Abstract
To investigate the effects of a dietary intervention on arthritis pain and disease severity, 44 adults previously diagnosed with rheumatoid arthritis were randomly assigned to a Diet phase (vegan diet for 4 weeks, elimination of additional foods for 3 weeks, and then reintroduction of the eliminated foods individually over 9 weeks) or a Supplement (placebo) phase for 16 weeks. After a 4-week washout, they then switched to the opposite phase. The Disease Activity Score-28 (DAS28) decreased from 4.5 to 2.5 (P < .001) in the Diet phase and from 3.2 to 2.9 (P = .41) in the Supplement phase (between-group P = .01). The mean number of swollen joints decreased from 7.0 to 3.3 in the Diet phase (P = .03) and increased from 4.7 to 5 in the Supplement phase (P = .63; between-group P = .047).
In a subanalysis excluding individuals who increased medications during the study, DAS28 decreased 1.9 points in the Diet phase (P = .003) and .4 points in the Supplement phase (P = .27; between-group P = .04). In a further subanalysis limited to participants making no medication changes, DAS28 decreased 1.5 points in the Diet phase (P = .009) and .3 points in the Supplement phase (P = .40, between-group P = .11). We conclude that the dietary intervention was associated with symptomatic improvements. TRIAL REGISTRATION: Clinicaltrials.gov NCT01544101, NCT01700881, NCT03417648, and NCT03580681
“...The observed improvements in DAS28 and VAS scores suggest that a plant-based elimination diet reduces arthritis pain and disease severity....”
Introduction
Rheumatoid arthritis is a chronic autoimmune disease that affects approximately 1% of the world’s population. 1 Inflammation of the hands, wrists, knees, and other joints typically causes pain, swelling, and eventually permanent joint damage 2 and is apparently attributable to genetic and environmental factors. 3 Studies have suggested that dietary changes, often including fasting, may reduce symptoms and signs of rheumatoid arthritis, presumably due to the elimination of specific proteins, nutritional influences on inflammatory processes, or general effects of energy restriction.4-10
The present study tested the hypothesis that a plant-based dietary intervention with no energy restriction, but with a reduced range of food exposures, reduces pain and disease activity and improves quality of life in individuals with a diagnosis of rheumatoid arthritis.
Patients and Methods
Using a randomized, controlled crossover design, individuals with a prior physician’s diagnosis of rheumatoid arthritis were assigned to a Diet phase or a Supplement (placebo) phase for 16 weeks, after which they switched to the opposite condition after a 4-week washout period.
The study was approved by Ethical and Independent Review Services. All participants gave informed consent. No inducements for participation were provided. The study was registered at clinicaltrials.gov with registration numbers NCT01544101, NCT01700881, NCT03417648, and NCT03580681.
Volunteers were recruited in multiple replications through newspaper, radio, and bus advertisements in the Washington, DC, area and mailings to patients of a local rheumatology practice. Eligible participants had a previous physician’s diagnosis of rheumatoid arthritis, with daily joint pain unless on pain medication, had no changes in pain medications within the preceding 6 weeks, and were able to participate in all components of the study. Exclusion criteria included current alcohol or drug abuse; pregnancy; unstable medical or psychiatric illness; current use of a low-fat, vegan diet; and lack of English fluency.
Volunteers were initially screened by telephone. Those appearing to meet the participation criteria were invited to in-person interviews during which the study was explained in detail. Volunteers were then asked to complete the evaluations described below. Diet records and questionnaires 3
The International Physical Activity Questionnaire short form assessed recent physical activity patterns. 12
Clinical Evaluations
Height was measured, using a wall-mounted stadiometer, to the nearest .5 cm.
Body weight was measured in light, indoor clothing without shoes, to the nearest .1 kg, using a digital scale.
“Worst pain” score was indicated using a visual analog scale (VAS). Participants were asked to make a vertical mark along a 10-cm line according to the severity of their worst joint pain in the preceding 2 weeks. The scale was anchored on the left side with the words “No pain” and on the right side with “Pain as bad as it could possibly be.”
An interview and physical examination, conducted by a rheumatologist who was masked to group assignment, included the following assessments: 1. The numbers of painful, tender, and swollen joints were noted. 2. The Modified Health Assessment Questionnaire (MHAQ) was completed.
13
3. Participants were asked, “How long does it take for your stiffness to go away in the morning?” 4. “Average pain” score was indicated similarly to the “worst pain” score described above, using a 10-cm VAS, except that this score was completed by the examining physician. 5. The Disease Activity Score-28 (DAS28), a combined score, was calculated based on tender joints, swollen joints, and C-reactive protein values.
14
Laboratory Methods
C-reactive protein was measured using high-sensitivity latex immunoturbidimetry.
Erythrocyte sedimentation rate was estimated by the Westergren method.
Serum cholesterol and triacylglycerol concentrations were measured using the Olympus Cholesterol Reagent on Olympus Chemistry Analyzers. HDL-cholesterol was measured directly using the HDL-C plus 3rd generation test with the Roche direct HDL-cholesterol assay. Low-density lipoprotein cholesterol (LDL-C) concentration was estimated using the Friedewald equation. 15 Plasma triglyceride concentrations were measured using Olympus Chemistry analyzers from Quest Diagnostics.
Group Assignment
Those completing these steps were assigned to 2 groups using a computer-generated random-number table. All participants were assigned simultaneously within each study cohort, so allocation concealment was not required. One group began the Diet phase for 16 weeks, followed by the Supplement phase for 16 weeks, with a 4-week washout period between them. The other group had the 2 phases in the reverse order.
The Diet phase consisted of the elimination of specific foods, followed by their reintroduction, as follows: During weeks 1 through 4, animal products (meat, dairy products, and eggs) were eliminated and added oils were minimized. During weeks 5 through 7, the same limitations were continued, and the diet was further restricted to eliminate gluten-containing grains (wheat, barley, and rye), soy products, white potatoes, sweet potatoes, chocolate, citrus fruits, nuts and peanuts, onions, corn, tomatoes, apples, bananas, celery, peppers, coffee, alcohol, and table sugar (either separately or as an ingredient). Participants were also asked to avoid any foods to which they had a known allergy or sensitivity.
Common staple foods during this period included grains (e.g., rice, oats, quinoa, or millet), vegetables (e.g., broccoli, kale, collards, Brussels sprouts, squash, and carrots), fruit (e.g., pears, apricots, blueberries, or plums), lentils, and beans, all of which could be consumed ad libitum. After week 7, the excluded foods were reintroduced, one at a time, every 2 days. Any food that was associated with pain or other symptoms upon reintroduction was eliminated for the remainder of the Diet phase, and symptoms were allowed to return to baseline before the next food was reintroduced, which in some cases took longer than 2 days. Participants were not required to reintroduce foods they did not consume prior to the study.
No meals were provided. Participants handled their own food preparation and purchases, with guidance from the research team. There were no caloric limits. During the Diet phase, participants were counseled to use the provided vitamin B12 supplement, with the exception of weeks 5–7. If participants took additional non-prescription vitamin supplements, they were counseled to discontinue them during weeks 5–7 and reintroduce them one at a time thereafter.
During the Supplement phase, participants followed an unrestricted diet, but were asked to take a daily capsule containing clinically insignificant amounts of alpha-linolenic acid (10 mcg) and vitamin E (10 mcg), packaged in a plain white bottle, which served as a placebo. The participants were told that the supplement contained omega-3 oils and vitamin E. It was chosen because it could credibly be presented as potentially having some clinical efficacy (in higher doses, omega-3 oils and vitamin E may have efficacy in inflammatory conditions), while having no actual effect in the current study.
For both groups, alcoholic beverages were initially to be limited to 1 per day for women and 2 for men, and participants were asked to eliminate alcohol entirely starting in week 5, until alcohol was reintroduced after week 7. Participants in both groups were asked to keep their medications constant and to add no new nutritional supplements, except as recommended by their personal physicians.
Participants in the Diet phase were asked to attend weekly, one-hour group sessions for support and education on meal planning, shopping, food preparation techniques, and everyday challenges, such as dining out and healthful snacking. No weekly sessions were provided during the Supplement phase. The study did not seek to separate the effects of the diet from those of regular group support, nor did the study design aim to construct isocaloric diets during the 2 study phases.
At the mid-point and end of each 16-week study phase, the above clinical determinations, except for height, were repeated. At the end of each phase, patients were asked to rate symptom changes during that period using a 5-point Likert-style scale, ranging from “much worse” to “much better.” In the 2nd replication, this scale was replaced by the Patient’s Global Impression of Change (PGIC) question, which measures subjective pain improvement by asking participants to rate symptom change on a scale of 1–7 from “no change” to “a great deal better.” 16 This change was made in order to utilize a validated scale.
Statistical Methods
A power analysis determined the appropriate sample size to test for effects of diet at week 16 on the visual analog scale pain score. Those in the Supplement phase were expected to show minimal changes. In a 1991 study using a vegan diet and a visual analog pain scale in arthritis patients, participants had a baseline mean score of about 5.2. 4 At 16 weeks, a pain score had fallen by 2.0 cm (SD = 2.9) for the vegan group. The comparison-group pain score change was about one-tenth that of the vegan group. If the effect continued to be one-tenth, the effect sizes would be .62 for pain score (9/10 times 2.0/2.9).
We used an alpha of .05 to test for group differences, and power of .80. The sample size needed to demonstrate a significant difference for effect size of .62 was 42, assuming a crossover design in which each participant completed both phases. Accounting for expected drop-out, 50 participants were deemed to be required.
An alpha level of .05 was used for all statistical tests. The hypothesis of the effectiveness of the dietary intervention vs. supplement was examined by performing a crossover ANOVA model with between-subject and within-subject factors and interactions. Factors treatment (diet or supplement), subject, time (week), and period (1 and 2) were included in the model for participants with complete data. Evidence of a carryover effect between the 2 treatment periods was evaluated for each outcome by assessing the significance of an interaction between period and initial treatment assignment in a linear mixed model of the outcome with treatment and period as fixed effects and participant as a random effect, fit for participants with complete data. For all variables for which this interaction was not statistically significant, the magnitude and significance of treatment effect were then evaluated using a one-sample t-test (2-sided with alpha level of .05) evaluating differences between change in outcome when in the Diet phase versus change in outcome in the Supplement phase for all participants with complete data. For any outcomes with evidence of a carryover effect, estimated treatment effect for the first period of the trial alone was deemed the more appropriate.
Results
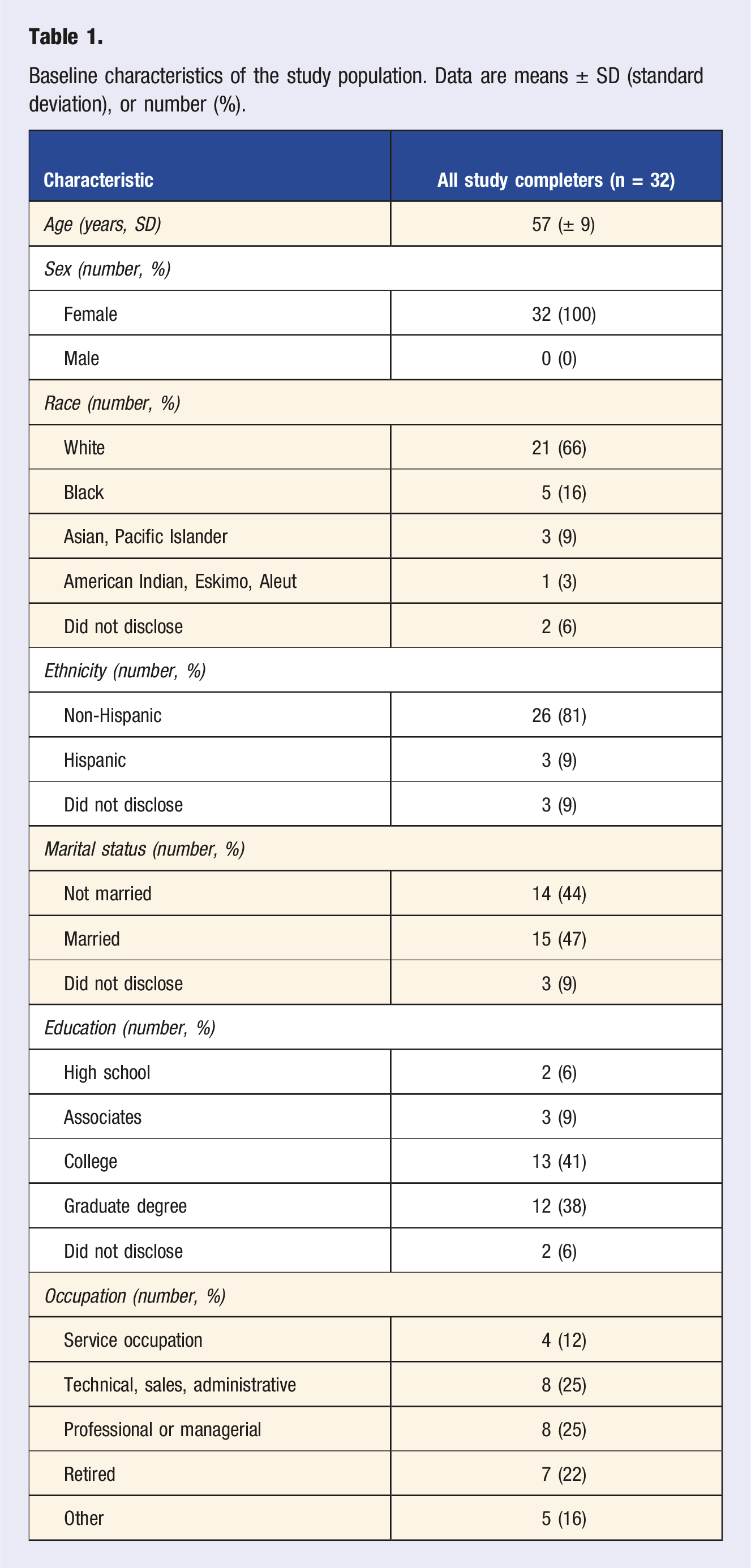
Baseline characteristics of the study population. Data are means ± SD (standard deviation), or number (%).
Diet and medication changes. Of the 22 participants who had the Diet phase first, 4 (18%) refused to fully resume their regular diets when the crossover protocol called for them to do so, and 2 of these participants completed the ensuing Supplement phase while continuing to follow a vegan or vegetarian diet. Additionally, 1 participant began the vegan diet in the first phase of the study while assigned to the Supplement phase. During the Diet phase, pain medication use was unchanged in 26 participants but was reduced in 4 participants, increased by 1, and mixed in 1. During the Supplement phase, medication use was unchanged in 30 participants, reduced in 1 participant, and increased in 1.
Diet Effects on Clinical Measures.
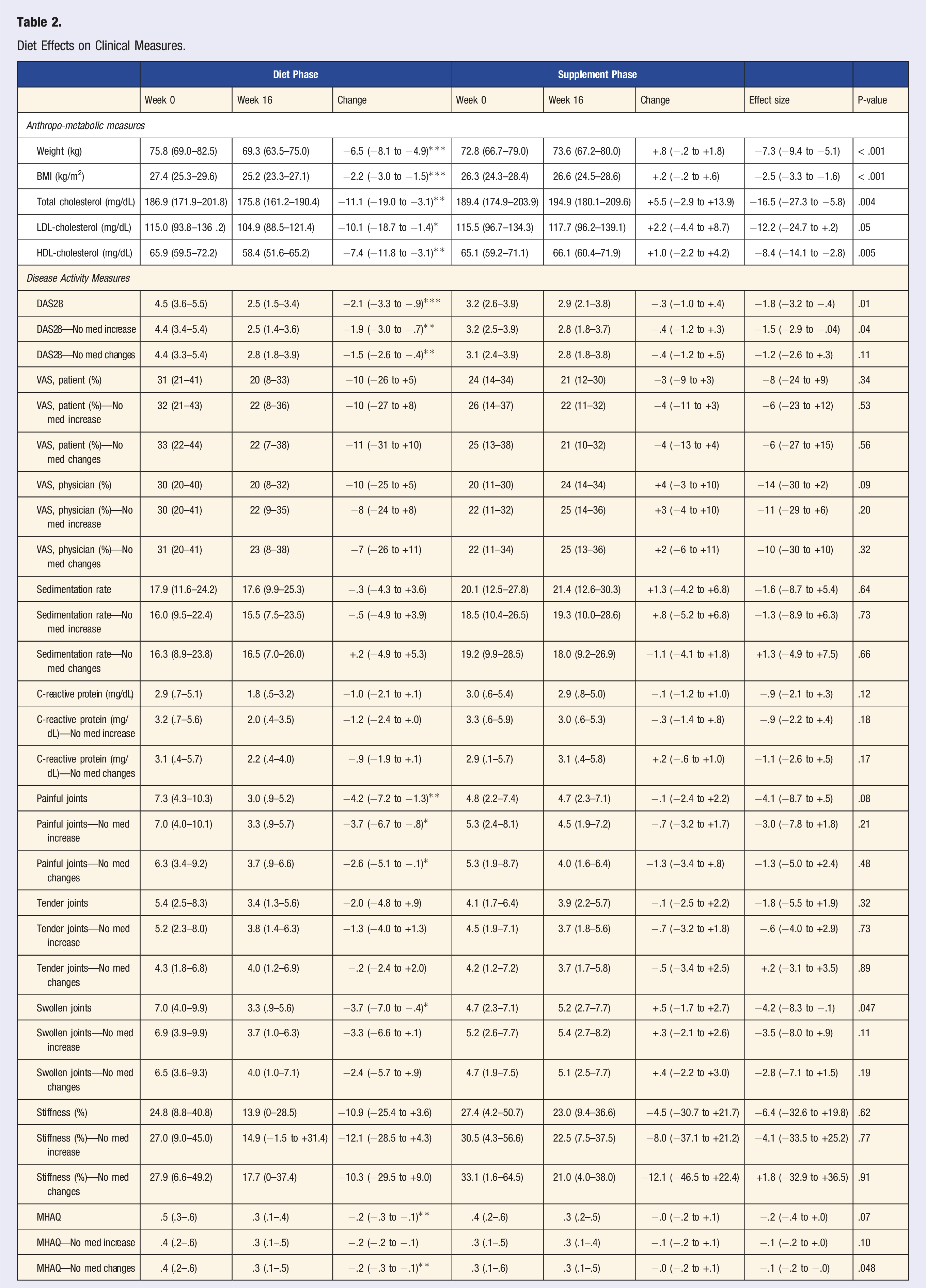
Disease activity. DAS28 score decreased in the Diet phase, compared with the Supplement phase (treatment effect, −1.8 [95% CI, −3.2 to −.4]; P = .01, Table 2), as did the number of swollen joints (treatment effect, −4.2 [95% CI, −8.3 to −.1], P = .047). The reductions in the number of painful and tender joints did not reach statistical significance (treatment effects, −4.1 [95% CI, −8.7 to +.5]; P = .08; and −1.8 [95% CI, −5.5 to +1.9]; P = .41, respectively).
For the overall study group, the inter-phase differences between the changes in the participant and physician VAS ratings were not significant (Table 2). However, in the analysis of study completers with a VAS of at least 20% at baseline (n = 16), both the participant’s and the physician’s VAS decreased significantly in the Diet phase compared with the Supplement phase (treatment effects, −27 [95% CI, −38 to −.17]; P < .001; and −30 [95% CI, −.45 to −.15]; P < .001, respectively).
Changes in outcomes and estimated treatment effects for the first and the second period of the study, comparing a low-fat vegan diet and a supplement
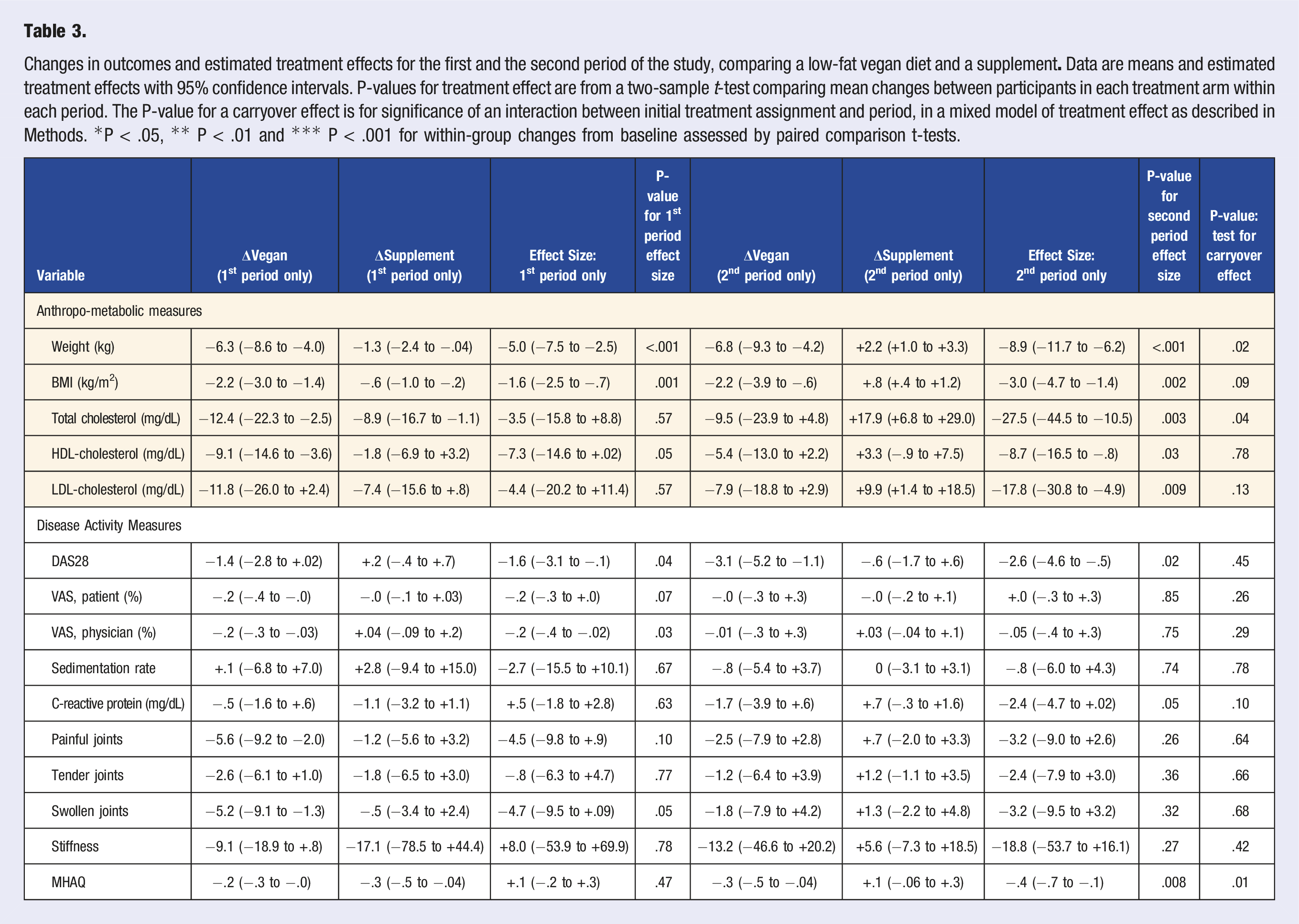
In an additional subanalysis excluding those who increased medications and also those whose symptomatic improvements led them to stop medications, DAS28 score decreased by 1.5 points in the Diet phase (P = .009), compared with a decrease of .3 points in the Supplement phase (P = .40; between-group P = .11). Other changes are reported in Table 2.
Discussion
The observed improvements in DAS28 and VAS scores suggest that a plant-based elimination diet reduces joint pain. Similar findings were noted in the analysis excluding the 2 participants who increased their medications and the additional participant with mixed medication changes. In the additional analysis that also excluded those whose symptomatic improvements led them to stop their medications, the clinical changes, while in the same direction, were no longer statistically significant.
Several studies have investigated the effects of vegan and lacto-vegetarian diets on rheumatoid arthritis. Some have included fasting. A one-year controlled trial evaluated the effect of 7–10 days of fasting followed by an individually adjusted gluten-free vegan diet for 3.5 months and then a gradual change to a lacto-vegetarian diet. The intervention group showed a significant improvement in the number of tender joints, Ritchie’s articular index, number of swollen joints, pain score, duration of morning stiffness, grip strength, erythrocyte sedimentation rate, and C-reactive protein at 1 month. The benefits were sustained after 13 months. 4
In another study, 20 rheumatoid arthritis patients fasted for 7–10 days and subsequently followed a vegan diet for 4 months. Pain and subjective functional capacity improved, but erythrocyte sedimentation rate and C-reactive protein did not. 6 A pooled analysis of 4 studies, examining the effect of fasting followed by a vegetarian diet for at least 3 months, reported a significant long-term improvement in rheumatoid arthritis symptoms. 7
In a 2001 study, Hafstrom et al examined the clinical effects of a gluten-free, vegan diet among rheumatoid arthritis patients in a randomized controlled 1-year trial. Of those who followed the diet, 40.5% showed improvement according to American College of Rheumatology criteria, compared with 4% in the control group. Immunoglobulin G (IgG) antibody and beta-lactoglobulin also decreased among diet responders. 8
In a 4-week study of a low-fat vegan diet in individuals with rheumatoid arthritis in 2002, McDougall et al. found reductions in pain, stiffness, joint swelling and tenderness, and functional limitations, along with nonsignificant reductions in C-reactive protein (2.08 to 1.74 mg/dl) and rheumatoid factor (371.5 to 333.3 IU/mL). 9
The presumed mechanisms by which such diets reduce joint symptoms relate to the removal of inflammatory elements of an omnivorous diet, the presence of anti-inflammatory constituents in a plant-based diet, and diet-induced reductions in gut permeability that may, in turn, reduce the passage of antigens into circulation.
Low-fat vegetarian and vegan diets are similar to other therapeutic diets in quantitative measures of acceptability, enjoyment, and eating behavior. 17 Additional evidence regarding sustainability comes from a University of Pittsburgh survey of young women who had tried both vegetarian and various calorie-restricted diets. The mean duration of adherence to vegetarian diets was at least 2 years, compared to only 4 months for calorie-restricted diets. 18
Strengths of the study include the fact that volunteers were not confined to a metabolic ward or otherwise restricted, so that results can be readily translated into the clinical setting, and no restriction on energy intake was imposed. The intervention was based on simple diet changes and was therefore free of medication side-effects. This study also has limitations. The participant sample was small and entirely female. While each participant had a prior physician’s diagnosis of rheumatoid arthritis, there was no further confirmation of the diagnosis, and diagnostic errors by prior physicians cannot be ruled out.
Participants who feel better sometimes wish to discontinue their medications, creating a confounding variable. Although participants were asked not to alter their use of medications, several participants nonetheless did so, in most cases because they felt less need for them, necessitating subanalyses of participants who did not change their medications. Such subanalyses may eliminate those who had the greatest effect of the intervention diet and have reduced statistical power. It is noteworthy that the growing pharmacologic armamentarium may increase the risk of such confounding in future diet studies in rheumatoid arthritis.
While a crossover design improves the statistical power of a small sample, several individuals who began the study with the Diet phase refused to fully return to their baseline diets as they began the Supplement phase. The same problem was reported during a crossover study of the effect of a plant-based diet on menstrual pain. 19 Although this phenomenon provides some reassurance regarding the efficacy and acceptability of the dietary intervention, it means that, in some individuals, the comparison is not between the intervention and the control condition, but rather between the intervention and a partial intervention.
Because participants were not confined and food was not provided, there remains a degree of uncertainty as to the extent to which participants adhered to their prescribed diets. Also, although the elimination of foods was well regimented and uniform across participants, the reintroduction of foods was individualized (foods associated with symptom recurrence were again eliminated).
In conclusion, the current study suggests that a low-fat vegan diet eliminating specific foods, without fasting and without caloric restriction, may improve joint pain. Additional studies are needed in which the diagnosis is confirmed by independent observers and medications remain stable in a larger sample. To prevent the problem of participants refusing to relinquish a dietary intervention they view as helpful at the end of the diet period, it may also be preferable to use a simple parallel design, rather than a crossover design.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Barnard is an Adjunct Professor of Medicine at the George Washington University School of Medicine. He serves without compensation as president of the Physicians Committee for Responsible Medicine and Barnard Medical Center in Washington, DC, nonprofit organizations providing educational, research, and medical services related to nutrition. He writes books and articles and gives lectures related to nutrition and health and has received royalties and honoraria from these sources. Ms. Crosby is the author of the nutrition blog Veggie Quest, and a trust for her benefit previously owned shares in Walgreen’s, 3M, and Johnson & Johnson. Dr. Kahleova, Ms. Levin, Ms. Crosby, Mr. Flores, and Dr. Holubkov received compensation from the Physicians Committee for Responsible Medicine for their work on this study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the study was supported by the Physicians Committee for Responsible Medicine.
