Abstract
Objectives
This study aimed to evaluate the effect of repetitive peripheral magnetic stimulation on edema of the hemiparetic hand in patients with stroke.
Methods
This single-center, open-label, crossover, randomized controlled trial was registered with the Japan Registry of Clinical Trials, included 18 post-stroke patients in the convalescent rehabilitation ward. Patients were randomly assigned to two groups and received either conventional rehabilitation (control) or conventional rehabilitation plus repetitive peripheral magnetic stimulation (intervention) for 2 weeks, followed by 2 weeks of the other treatment. The repetitive peripheral magnetic stimulation intervention consisted of 6,000 pulses per day at a frequency of 30 Hz, 5 days per week. The primary outcome was changes in hand edema. The secondary outcomes were circumference of the hand, passive range of motion of flexion and extension of the metacarpophalangeal joint, hand pain and numbness, and grip strength. The outcomes were analyzed using mixed-effects models for repeated measures.
Results
Sixteen patients who completed the whole phase were included in the analysis. The changes in hand edema showed significant differences between the groups (p < .01). Metacarpophalangeal joint flexion also had significant differences between the groups (p < .01). The left-right difference in hand volume increased from 48.8 ± 27.6 mL to 59.1 ± 26.3 mL after the control but significantly decreased from 56.3 ± 31.6 mL to 39.7 ± 30.2 mL after the intervention (p < .01).
Conclusions
Repetitive peripheral magnetic stimulation is effective in reducing hemiparetic hand edema and increasing metacarpophalangeal joint flexion after stroke.
Introduction
Post-stroke patients with hemiplegia tend to have edema of the paretic hand. The incidence of edema in the hemiparetic hand varies from 18.5%–50% depending on the methods of evaluation (Gebruers et al., 2011; Leibovitz et al., 2007; Post et al., 2003). Edema derives from impairment of venous and lymphatic return caused by muscle pump dysfunction resulting from muscle paralysis (Iwata et al., 2002). The edema in the hand will persist and lead to pain and joint contractures (Boomkamp-Koppen et al., 2005; Geurts et al., 2000). In addition, heaviness of the hand and loss of movement due to edema are major impediments to rehabilitation (Geurts et al., 2000; Post et al., 2003). Therefore, early intervention against edema is preferable.
Treatment for hand edema includes manual edema mobilization (Knygsand-Roenhoej & Maribo, 2011), elevation (Faghri, 1997), continuous passive motion (Dirette & Hinojosa, 1994), a combination of elevation and continuous passive motion (Giudice, 1990), intermittent pneumatic compression (Roper et al., 1999), taping (Bell & Muller, 2013), orthosis (Meijer et al., 2009; Ring & Rosenthal, 2005), whirlpool bath (Devrimsel et al., 2015), and neuromuscular electrical stimulation (NMES) (Devrimsel et al., 2015; Faghri, 1997; Ring & Rosenthal, 2005). A systematic review for subacute hand edema revealed that manual edema mobilization in conjunction with conventional therapies was effective for hand edema after trauma or surgery, but it was not advocated for edema after stroke due to the lack of randomized controlled or controlled trials (Miller et al., 2017). Similarly, no firm conclusion could be drawn for clinical effectiveness studies in managing post-stroke hand edema because of short intervention periods and the fact that hand edema is not the primary outcome measure (Giang et al., 2016).
NMES is used for the upper and lower limbs, but sometimes it gives pain to the patients. Moreover, the presence of edema increases the requirement of current needed for NMES (Harper et al., 2001). Therefore, pain and discomfort during stimulation are inevitable for high-intensity stimulation by NMES (Bergquist et al., 2011; Harper et al., 2001; Vanderthommen et al., 2000).
Recently, repetitive peripheral magnetic stimulation (rPMS) has been applied as a new technique for neuromodulation (Bustamante et al., 2010; Fujimura et al., 2020, 2024; Krewer et al., 2014). Magnetic stimulation induces muscle contraction by the same mechanism as electrical stimulation; however, because it does not stimulate nociceptors in the skin, instead it can stimulate α-motor nerves and Ia-afferents without excitability of pain nerves in the superficial skin, resulting in less pain than NMES (Han et al., 2006; Struppler et al., 2007).
One study reported that rPMS from the dorsum of the hand produces the same extent of MCP joint flexion and abduction as active movement (Fujimura et al., 2022). Therefore, we hypothesized that repetitive contraction and relaxation of the intrinsic muscles of the hand by rPMS would reconstruct muscle pump action and promote metabolism, thereby improving edema. Paretic edema of the hand is an important issue for referral to occupational therapy aiming at improving the motor function of the hand and integrating these improvements in the activities of daily living for patients with stroke. The rPMS treatment emerges as a prospective innovation in occupational therapy, potentially offering a novel approach to addressing this issue. Therefore, the purpose of this study was to evaluate the effect of rPMS on the hemiparetic hand edema after a stroke.
Materials and methods
This prospective exploratory study was conducted according to the guidelines of the Declaration of Helsinki, approved by our Certified Clinical Research Review Board in our institution, and was registered with the Japan Registry of Clinical Trials (no. jRCTs042180062). The patient or a proxy provided written informed consent.
Patients
Inclusion criteria were post-stroke inpatients in the convalescent rehabilitation ward between January 2020 and August 2021, over 20 years of age, and edema of the paretic hand with more than 15 mL in hand volume compared to the opposite side. Exclusion criteria were: unstable general condition, edema of the hand caused by conditions other than stroke, history of epilepsy, use of cardiac pacemakers, magnetic materials near the intended stimulation site, patients who were expected to be discharged within 2 weeks, and pregnant patients.
rPMS therapy
The rPMS therapy was implemented after conventional rehabilitation. A peripheral magnetic stimulator (PathleaderTM; IFG, Sendai, Japan) that generates a biphasic 350 μs magnetic gradient of up to 20 kT/s was used. The patients sat on a chair, and the paretic hand was set on a pillow placed on the desk. Since the aim of the present study was to reconstruct muscle pump action with rPMS, the center of the coil was placed between the second and third metacarpal bones on the dorsum of the hand to stimulate the intrinsic muscles that produced flexion of the metacarpophalangeal (MCP) joints (Fujimura et al., 2022) (Figure 1). We stimulated the dorsum of the hand as it was less painful than palmar stimulation. Moreover, stimulation from the dorsum of the hand by using the coil for rPMS does not interfere with the flexion of the MCP joint. Based on a previous study reporting rPMS to intrinsic muscles (Fujimura et al., 2022), 6,000 pulses per day, 5 days a week, with an on-time of 2 s and an off-time of 2 s at a frequency of 30 Hz. Therefore, it took 400 s to complete the rPMS therapy. Typically, the stimulus intensity was set at 100%, the maximum output value of the peripheral magnetic stimulator used, but when the patients felt pain or discomfort, it was adjusted to between 80%–100% (approximately 0.75–0.9 T). rPMS therapy to the intrinsic muscles. The paralyzed hand was placed on a pillow. The center of the coil for peripheral magnetic stimulation was set between the second and third metacarpal bones on the dorsum of the hand to stimulate the intrinsic muscles.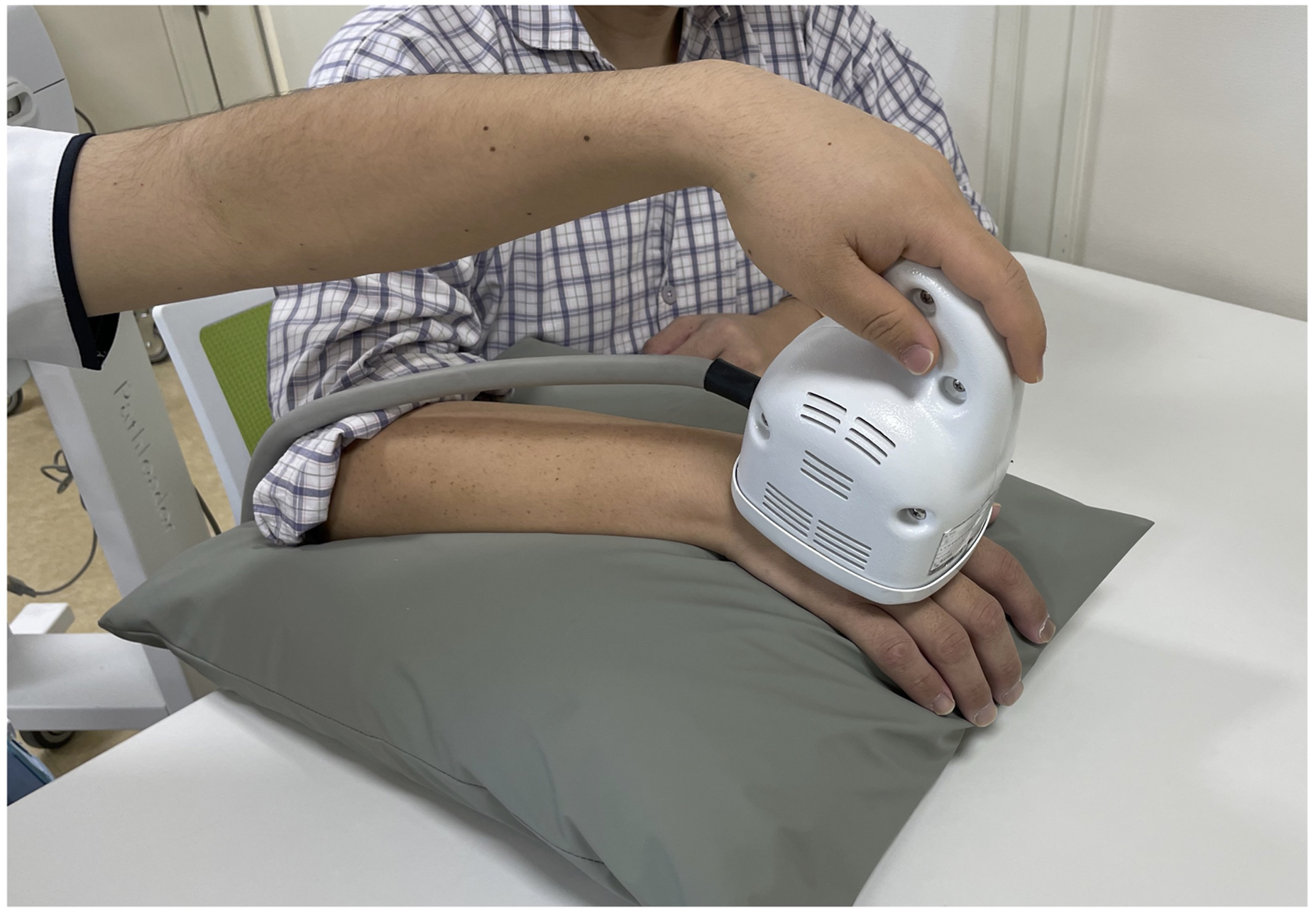
Study protocol
This study was a single-center, open-label, crossover, randomized controlled trial. Patients were randomized to either Group A or Group B according to a computer-generated sequence. Group A underwent a conventional rehabilitation (control) for 2 weeks in the initial phase followed by a conventional rehabilitation plus rPMS (intervention) for 2 weeks in the second phase. In contrast, the patients in group B had the intervention for 2 weeks in the initial phase followed by control for 2 weeks in the second phase. No washout periods were set between the initial and the second phase. Conventional rehabilitation was performed 7 days/week and included joint mobilization, muscle stretching, muscle strengthening, gait, balance exercises, and task-related training in activities of daily living depending on the function of the patients. Both groups had 180 minutes/day of rehabilitation which consisted of conventional rehabilitation or conventional rehabilitation plus rPMS. To ensure equivalence of rehabilitation programs in the intervention and control phases, therapists did not provide other direct interventions to reduce hand edema in both phases, and interventions were kept as consistent as possible.
Study endpoints
The primary outcome, changes in hand edema, was calculated from the difference in hand volume between the left and right sides by using a volumetric meter. This method showed high reliability (Boland & Adams, 1996; Post et al., 2003). A hand volumeter set (KN-940-1, Natsume Seisakusho Co, Ltd., Tokyo, Japan) was used and consisted of a volumeter (L90 × W130 × H280-mm), a plastic beaker, and 500-mL graduated cylinders with 5-mL increments to measure the amount of water displaced. At first, the volumeter was filled with room temperature water until water began to overflow into a beaker positioned beneath the spout. Once water stopped dripping from the overflow spout, the beaker was replaced with a second empty beaker. The thumb was positioned to face the overflow spout with the fingers adducted, and the forearm pronated and positioned perpendicular to the bottom of the volumeter. Next, the patient lowered the hand slowly into the center of the volumeter until the web of the middle and ring fingers rested on the stop dowel. As the patient lowered the hand into the volumeter, displaced water spilled out through the overflow spout into the beaker. The patient’s hand was left in the volumetric meter until no water came out from the spout. The data recorder poured the water that had collected in the beaker into the graduated cylinder and measured the amount of water in the cylinder (Leard et al., 2004). The non-edema side was measured first, and the edema side was measured later. Since the sizes of the hands differed among the patients, the difference in size between the left and right hands was used (Vasiliauskas et al., 1995). Additionally, the secondary outcomes were circumference of the hand, passive range of motion (ROM) of flexion and extension of the MCP joint of the index finger, hand pain and numbness, and grip strength. The goniometer (TTM-KS, SAKAI Medical Co., Ltd., Tokyo, Japan) was used to measure ROM, and the grip strength was measured using the grip strength instrument (EKJ094, EVERNEW INC., Tokyo, Japan). The circumference of the hand was measured using the figure-of-eight method with a tape measure (Dewey et al., 2007; Leard et al., 2004; Maihafer et al., 2003). The MCP joint angles were measured by a goniometer for the fingers. Hand pain and numbness were evaluated from 0 to 10 using a numerical rating scale (NRS) (0 = no pain and 10 = the most severe pain) (Hjermstad et al., 2011). Hand muscle strength was measured using a grip strength meter. In addition, the variables that had no interaction between the control and the intervention were compared as a secondary analysis. Assessment was performed three times in each patient: T0 was just before the initial phase, T1 was just after the initial phase, and T2 was just after the second phase. Since edema may change depending on the time of day, all measurements were conducted from 17:00 to 18:00.
Moreover, the motor function of the affected upper arm at T0 was assessed by Fugl-Meyer Assessment upper extremity (FMA-UE) total score and its subscales (Fugl-Meyer et al., 1975). Sex, age, etiology, days from stroke onset, and paretic side were also obtained from the medical records.
Statistical analyses
A previous study using NMES on paralytic edema of the hand showed an effect size of 1.76 (Faghri, 1997). We assumed α = 0.05, a detection power of 0.95, and a dropout rate of 10%. The estimated sample size was calculated as 18 cases by using G*Power software (ver. 3.1.9.6; Heinrich-Heine-Universität Düsseldorf, Düsseldorf, Germany) (Faul et al., 2007).
Fisher’s exact test was used to compare sex, etiology, and paretic side, while the unpaired t test was performed to compare age, days from stroke onset, and FMA-UE between Groups A and B. The values are expressed as mean ± SD. The changes in hand edema, hand circumference, passive ROM of flexion and extension of the MCP joint, hand pain and numbness, and grip strength were analyzed using mixed-effects models for repeated measures. The fixed effects were time, group, and time-by-group interactions, whereas the random effect was patients. A paired t test was used to compare the changes between control and intervention when there was no interaction between time and group. Statistical analysis was performed using JMP version 14 (SAS Institute Japan Inc, Tokyo). The significance level was set at 5%.
Results
Eighteen patients were randomly assigned to the two groups. The dominant hand of all patients was the right. One patient in Group A and five in Group B had paralysis of the dominant hand. In Group B, one patient refused to continue the intervention in the initial phase, and one patient dropped out due to a sudden discharge from the hospital in the second phase. Therefore, nine patients in group A and seven patients in Group B completed the protocol (Figure 2). We included 16 patients who completed both the T1 and T2 assessments. The baseline characteristics in each group are shown in Table 1. None of the parameters had significant differences between the groups. The total FMA-UE score for all patients was 15 ± 17. Study flow diagram. The flow diagram shows the randomization and the flow of patients during the study period. Seventeen patients who completed at least the initial phase were analyzed. Patient characteristics. FMA-UE, Fugl-Meyer Assessment Upper Extremity; mean ± SD (median).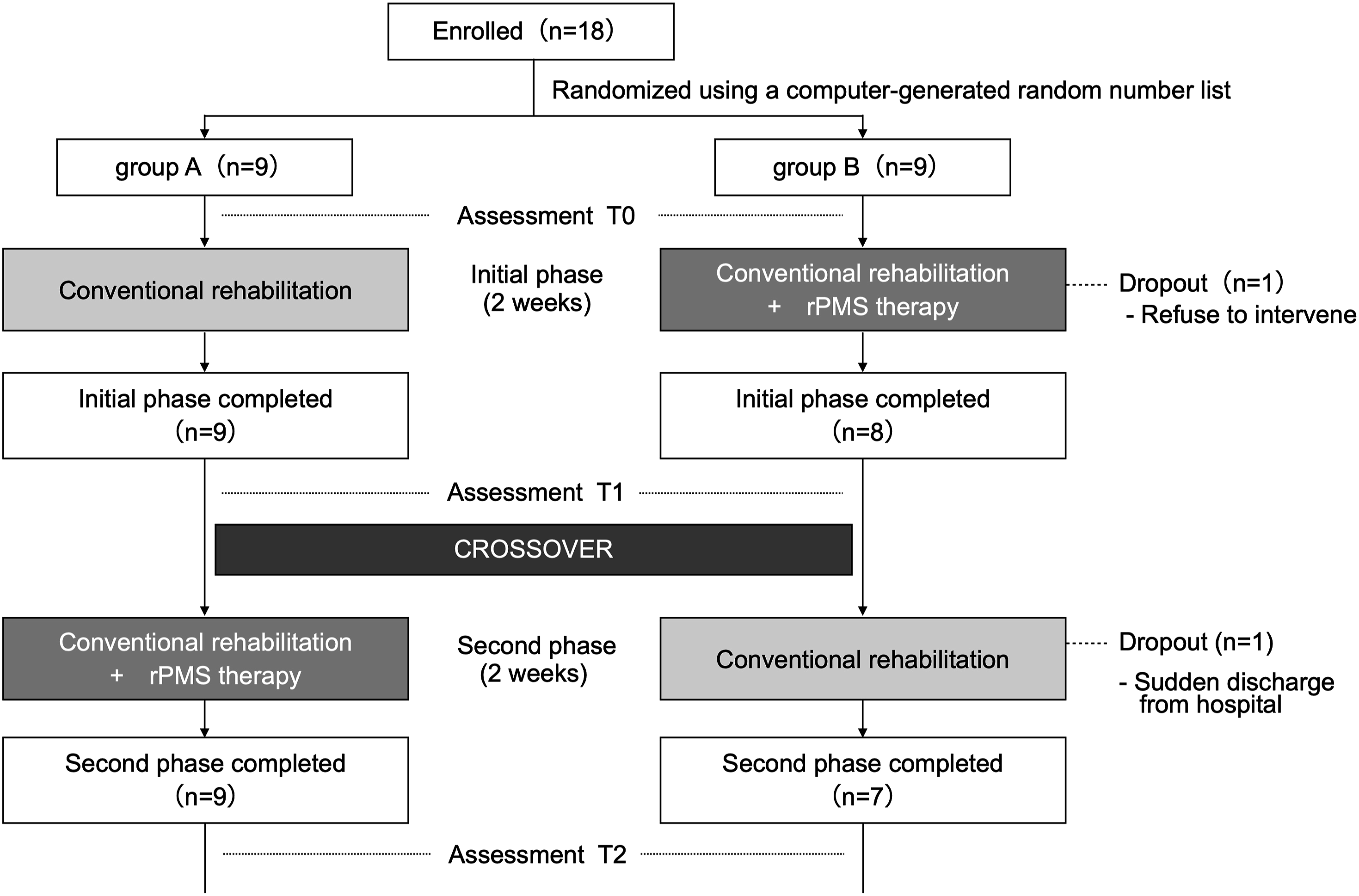

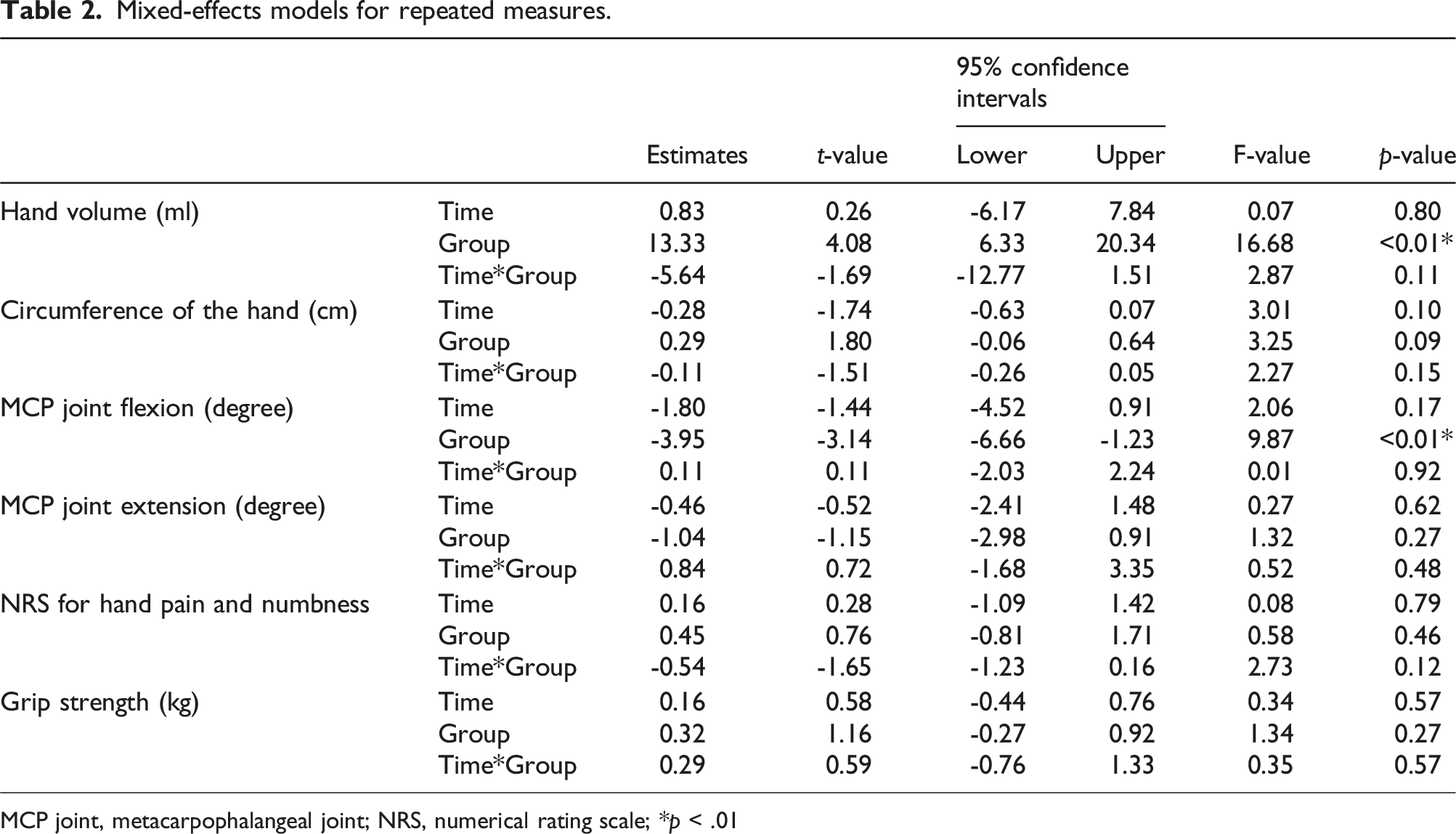
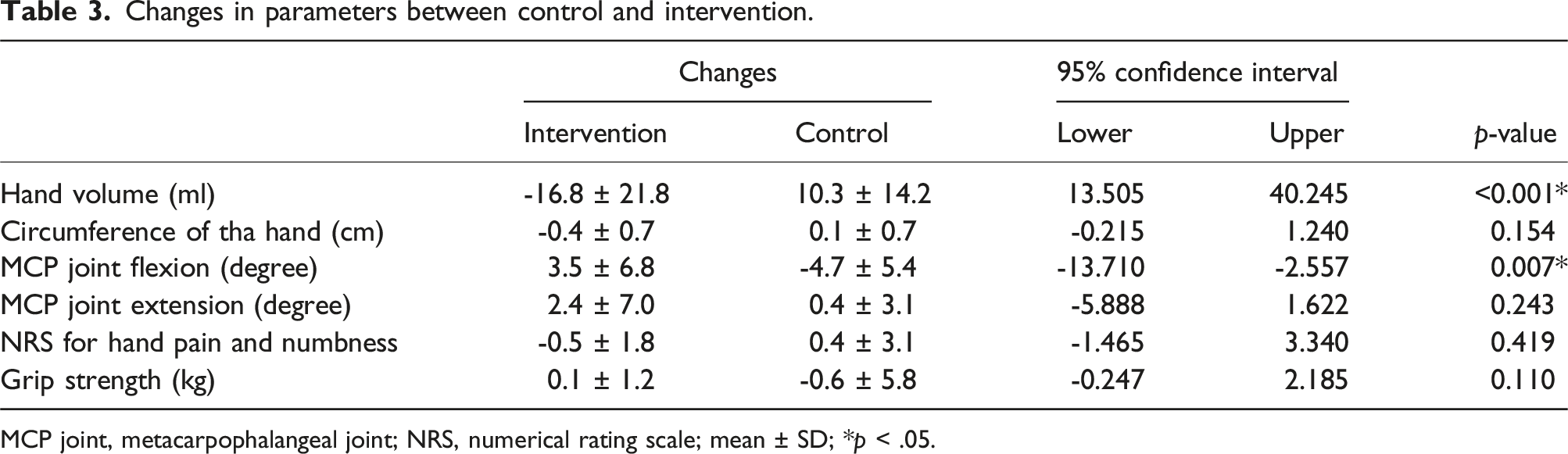
Time-dependent changes in each variable are shown in Figure 3. The difference in hand volume (mL) between the left and right hand in T0, T1, and T2 was 50.6 ± 28.2, 56.7 ± 21.7, and 34.4 ± 31.9, respectively, for Group A and 55.7 ± 43.3, 46.4 ± 28.8, and 62.1 ± 33.0, respectively, for Group B. The difference in hand volume showed significant differences between the groups (t = 4.08; p < .01). Among the secondary outcomes, MCP joint flexion had significant differences between the groups (t = −3.14; p < .01) (Table 2). There were no interactions between time and groups in all variables. Comparing the changes between intervention and control, the left-right difference in hand volume significantly decreased (d = 1.43; p < .01) and the MCP joint flexion angle significantly increased (d = 1.31; p < .01) in the intervention time (Table 3). Time-dependent changes in each variable. Group A underwent a conventional rehabilitation (control) for 2 weeks between T0 and T1 that included conventional rehabilitation plus rPMS (intervention) for 2 weeks between T1 and T2. Group B had intervention for 2 weeks between T0 and T1 followed by control for 2 weeks between T1 and T2. Mixed-effects models for repeated measures. MCP joint, metacarpophalangeal joint; NRS, numerical rating scale; *p < .01 Changes in parameters between control and intervention. MCP joint, metacarpophalangeal joint; NRS, numerical rating scale; mean ± SD; *p < .05.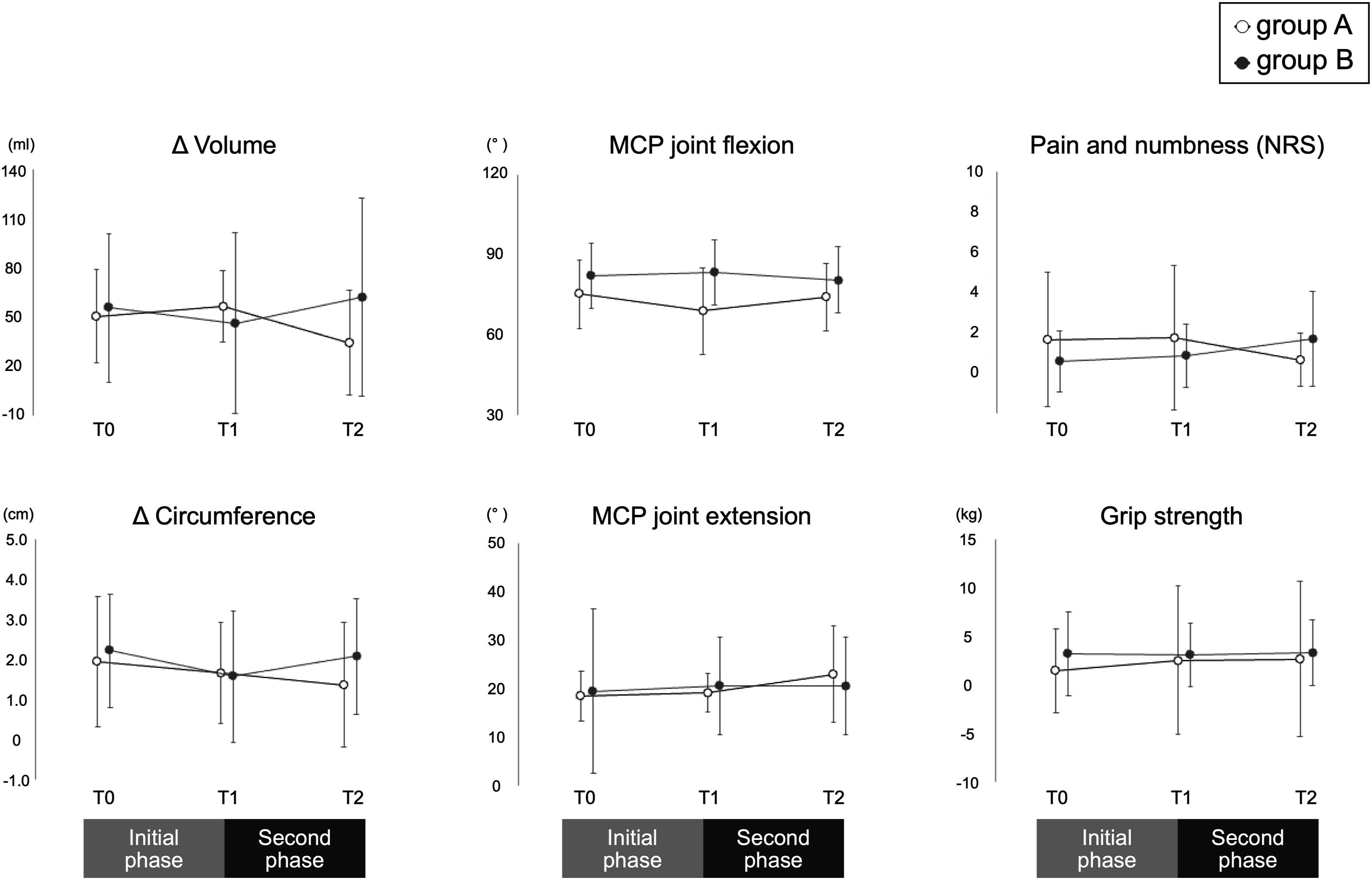
Discussion
This study was a randomized crossover trial with or without 2 weeks of rPMS in addition to conventional rehabilitation in stroke patients who had edema in the paretic hand in a convalescent rehabilitation ward. The results showed that hand edema was reduced and MCP joint flexion increased with rPMS, suggesting the effectiveness of rPMS for edema of the hemiparetic hand after stroke.
Various therapeutic procedures have been investigated for hand edema, but it is not possible to draw a firm conclusion on their clinical significance (Giang et al., 2016; Miller et al., 2017). The major factor for paralytic edema is the disruption of the pumping action, so it is important to reconstruct the muscle pumping action to treat the condition (Iwata et al., 2002). NMES is expected to improve muscle pump action, and its effectiveness has been reported (Burgess et al., 2019). However, few studies have examined NMES for paralytic edema of the hand or quantified the changes in hand volume as in this study. Conventionally, flexors and extensors of the fingers in the forearm have been stimulated by NMES (Devrimsel et al., 2015; Faghri, 1997; Meijer et al., 2009; Ring & Rosenthal, 2005). A 30-min treatment of NMES in stroke patients within 6 months of onset resulted in an average of 13.4 mL reduction in hand volume (Faghri, 1997). Another report states that hand volume was reduced by 16.6 mL on average in patients with complex regional pain syndrome (CRPS) type 1 who had NMES for 20 minutes per day, 5 times per week for 3 weeks (Devrimsel et al., 2015). Given that the muscle pump action promotes return of adjacent veins and lymphatic vessels through the contraction of skeletal muscles, it may be more beneficial to stimulate the muscles directly where the edema exists. Moreover, NMES for the intrinsic muscles of the hand is not practical due to high impedance caused by edema and pain during stimulation (Bergquist et al., 2011; Carroll et al., 1992; Harper et al., 2001; Vanderthommen et al., 2000).
To the best of our knowledge, we are the first to report on the use of rPMS for treating hand edema. Repetitive contraction of the muscles due to the voluntary movement of the joints pushes blood through the muscle pump action and promotes venous return (Ludbrook, 1962; Lyons et al., 2002). The repetitive contraction and relaxation of the intrinsic muscles of the hand by rPMS seem to promote the return of blood and lymphatic fluid around the stasis in the tissues. In addition, muscle contraction may increases the metabolic rate, increasing the demand for oxygen and nutrients and the amount of waste products, including metabolic products. These metabolites dilate capillaries and small arteries, thus markedly enhancing blood circulation. Since rPMS reaches deeper muscles than NMES while causing less pain (Harper et al., 2001; Struppler et al., 2007), we could stimulate the intrinsic muscles of the hand directly and obtained similar effects in patients with more severe hand edema within a shorter period of time compared to the results of a previous study using NMES (Devrimsel et al., 2015).
We also found significant improvement in the MCP joint flexion angle by rPMS. Although joint mobilization in conventional rehabilitation improves mobility, edema reduction is necessary to alleviate decreased mobility caused by edema. Additionally, the intrinsic muscles of the hand include dorsal interosseous, palmar interosseous, and lumbrical muscles, and all these muscles flex the MCP joint (Fujimura et al., 2022). The repetitive ROM exercise of MCP by rPMS can improve the MCP joint flexion angle along with a reduction of edema. As hand edema causes joint contractures (Boomkamp-Koppen et al., 2005; Geurts et al., 2000), our rPMS treatment may be useful to prevent or treat extension contracture in the MCP joint.
The hand circumference did not change in this study. Although the figure-of-eight method of using a tape measure showed similar reliability and validity to those of the water volumetry method (Leard et al., 2004; Maihafer et al., 2003; Dewey et al., 2007), the circumferences are affected by the placement and tension of the tape (Bear-Lehman & Abreu, 1989). Moreover, the tape was placed between the radial and ulnar styloid processes and MCP joints in the figure-of-eight method (Leard et al., 2004). Therefore, the figure-of-eight method does not reflect the changes in edema that are more distal to the MCP joint.
The magnetic field from magnetic stimulation decays in proportion to the square of the distance. It becomes difficult to stimulate the intrinsic muscles by rPMS from dorsum of the hand for patients with severe edema. Future studies are expected for such patients.
Edema worsened in the control during the conventional rehabilitation. The natural history of edema of the paralyzed hand after stroke is unclear, but one report indicates that hemiplegic hand edema is most pronounced 2–4 months after stroke (Iwata et al., 2002). Therefore, it is possible that rPMS in this study prevented the progress of hand edema in addition to decreasing it.
This study has several limitations. The optimum conditions for rPMS, such as frequency, intensity, and intervention period, were unclear. In addition, muscle pump action depends on the given contractile force and muscle volume, but we failed to measure them induced by rPMS. Muscle activity in the target and associated forearm muscles of the rPMS could not be measured using electromyography. Furthemore, rPMS seems to give less pain during stimulation than NMES, but we did not assess patient-reported pain during rPMS in this study. The patients in this study had severe hemiparalysis with a mean FMA-UE score of 18 (Woodbury et al., 2013). We need to know the effect of rPMS for hand edema in patients with mild to moderate paralysis. Since the amount of activity in daily life was not assessed, its relationship to changes in edema could not be examined. We used a crossover design to facilitate recruitment and reduce the effects of individual variability. A washout period is usually needed in a crossover design; however, we did not set this period because this study was the first to use rPMS treatment for hand edema and the appropriate washout period was unknown. A long-term intervention of more than 2 weeks has not been investigated. Therefore, further studies are required to ensure and validate the effectiveness of rPMS in treating stroke-induced hand edema.
Conclusions
The rPMS treatment was effective in reducing hand edema caused by post-stroke hemiparalysis and increasing MCP joint flexion. We could activate the intrinsic muscles of the hand directly by rPMS because it can stimulate deep muscles without causing pain. This new treatment may replace NMES for hand edema, and we can anticipate the use of rPMS to treat edema in other areas.
Footnotes
Acknowledgements
We thank Haruna Miwa (OTR) and Chiharu Endou (OTR) from the Department of Rehabilitation at Fujita Health University Hospital for their assistance with the measurements. We also thank Professor Ayumi Shintani at Osaka City University and Assistant Professor Takuma Ishihara at the Center for Innovative Clinical Research, Gifu University Hospital, for their advice as biostatisticians regarding the statistical analysis in this study.
Author Contributions
Kenta Fujimura and Hitoshi Kagaya have given substantial contributions to the conception or the design of the manuscript, Takuya Suzuki, Hiroki Tanikawa, Kazuhiro Tsuchiyama and Hirofumi Maeda. to acquisition, analysis and interpretation of the data. All authors read and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded in part by a grant from the TERUMO LIFE SCIENCE FOUNDATION [21-III1027].
Data Availability Statement
The data collected and analyzed during the current study are available from the corresponding author on reasonable request.
