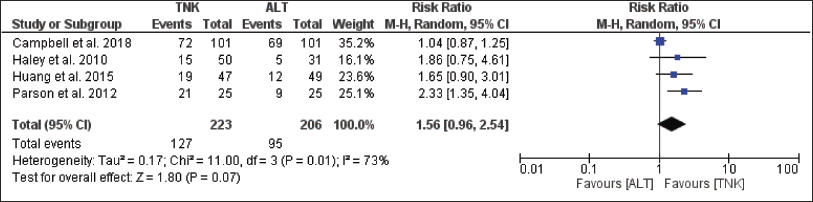
For the efficacy endpoint, i.e., the proportion of patients with freedom from disability at 90 days, publication bias was suspected based on an asymmetric funnel plot. Further, significant heterogeneity was observed (I 2 = 43 per cent, p = .08) (Figure 3). TNK group had more disability-free patients than the alteplase group (47.8 per cent vs. 46 per cent); RR = 1.04, 95 per cent CI = 0.92–1.17; p = .53 (Figure 3). The proportion of patients with functional independence at 90 days were higher in TNK group compared to Alteplase (59.7 per cent vs. 56 per cent); RR = 1.12, 95 per cent CI = 0.96–1.31; p = .14 (Figure 4). For this outcome publication bias was observed, and significant heterogeneity was observed (I 2 = 59 per cent, p = .05) (Figure 4). The proportion of patients showing early neurological improvement was considerably higher in TNK group than alteplase group (56.9 per cent vs. 46.1 per cent); RR = 1.56, 95 per cent CI = 0.96–2.54; p = .07 (Figure 5). For this outcome, no publication bias was observed, but significant heterogeneity was observed (I 2 = 73 per cent, p = .01) (Figure 5). Funnel plots for individual outcomes have been presented as supplementary files (S1–S6).
Forest Plot for no Disability.
Forest Plot for Functional Independence.
Forest Plot for Neurological Improvement.
Safety Endpoints
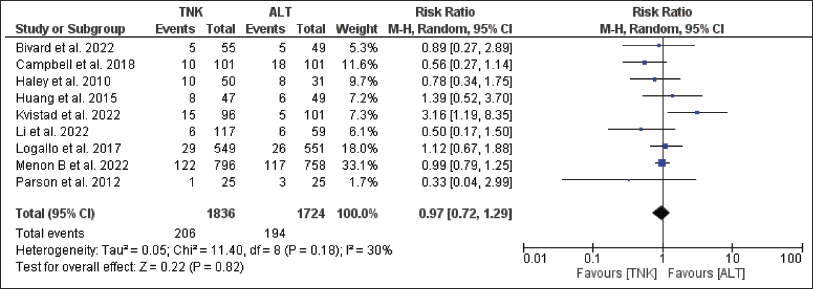
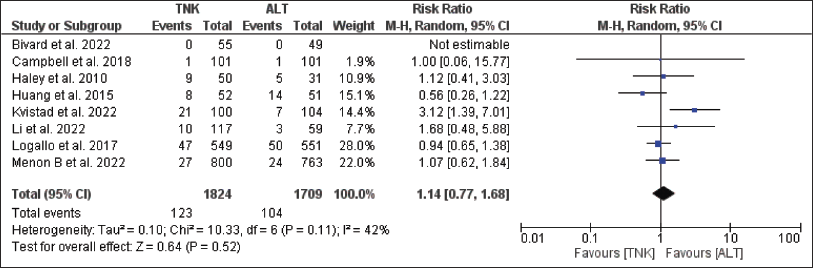
Among the safety endpoints, all-cause mortality at 90 days was higher in the alteplase group compared to TNK (11.3 per cent vs. 11.2 per cent), though statistically not significant; RR = 0.97; 95 per cent CI = 0.72–1.29; p = .82 (Figure 6). The publication bias was suspected as the funnel plot was asymmetrical. Heterogeneity was observed but was not significant (I 2 = 30 per cent, p = .18) (Figure 6). The patients of the TNK group had more ICH than the alteplase group (6.7 per cent vs. 6.1 per cent); RR = 1.14, 95 per cent CI = 0.77–1.68; p = .52 (Figure 7). The publication bias was suspected as funnel plot was asymmetrical. Heterogeneity was observed but was not significant (I 2 = 42 per cent, p = .11) (Figure 7). The TNK group had fewer patients with severe disability than alteplase group (3.9 per cent vs. 4.5 per cent); RR = 0.84, 95 per cent CI = 0.53–1.32; p = .44 (Figure 8). The publication bias was not suspected as funnel plot was symmetrical and heterogeneity was observed but was not significant (I 2 = 17 per cent, p = .30) (Figure 8). None of the studies addressed the last two endpoints, that is, the incidence of TEAEs and proportion of patients with deterioration in mRS at least by one point at 90 days.
Forest Plot for all-cause Mortality.
Forest Plot for Intracerebral Hemorrhage.
Forest Plot for Severe Disability.
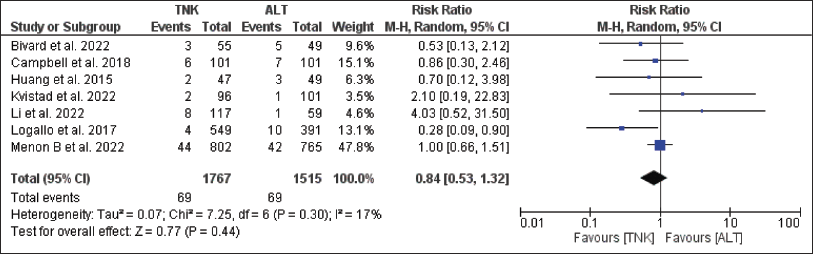
The sensitivity analysis was performed for each outcome, and the results were consistent except for the proportion of patients with early neurological improvement. Similar results were obtained when we analysed using a fixed-effect model instead of a random-effects model, except for the proportion of patients with early neurological improvement. Detailed estimates of all the endpoints and certainty of evidence have presented a table of SoF (Table 3).
Summary of Findings.
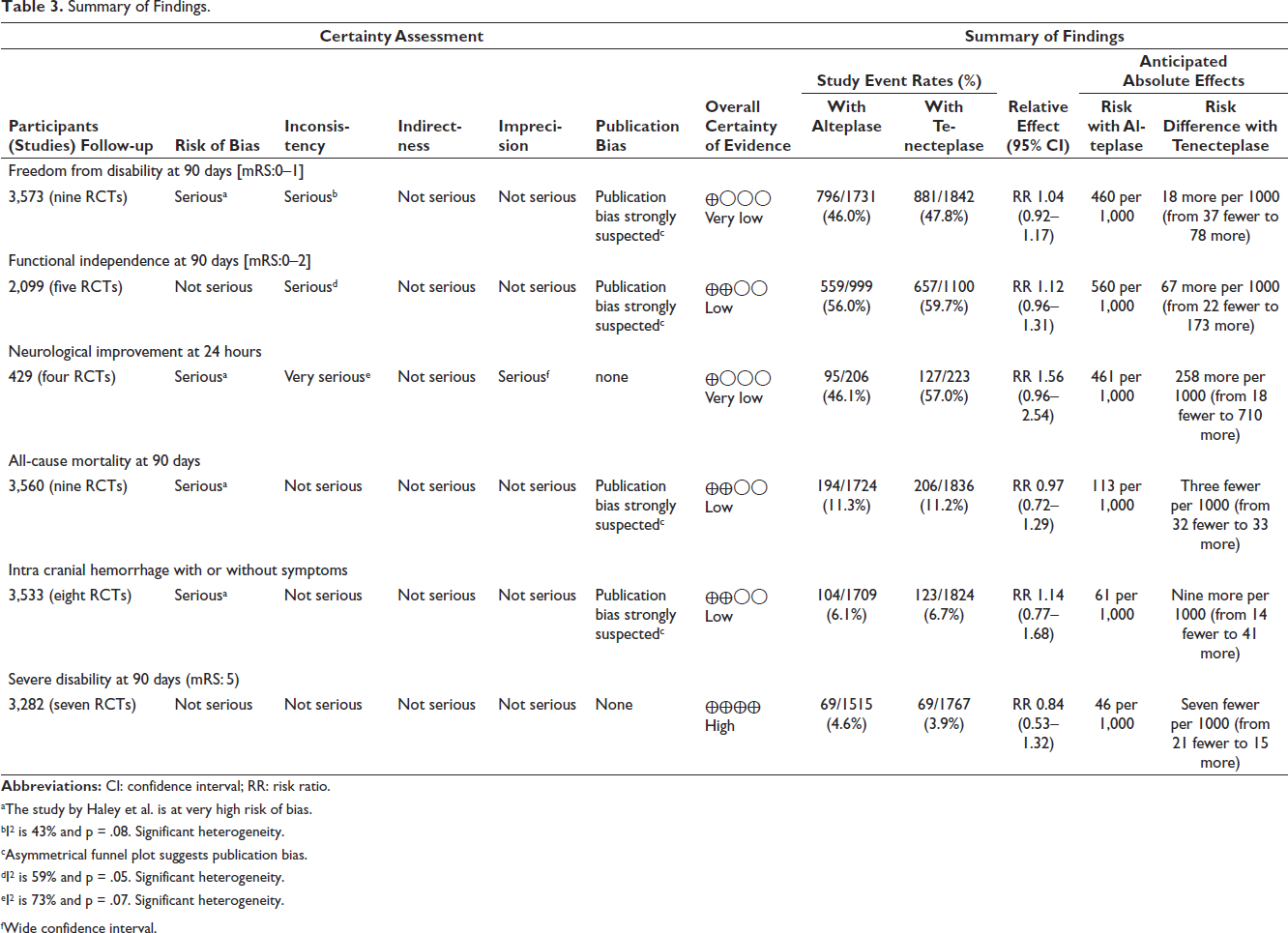
aThe study by Haley et al. is at very high risk of bias.
bI 2 is 43% and p = .08. Significant heterogeneity.
cAsymmetrical funnel plot suggests publication bias.
dI 2 is 59% and p = .05. Significant heterogeneity.
eI 2 is 73% and p = .07. Significant heterogeneity.
fWide confidence interval.
Discussion
In the current study, most of the patients were male and above 70 years, following the usual epidemiologic distribution. 32 Furthermore, only two studies contributed the maximum number of patients (75 per cent), which creates an imbalance; these studies had disproportionately higher weight than those. In the present meta-analysis, we observed that the TNK-treated group had slightly higher proportion of patients showing early neurological improvement at 24 hours, a greater proportion of disability-free patients, and a greater degree of functional independence than the alteplase group at 90 days. ICH was seen more frequently in the TNK group than in the alteplase group. The all-cause mortality was similar in both groups. Also, comparatively fewer patients had a severe disability in the TNK group.
In different RCTs, TNK was administered in AIS patients at varying doses, that is, 0.20, 0.25, 0.32, and 0.40 mg/kg.23–31 The review authors have included studies in which TNK was administered at a dose of 0.25–0.4 mg/kg in patients of AIS since most of the studies showed its efficacy at a dose of 0.25 mg/kg.
TNK has been shown to favorably affect all endpoints that must be balanced with an increased risk of ICH (Table 3). Although early neurological improvement was observed with TNK, that should be carefully evaluated. This endpoint had the least number of patients and studies, maximum heterogeneity, and results were affected during sensitivity analysis (Figure 5). Upon removing the study by Campbell et al., the results were statistically significant. Similarly, the results were highly significant when the authors changed to a fixed-effect model (Supplementary file S7, S8). Hence, early improvement in neurological symptoms cannot be substantiated based on currently available evidence.
Several systematic reviews and meta-analyses have compared the effectiveness and safety of TNK and alteplase for treating AIS with superior and non-inferiority designs.33–45 Overall, the studies suggested that TNK may be more effective than Alteplase in improving early neurological function and higher recanalization rates; there have been no significant differences between the two drugs regarding functional outcomes, disability-free three-month outcomes, or mortality. The optimal dose of TNK appears to be 0.25 mg/kg, which has been shown to have the greatest odds of achieving early neurological improvement and an excellent functional outcome at three months, with reduced odds of ICH.33–45
A recently published randomized controlled non-inferiority trial involving 1,430 participants conducted in China evaluated the efficacy and safety of TNK compared to Alteplase in adults with an AIS who were not eligible for endovascular thrombectomy. The results showed that the TNK and Alteplase were comparable in efficacy (mRS score of 0–1 at 90 days) and safety (mortality and ICH). Therefore, TNK could be an alternative to Alteplase for such patients. 46
Current guidelines recommend TNK (0.4 mg/kg) as an alternative to Alteplase (dose) in limited patients with minor AIS with no significant intracranial occlusion (level of evidence B-R and class of recommendation IIb). 15 The class IIb recommendations indicate weak evidence with benefit may be equal to or more than risk, whereas the level of evidence B-R indicates moderate quality evidence. 15 The findings of the present review further substantiate the earlier observations, that is, low to moderate evidence of efficacy comparable to Alteplase. The ease of administration and better pharmacokinetic profile favors TNK over alteplase for thrombolysis in AIS. The limitation of our study includes that we have not quantified the publication bias. There was variability in the dose and duration of administration of TNK in different studies, so that it may result in heterogeneity.
Conclusion
In the present review, the authors noted relatively better outcomes in the TNK group among efficacy parameters which were not statistically significant with low-moderate certainty of evidence. Safety parameters were generally comparable, but the TNK group had more risk of ICH. TNK administered at a dose range of 0.25–0.4 mg/kg in AIS patients showed comparable efficacy and safety with Alteplase. From this, it is clear that TNK is comparable to Alteplase with low certainty of the evidence as the total number of participants in included trials is less, and more studies, especially non-inferior trials are required to find the exact place of TNK in AIS.
Abbreviations
AIS: Acute Ischemic Stroke.
ICH: Intra Cranial Hemorrhage.
RT-PA: Recombinant Tissue-plasminogen Activator.
USFDA: United States Food and Drug Administration.
TNK: Tenecteplase.
PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
ALT: Alteplase.
RoB: Risk of Bias.
mRS: Modified Rankin Scale.
NIHSS: National Institutes of Health Stroke Scale
TEAE: Treatment-emergent Adverse Event.
RR: Relative Risks.
CI: Confidence Interval.
Authors’ Contribution
All authors framed the concept. Search and meta-analysis were performed by AS and MPS. AS and MPS drafted the manuscript. NRG and PKJ critically evaluated the manuscript and all authors approved the manuscript.
Statement of Ethics
The study does not require ethical approval and was exempted from ethical approval.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
ICMJE Statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article and no funding was received.
