Abstract
Healthy dietary interventions are well established in cardiovascular disease prevention, but their effects on the brain remain underexplored. This scoping review aims to investigate how adherence to core components of a whole-food plant-based diet (WFPBD) may impact neuroimaging outcomes across different brain conditions. We searched PubMed and MEDLINE for studies published in the past 20 years evaluating the effects of a predominantly or exclusively WFPBD, alone or combined with other lifestyle factors, on neuroimaging biomarkers. Selected studies were grouped into three domains: (1) demyelinating disease, (2) dementia and Alzheimer’s disease, (3) cognitive aging and cerebrovascular burden. The dataset comprised heterogeneous study designs and follow-up durations, including randomized-controlled trials (RCTs; n = 9; 14 days - 3 years), longitudinal observational studies (n = 7; 2-20 years), and cross-sectional studies (n = 6). Observational evidence suggests associations between plant-based dietary patterns and favorable neuroimaging outcomes, whereas RCTs have generally not confirmed these effects. Residual confounding and healthy user bias are potential limitations. Dietary patterns aligned with a WFPBD may be important components of broader lifestyle interventions supporting brain health. Confirming observational findings will require adequately powered randomized trials with standardized neuroimaging endpoints and well-designed dietary interventions.
“Observational evidence suggests associations between plant-based dietary patterns and favorable neuroimaging outcomes”
Introduction
Extensive research has explored the impact of a whole-food, plant-based diet (WFPBD) on cardiovascular disease and non-brain tumors,1-5 yet studies on neurological effects remain scarce. Recently, growing evidence is addressing this gap, supporting the role of lifestyle interventions in preserving brain health and cognitive function. The latest randomized-controlled trial (RCT) by Ornish and colleagues, for instance, demonstrated that intensive lifestyle changes, centered on a WFPBD, regular exercise, and daily meditation, can reverse cognitive decline and reduce amyloid biomarkers in Alzheimer’s disease (AD) over just 20 weeks. 6 Given its anti-inflammatory and vasoprotective properties, it is reasonable to assume that a WFPBD may also induce measurable neuroimaging changes across the lifespan, positioning it as a novel therapeutic approach. One of the most ambitious ongoing studies to date, the U.S. POINTER, has enrolled more than 2,000 cognitively normal older adults at elevated risk for dementia and employed multimodal imaging biomarkers to examine associations with adherence to the Mediterranean-DASH Intervention for Neurodegenerative Delay (MIND) diet over a 2-year observation period. 7 In the imaging baseline cohort, higher cardiovascular risk was associated with more abnormal brain structure and poorer cognitive performance. 8 Nonetheless, conclusive longitudinal neuroimaging data are still pending publication. 9 In this emerging scenario, our scoping review aims to investigate how dietary adherence to the core components of a WFPBD, alone or combined with other lifestyle factors, may influence neuroimaging outcomes across different brain conditions. To our knowledge, this is the first comprehensive review on the topic.
Methods
The detailed literature selection process is displayed in Figure 1. Prisma flow-diagram.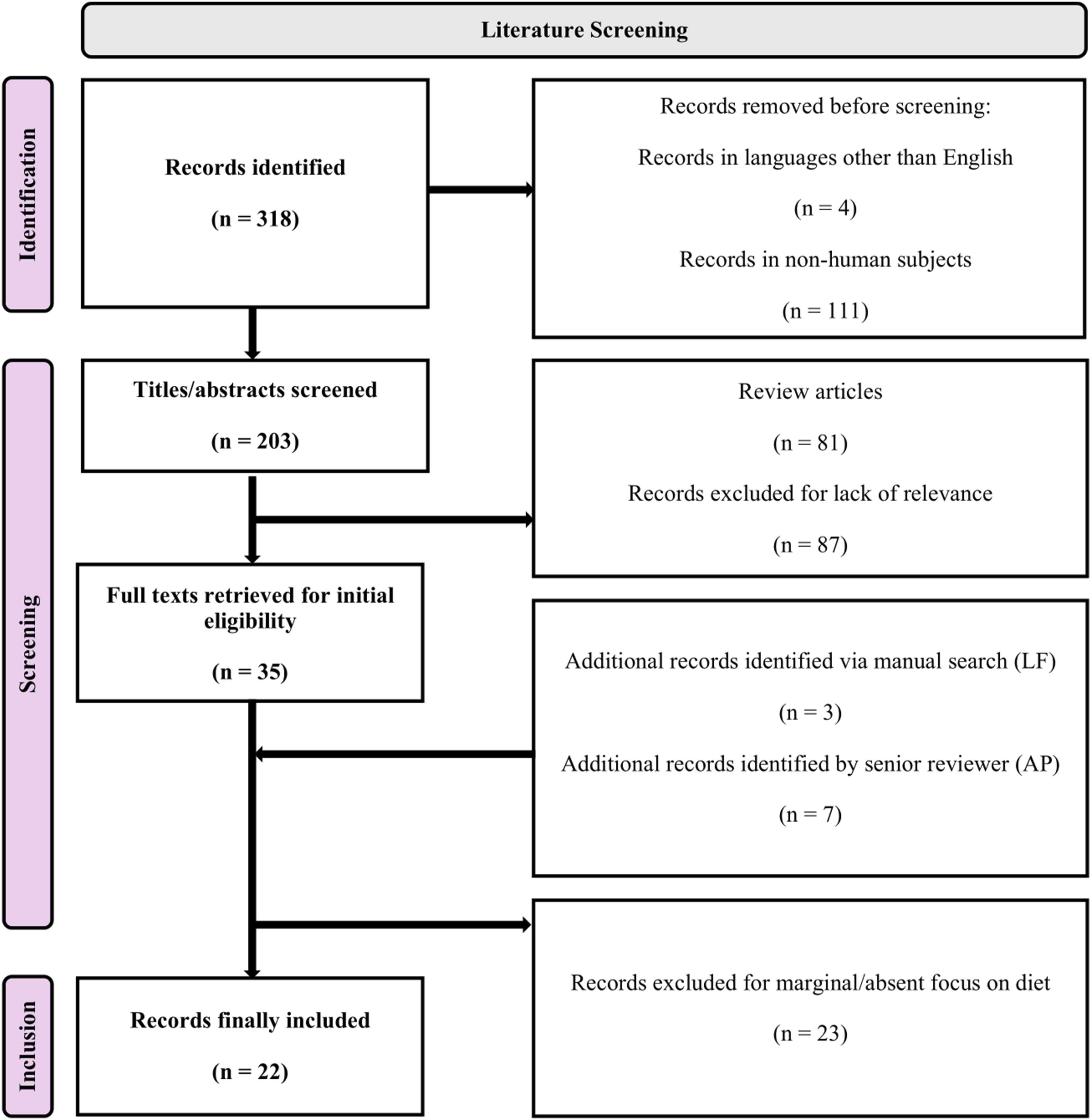
We searched PubMed and MEDLINE for papers from January 2005 to all March 2025, aiming to identify published studies focused on the impact of a predominantly WFPBD, alone or combined with other lifestyle factors, on neuroimaging biomarkers. We tested multiple search strategies with concept variations (e.g., “vegetarian” vs “plant-based” vs “Mediterranean,” “advanced imaging” vs “fMRI” vs “DTI”), combined with MeSH terms. The term “Mediterranean” was finally excluded to avoid dispersive results, maintain specific focus on plant-based food groups, and minimize bias towards fish consumption. After applying these criteria, we finalized the following research string to retrieve relevant titles and abstracts: (lifestyle[Title/Abstract] AND (plant-based[Title/Abstract] OR vegan[Title/Abstract] OR diet[Title/Abstract] OR exercise[Title/Abstract] OR sleep[Title/Abstract] OR stress[Title/Abstract]) AND (neuroimaging[Title/Abstract] OR fMRI[Title/Abstract] OR brain function[Title/Abstract] OR brain structure[Title/Abstract] OR brain connectivity[Title/Abstract])
We limited search results to articles published in English, on human subjects. The obtained records were evaluated for replicates/updates and individually screened in their titles and abstracts. Abstracts not reporting dietary assessments or neuroimaging outcomes were excluded, as well as review studies. For the remaining papers, the full texts were retrieved for evaluation and final inclusion. To screen for further relevant records, we complemented the database with a manual search from the reference lists of all the selected articles. The whole process was carried out following PRISMA guidelines for scoping reviews. 10 No automated tools were used. Titles and abstracts were retrieved by a single reviewer (LF). To ensure consistency in the application of the inclusion criteria, two reviewers (LF, AP) independently screened an initial 10% sample of records as a calibration exercise. Discrepancies were discussed and resolved by consensus. The remaining records were screened by a single reviewer (LF). Results were finally clustered into three domains: 1. Demyelinating Disease, 2. Dementia and Alzheimer’s Disease, 3. Cognitive Aging and Cerebrovascular Burden.
Clusters of Results and Discussion
Summary of Findings, in Alphabetical Order by First Author. Studies Shown in Italics are Still Underway, With Results Pending Publication.
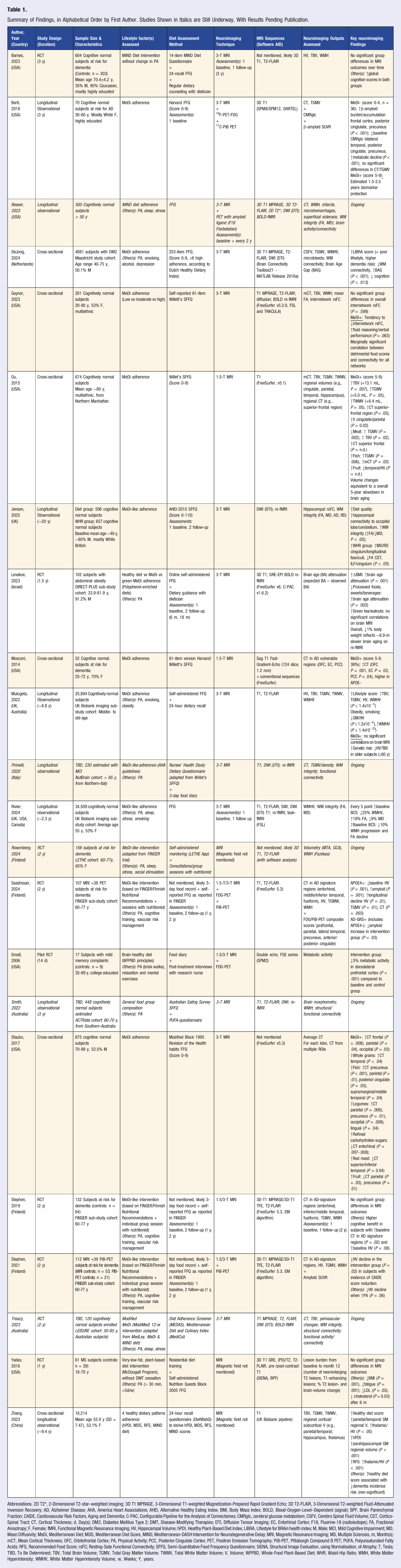
Abbreviations. 2D T2*, 2-Dimensional T2-star–weighted imaging; 3D T1 MPRAGE, 3-Dimensional T1-weighted Magnetization-Prepared Rapid Gradient Echo; 3D T2-FLAIR, 3-Dimensional T2-weighted Fluid-Attenuated Inversion Recovery; AD, Alzheimer Disease; AHA, America Heart Associations; AHEI, Alternative Healthy Eating Index; BMI, Body Mass Index; BOLD, Blood-Oxygen-Level–Dependent (signal); BPF, Brain Parenchymal Fraction; CAIDE, Cardiovascular Risk Factors, Aging and Dementia; C-PAC, Configurable Pipeline for the Analysis of Connectomes; CMRglc, cerebral glucose metabolism; CSFV, Cerebro Spinal Fluid Volume; CST, Cortico-Spinal Tract; CT, Cortical Thickness; d, Day(s); DM2, Diabetes Mellitus Type 2; DMT, Disease-Modifying Therapies; DTI, Diffusion Tensor Imaging; EC, Entorhinal Cortex; F18, Fluorine-18 (radioisotope); FA, Fractional Anisotropy; F, Female; fMRI, Functional Magnetic Resonance Imaging; HV, Hippocampal Volume; hPDI, Healthy Plant-Based Diet Index; LIBRA, LIfestyle for BRAin health index; M, Male; MCI, Mild Cognitive Impairment; MD, Mean Diffusivity; MeDi, Mediterranean Diet; MDS, Mediterranean Diet Score, MIND, Mediterranean-DASH Intervention for Neurodegenerative Delay; MRI, Magnetic Resonance Imaging; MS, Multiple Sclerosis, m, Month(s); mCT, Mean Cortical Thickness; OFC, Orbitofrontal Cortex; PA, Physical Activity; PCC, Posterior Cingulate Cortex; PET, Positron Emission Tomography; PiB-PET, Pittsburgh Compound B PET; PUFA, Polyunsaturated Fatty Acids; RFS, Recommended Food Score; rsFC; Resting-Sate Functional Connectivity; SFFQ, Semi-Quantitative Food Frequency Questionnaire; SIENA, Structural Image Evaluation, using Normalisation, of Atrophy; T, Tesla; TBD, To Be Determined; TBV, Total Brain Volume; TGMV, Total Gray Matter Volume; TWMV, Total White Matter Volume; V, Volume; WFPBD, Whole-Food Plant-Based Diet; WHR, Waist-Hip Ratio; WMH, White Matter Hyperintensity; WMHV, White Matter Hyperintensity Volume; w, Weeks; Y, years.
Cluster 1: Demyelinating Disease
Demyelinating diseases of the brain, such as multiple sclerosis (MS), are characterized by damage to the myelin sheath around nerve fibers, leading to impaired conduction of nerve impulses. This inflammatory-driven process results in progressive neurological dysfunction and disability. 11 Limited data are available regarding the impact of diet on demyelinating disease activity as observed on brain MRI, and those that exist focus primarily on MS. This may be because standard medication outcomes are preferred objectives in study designs, and any improvement in MRI findings may take longer than any symptom improvement experienced by the patient. Nonetheless, diet may plausibly influence disease activity by modulating neuroinflammatory pathways. Arachidonic acid, for instance, a pro-inflammatory omega-6 fatty acid predominantly found in fatty animal products, contributes to the central nervous system inflammation. 11 Excessive meat intake may thus exacerbate neuroinflammation, central to MS pathogenesis, while diets low in animal fat and rich in anti-inflammatory components could help attenuate symptoms.
Building on this, Yadav et al conducted the first RCT on 61 patients with MS to evaluate adherence, safety and measurable effects on brain MRI of a very-low-fat, WFPBD and daily exercise.12,13 Although the diet was well tolerated and adhered to, it resulted in no significant improvement neither on brain MRI, nor in relapse rate or disability over 1 year. Specifically, after controlling for baseline MRI differences, the number of new T2, newly enlarging T2, T1 enhancing or combined unique active lesions revealed no differences between the two groups. One possible explanation is that the intervention group exhibited more baseline lesions, possibly obscuring dietary effects between groups. It is also possible that time-related factors, in combination with the limited sensitivity of standard MRI, constrained the detection of subtle effects observable only with more advanced techniques. The diet group, however, did show significant improvements in BMI, metabolic biomarkers like LDL, and fatigue. Growing evidence suggests that vascular disease risk factors, like hyperlipidemia, are common in people with MS,14,15 implying that the cholesterol-lowering benefits supported by a WFPBD may still confer some advantages in the long term. Additional limitations include the small sample size and the concomitant use of disease-modifying therapies (DMTs) among most participants, which may have altered results. Evidence relating to outcomes after discontinuation of DMTs remains purely anecdotal, 16 and longer controlled trials are needed to rigorously evaluate the direct anti-inflammatory and immunomodulatory effects of a WFPBD on MS, particularly on those pathways influenced by the gut microbiome.17,18
Cluster 2: Dementia and Alzheimer’s Disease
AD pathogenesis has been linked to multiple pathways beyond amyloid accumulation, such as inflammation, microglial dysregulation, and lipid dysmetabolism. Diets emphasizing high intake of fiber, like the Mediterranean diet (MeDi), are known to be associated with less age-related cognitive decline thanks to their antioxidant effect.19-21 In addition, the emphasis on low-glycemic index carbohydrates and the abstention from alcohol and refined foods have shown to reduce the risk of dementia. 22 The Finnish Geriatric Intervention Study to Prevent Cognitive Impairment and Disability (FINGER) is a landmark RCT that assessed the effects of a 2-year multidomain lifestyle intervention on cognitive outcomes in 2,654 older adults at increased risk for dementia. While not strictly vegetarian, the trial promoted increased intake of vegetables, fruits, legumes, whole grains, and nuts, with reduced consumption of refined sugar and saturated fats, red and processed meats, aligning closely with a WFPBD. Dietary adherence was assessed through self-reported questionnaires and structured diet quality scores. 23 Several key exploratory sub-studies within FINGER later integrated neuroimaging biomarkers to evaluate structural and functional brain changes associated with the intervention.24-26
One of these, by Stephen et al, 24 is a RCT assessing the effects of the FINGER multidomain lifestyle intervention on brain MRI outcomes in a subset of 132 older adults. Imaging protocols were performed on 1.5/3-T MRI scanners, with high-resolution T1-weighted imaging for brain volumetrics and T2-FLAIR imaging for white-matter hyperintensities (WMH). After 2 years, no significant differences were observed between the intervention and control group involving regional brain volumes, cortical thickness, or white-matter (WM) lesion volume, although post hoc subgroup analyses revealed increased cognitive benefits in individuals with higher baseline cortical thickness in AD signature regions. A similar, though non-significant trend was found in participants with higher baseline hippocampal volume. These secondary findings should be interpreted cautiously given the elevated risk of false-positive results; taken together with the absence of significant MRI differences between groups, the overall pattern is more consistent with a null effect, underscoring the need for future, adequately powered studies.
In a separate sub-study by the same author, 25 researchers evaluated the specific relationship between changes in the Cardiovascular Risk Factors, Aging, and Dementia (CAIDE) risk score and neuroimaging biomarkers over the 2-year intervention period. This analysis included a subset of 112 individuals with MRI data and 39 with Pittsburgh Compound B PET (PiB-PET) scans. In this case, a reduction in CAIDE score, indicating improved dementia risk profile, was significantly associated with attenuated hippocampal volume loss in the intervention group but not in the control group, implying a possible enhanced protective effect. No significant associations were observed for gray matter (GM) volume, WM lesion volume or cortical thickness. Considering their preliminary nature, findings need to be confirmed in larger cohorts.
Another FINGER sub-study by Saadmaan and colleagues 26 utilized structural MRI to assess hippocampal volume, GM volume, WM lesion volume, and cortical thickness in AD’s signature regions. Fluorodeoxyglucose Positron Emission Tomography (FDG-PET) was used to evaluate glucose hypometabolism, a marker of neurodegeneration, while PiB-PET was used to quantify amyloid deposition. Brain imaging was performed at baseline and after 2 years in 107 patients from the MRI subgroup and 38 patients from the PET subgroup. APOE4-allele carriers showed significantly lower hippocampal volumes and higher amyloid deposition at baseline; the gene also showed a significant association with higher longitudinal decline of hippocampal volume, total GM volume, and cortical thickness in AD signature regions. Additionally, individuals with a high AD genetic risk score (including non-APOE4 carriers) in the intervention group showed less amyloid accumulation on PiB-PET and less FDG decline over time, suggesting that diet and lifestyle may differently modulate superimposed genetic risk factors through metabolic pathways. No significant differences in the rate of change of hippocampal volume, cortical thickness, GM, or WM lesion volume were observed between the intervention and control groups overall. The use of different MRI scanners across sites may have reduced consistency in volumetric and structural measurements; furthermore, the intervention impact on amyloid burden and brain atrophy may require longer follow-ups to detect striking changes.
Similarly to the FINGER sub-studies, a 3-year longitudinal trial by Berti et al 27 evaluated whether adherence to MeDi influences AD biomarker trajectories in 70 cognitively normal, middle-aged adults at increased risk for dementia. Dietary intake was assessed at baseline using a validated FFQ, with a greater score indicating greater intake of fruits, vegetables, legumes, cereals and healthy fats, and lower scores reflecting higher consumption of detrimental foods such as dairy, meat, and alcohol. Participants underwent multimodal neuroimaging at baseline and follow-up, including 3-T MRI, 18F-FDG-PET to quantify cerebral metabolic rate of glucose (CMRglc), and 11C-PiB-PET to assess β-amyloid deposition. Compared with high MeDi adherence, low adherence was associated with reduced baseline glucose metabolism, higher baseline amyloid burden, and greater longitudinal amyloid accumulation and metabolic decline, whilst no significant longitudinal group differences were observed on structural MRI. Nonetheless, the study found an estimated 1.5-3.5 years of biomarker protection against dementia in the high-adherence group. Considering that diet-related effects may become measurable only at later stages of life, the relatively younger average age of this cohort (approximately 50 years vs 70+ years in other studies) may represent a limitation. Other limitations include reliance on self-reported dietary assessments, the small, highly educated and health-screened cohort, and the absence of data on important lifestyle confounders such as smoking.
To better delineate the role of dementia genetic predisposition, Mosconi and colleagues 28 conducted a cross-sectional analysis to examine whether adherence to the MeDi is associated with structural MRI markers of brain atrophy in cognitively normal adults at increased risk for AD. Fifty-two participants completed a validated FFQ categorized into five beneficial food groups (fruits, vegetables, legumes, cereals and fish), and two detrimental groups (meat and dairy products), from which MeDi adherence scores were derived ranging from 0 to 9. Structural MRI was acquired and images were processed with FreeSurfer to obtain cortical thickness in AD-vulnerable regions. MeDi-adherent subjects showed greater thickness in these regions, most robust in the left hemisphere. APOE non-carriers with high MeDi adherence exhibited the greatest cortical thickness, while APOE carriers showed reduced thickness irrespective of dietary adherence, suggesting that genetic predisposition may induce atrophy regardless of lifestyle interventions. The authors concluded that lower MeDi adherence is associated with greater cortical thinning in regions typically affected in prodromal AD, highlighting food quality as a valuable modifiable factor in preventing neurodegeneration. Limitations include the small sample size, the cross-sectional design, reliance on self-reported dietary intake, and the use of a 1.5-T MRI scanner, providing lower spatial resolution.
The Mediterranean-DASH Intervention for Neurodegenerative Delay (MIND) by Barnes et al 29 is an example of RCT specifically designed to test whether the consumption of a predominantly WFPBD, with emphasis on green leafy vegetables, nuts, berries, and olive oil, and limited intake of sugars and animal-derived saturated fats, could slow cognitive decline among 604 older adults with elevated risk associated with a family history of dementia. The protocol included MRI scans at baseline and 3-year follow-up in a sample of 604 participants to assess brain structure, WMH, and hippocampal atrophy. While both MIND and control diet groups showed modest cognitive improvement over time, no significant differences were detected in MRI outcomes over 3 years, including hippocampal volume and total GM or WM measures. One possible explanation for the largely null findings is the limited diversity of the study population and the likelihood that participants in the control group also improved their dietary patterns in response to caloric-restriction guidance, thereby reducing the contrast between groups.
Additional evidence from at-risk subjects comes from a small randomized-controlled pilot study by Small et al, 30 which evaluated the effects of a 14-day multimodal healthy lifestyle program on cognitive function and brain metabolism in 17 middle-aged adults with self-reported memory complaints. Participants were randomly assigned to either an intervention group that followed a structured program (including a brain-healthy diet, cardiovascular exercise, relaxation techniques, and memory training) or a control group that continued their usual lifestyle. Diet emphasized antioxidant-rich fruits and vegetables, omega-3 fatty acids, and low-glycemic index carbohydrates. Objective cognitive tests and brain imaging using FDG-PET and 3-T MRI were conducted at baseline and after 2 weeks. Neuroimaging revealed a statistically significant reduction in resting metabolic activity in the left dorsolateral prefrontal cortex in the intervention group only. Although interpreted as greater neural efficiency in a region central to working memory, executive function, and verbal fluency, reduced prefrontal metabolism is not always unequivocally beneficial and may also be indicative of reduced neural engagement or dysfunction. Given the very small sample and short duration, as well as the lack of objective adherence measures, the findings should be viewed as a provisional proof-of-concept rather than meaningful evidence for or against the hypothesis.
Cluster 3: Cognitive Aging and Cerebrovascular Burden
Studies outlined in the previous section lack to directly link specific food or nutrient subgroups (e.g., green leafy vegetables, polyphenol intake) to changes in specific brain biomarkers. Moreover, the absence of advanced neuroimaging assessments, such as brain-age prediction models, diffusion MRI (dMRI), or functional MRI (fMRI), may further reduce sensitivity to subtle microstructural or functional changes. Rather than relying on cohorts enriched for genetic or clinical risk of dementia, these evaluations are better addressed in studies involving cognitively normal individuals or those with cardiometabolic risk factors (e.g., diabetes, obesity). Such studies aim to more accurately characterize patterns of normal brain aging and to identify early signs of cerebrovascular disease or neurodegeneration.
Volumetry and Functional Connectivity in Larger Cohorts
A large study by Zhang et al 31 retrieved data from the UK Biobank to investigate whether four different dietary patterns in midlife were associated with higher structural brain health at baseline and higher incidence of dementia long-term. Dietary intake was assessed using a validated 24-hour recall questionnaire, from which four diet quality indices were derived, with hPDI reflecting high adherence to a fully WFPBD, and the Mediterranean Diet Score (MDS), the Recommended Food Score (RFS), and the MIND diet scores capturing variations of MeDi eating patterns. Brain structure was cross-sectionally evaluated in a subset of 18,214 participants using structural MRI to generate normalized global and regional volumetrics of total brain, GM and WM, and subcortical structures such as the hippocampus and thalamus. The study found that higher diet quality was consistently associated with larger volumes in multiple brain regions relevant to cognitive aging, such as the hippocampus and thalamus (RFS group), and the parahippocampal GM (hPDI group). However, important limitations include the cross-sectional design for imaging outcomes, reliance on self-reported dietary recalls, potential selection bias within the UK Biobank cohort, and the possibility of reverse causation.
Mulugeta et al conducted an additional large-scale, prospective observational study using imaging data from the UK Biobank.32,33 The sample included up to 25,894 middle- to older-aged participants, making this one of the largest neuroimaging datasets to date. The study aimed to investigate both the independent and interactive effects of genetic risk and healthy lifestyle behaviors on preclinical markers for brain health, such as total brain volume, GM and WM volumes, hippocampal volume, and WMH burden. The dietary component was defined by three indicators: sufficient intake of fruits and vegetables, adequate omega-3 consumption, and limited intake of processed foods and red meat. Neuroimaging outcomes were derived from structural T1-weighted and T2-FLAIR MRI sequences. The main findings revealed that a higher overall healthy lifestyle score was associated with greater total brain, GM, and hippocampal volumes, as well as lower WMH volume, with these benefits observed independently of age or genetic risk for AD. However, when the components of the lifestyle score were examined individually, only obesity and smoking were consistently associated with brain volumetrics. Specifically, both risk factors were linked to lower GM and hippocampal volumes and increased WMH burden. The null association between the diet component of the lifestyle score and brain volumetrics highlights again the current limitations in the sensitivity and consistency of dietary assessment tools used in large cohorts, reliant on variable self-reported intake. Furthermore, no interaction effects were observed between genetic risk scores and lifestyle factors, suggesting that lifestyle modifications benefit brain health regardless of underlying genetic predisposition, especially in younger subjects (< 60 years old).
Similarly, Rivier and colleagues retrieved data from the UK Biobank to further investigate the relationship between health-related behaviors and neuroimaging markers of brain health in a large cohort of middle-aged adults without history of prior stroke or dementia.33,34 Researchers applied the Brain Care Score (BCS), a measure encompassing 12 modifiable risk factors, including diet quality, physical activity (PA), and cardiovascular health, to assess participants’ brain health behaviors. Dietary habits, as part of the BCS, scored higher if based on abundance of fruits and vegetables, considerable grain intake, and reduced/null red meat, salt and alcohol consumption. Neuroimaging outcomes included WMH volume to assess vascular burden, as well as fractional anisotropy (FA) and mean diffusivity (MD) to evaluate WM integrity, all measured via MRI. 35 Results showed that higher baseline BCS scores, hence higher healthy lifestyle adherence, are consistently associated with better neuroimaging profiles at baseline (lower WMH burden, better WM integrity) and reduction in WMH progression and FA decline over time. Although the individual effect of diet was not isolated, this study is the first to extend the impact of healthier behaviors on neuroimaging markers of clinically silent cerebrovascular disease, potentially useful in stroke prevention. A major strength of the study is its large, well-characterized neuroimaging cohort, though its generalizability is limited by the restricted age range (40-69 years) and the predominantly healthy demographic of the UK Biobank, who generally tend to have higher BCS.
In cohorts beyond the UK Biobank, Gaynor et al performed a cross-sectional neuroimaging study within the Reference Ability Neural Network (RANN) project to investigate whether MeDi adherence modulates the association between resting-state functional connectivity (rsFC) and cognitive function. 36 The study included 201 cognitively healthy adults who underwent rs-fMRI while completing a battery of functional tasks. Dietary data were collected using a validated semi-quantitative FFQ, and participants were categorized into low, moderate, or high MeDi adherence groups. The MeDi scoring system emphasized higher consumption of wholesome plant-based food groups including vegetables, fruits, legumes, whole grains, and unsaturated fats, while discouraging full-fat dairy products, red and processed meats. The study found that higher internetwork rsFC (often a marker of cognitive decline due to reduced segregation between brain networks) was associated with worse fluid reasoning and verbal performance in individuals with low adherence to the MeDi, indicating a potential protective effect when certain food groups are avoided. In fact, analyses of dietary subcomponents suggested that the observed protective effects were more strongly associated with reduced consumption of detrimental foods rather than increased intake of beneficial ones, reinforcing the importance of minimizing processed and high-saturated-fat foods in supporting brain function. Limitations include the cross-sectional design, small sample, and reliance on in-scanner cognitive tasks. Nonetheless, the use of rs-fMRI to assess internetwork connectivity adds a novel methodological dimension, offering insights into how diet may modulate brain network dynamics.
The latest longitudinal study by Jensen and colleagues 37 also implemented rs-fMRI to examine associations between midlife diet quality and central obesity with later-life hippocampal connectivity and cognition, following participants within the Whitehall II study from their 50s into their 70s. The final sample comprised 657 subjects for waist-hip ratio (WHR) analyses and 506 subjects for diet quality analyses. Neuroimaging outcomes included hippocampal rsFC and markers of WM integrity such as FA and MD. Better diet quality in midlife and from midlife to late life, reflecting higher intakes of wholesome plant-foods and healthy fats, and lower consumptions of unhealthy foods, were linked to stronger hippocampal connectivity to occipital and cerebellum and better WM integrity; conversely, higher midlife central obesity predicted widespread WM microstructural decline in the CST, cingulum and inferior longitudinal fasciculus. Notably, leveraging more than 20 years of observational data, this study is the first to identify midlife as a key window for early dietary interventions in support of brain connectivity. However, limitations to consider include the predominantly male and Caucasian study population, reliance on self-reported dietary data, and its observational design.
Cortical Thickness and Brain-Age Gap in Relation to Food Subgroups and Cardiovascular Risk
Based on a large cross-sectional analysis of 674 dementia-free participants within the multiethnic WHICAP cohort, Gu et al 38 demonstrated how a higher adherence to the MeDi is associated with a healthier brain structure in older adults (mean age 80 years). Dietary intake was assessed using the validated 61-itemWillet’s semi-quantitative FFQ, which assigns higher scores for greater consumption of vegetables, legumes, fruits and nuts, cereals, fish, a higher monounsaturated-to-saturated fat ratio, moderate alcohol use, and lower intake of detrimental foods such as meat and dairy. Structural brain MRI and FreeSurfer served to derive brain volumes and cortical thickness. A higher MeDi adherence (score 5-9), driven particularly by greater fish intake and lower meat intake, was linked to larger total brain, GM and WM volumes, greater regional volumes in areas vulnerable to aging and AD (cingulate, parietal), and larger cortical thickness of the superior-frontal region. Surprisingly, fruit intake was associated with lower temporal and hippocampal volumes, although this finding was not clearly detailed nor addressed in the paper. These volumetric differences corresponded to approximately five fewer years of brain aging, and the associations remained consistent across sex, ethnicity, education, vascular factors, and APOE genotype. Despite its cross-sectional design, reliance on self-reported dietary data, and the fact that cortical thickness was not adjusted for intracranial volume, the study’s integration of a detailed dietary assessment with brain volumetry in a large and ethnically diverse cohort suggests that higher consumption of plant-based food groups, alongside seafood-derived omega-3s, may help preserve brain volume and cortical integrity in later life, especially in AD-vulnerable regions like the frontal lobe.
More recently, Staubo et al 39 performed a comparable cross-sectional analysis in 672 cognitively normal adults aged over 70 from the Mayo Clinic Study of Aging, investigating the relationship between MeDi adherence and cortical thickness. Dietary intake over the prior year was assessed using a validated 128-item FFQ, from which MeDi score and individual macro- and micronutrient intakes were calculated. High-resolution 3-T MRI scans were processed with Freesurfer to derive cortical thickness measures for each lobe. Higher MeDi adherence and greater intakes of legumes, whole grains, vegetables and fish were associated with larger cortical thickness in parietal and frontal lobes, as well as in multiple AD-vulnerable regions across all lobes; conversely, higher glycemic index foods, including highly refined carbohydrates and, surprisingly again, fruit, were associated with lower entorhinal and parietal thickness respectively; red meat was also associated with lower cortical thickness, specifically in the temporal lobe. Though impactful, results are limited by the cross-sectional design, the potential reporting bias inherent to FFQs, and healthier and more educated MRI participants relative to non-participants, which may underestimate associations. Moreover, the observed negative association between fruit intake and cortical thickness, in line with findings by Gu et al, 38 may reflect older adults’ more monotonous dietary habits and their low PA, thus it may not apply to younger, more active populations with varied diets. Overall, while fruit has widely recognized health benefits, non-starchy vegetables appear to offer more robust protection against cognitive decline. 40
Regarding brain integrity outcomes in subjects with increased cardiometabolic risk, Levakov et al demonstrated that lifestyle-induced weight loss, especially when accompanied by reduced consumption of animal products and processed foods, can attenuate functional brain aging. 41 This study was a sub-analysis of an 18-month Dietary Intervention RCT which evaluated baseline and follow-up neuroimaging data of a total of 102 middle-aged adults with abdominal obesity and/or dyslipidemia. Based on self-reported questionnaire data, participants were randomized into one of the following three arms: general healthy dietary guidelines, non-green Mediterranean diet (MED), green-Mediterranean diet (green-MED). Despite their inclusion of fish and poultry, both MED and green-MED diets encouraged an increased intake of fiber and plant-derived polyphenols (i.e., 28 g walnuts and 3-4 cups/day green tea plus 100 g/day Wolffia globosa shake respectively) and a lower intake of meat, processed foods, sweets, and sugar-sweetened beverages. fMRI was employed to assess brain rsFC and to estimate brain age using machine learning datasets, with less network segregation reflecting increased brain age. Among specific dietary variables, reduced intake of processed foods, sweets, and sugary beverages was associated with greater brain-age attenuation. Contrary to expectations, increased intake of polyphenol-rich foods was not independently associated with improved brain-age metrics. Significant associations were also observed between brain-age attenuation and reductions in waist circumference, visceral and liver fat, liver and kidney function markers, and metabolic inflammation. Overall, findings showed that a 1% reduction in body weight was associated with an estimated 8.9-month attenuation in predicted brain age on rs-fMRI, providing indirect evidence for the neuroprotective potential of a WFPBD, widely known to help weight loss. 42 Key limitations include the short duration of the study, the lack of a no-intervention control arm, substantial gender imbalance (91.2% male), and a predominantly obese cohort with comorbidities.
Finally, in their large observational cohort from The Maastricht Study, DeJong et al more recently investigated whether brain structure and connectivity reflect lifestyle choices and cognitive health among 4881 adults aged 40 to 75 years with type 2 diabetes.43,44 Lifestyle risk was quantified using the LIfestyle for BRAin health (LIBRA) score, which integrates modifiable dementia risk factors, including diet assessed through both the Dutch Healthy Diet Index 2015 and MeDi adherence. 45 Participants underwent extensive cognitive testing and 3-T brain MRI to assess both structural and functional neuroimaging outcomes, including WMH, brain volumetrics, WM tractography and connectivity evaluation. The Brain Age Gap (BAG) was calculated as the difference between the predicted brain age, based on structural imaging markers, and chronological age, a higher gap meaning that the brain looks older than its actual age. The model predicting brain age used total GM volume, WMH volume, cerebrospinal fluid (CSF) volume, and the presence of cerebral microbleeds. Higher LIBRA scores, reflecting a higher dementia risk due to poor lifestyle choices, were significantly associated with older brain age, lower WM connectivity, and poorer cognitive performance. Although a higher diet score contributed to a better neuroimaging profile, the study did not report separate correlations between diet alone and MRI measures; moreover, although higher diet scores reflected a predominantly plant-centered diet, the study did not specifically evaluate individual food subgroups. Other limitations include the cross-sectional nature of the study, and the use of whole-brain average measures that may obscure effects that are more region-specific.
Overall, observational evidence in cognitively healthy individuals suggests associations between higher adherence to plant-based dietary patterns and more favorable neuroimaging biomarkers (e.g., greater brain volumes, improved integrity and connectivity, reduced amyloid accumulation), though residual confounding and healthy user bias may largely contribute to these positive findings. In fact, existing RCTs in at-risk or clinically affected populations (Clusters 1-2) have largely failed to confirm these outcomes; nonetheless, the promising signals from observational studies highlight an emerging theme deserving further investigation. A conceptual framework outlining the hypothesized neuroprotective pathways supported by observational evidence are outlined in Figure 2. Hypothesized neuroprotective pathways supported by observational evidence, requiring confirmation through adequately powered RCTs. Abbreviations. BBB, Blood Brain Barrier; FA, Fractional Anisotropy; FDG, Fluorodeoxyglucose; MD, Mean Diffusivity; ML, Machine Learning; MRI, Magnetic Resonance Imaging; PET, Positron Emission Tomography; PiB, Pittsburgh Compound B; rs-fMRI, Resting-State Functional MRI; SCFA, Short-Chain Fatty Acids; WM, White Matter; WMH, White Matter Hyperintensities.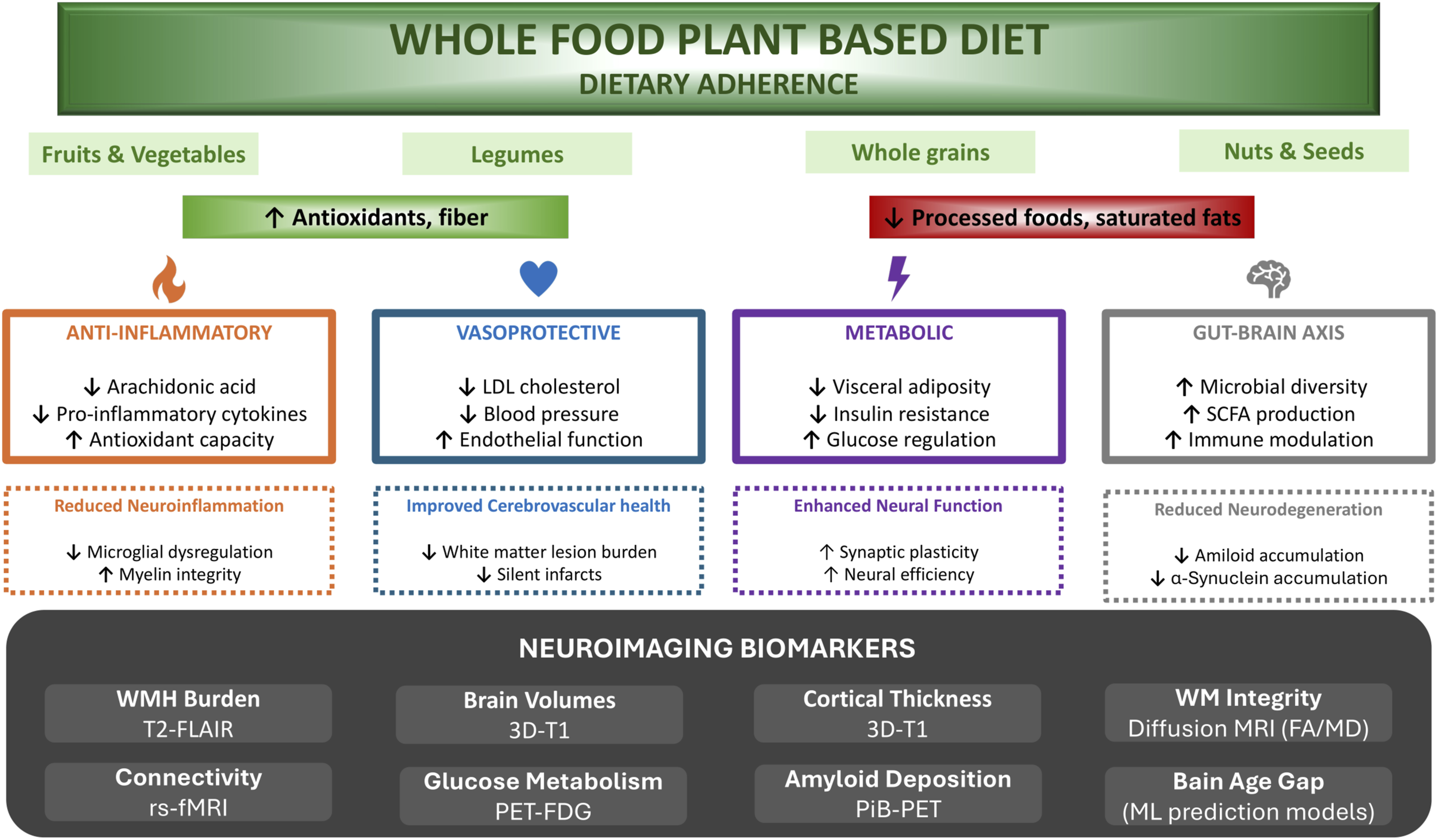
Cluster 4: Ongoing Projects
Beyond the U.S. POINTER,7-9 other several ongoing longitudinal trials and related sub-studies are currently investigating via neuroimaging techniques how lifestyle habits may impact brain integrity over time.46-55 These studies emphasize adherence to healthy dietary patterns, assigning higher scores to increased intakes of core WFPBD components. The main characteristics are summarized in Table 1 (in italics), with a detailed description provided in the Supplemental Material section.
The Evidence Gap: Brain Tumors, Movement Disorders and Stroke
Our review disclosed a notable gap in literature on studies specifically investigating the effects of a WFPBD on brain tumors, movement disorders and stroke-related outcomes assessed through neuroimaging techniques.
Current glioma imaging studies predominantly focus on tumor size, growth, edema, and response to conventional treatments such as surgery, chemotherapy, radiotherapy, and emerging targeted therapies. Metabolic imaging studies in brain tumors have primarily explored ketogenic or calorie-restricted diets, which differ substantially from a WFPBD.56,57 Nonetheless, dietary patterns rich in plant-derived antioxidants and anti-inflammatory compounds are theorized to support brain tumor prevention.58-63 The studies by Mousavi et al and Zhang et al represent important early efforts to investigate the relationship between a WFPBD and glioma incidence, though they lack to report neuroimaging outcomes.64,65 Mousavi et al conducted a hospital-based case-control study involving 128 glioma patients and 256 glioma-free controls, showing that higher adherence to a healthy plant-based diet, with low consumption of refined grains, sugar-sweetened beverages, fruit juices, and sweets/desserts, was associated with significantly reduced odds of glioma, in line with other studies. 66 The study employed comprehensive dietary assessment tools to differentiate between healthy and unhealthy plant-based components. 67 Similarly, Zhang et al examined 506 matched glioma cases and controls in a Chinese population, reporting a 58% reduced risk associated with a healthy WFPBD and an 8-fold increased risk of glioma associated with unhealthy plant-based diet. This study further analyzed dietary components, identifying refined grains particularly as a major contributor to increased glioma risk and potatoes as a potential benefit for its antioxidant properties.68,69 Despite robust dietary characterization and statistical controls, both studies were entirely observational and lacked neuroimaging data assessing tumor progression, structure, or metabolic changes in relation to diet. Therefore, while these findings strengthen the hypothesis that diet may influence glioma risk, they offer no evidence on whether such effects are reflected in measurable imaging biomarkers or align with clinical criteria like the Response Assessment in Neuro-Oncology (RANO).
Building on the growing interest in the brain-gut axis and its role in neurodegeneration, Hegelmaier et al conducted a case-control study examining the effects of a short-term, well-controlled high-fat lacto-ovo-vegetarian diet on the gut microbiome in 16 patients with PD. 70 Over 14 days, data showed increased microbial diversity, enclosing a potential therapeutic role in modulating the gut-brain axis; specifically, at a 1-year follow-up, the difference in gut microbiome was associated with significant improvements in motor symptoms and reductions in levodopa daily doses, particularly when combined with fecal enemas. Most dietary studies in PD have centered on ketogenic or low-fat diets, making the adoption of a plant-rich diet a noteworthy advancement in this case, although it did include controversial sources of saturated fat such as ghee. 71 However, no neuroimaging outcomes were reported. Given the challenges of directly visualizing the substantia nigra, future research should incorporate advanced imaging modalities such as neuromelanin-sensitive MRI to detect dopaminergic neuron loss, iron-sensitive techniques like susceptibility-weighted imaging (SWI) or quantitative susceptibility mapping (QSM) to track pathological iron accumulation, and DTI to assess microstructural changes in motor pathways. Rs-fMRI and volumetric analysis may also capture early network alterations linked to disease progression.
Finally, although a substantial body of literature supports the role of dietary interventions in reducing stroke risk,72,73 particularly in secondary prevention through MeDi diets centered on high intakes of fruits, vegetables and fiber,74-78 there remains a striking absence of studies incorporating neuroimaging data to assess their effects on brain structure and function in stroke survivors.
The Role of Physical Activity
Across the several studies reviewed, PA is consistently integrated with diet and other healthy behaviors in multidomain lifestyle interventions. According to the American Heart Association (AHA), adults should aim for at least 150 minutes per week of moderate-intensity aerobic activity (or 75 minutes of vigorous activity), combined with muscle-strengthening exercises at least 2 days per week, while reducing sedentary time. In studies like FINGER23-26 and POINTER, 8 PA is part of structured programs combining supervised aerobic and resistance training with tailored home-based exercises, which show to improve cognitive outcomes alongside diet. Notably, the ACTIVate, 49 LETHE 48 and LEISURE 46 studies implement the use of accelerometers, smartwatches, app-guided sessions, and group support to track and promote adherence to exercise. In the DIRECT-PLUS study, 41 PA promotion accompanies dietary changes in supporting weight loss and leading to attenuation of brain age on rs-fMRI. Observational studies from the UK Biobank dataset32,34 assess PA through self-reported questionnaires and composite lifestyle scores, showing strong associations between synergic healthy behaviors and better neuroimaging biomarkers. Incorporating daily brisk walks to a short-term dietary intervention also proved to enhance neural function on PET-FDG. 30 In line with this, a preliminary analysis from the HBI found that just 20 minutes of daily walk is associated with a lower WMH burden. 55 In MS studies, PA is an additional part of the intervention adopted for its beneficial effects. 12 Overall, extensive evidence highlights PA as a key lifestyle factor, with positive outcomes reflecting the combined influence of multiple virtuous behaviors. Nonetheless, there is observational evidence that has independently linked higher adherence to the Healthy Plant-Based Diet Index, fully aligned with a WFPBD, to larger parahippocampal volumes even after adjustment for PA, APOE genotype, and other relevant covariates. 31 This suggests that, while PA can enhance benefits in most contexts, diet adherence alone may also have robust and independent effects on brain integrity.
Nutritional Recommendations
Plant-exclusive diets usually require careful planning to ensure adequate intake of nutrients that are either absent or present in low bioavailability in plant foods, like vitamin B12 and DHA/EPA. These considerations are largely absent in the reviewed literature, yet essential in the context of brain health, as adequate vitamin B12 and DHA levels are crucial for preventing adverse neurological outcomes.79-81 Thus, while a WFPBD may offer significant neuroprotective benefits by completely avoiding animal-derived saturated fats and cholesterol, heavy metals and neurotoxins (including fish-derived ones),82-85these advantages may be diminished if nutritional adequacy is not simultaneously addressed. Current guidelines86,87 recommend a reliable source of vitamin B12 through supplements or fortified foods in all meatless diets, including the widespread lacto-ovo-vegetarian, and recognizes the potential need for microalgae-derived DHA in all those individuals with low omega-3 status or higher physiological needs (e.g., pregnancy, childhood, diabetes). For individuals who do not fall into these categories, sufficient omega-3 biosynthesis can generally be achieved through ALA-rich nuts and seeds, particularly flaxseed oil, chia seeds, and walnuts. In fact, observational data from the EPIC-Norfolk cohort indicate that even in the absence of direct EPA and DHA intake from seafood, and overall lower blood levels, vegans can still efficiently convert shorter-chain fatty acids into long-chain EPA/DHA. 88 Other micronutrients such as calcium, iron, zinc, selenium, magnesium, and iodine are typically of lesser concern in vegan populations due to their broad availability in plant foods like green leafy and cruciferous vegetables, whole grains and legumes (including soy products), and seaweed, while also reducing the risk of unnecessary overloads.
With this in mind, the precise “dose-response” relationship between adherence to a fully WFPBD and neuroimaging outcomes remains unclear, including whether it offers benefits beyond plant-rich diets that still allow small amounts of animal foods (e.g., MeDi, DASH, or MIND diets). In fact, contrary to the common misconception that the Mediterranean is merely a “balanced omnivore” diet, the traditional MeDi is by its newest definition largely a WFPBD, allowing only moderate weekly amounts of small-sized fish and generally minimizing to low or zero intake red meat, full-fat dairy, highly processed foods, and alcohol. 89 Rajaram et al 90 arguments that many of the beneficial associations found on structural and functional MRI of the brain mainly arise from vitamin- and polyphenol-rich foods such as citrus fruits, grapes, berries, cocoa, nuts, green tea, and small amounts of coffee, largely through mechanisms involving reduced inflammation and improved vascular health. On the other hand, it also seems that reduced consumption of detrimental foods, rather than increased intake of beneficial ones, may offer greater protection.36,41 Available evidence openly supporting regular fish consumption seems to suggest that the cumulative benefits of an elevated and diverse intake of wholesome plant-based food groups, together with a high fish-derived omega-3 component, may still outweigh the potential negative effects associated with scattered amounts of animal products.28,37-39 Conversely, when diets are low in plant foods, the cognitive benefits of fish are no longer observed.91,92 In line with this, existing comparisons among pesco-vegetarian, lacto-ovo-vegetarian and vegan groups tend to indicate that an exclusively WFPBD may offer greater advantages than mixed diets, at least from a metabolic perspective.93-96
From a “time-response” standpoint, future trials are needed to clarify how the timing and duration of a WFPBD adherence influence brain structure and function. In the study by Mosconi and colleagues, 28 most participants (≈ 90%) reported stability in their diets over the previous 5 years or longer, and individuals showing reduced brain atrophy had often been following a plant-centered diet since childhood. Although the reported dietary adherence likely reflects long-term dietary habits, the cross-sectional design limits conclusions about its temporal effects. Overall, several studies seem to suggest that the earlier the lifestyle change, the more pronounced and durable the neuroprotective effects may be, with midlife emerging as a potentially critical window in which dietary interventions may exert maximal influence,24-27 sometimes even defying a superimposed genetic risk.26,32 Nonetheless, even a shorter-term adherence to healthier choices appears to provide some anti-inflammatory and neuroprotective benefits,30,41 although results need to be confirmed on more robust trials.
Common Limitations in the Current Body of Literature and Future Directions
Available evidence combining plant-based dietary patterns to brain imaging biomarkers predominately comes from heterogeneous cross-sectional, longitudinal or interventional studies of 2-3 years duration. Only three longitudinal observations collected data over a period of 10 years or more,31,32,37 which would be preferable for detecting subtle structural or functional changes. In addition, existing RCTs mostly rely on data from small sample sizes (less than 150 subjects overall, with only one exception 29 ), whilst powered trials would rather need several hundreds of participants per arm. Moreover, beyond brain-volume measures and cortical thickness, an emerging sensitive neuroimaging outcome for dietary effects appears to be white-matter integrity,34,37,43 derived from 3T-dMRI, a technique largely neglected in current RCTs.
Active interventions, where implemented, commonly rely on self-reported dietary data, prone to misclassification, and do emphasize Mediterranean-inspired patterns rather than a fully WFPBD. High-quality dietary interventions should include dietician-led counseling, standardized meal plans, and frequent adherence monitoring, with integration of metabolomic biomarkers to verify true adherence. Moreover, dietary outcomes are often examined alongside other lifestyle factors, introducing confounding bias and making it difficult to isolate the impact of diet and specific food groups alone.
As more positive correlations emerge from observational studies, which cannot establish causation and are particularly susceptible to healthy user bias, it is important to interpret these findings prudently. For example, the only large-scale studies currently available rely on UK Biobank data, a valuable epidemiological resource that is nonetheless limited by low ethnic diversity and a participant cohort that is generally healthier, wealthier, and more educated than the general population.
Finally, the potential for publication bias must also be acknowledged, as the existing literature is often weighted toward studies reporting beneficial outcomes, whereas null or unfavorable findings, such as nutrient deficiencies, may be underreported. Together, these findings underscore the need for well-powered, multi-year RCTs that combine a standardized plant-based dietary intervention, rigorous adherence verification, and advanced neuroimaging to detect subtle yet clinically meaningful effects on brain health. To achieve meaningful population-level impact, future research should be paired with coordinated efforts among clinicians, policymakers, and communities to support sustainable, person-centered lifestyle changes. Lifestyle medicine offers a scientifically supported and cost-effective strategy to reduce the growing burden of cardiometabolic and neurovascular diseases, but its benefits depend on broad, systemic adoption. 97
Review Limitations and Potential Sources of Bias
Several methodological limitations should be acknowledged in the present review. First, the exclusion of the term “Mediterranean diet” from keyword search strategy may itself introduce selection bias by omitting a substantial body of relevant dietary literature. While this approach aligns with our intent to maintain a more conservative focus on plant-based food groups, it may reduce comparability with existing nutritional epidemiology and should be interpreted as a deliberate restrictive methodological choice. Second, the full literature screening was conducted by a single reviewer; however, to mitigate potential bias, a calibration exercise was performed on 10% of retrieved titles and abstracts, and the final study set was evaluated by a second expert to support selection accuracy and completeness. Finally, the lack of a formal quality assessment across included studies constrains the strength of inferences that can be drawn, emphasizing hypothesis generation rather than causal inference.
Conclusions
To our knowledge, this is the first comprehensive review to integrate evidence on plant-based dietary patterns, alone or in combination with other lifestyle factors, in relation to neuroimaging biomarkers across diverse brain conditions. Overall, observational evidence suggests associations between plant-based dietary patterns and favorable neuroimaging outcomes, such as greater brain volumes and cortical thickness, improved white-matter integrity, lower vascular burden, better functional connectivity, and reduced amyloid accumulation; however, RCTs have widely not confirmed consistent group-level MRI effects, with only few trials showing attenuated hippocampal and cortical decline, reduced amyloid progression, or lower brain-age metrics in high-adherence subgroups. Consequently, it remains unclear to what extent these associations reflect direct dietary effects vs residual confounding, including healthy user bias. Existing literature supports an emerging theme worth investigating, rather than definitive conclusions, stressing the need for rigorously designed RCTs implementing a standardized WFPBD intervention and advanced neuroimaging techniques to clarify causality and detect subtle, longer-term brain changes that may currently go undetected.
Supplemental Material
Supplemental Material - Plant-Based Dietary Patterns and Neuroimaging Biomarkers of Brain Health: A Scoping Review of Observational and Interventional Evidence
Supplemental Material for Plant-Based Dietary Patterns and Neuroimaging Biomarkers of Brain Health: A Scoping Review of Observational and Interventional Evidence by Lydia Viviana Falsitta, Aleksandra Pikula in American Journal of Lifestyle Medicine.
Footnotes
Acknowledgments
The authors acknowledge that AI (ChatGPT-5.2) was employed solely as a final assistive tool to improve writing fluency and accuracy. R software was used as a supporting tool for formatting and inserting references. Microsoft Word and PowerPoint were used to manually create figures. All contents have been reviewed, verified, and approved by both authors.
Ethical Considerations
This is a review article, and ethics approval was not required.
Consent for Publication
This article does not contain any individual person’s data, images, or other identifiable materials.
Author Contributions
L.F. served as the main reviewer, drafted the manuscript, and integrated revisions. A.P. supervised the project, contributed to conceptualization and overall ideation, and provided critical feedback throughout all stages. Both authors reviewed and approved the final version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data sharing is not applicable to this article as no new data were created. All analyzed studies are included in the reference list.
Supplemental Material
Supplemental material for this article is available online.
