Abstract
Objective:
Chemotherapy-induced peripheral neuropathy is one of the major toxicities in multiple myeloma patients, often resulting in dose reductions or treatment interruptions. Repetitive transcranial magnetic stimulation is a safe non-invasive neuromodulation therapy with potential benefits for chemotherapy-induced peripheral neuropathy. The objective of this study was to investigate the efficacy of repetitive transcranial magnetic stimulation treatment on chemotherapy-induced peripheral neuropathy in multiple myeloma patients.
Materials and methods:
We screened 30 multiple myeloma patients with chemotherapy-induced peripheral neuropathy who underwent repetitive transcranial magnetic stimulation treatment in this study. Prior to and following repetitive transcranial magnetic stimulation treatment, patients were assessed with nerve conduction velocity, visual analog scale and the European Organization of Research and Treatment of Cancer Quality of Life Questionnaire-CIPN 20-item scale (EORTC QLQ-CIPN20). Categorical and continuous variables were analyzed using Fisher’s exact test and Mann-Whitney U test respectively. A p-value < 0.05 (2-tailed) was considered statistically significant.
Results:
Following repetitive transcranial magnetic stimulation treatment, 24/30 (80.0%) patients reported a reduction in chemotherapy-induced peripheral neuropathy symptoms. Meanwhile, all 15 patients with grade 2 chemotherapy-induced peripheral neuropathy experienced improvements about themselves, compared to 8/10 patient with grade 3 chemotherapy-induced peripheral neuropathy and 1/5 with grade 4 chemotherapy-induced peripheral neuropathy. Visual analog scale scores decreased after repetitive transcranial magnetic stimulation treatment (5.40 ± 1.94 vs 3.10 ± 1.60, p < 0.001). We also observed enhancements in both motor conduction velocity and sensory conduction velocity of patients in bilateral median nerves, posterior tibial nerves, common ulnar nerves and peroneal nerves following repetitive transcranial magnetic stimulation treatment. Analysis of the European Organization of Research and Treatment of Cancer Quality of Life Questionnaire-CIPN 20-item scale data (17.68 ± 8.14 vs 10.50 ± 9.55, p < 0.001) revealed significant reductions in scores. Patients with grade 2–3 (n = 25) exhibited a mean reduction of 8.89 ± 4.24 points, while those with grade 4 (n = 5) showed a difference value of 3.54 ± 3.45, p < 0.001. No adverse events were observed.
Conclusion:
Our findings suggest that repetitive transcranial magnetic stimulation is a safe and effective therapeutic approach for ameliorating peripheral nerve injury and alleviating the chemotherapy-induced peripheral neuropathy symptoms in multiple myeloma patients. Early initiation of repetitive transcranial magnetic stimulation treatment may yield more favorable outcomes for these patients.
Keywords
Introduction
Multiple myeloma (MM) is a malignancy originating from monoclonal plasma cells of B cell origin; patients with MM are considered to have poor outcomes. The introduction of novel agents, including proteasome inhibitors (PIs) and immunomodulatory drugs (IMiD), has dramatically improved the prognosis of MM. 1 However, the increasing use of these new agents has drawn attention to their associated toxicities in recent years. Chemotherapy-induced peripheral neuropathy (CIPN) as one of the major toxicities of the treatments, often leads to dose reduction or treatment interruptions. CIPN typically manifests as sensory and motor neuropathy, with a predominant sensory component, resulting in symptoms like limb pain and numbness; 2 it significantly impacts the patient’s quality of life and can lead to adverse outcomes. 3 For some patients, particularly the elderly, who are unable to tolerate high-intensity treatment, the current clinical treatment mainly focuses on sequential chemotherapy. Severe CIPN can necessitate dose reductions or less frequent chemotherapy treatment, or even discontinuation of treatment, potentially leading to disease relapse or progression. 4
The current treatment such as pharmacological interventions often offer limited effectiveness, therefore, there is a need for more effective therapeutic approaches. In order to find more effective treatment methods, we designed this study. Repetitive transcranial magnetic stimulation (rTMS) is a safe, non-invasive neuromodulation therapy that has found broad application in managing peripheral neuropathic disease, especially neuropathic pain,5,6 and it holds the potential to be a beneficial and effective treatment for CIPN. 7 The rTMS equipment comprises one or two copper wire coils. During treatment, the coil is precisely positioned on the brain surface and generates electromagnetic pulses non-invasively, then these pulses are transmitted through the skull without attenuation to the cerebral cortex, followed by axonal depolarization. This depolarization activates both cortical and subcortical networks, enhances the excitability of the corticospinal system, promotes neuronal plasticity, improves synaptic transmission, and enhances the responsiveness of the nervous system, ultimately achieving therapeutic goals. 8
In this pilot study we monitored the nerve conduction velocity (NCV) and changes of CIPN symptoms in 30 MM patients before and after receiving rTMS treatment to investigate the efficacy of rTMS treatment for CIPN in MM patients.
Methods
Study design and patients
Thirty MM patients with CIPN from April 2019 to August 2022 who were treated with rTMS at the first people’s hospital of Lianyungang were screened in this study. Patients had to meet specific inclusion criteria: 1. Diagnosis of MM and receipt of PIs (bortezomib or ixazomib) and/or IMiD (lenalidomide), 2. Development of CIPN of grade 2 or higher, graded according to the grading system of National Cancer Institute Common Toxicity Criteria Adverse Event, 9 an expected survival period of more than 6 months. Exclusion criteria were as follows: 1. Patients with pacemakers, 2. Epileptic patients, 3. Recent acute cerebral hemorrhage, 4. Patients with metal objects in their heads, and 5. Active infections.
The medium time of CIPN occupied after chemotherapy was 2.5 months. All patients received pharmacological interventions, which included Vitamin B12, Vitamin B1, gabapentin, amitriptyline, pregabalin, and opioids when CIPN occurred. However, these treatments yielded limited benefits. Our study obtained approval from the institutional review committee of First Peoples’ Hospital of Lianyungang (approval number: LW-20230320001) and was conducted in accordance with the Declaration of Helsinki; informed consent was obtained from all subjects and/or their legal guardians. Figure 1 provides a flowchart illustrating the organizational structure of this study.

The flowchart of this study.
RTMS treatment
All patients underwent rTMS treatment. While most patients exhibited symptoms affecting only one side of the upper and/or lower extremities, the peripheral nerves of these patients displayed varying degrees of damage. As a result, we employed a standardized treatment approach that encompassed all limb nerves for every patient. Patients were positioned in supine position, and an “8” shaped magnetic stimulation coil was utilized. The coil was consistently aligned with the M1 region of the brain, alternating between sides. The magnetic stimulation parameters included a frequency of 10 Hz, set at 80% of the resting motor threshold, 15 pulses per sequence, 1.5 s of stimulation, a sequence interval of 3 s, and a total of 1400 magnetic pulses, equivalent to a 7 min treatment session. Bilateral M1 regions were stimulated alternately, and the treatment sessions were administered once a day for 14 min each, five times a week, for a total of 6 weeks per treatment course. Patients were closely monitored for any discomfort or adverse events during and following treatment. rTMS was administered using a transcranial magnetic stimulator (Yiruide, CCY-II, China).
Assessments
Prior to and following rTMS treatment, patients underwent a series of assessments to evaluate treatment outcomes including visual analog scale (VAS), NCV and the European Organization of Research and Treatment of Cancer Quality of Life Questionnaire-CIPN 20-item scale (EORTC QLQ-CIPN20). Assessments were conducted at two time points: (1) pre-treatment (1–3 days before the commencement of rTMS treatment), and (2) post-treatment (1 week after the completion of the last rTMS session). Differences in the severity of CIPN could be objectively and quantitatively accessed by NCV. 10 We monitored the motor conduction velocity (MCV) and sensory conduction velocity (SCV) of patients’ bilateral median nerves, tibial nerves, ulnar nerves and peroneal nerves by using key point full-function electromyograph, the normal value of NCV (both MCV and SCV) in the upper limb is ⩾50 m/s, while in the lower limb, it is ⩾40 m/s. EORTC QLQ-CIPN20 life questionnaire is validated and reliable, which is used to assess the severity of CIPN symptoms experienced by patients.11,12 This validated and reliable questionnaire comprises three subscales: sensory (9 items), motor (8 items), and autonomic symptoms and functioning (3 items). It includes 20 items with a 4-points Likert scale 13 (1 = “not at all” to 4 = “very much”). Scale scores were linearly converted to a 0 to 100 scale, with higher scores indicating a greater symptom burden. 12 Pain experienced by CIPN patients was assessed using VAS. This scale is widely used in clinical practice in China. The methodology involves employing a 10 cm long swimming ruler with 10 scales on one side and “0” and “10” points at both ends. A score of 0 represents the absence of pain, while a score of 10 indicates the most severe, unbearable pain. 14
Statistical analysis
GraphPad Prism and SPSS 26.0 software were used to analyze the data and plot. Fisher’s exact test and Mann-Whitney U test were used to assess categorical and continuous variables. p value < 0.05 (2-tailed) was statistically significant.
Results
A summary of the subjects’ clinical characteristics can be found in Table 1. Patients presented with a range of CIPN symptoms, including pain, dysesthesia, and dyskinesia. The predominant symptom among patients was somatosensory-related. Following rTMS treatment, 24/30 (80.0%) patients reported a reduction in their CIPN symptoms. Among these 24 patients, two ceased pharmacological CIPN intervention, five reduced their medication dosage, while 18 of them continued with the same medication dose. Furthermore, all 15 patients with grade 2 CIPN experienced symptom improvement for themselves, compared to 8/10 patient with grade 3 CIPN and 1/5 with grade 4 CIPN. We assessed VAS for 24 patients with pain. For the 24 patients experiencing pain, their VAS scores decreased significantly following rTMS treatment (5.40 ± 1.94 vs 3.10 ± 1.60, p < 0.001), indicating significant pain relief. Importantly, no noticeable adverse events were observed during the treatment.
Clinical characteristics of the patients (n = 30).

VRD: bortezomib, lenalidomide and dexamethasone; IRD: ixazomib, lenalidomide and dexamethasone; PCD: bortezomib, cyclophosphamide and dexamethasone.
We detected the NCV of bilateral median nerves, posterior tibial nerves, common ulnar nerves and peroneal nerves of patients before and after rTMS treatment. The results indicated varying degrees of reduction in SCV of limbs, even among patients not experiencing symptoms in all limbs. Additionally, partial patients exhibited varying degrees of reduction in MCV despite the absence of dyskinesia-related symptoms. For instance, patient No. 11 with grade 3 CIPN primarily complained of right upper limb pain, but showed no motor impairment. Nonetheless, SCV in all extremities and MCV in both upper decreased during NCV examination. Tables 2 and 3 summarize the SCV and MCV for each nerve injury of the patients. The comparisons of bilateral SCV and MCV before and after rTMS are presented in Figures 2 and 3, which show improvements in both SCV and MCV following rTMS treatment.
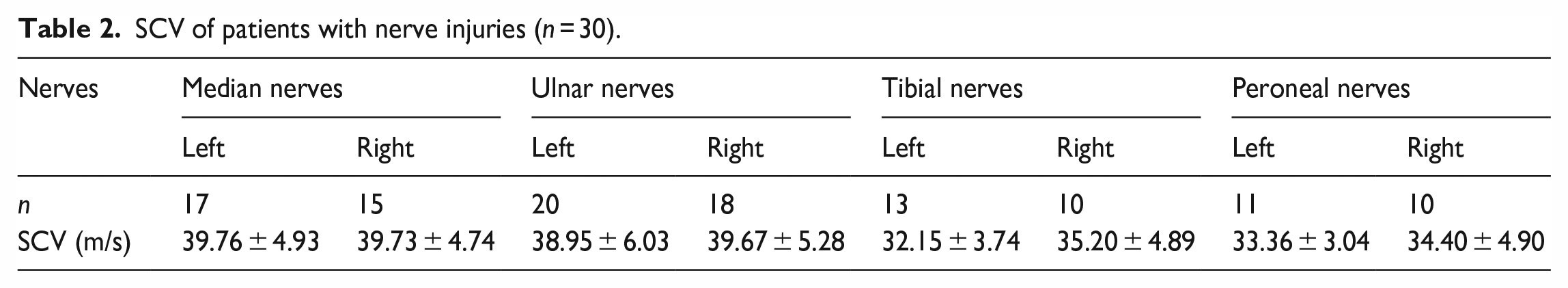
SCV of patients with nerve injuries (n = 30).
MCV of patients with nerve injuries (n = 30).


Comparisons of SCV of patients with nerve injuries before and after rTMS. Median nerve: left: 39.76 ± 4.93 m/s versus 44.29 ± 4.34 m/s (p = 0.013); right: 39.73 ± 4.74 m/s versus 43.4 ± 5.65 m/s (p = 0.045). Ulnar nerve: left: 38.95 ± 6.03 m/s versus 43.60 ± 5.72 m/s (p = 0.023); right: 39.67 ± 5.28 m/s versus 43.44 ± 5.22 m/s (p = 0.051). Tibial nerve: left: 32.15 ± 3.74 m/s versus 36.46 ± 4.99 m/s (p = 0.029); right: 35.20 ± 4.89 m/s versus 39.10 ± 5.69 m/s (p = 0.023). Peroneal nerves: left: 33.36 ± 3.04 m/s versus 36.55 ± 3.80 m/s (p = 0.023); right: 34.40 ± 4.90 m/s versus 38.20 ± 5.22 m/s (p = 0.105).
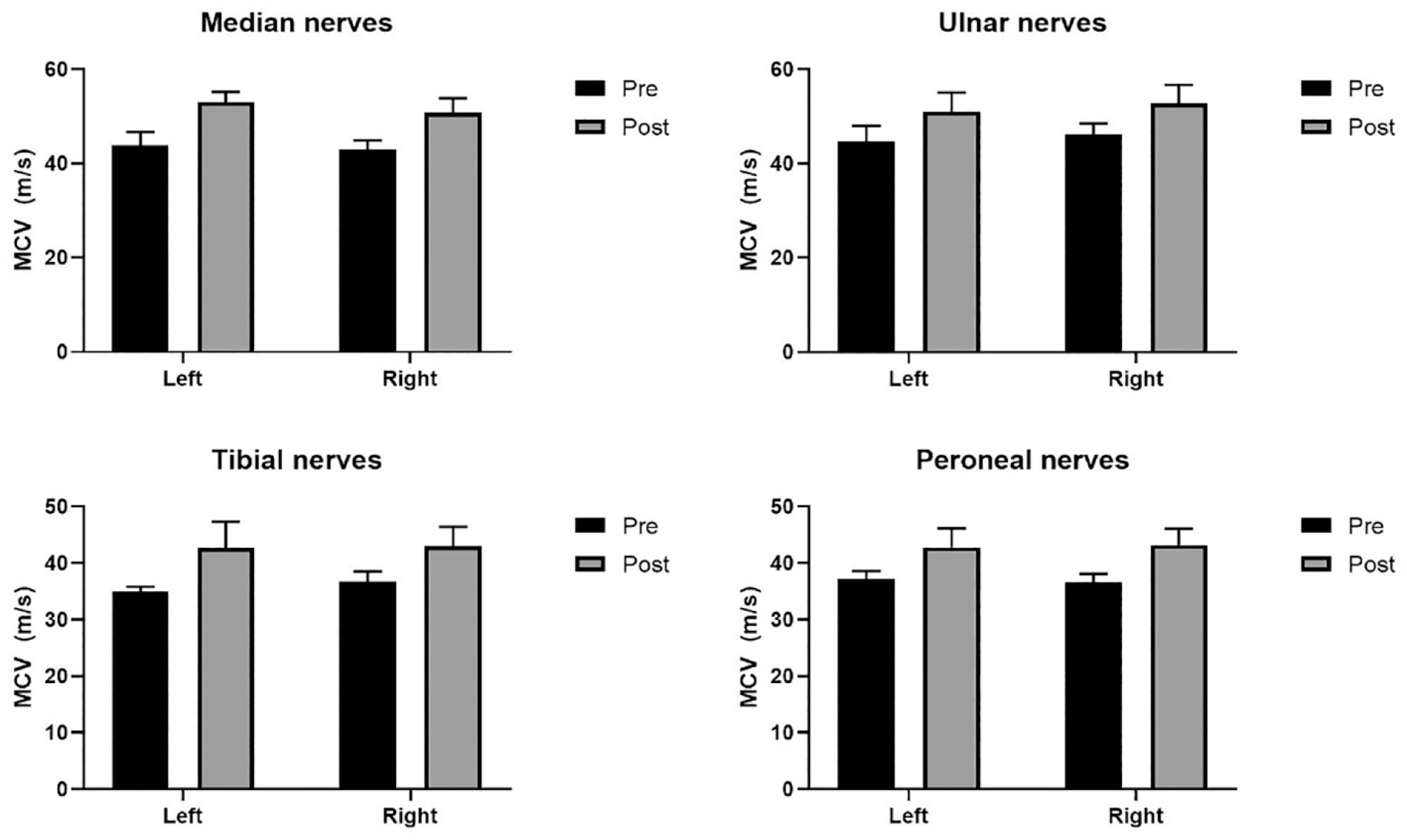
Comparisons of MCV of patients with nerve injuries before and after rTMS. Median nerve: left: 43.83 ± 2.86 m/s versus 53.00 ± 2.19 m/s (p = 0.002); right: 42.88 ± 2.03 m/s versus 50.75 ± 3.05 m/s (p < 0.001). Ulnar nerve: left: 44.78 ± 3.19 m/s versus 51.00 ± 4.03 m/s (p = 0.002); right: 46.20 ± 2.28 m/s versus 52.80 ± 3.90 m/s (p = 0.008). Tibial nerve: left: 35.00 ± 0.82 m/s versus 42.75 ± 4.57 m/s (p = 0.029); right: 36.67 ± 1.86 m/s versus 43.00 ± 3.41 m/s (p = 0.004). Peroneal nerves: left: 37.20 ± 1.40 m/s versus 42.80 ± 3.36 m/s (p < 0.001); right: 36.60 ± 1.52 m/s versus 43.20 ± 2.86 m/s (P = 0.008).
All patients underwent EORTC QLQ-CIPN20 assessments before and after treatment. The data (Pre: 17.68 ± 8.14 vs Post: 10.50 ± 9.55, p < 0.001) revealed statistically significant reductions in scores across all items, which indicates a reduction in CIPN symptoms following rTMS treatment. The results for the EORTC QLQ-CIPN20 subscales are depicted in Figure 4, highlighting significant differences before and after treatment across all three CIPN20 subscales. After rTMS treatment, the EORTC QLQ-CIPN20 scale of the patients with grade 2–3 (n = 25) decreased by 8.89 ± 4.24 points, while the difference value of patients with grade 4 (n = 5) was 3.54 ± 3.45, the difference was statistically significant (p < 0.001).

Comparisons of EORTC QLQ-CIPN20 subscale scores before and after rTMS. All subscale scores were linearly converted to a 0 to 100 scale; data are presented as mean ± standard deviation. Sensory symptoms: 24.35 ± 11.22 versus 16.48 ± 13.85, p = 0.002; Motor symptoms: 11.70 ± 8.10 versus 4.69 ± 6.97, p < 0.001; Autonomic symptoms: 12.92 ± 9.87 versus 7.50 ± 9.56, p = 0.022.
Discussion
We have retrospectively analyzed the clinical data of 30 MM patients with CIPN to assess the efficacy and safety of rTMS as a therapeutic approach. Our findings strongly indicate that rTMS is a secure and efficacious therapeutic approach for ameliorating peripheral nerve injury and alleviating the CIPN symptoms in these patients.
It’s important to note that MM remains an incurable disease. 15 Despite the emergence of new drugs such as CD38 monoclonal antibody daritozumab, 16 SLAMF7 monoclonal antibody elotuzumab 17 and histone deacetylase (HDAC) inhibitor panobinostat 18 in recent years, first-line treatment still revolves around sequential chemotherapy regimen, primarily incorporating PI and/or IMiD. Both PI and IMiD exhibit substantial anti-MM activity. The sequential chemotherapy effectively extends the survival time of MM patients and ameliorates the clinical symptoms caused by MM. 19 Notably, the incidence of CIPN in MM patients treated with PI and IMiD can be as high as 75% 20 and 30%~60%, 21 respectively. The precise pathogenesis of CIPN in MM patients remains elusive. Metabolic changes resulting from bortezomib accumulation in dorsal root ganglia cells, mitochondrial-mediated dysregulation of calcium homeostasis, and dysregulation of neurotrophins may contribute to the pathogenesis of CIPN. 22 Furthermore, CIPN might be influenced by gene polymorphisms related to the repair mechanisms and peripheral nervous system inflammation. 23 While the availability of some new targeted medicines had improved treatment safety, 24 their high cost has discouraged many patients, particularly those from economically disadvantaged regions. Given that CIPN can be refractory, it poses a significant burden on patients undergoing chemotherapy and can even lead to treatment abandonment. 25 In our study, 15 patients had to postpone or even discontinue treatment due to grade 3–4 CIPN, particularly grade 4 CIPN. Following rTMS treatment, 6 (40%) patients could resume chemotherapy, significantly reducing the risk of MM progression or relapse. This underscores the potential effectiveness of rTMS in managing CIPN in MM patients. Nevertheless, owing to the limitations of this retrospective study, prospective clinical trials were warranted for further validation. Additionally, for the remaining nine patients, exploring new treatment remains imperative to enhance the prognosis of these patients.
Previous clinical studies have highlighted the considerable therapeutic potential of rehabilitation treatments for individuals experiencing peripheral neuropathy due to neurotoxic medicine.26,27 In the context of MM, acupuncture has emerged as a promising approach to address the diverse symptoms associated with CIPN. 28 Furthermore, rTMS, as a novel and safe method of brain stimulation, has demonstrated significant efficacy in managing neuropathic pain.29,30 A pilot study suggested that rTMS could be potentially beneficial and effective for addressing not only pain but also dysesthesia in CIPN patients with breast or gynecologic cancer. 7 In our study, the assessment via the EORTC QLQ-CIPN20 life questionnaire, which evaluated patients’ symptoms, revealed that rTMS effectively reduced CIPN symptoms induced by PI and IMiD. This finding suggests that rTMS holds promise for alleviating CIPN symptoms in MM patients. Additionally, the results of NCV before and after treatment demonstrated improvements in both MCV and SCV of patients, further supporting the potential of rTMS to enhance peripheral nerve injury. The underlying therapeutic mechanisms of rTMS remain under investigation, proposed therapeutic mechanisms of rTMS include: (1) the regulation of cerebral cortex excitability 31 ; (2) modulation of local brain blood flow 32 ; and (3) enhancement of neurotransmitter levels 33 ; etc. Besides, there is no more data to suggest how to choose the stimulation frequency, stimulation intensity, etc. These problems require more research in the future.
CIPN can persist for extended periods, significantly affecting the quality of life of MM patients. 34 Previous studies have indicated that rTMS may be less effective in cases of severe dysesthesia. 35 Our study demonstrated that patients with grade 2 CIPN achieved a high rate (100%) of symptom improvement following rTMS treatment. On the contrary, only 1 (20%) patient with CIPN4 achieved remission. Furthermore, the results of EORTC QLQ-CIPN20 before and after treatment suggested that patients with lower-grade CIPN tended to experience more significant treatment effects. These findings underscore the importance of monitoring the severity of patients and initiating early interventions for CIPN. In addition, the authors hypothesize that preventive rTMS treatment may reduce the incidence of CIPN, but the exact mechanism has not been clarified; more researches are needed to validate this viewpoint.
The following information provides details on the limitations of the study. The small patient cohort, the absence of a power analysis for sample size calculation, and its retrospective nature, may impact the robustness of the results. Therefore, our results suggest the potential for clinical treatment, rather than definitive conclusions. Given the low incidence rate of the disease, obtaining large samples at a single center is challenging, highlighting the need for larger-scale multicenter clinical trials.
Conclusion
Our study demonstrates that rTMS is effective and safe for patients diagnosed with CIPN in the context of MM. The evaluation and monitoring of CIPN are pivotal aspects of clinical practice; early initiation of rTMS treatment could yield superior therapeutic outcomes for MM patients grappling with CIPN. To firmly establish these findings and expand our understanding, further randomized controlled studies are imperative.
Supplemental Material
sj-pdf-1-smo-10.1177_20503121231209088 – Supplemental material for Repetitive transcranial magnetic stimulation for chemotherapy-induced peripheral neuropathy in multiple myeloma: A pilot study
Supplemental material, sj-pdf-1-smo-10.1177_20503121231209088 for Repetitive transcranial magnetic stimulation for chemotherapy-induced peripheral neuropathy in multiple myeloma: A pilot study by Zhenzhuang Yan, Weiwei Cao, Lei Miao, Juan Li, Hui Wang, Dandan Xu, Hainan Yu and Yuanxin Zhu in SAGE Open Medicine
Footnotes
Acknowledgements
The authors thank the patients. They also thank the support from the Kangda College of Nanjing Medical University and the First People’s Hospital of Lianyungang City.
Authors’ contributions
ZZ.Y, WW.C, and H.W collected the clinical data. L.M, WW.C and DD.X analyzed the clinical data, made the summary. ZZ.Y and YX.Z designed this study and wrote this manuscript. L.J, YX.Z revised this manuscript. HN.Y provided administrative assistance for this study. All authors approved the manuscript.
Data availability
The data are not publicly available due to (restrictions e.g., their containing information that could compromise the privacy of research participants).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
Ethical approval for this study was obtained from * the institutional review committee of First Peoples’ Hospital of Lianyungang (Approval Number: LW-20230320001)*.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by funding from the First People’s Hospital of Lianyungang City ((grant QN202116) and the Kangda College of Nanjing Medical University (grant KD2021KYJJZD067 and KD2021KYJJZD033)).
Informed consent
Verbal informed consent was obtained from all subjects before the study, written informed consent was not obtained from patients, this requirement was waived off by the Ethical Review Committee. (IRB: the institutional review committee of First peoples’ hospital of Lianyungang).
Trial registration
*Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
