Abstract
Background
Current guidelines recommend that eligible acute ischemic stroke (AIS) patients receive intravenous alteplase (IVT) prior to endovascular treatment (EVT). Six randomized controlled trials recently sought to determine the risks of administering IVT prior to EVT, five of which have been published/presented. It is unclear whether and how the results of these trials will change guidelines. With the DEBATE survey, we assessed the influence of the recent trials on physicians’ IVT treatment strategies in the setting of EVT for large vessel occlusion (LVO) stroke.
Methods
Participants were provided with 15 direct-to-mothership case-scenarios of LVO stroke patients and asked whether they would treat with IVT + EVT or EVT alone, a) before publication/presentation of the direct-to-EVT trials, and b) now (knowing the trial results). Logistic regression clustered by respondent was performed to assess factors influencing the decision to adopt an EVT-alone paradigm after publication/presentation of the trial results.
Results
289 participants from 37 countries provided 4335 responses, of which 13.5% (584/4335) changed from an IVT + EVT strategy to EVT alone after knowing the trial results. Very few switched from EVT alone to IVT + EVT (8/4335, 0.18%). Scenarios involving a long thrombus (RR 1.88, 95%CI:1.56–2.26), cerebral micro-hemorrhages (RR 1.78, 95%CI:1.43–2.23), and an expected short time to recanalization (RR 1.46 95%CI:1.19–1.78) had the highest chance of participants switching to an EVT-only strategy.
Conclusion
In light of the recent direct-to-EVT trials, a sizeable proportion of stroke physicians appears to be rethinking IVT treatment strategies of EVT-eligible mothership patients with AIS due to LVO in specific situations.
Introduction
In 2015, endovascular treatment (EVT) became the standard of care for acute ischemic stroke (AIS) due to large vessel occlusion (LVO) of the anterior circulation.1–5 For eligible patients, current guidelines recommend prior administration of intravenous alteplase (IVT) if there are no contraindications 6 . Although the rates of successful reperfusion with IVT are generally low in LVO,7,8 IVT can in some cases recanalize the vessel before EVT. 9 It is also thought to promote dissolution of distal emboli, which may occur either spontaneously or due to fragmentation of the thrombus during EVT. 10 On the other hand, IVT may be associated with an increased risk of intracranial hemorrhage and other bleeding complications, 11 and due to the numerous contraindications, determining IVT eligibility can result in treatment delays.12,13 Recently, five randomized trials sought to investigate whether EVT alone resulted in comparable outcomes to IVT followed by EVT: both DIRECT-MT 14 and DEVT 15 were able to demonstrate non-inferiority of the direct to EVT paradigm, while the MR CLEAN-NO IV (presented at ISC 2021), SWIFT-DIRECT (presented at ESOC 2021), and SKIP 16 results did not reach statistical significance for non-inferiority, despite comparable patient outcomes. While DIRECT-SAFE (NCT03494920) is ongoing, it is currently unclear whether the available data would result in a change of guidelines.
Therefore, with this survey-based study, we aimed to assess the influence of the recent direct-to-EVT trial results on treatment strategies with respect to IVT for AIS due to LVO.
Methods
Survey design
We conducted an international online, cross-sectional, anonymous, invitation-only survey (DEBATE – [Decision-making for Borderline cases After tPA/EVT Trial Evidence]) among stroke physicians using Qualtrics (www.qualtrics.com). The survey took approximately 10 min to complete and consisted of 15 brief scenarios that represented borderline and/or difficult cases for IVT decision-making (7 requiring special consideration, 3 contraindicated, as according to guidelines,
6
Table 1). Five cases had a relative (level IIB) recommendation for EVT according to current guidelines.
6
For each scenario, participants were asked how they would treat the patient in question. Due to the nature of the study (survey) and the use of anonymized data, the need for ethics approval was waived by the Conjoint Health Research Ethics Board (CHREB) of the University of Calgary. Data used in the current study are available from the author upon reasonable request (see also
Selected scenarios (total n = 15) of difficult/borderline thrombolysis decision-making and the associated level of evidence.

A: high quality evidence (e.g. from >1 RCT); Acom: anterior communicating artery; ASPECTS: Alberta Stroke Program Early CT Score; C-EO: expert opinion; B-NR: moderate quality evidence from non-randomized studies; B-R: moderate quality evidence from ≥1 RCT) CI: confidence interval; C-LD: limited data; EVT: endovascular treatment; LOE: level of evidence 6 ; mRS: modified Rankin Scale; SAH: subarachnoid hemorrhage; NA: not available.
+ Associated risks.
- No known associated risks.
See Supplementary Material.
Class of recommendation and LOE determined by presence of M2 thrombus.
Guidelines state that it is reasonable to maintain blood pressure at ≤180/105mmHg during and for 24 h following EVT, but do not comment on pre-therapeutic blood pressure for EVT.
The usefulness/risks of IVT are not well established in these scenarios.
Survey participants
Approximately 2300 stroke physicians (neurologists, interventional neurologists, interventional neuroradiologists, neurosurgeons, and other physicians directly involved in AIS care) from 50 countries were invited to participate in this web-based survey in April 2021. There were no restrictions with respect to personal experience with EVT or IVT. Prior to accessing the case scenarios, participants were asked to provide their age, gender, specialty, career stage (e.g. resident, fellow, junior staff, senior staff), and geographic region of practice.
Clinical case scenarios
All 15 narrative survey cases took place within a mothership scenario (e.g. describing patients presenting directly at a comprehensive stroke center with EVT capabilities; no transferred patients). Information was provided on vessel occlusion site, time from symptom onset, patient age, sex, National Institutes of Health Stroke Scale (NIHSS) score, and Alberta Stroke Program Early CT Score (ASPECTS). For each scenario, participants were asked what treatment approach they would have chosen a) prior to publication of the direct-to-EVT trials and b) what approach they would choose now, knowing the results. Response options were either “direct to EVT” or “IVT + EVT”. For a detailed description of all case scenarios, see the
Statistical analysis
The primary outcome was the binarized change from IVT + EVT to EVT alone (i.e. if the participant would have chosen IVT + EVT prior to publication of the trials and then changed to EVT alone with their current knowledge). This outcome was chosen in line with the hypotheses and findings of the trials, and because the vast majority of participants who changed their treatment approach chose to adopt an EVT alone strategy.
Participant characteristics and response data were analyzed using descriptive statistics and group differences in willingness to change to an EVT alone approach were assessed using the chi-square test. Respondent age was dichotomized at 45 years (the median). Univariable logistic regression clustered for respondent identity was used to provide measures of effect size for each specific scenario (
We report risk ratios (RR) derived from binary logistic regression. P-values < 0.05 were considered statistically significant. Data analysis was performed with Stata 15.1. Figures were created with Microsoft Power BI Platform.
Results
Respondent demographics
A total of 289 participants (n = 48, 16.6% women) from 37 countries provided 4335 responses to the survey. The majority (n = 129, 45%) were neurologists, followed by interventional neuroradiologists (n = 79, 27%), interventional neurologists (n = 43, 15%), neurosurgeons (n = 35, 12%), and other (n = 9, 3, 1%). Detailed participant characteristics are listed in Supplementary Table 1 .
Change in treatment strategy
Based on the results of the direct to EVT trials, 13.7% (592/4335 responses) changed their treatment approach. Of these, the vast majority (n = 584, 98.6%) indicated a switch from a combined IVT + EVT approach to an EVT only paradigm. Furthermore, for every scenario, a greater proportion of physicians elected to perform EVT alone following publication/presentation of the trials (Figure 1) compared to before. The scenarios that had the highest rates of adoption of an EVT only approach were those involving an intracranial internal carotid T occlusion with a long M1 thrombus (23.9%, scenario 5), a patient with multiple micro-hemorrhages (22.8%, scenario 14), and a patient with an expected short time to recanalization of ≤30 min (19.3%, scenario 6) (Table 2, Figure 1, Supplementary Material).
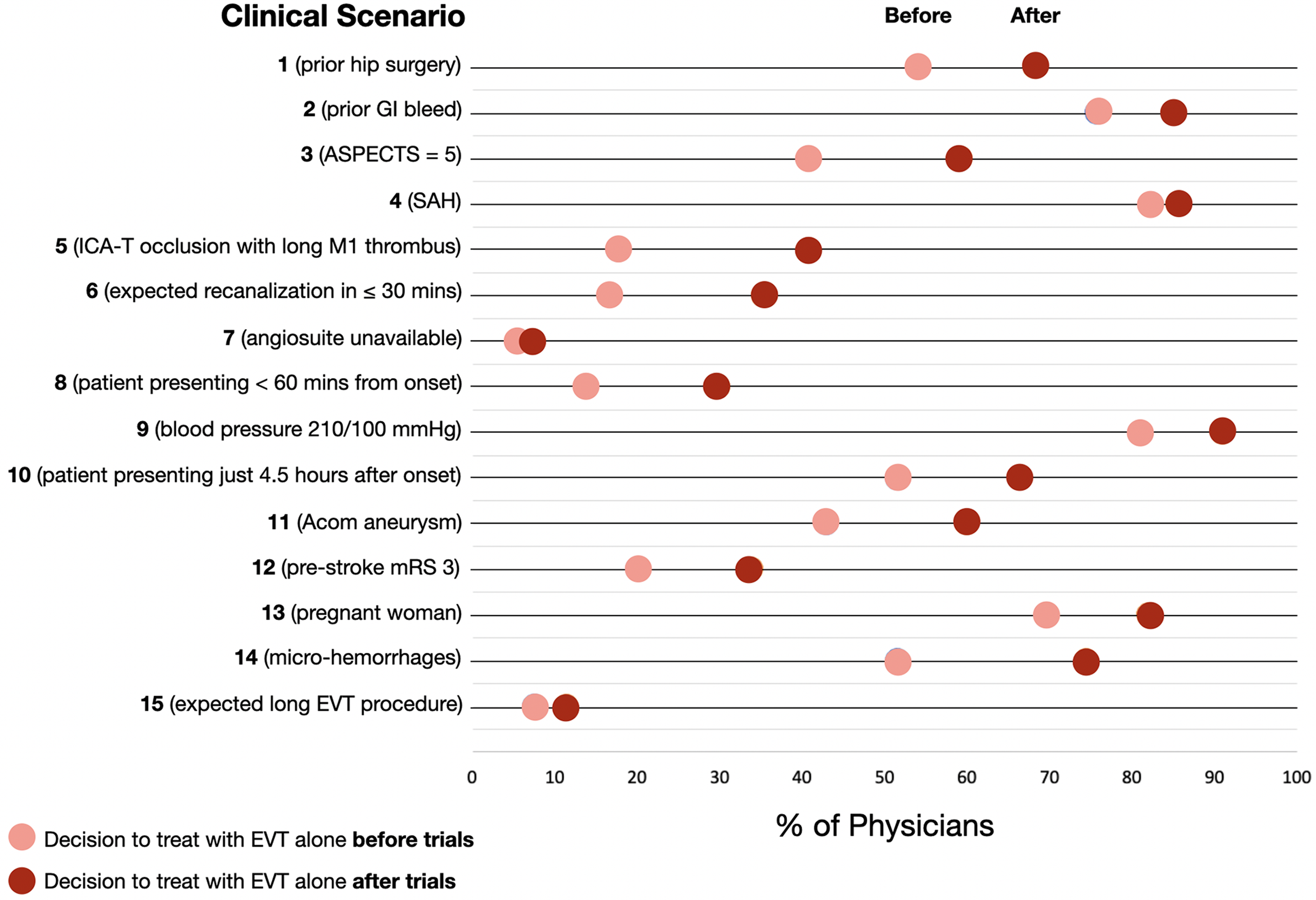
The rates (in percent) of responses to treat with EVT alone both before and after publication/presentation of the direct to EVT trials, per scenario. A form a ceiling effect can be observed for scenarios 2, 4, and 9, affecting their influence on the primary outcome of a change from an IVT + EVT to an EVT alone strategy. GI: Gastrointestinal; ASPECTS: Alberta Stroke Program Early CT Score; SAH: Subarachnoid hemorrhage; ICA-T: Internal carotid artery T; M1: M1 segment of the middle cerebral artery; Acom: Anterior communicating artery; mRS: Modified Rankin Scale; EVT: Endovascular treatment.
Decision to adopt an EVT only strategy, to continue with IVT + EVT, or to continue with EVT alone, by respondent characteristics.
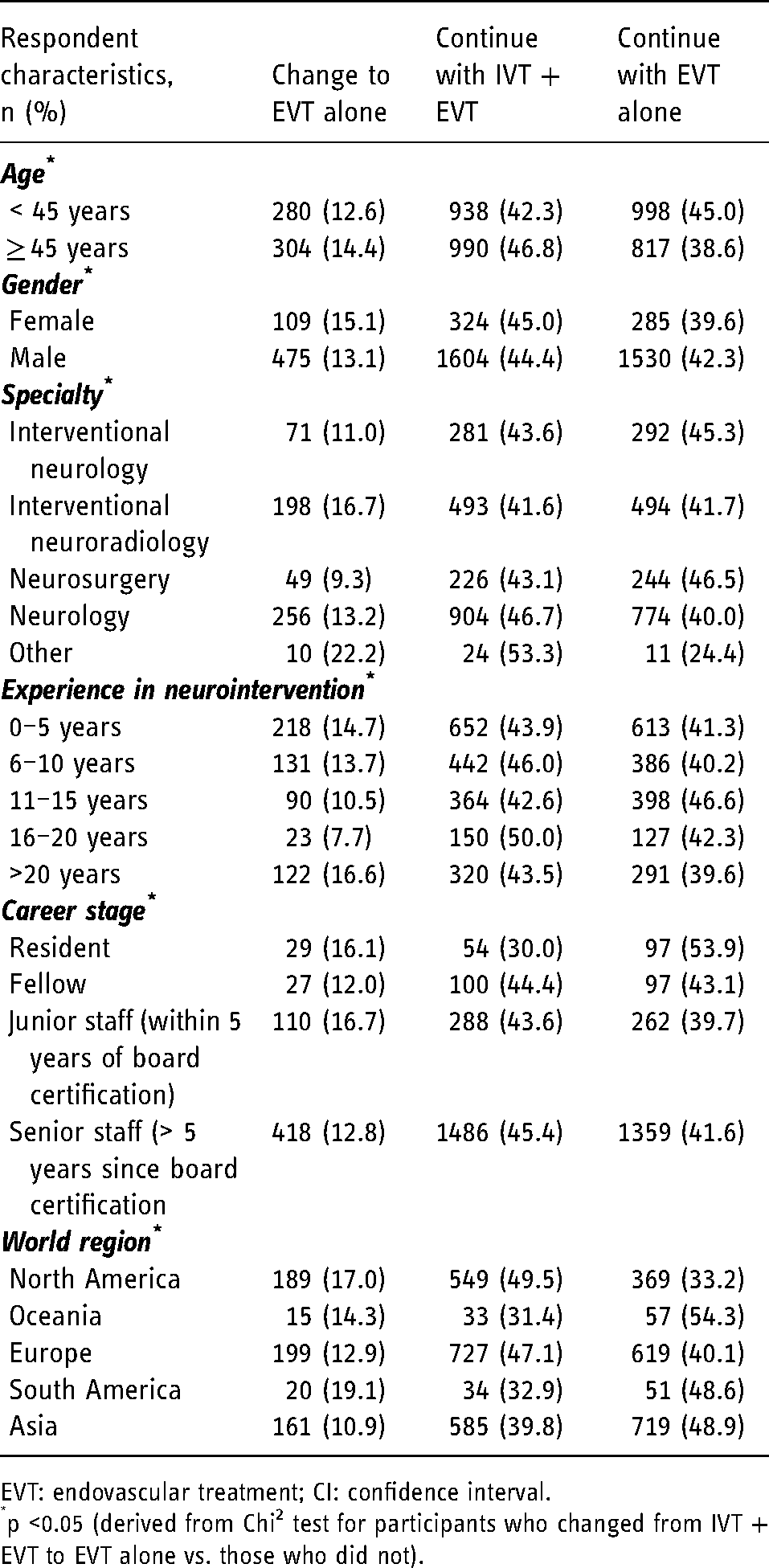
EVT: endovascular treatment; CI: confidence interval.
p <0.05 (derived from Chi 2 test for participants who changed from IVT + EVT to EVT alone vs. those who did not).
Scenario factors influencing a change in treatment strategy to EVT alone
In accordance with the above, the characteristic features of the scenarios that showed the highest rates of change to an EVT only strategy also had the greatest effect sizes in univariable logistic regression (carotid T occlusion with a long thrombus of the M1 segment: RR 1.88, 95%CI: 1.56–2.26; patient with microhemorrhages: RR 1.78, 95%CI: 1.43–2.23; expected short recanalization time: RR 1.46 95%CI: 1.19–1.78) (Table 2). In contrast, the scenario features that were associated with a reduced likelihood to change to an EVT only strategy were unavailability of the neuroangiography suite (RR 0.12, 95%CI: 0.05–0.29), subarachnoid hemorrhage (RR 0.22, 95%CI: 0.12–0.41), and expected long and/or difficult EVT procedure (RR 0.29, 95%CI: 0.17–0.50). Increased bleeding risk (scenarios 1, 2, 3, 4, and 14 [see Supplementary Material, Table 2]), had no significant effect on the change to an EVT only strategy (RR 1.02, 95%CI: 0.88–1.18). Longer expected EVT procedure time (scenarios 7 and 15 [see Supplementary Material, Table 2]) and scenarios depicting absolute contraindications to IVT (scenarios 3, 4, and 9 [see Supplementary Material, Table 1]), however, were associated with a significantly reduced likelihood of change to an EVT only strategy (RR 0.19, 95%CI: 0.12–0.31 and RR 0.75, 95%CI: 0.63–0.89, respectively).
Physician factors influencing a change in treatment strategy to EVT alone
Older (≥ 45 years) physicians were more likely to adopt an EVT only strategy compared to their younger counterparts (14.4% vs. 12.6%, p = 0.016), as were male respondents (15.1% vs. 13.1% for females, p = 0.042) (Table 2). Those with a listed specialty of “other” had the highest rate of adoption for an EVT only approach (22.2%), followed by interventional neuroradiologists (16.7%), neurologists (13.2%), interventional neurologists (11.0%), and neurosurgeons (9.3%) (Table 2). Participants with varying years of experience in neurointervention, from different world regions, and at different career stages also significantly differed in their propensity to change to an EVT alone strategy (Table 2). Specifically, those with >20 years of neurointervention experience (16.6%), who were junior staff within five years of their board examination (16.7%), and those from South America (19.1%) were the most likely to switch to EVT alone in their respective categories (Table 2).
Discussion
Our survey shows that, following publication/presentation of the recent direct-to-EVT trials, roughly 1 out of 7 stroke physicians appear to be rethinking their IVT treatment strategies of EVT-eligible patients with AIS due to LVO. While an overall change in treatment strategy favoring EVT alone of 13.5% as observed in this study may be considered low by some, it is interesting that the conflicting results of the recent direct-to-EVT randomized trials may have already altered the approach of a substantial number of physicians to IVT in a mothership setting.
Although MR CLEAN NO-IV (presented at ISC 2021) showed neither superiority nor non-inferiority of EVT only over a combined approach, functional outcomes and rates of any or symptomatic intracranial hemorrhage did not substantially differ between the arms. Similarly, the results of SKIP did not significantly show non-inferiority, which was likely due to the unexpectedly high rates of early recanalization and good outcomes of the IVT + EVT group. 16 Conversely, both DIRECT-MT 14 and DEVT 15 were able to establish noninferiority of the direct to EVT approach, with comparable 90-day functional outcomes in both arms, although some argue that the non-inferiority margins (20% in DIRECT-MT and 10% in DEVT) were too generous, and the trial results therefore cannot truly exclude superiority of a combined approach. 17
These controversial results, in combination with the fact that IVT has been a mainstay of stroke therapy since the early 1990s 18 and was, until 2015, the only approved form of treatment, led us to intentionally include difficult cases of IVT decision-making in this survey. We include scenarios with varying levels of evidence for EVT and both relative and absolute contraindications for IVT, as listed by the current guidelines 6 (Table 1).
An analysis of the specific scenarios revealed that factors such as increased thrombus length, the presence of cerebral micro-hemorrhages, and faster recanalization times were most likely to lead to a change in treatment strategy in favor of EVT alone. For the latter, it is possible that physicians would be less willing to lose time determining IVT eligibility/administering IVT in situations in which there was a high likelihood of quick recanalization with EVT. Indeed, this finding held true following grouping of the “fast recanalization” scenarios. Interestingly, some of the scenarios involving a presumed increased risk of bleeding (e.g. SAH or prior history of gastrointestinal hemorrhage requiring blood transfusion) were significantly associated with a decreased likelihood of switching to an EVT only approach (likely explaining why the grouping of all scenarios with potentially increased bleeding risk was not significantly associated with a change in strategy). While recent (within the last 21 days) history of a gastrointestinal or genitourinary hemorrhage is listed as a relative contraindication, the guidelines state that the literature reports a low bleeding risk for IVT in such patient groups. 6 Furthermore, our scenario involved a bleed that took place four weeks (28 days) prior to stroke onset. The decision to continue with IVT + EVT in the scenario involving a patient with a minimal SAH following a fall during stroke onset was likely swayed by other scenario factors such as thrombus location within the M2 segment and a relatively quick time from onset (100 min), despite SAH being listed as an absolute contraindication 6 . Finally, and importantly, our analysis was focused on a change from an IVT + EVT strategy to one of EVT alone. In the scenarios involving SAH and a GI bleed, the majority of physicians elected to perform EVT alone, both before and after publication/presentation of the trials (83.4% and 75.8%, respectively, Table 3, Figure 1), resulting in an overall lower proportion of physicians adopting an EVT alone strategy. This form of “ceiling effect” for our chosen primary outcome measure was also reflected in the scenarios that involved absolute contraindications to IVT, particularly those depicting a patient presenting with an SAH and one with therapy-resistant high blood pressure.
Decision to continue with or change original treatment strategy and risk ratios for changing treatment approach to adopt an EVT only strategy, by scenario.
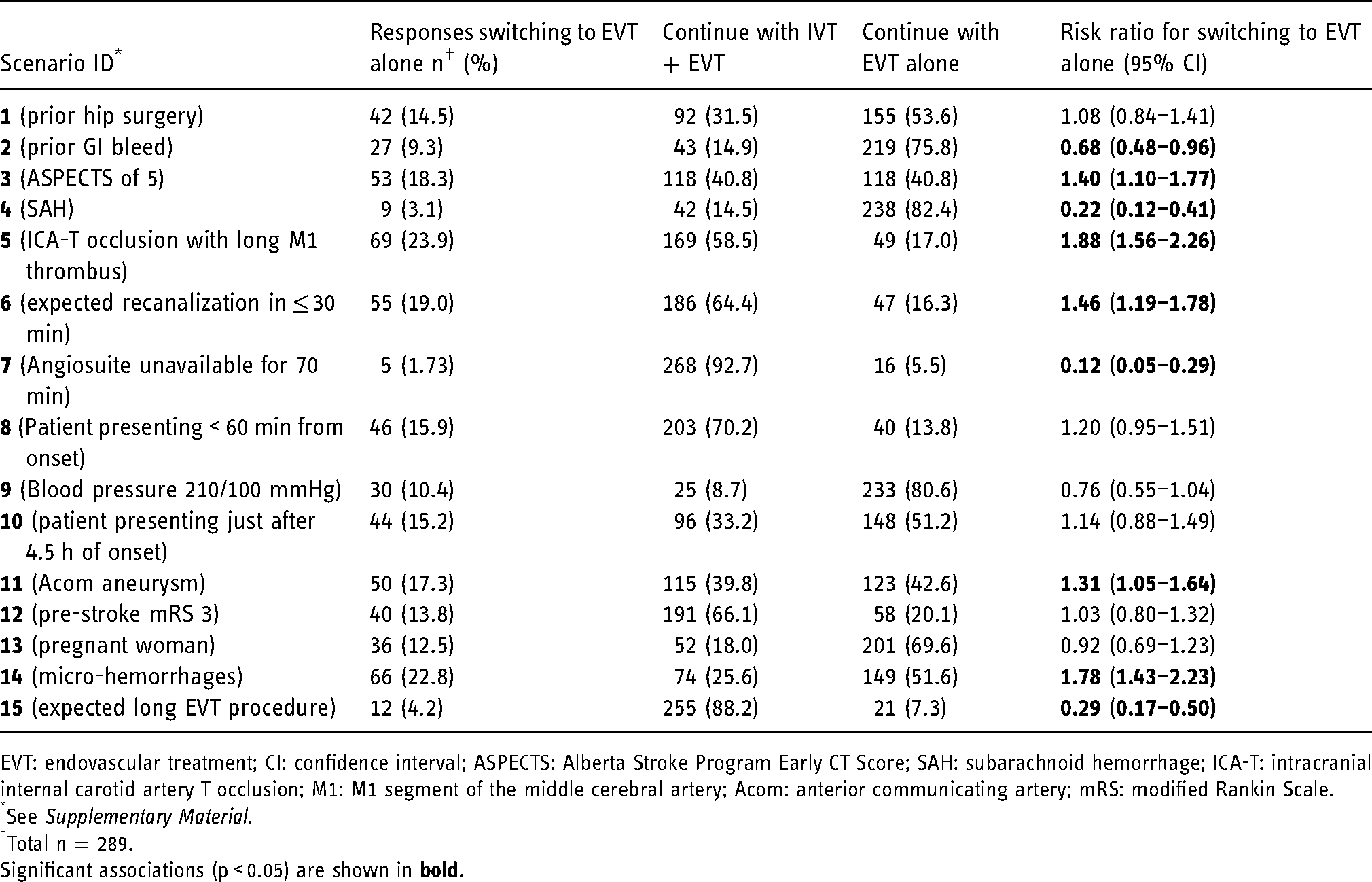
EVT: endovascular treatment; CI: confidence interval; ASPECTS: Alberta Stroke Program Early CT Score; SAH: subarachnoid hemorrhage; ICA-T: intracranial internal carotid artery T occlusion; M1: M1 segment of the middle cerebral artery; Acom: anterior communicating artery; mRS: modified Rankin Scale.
See Supplementary Material.
Total n = 289.
Significant associations (p < 0.05) are shown in
With respect to participant characteristics, significant differences were observed for each category, with participant experience in neurointervention (in years), world region of practice, and specialty showing the greatest numerical differences in rates of change to an EVT alone strategy among their respective subcategories. This could be a result of regional resources, healthcare system cost structures, and differences in local guidelines, 19 as well as individual physician experience with such borderline cases. However, as we present only a descriptive statistical analysis, limited conclusions can be drawn from the data regarding participant characteristics.
Our study has limitations. First, participation was based on personal/institutional networks and co-operations, so sampling, ascertainment, and expectancy biases may have occurred. Although we tried to limit the latter two by making the survey anonymous, our results may not be representative of the entire stroke community. Second, while survey data may reflect certain attitudes, they are unable to truly depict decision-making in routine practice. To this end, we constructed the case scenarios to be simple and as realistic as possible. However, the simple design and brevity of the survey (chosen to increase participation) may have introduced confounding factors. For this reason, we were unable to perform an analysis of the influence of patient factors, such as age, sex, occlusion location, etc. on the likelihood of adopting an EVT alone strategy. Finally, these results represent decision making based on the knowledge base at the time (i.e., after the results of the first four direct to EVT trials became known) and before presentation of the results of the two ongoing trials and pooled data. Physicians’ approach with regard to IVT prior to EVT may change after these results are made available.
Despite these limitations, this study provides important insight on the impact of the recent direct to EVT trials and their ability to influence real-world practice. While the available data from the currently published and presented direct-to-EVT trials are insufficient to change guidelines now, DIRECT SAFE is still underway, and the results are expected soon. From the DEBATE study, it appears that a subset of stroke physicians is already reconsidering treatment strategies with respect to IVT within specific, difficult decision-making scenarios, suggesting a move towards a more individualized risk/benefit-based approach.
Conclusion
Despite the absence of overwhelmingly convincing evidence, one in seven stroke physicians chose to change their LVO approach for AIS from IVT + EVT to EVT alone, based on the recently published/presented direct-to-EVT trial results. This suggests that the direct-to-EVT approach is already considered by some to be a viable and appropriate option in specific, individual situations.
Supplemental Material
sj-pdf-1-ine-10.1177_15910199211057984 - Supplemental material for Influence of recent direct-to-EVT trials on practical decision-making for the treatment of acute ischemic stroke patients
Supplemental material, sj-pdf-1-ine-10.1177_15910199211057984 for Influence of recent direct-to-EVT trials on practical decision-making for the treatment of acute ischemic stroke patients by Rosalie McDonough, Johanna Ospel, Nima Kashani, Manon Kappelhof, Jianmin Liu, Pengfei Yang, Charles Majoie, Yvo Roos and Mayank Goyal in Interventional Neuroradiology
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Mayank Goyal: consultant for Medtronic, Stryker, Microvention, Mentice; University held grants: Medtronic, Cerenovus; Licensing agreements: GE healthcare, Microvention; stockholder: Circle Neurovascular Inc.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
