Abstract
Low back pain is a pervasive global public health problem. As with other chronic non-communicable diseases, dietary intervention can improve clinical outcomes and reduce health care costs. Pharmacologic and non-pharmacologic treatments are included in non-invasive treatment clinical practice guidelines. Nutrition recommendations can also be added. This review summarizes existing information on nutrition and low back pain, identifies areas for further investigation, and proposes a role for dietary recommendations in treatment and prevention from a public health and patient care perspective. Nutritional goals for decreasing low back pain include maintaining a healthy weight, following an anti-inflammatory diet, optimizing micronutrient intake, and promoting gut microbiome health. Adherence to nutrition and health recommendations is influenced by social determinants of health, including neighborhood design, structural racism, access to healthy food, socioeconomic status, and educational level. Achieving nutritional health equity must be a top priority to reduce existing health disparities. Future research on the role of nutrition in low back pain, including optimal dietary patterns, should focus on developing recommendations to be integrated into an interdisciplinary approach to low back pain.
Keywords
“Interdisciplinary rehabilitation, exercise, mind–body therapies, and other treatment modalities can be augmented with nutrition education.”
Introduction
Low back pain (LBP), defined as pain that originates below the costal margin and above the inferior gluteal folds that may or may not radiate into the lower extremities, is a global public health problem affecting all age groups. Years lived with disability increased by 54% between 1990 and 2015 due to the aging of the population and increasing prevalence in developing countries. 1 As the leading cause of disability worldwide, LBP is a pervasive and costly condition. Combined, health care costs attributable to LBP and neck pain totaled approximately $134.5 billion in 2016. 2 In 2019 alone, 39% of adults aged 18 years or older in the United States (US) experienced back pain. 3 Pathological compromise to anatomical structures, including the intervertebral discs, zygapophyseal joints, soft tissues, sacroiliac joints, and neurovascular structures, can lead to LBP. This can present as a complex intermix of nociceptive, neuropathic (radicular), and nociplastic (central nervous system-induced amplification of pain). Most cases of LBP are nonspecific (i.e., without identified anatomic pathology) and account for approximately 80%-90% of cases. 4 Imaging studies and other diagnostic tests are often subject to poor specificity, which can lead to treatment challenges. 5 Given the complexity of LBP, comprehensive, interdisciplinary care encompasses a range of treatment approaches that address biological, psychological, and social factors. This multi-modal approach is necessary and can optimize outcomes of pharmacologic, interventional, and surgical treatments. Additionally, primary and secondary prevention efforts can reduce health care costs. 5
Primary treatment of LBP includes self-care and non-pharmacological therapies, 5 such as nutritional interventions. However, evidence-based recommendations for nutrition and LBP remain limited. While current dietary recommendations for spine health are nonspecific, they include maintaining a healthy body weight and consuming a nutritious diet. 6 However, it remains unclear how to most effectively inform and advise the public about the role of nutrition in preventing and managing LBP. Targeted dietary recommendations may enhance clinical outcomes and reduce costs in both conservative and surgical spine care. Despite these potential benefits, integrating nutritional education into spine care encounters significant barriers, including clinician and patient-related obstacles.
One of the main perceived limitations for incorporating nutrition into health care is the lack of time, reimbursement, and education among health care professionals. 7 Furthermore, the lack of nutrition literacy within the public can further hinder the ability to incorporate and act on dietary recommendations, even when provided. These barriers are further amplified by the socioeconomic and cultural disparities that limit access to nutritious food. Public health and health care professionals must therefore work collaboratively to promote efforts aimed at understanding the individual, cultural, and structural barriers that affect patients and prioritizing effective interventions to address these challenges.7,8
This article aims to elucidate the current understanding of the role of nutrition in managing LBP by drawing on the existing body of literature and to identify areas for future study that will refine dietary recommendations for preventing and treating LBP optimally. The key questions to be addressed include: (1) What is known about nutrition concerning spine care? (2) What dietary advice should patients receive that may help reduce pain directly (through pain pathways) or indirectly (by addressing obesity or other factors)? (3) Are there optimal dietary plans for reducing pain in adults with spine-related pain, focusing on macronutrients and micronutrients? A broader perspective beyond these questions is necessary, as societal barriers exist and must be considered to optimize the successful implementation of recommendations.
Subtopics of interest include the role of diet in reducing inflammation, specific dietary patterns (such as Mediterranean, vegetarian, vegan, ketogenic, whole-food plant-based, paleolithic, etc.), essential macronutrients and micronutrients, and the impact of deficiencies. Additionally, obesity is examined as a risk factor for increased pain and poor outcomes, as well as barriers to optimizing nutritional status, such as food insecurity and structural/environmental obstacles. This narrative review aims to identify and summarize existing information on nutrition and LBP, pinpoint areas in need of focus and further investigation, and propose ways in which attention to nutrition can augment LBP treatment and prevention from both public health and patient care perspectives.
Search Methodology
Search Parameters
This review was conducted as a narrative review instead of a systematic review, given the scarcity and novelty of the literature regarding this topic. A comprehensive search of databases was performed utilizing combinations of search terms relevant to distinct concepts in the topic of nutrition and low back pain, as well as variant search term endings and synonyms. The Medical Subject Headings (MeSH) database was referenced for each concept. Terms were selected from the database: (diet OR diets OR dietary OR nutrition OR nutritional OR micronutrient OR micronutrients OR macronutrient OR macronutrients OR microbiome OR microbiota) AND (“low back pain” OR “low back pain” OR “lower back pain” OR “lumbar pain” OR “lumbar radiculopathy” OR “spine pain” OR “spinal pain” OR “vertebral pain” OR “musculoskeletal pain”).
This search yielded 1490 articles. Titles and abstracts were screened by two independent reviewers for relevance based on inclusion criteria of topics. Reviewers then convened to arrive at a shared consensus by identifying overlapping studies deemed relevant across reviewers. Any identified conflicts were discussed, taking into consideration the potential contribution of the article to thematic clarity and scientific rigor before including it in the review. Articles that fell outside of central themes, including those that examine obscure or less frequently studied micronutrients or niche mechanisms, were excluded. However, many of these topics were acknowledged as potential areas of future investigation. To balance comprehensiveness with clarity, subtopics were selected based on their frequency of occurrence in the literature search, while recognizing that not every potentially relevant article was included (Figure 1). Schematic for study selection for narrative review. Funnel chart of study selection for the narrative review. A leaky-funnel diagram showing the number of records at each screening step: 1,490 records identified; 110 duplicates removed; 1,380 records screened; 355 full texts retrieved; one reviewer included 137 studies in the final review. The second reviewer included 184 studies in the final review. Final consensus articles resulted in 56 after compiling overlapping articles and resolving conflicts on other articles.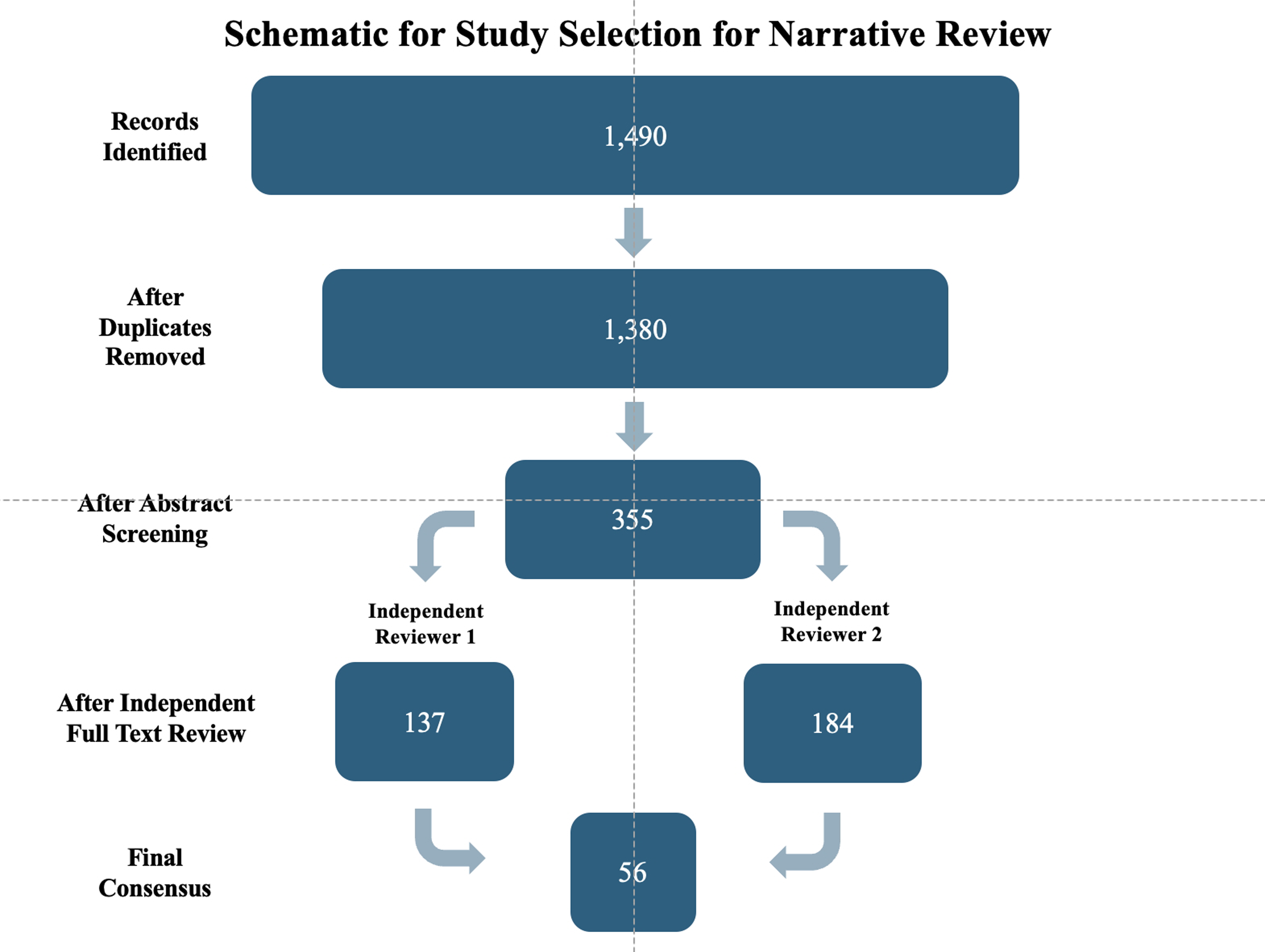
Inclusion Criteria
Studies focusing on either LBP or general musculoskeletal conditions, with a specific application to LBP, were included. This includes nonspecific LBP or more specific pathological diagnoses (e.g., disc pathology, vertebrogenic, radicular, spondylosis, etc.). Axial LBP (acute, chronic, mechanical, nonspecific), along with radicular or spinal stenosis, was also included. Studies that feature a clear nutritional variable, such as dietary patterns or specific nutrient intake or deficiencies (e.g., vitamin D, calcium, magnesium), were considered. Studies discussing obesity or body mass index (BMI) as a nutritional or metabolic factor were included as well. Articles exploring the mechanistic relationship between gut microbiota, systemic inflammation, and musculoskeletal pain were included, especially those connecting nutrient or microbiota pathways to spinal health. Study designs encompassed clinical trials (randomized controlled trials, cohort, cross-sectional, case-control) and relevant reviews. Studies were also selected based on article availability in English in either the Duke University or the University of North Carolina libraries.
Exclusion Criteria
Studies that did not directly examine the relationship of nutrition and pain, had a monetary focus on a proprietary supplement blend, and were not available in English were excluded.
Search Limitations
This study is a narrative review and not a systematic review, and therefore, is inherently limited. It does not aim to identify and systematically evaluate all existing studies comprehensively, nor does it intend to apply strict pre-defined inclusion or meta-analysis criteria, which are standard in systematic reviews. Notably, due to the nature of this study, this review is not exhaustive and may omit less commonly studied concepts that could have potential relevance. Some articles were excluded, especially if they highlighted niche or individual micronutrients or concepts with limited support or a lack of replication. As the body of evidence in the field expands and progresses, a systematic review will likely become a valuable next step and will be more feasible. That said, the purpose of this narrative review is to synthesize existing literature related to nutrition and LBP to provide a broad overview of current knowledge, highlight key areas of contention, identify gaps, and suggest future research directions and clinical recommendations.
Emerging Themes
The literature reviewed in this search produced topics of interest, including but not limited to the following: (1) mechanisms relevant to dietary intervention, (2) dietary patterns and inflammation, (3) micronutrient deficiency and polyphenol intake, (4) gut microbiome influence, and (5) the role of advanced glycation end-products. There was considerable overlap among these themes, which is expected as nutrition is a complex topic. While nutritional factors can be studied in isolation through controlled clinical trials, challenges arise when examining exposures of interest.
Mechanisms Relevant to Dietary Intervention
Being overweight or obese can lead to a pro-inflammatory state and metabolic syndrome, both of which are linked to a higher prevalence of chronic pain. Increased fat mass and BMI are also associated with reduced quality of life and functional capacity.9,10 Obesity is a recognized risk factor of LBP (including vertebral disc degeneration), 11 and evidence links higher BMI, body fat percentage, and other anthropometric measures to increased episode frequency, intensity, and level of disability. 12 This connection warrants further investigation and remains the focus of ongoing research. Additional research on lifestyle interventions for reducing obesity is warranted and may hold promise for lowering the risk of LBP and improving function and quality of life.13,14
Notably, people with obesity have a high prevalence of chronic pain. For example, Eichwald and Talbot 15 noted that approximately 40% of people with obesity also suffered from chronic pain, with most rating pain as severe. This correlation is likely multifactorial. The immune system has a role in the regulation of inflammation in acute and chronic pain via the release of inflammatory cytokines. Excess adipose tissue leads to the upregulation of the production of pro-inflammatory cytokines such as interleukin-6 (IL-6), tumor necrosis factor alpha (TNF-α), and C-reactive protein (CRP), which promote a chronic inflammatory state and can lead to the sensitization of pain pathways. 16 These elevated levels of CRP and IL-6 have also been demonstrated in individuals with LBP, suggesting a potential shared pro-inflammatory pathway that may play a contributory role in pain generation and chronicity. 16 In addition to pro-inflammatory cytokines, oxidative stress markers (e.g., reactive oxygen species, low-density lipoprotein, glutathione) contribute to pain sensitivity and acceleration of tissue degeneration.9,17 Furthermore, the burden of carrying extra weight can lead to mechanical impairment of the musculoskeletal system, particularly affecting weight-bearing joints such as the spine and knees. 9 At the same time, lifestyle factors that promote obesity, such as consuming pro-inflammatory substances in the diet and sedentary practices, can further reinforce this inflammatory process. 18
Acute pain stimuli trigger a rise in pro-inflammatory cytokines. In contrast, patients with diffuse and widespread pain show decreased levels of anti-inflammatory cytokines, indicating that both mechanisms likely contribute to chronic pain. 19 Since LBP often becomes chronic, and dietary intervention might help prevent it or improve treatment outcomes, research on the subgroup of people with chronic LBP should be a priority for nutrition studies.
Dietary Patterns
Diet quality has an impact on systemic inflammation. For example, emerging evidence suggests that the Standard American Diet (SAD), characterized by high intake of processed foods, saturated fats, refined carbohydrates, and added sugar, can exacerbate inflammation and alter pain perception in patients with chronic LBP. 18 Bodai et al. describe in their review how unhealthy lifestyle factors, including poor diet quality, accelerate the pathogenesis of chronic inflammation. Western diets in particular promote oxidative stress, gut microbiome dysbiosis, and cell injury, leading to the production of IL-6 and TNFα. By promoting sustained immune activation, this drives the development of inflammatory-driven comorbidities such as diabetes, obesity, cardiovascular disease, and cancer. This leads to a self-perpetuating cycle of chronic inflammation. 19
The link between adherence to a specific dietary pattern and LBP has been explored, but no particular diet has been identified as optimal. Several dietary plans aim to reduce chronic inflammation; these diets are generally plant-predominant and reduce or eliminate highly refined and processed foods, showing health benefits for various chronic diseases.20-22 Dietary inflammation can be assessed using several indices, including the Healthy Eating Index (HEI), the Empirical Dietary Inflammatory Index (EDI), the Dietary Inflammatory Index (DII), and the Alternate Healthy Eating Index (AHEI).23-28
Inflammation is increasingly recognized as a modern instigator of nonspecific pain and numerous chronic diseases. Given the growing evidence that diet can modulate inflammatory pathways, numerous studies have emerged to explore the role of nutritional intervention as a strategy to reduce systemic inflammation, mitigate pain, and ultimately improve long-term health outcomes. For example, in a prospective study of the Chinese population, individuals who consumed 4-5 servings of fruits, vegetables, and legumes daily had a decreased risk of all-cause mortality. As demonstrated in a study using CRP as a marker of inflammation, diets that adhere to the HEI, with higher levels of fruits, vegetables, grains, and high-quality proteins, are associated with lower levels of inflammation. 29 In a systematic review and meta-analysis evaluating the impact of nutrition interventions on chronic, non-cancer pain, nutrition interventions, including alterations to overall diet or changes in at least one nutrient, were found to affect the outcome of pain reduction significantly. 30 In an analysis of population-based data from NHANES 2009-2010, involving more than 4000 individuals with a mean age of 43, Zick and colleagues 31 examined the association between chronic spinal pain and diet quality. The HEI 2015 was calculated to measure diet quality. Individuals in the highest diet quality tertile were 24% less likely to experience spinal pain. Higher intake of fruit, whole grains, and dairy products was associated with a 20%-26% lower likelihood of having chronic spinal pain. In contrast, the higher presence of added sugars was associated with 49% increased odds of having chronic spinal pain. 31
Dietary patterns are of interest in multiple disease processes and longevity and can be further investigated in the context of LBP. The Mediterranean diet has been recommended for painful conditions, including rheumatoid arthritis (RA). However, a systematic review of human studies of patients with RA did not support the widespread recommendation of this dietary plan. 32 Still, the Mediterranean diet and other anti-inflammatory dietary plans have been shown to reduce systemic diseases prevalent in people with LBP, such as cardiovascular disease and diabetes mellitus. In a cross-sectional study involving over 7000 adults in Iran, high-protein dietary patterns were found to be inversely associated with LBP. 33 In contrast, nutrient-poor, energy-dense diets, such as the SAD, which comprises high consumption of red meats and low fiber intake, were positively associated with LBP, controlling for confounders of smoking, alcohol consumption, exercise, BMI, and waist circumference. Causality could not be established due to the study design. 33
A systematic review of nutritional factors in musculoskeletal pain found that protein, fat, and sugar intake were associated with pain intensity and threshold. 34 These SAD diets, especially in the context of comorbid obesity or diabetes, have demonstrated increased risk in the development of various cancers, including colorectal, breast, and prostate cancer. These cancers have the risk of distant metastasis, such as vertebral metastasis, which can lead to LBP. 35 Recent studies have shown an inverse relationship between dietary fiber intake and risk of breast cancer. 36 Thus, further investigation of dietary patterns is necessary and will advance the ability to inform patients. In general, nutrient-rich diets low in sugar and processed grains may be considered appropriate, with individualized recommendations made as needed.
Micronutrients
Vitamin D
The role of vitamin D in LBP has garnered interest because it possesses neuromodulating and anti-inflammatory properties. Vitamin D (1,25 (OH) 2D) promotes an anti-inflammatory immune response, which may lead to antinociceptive effects. 37 The most common dietary sources of vitamin D in the U.S. include fortified cereals and dairy products such as milk and yogurt, fatty fish, and cod liver oil. Despite the breadth of studies in the literature related to this topic, a systematic review and meta-analysis of clinical trials did not reveal that vitamin D supplementation is more effective than a placebo or other pharmacological treatments. This suggests that vitamin D supplementation for LBP should not be routinely recommended without well-designed and adequately powered studies showing some benefit. 38 Low vitamin D levels are more prevalent in individuals identifying as non-Hispanic black compared to non-Hispanic white individuals, and vitamin D deficiency may be associated with more severe LBP (especially in younger women), raising concern about health disparities that exist in chronic pain and vitamin D deficiency. 39 Given the bulk of literature regarding vitamin D and LBP, heterogeneity of findings, the prevalence of lower quality studies, and lack of clear consensus within the literature concerning the role of dietary Vitamin D in LBP, further research is essential. It is possible that optimizing levels may confer some benefit. Vitamin D levels appear to play an important role in the two-way relationship observed between sleep and pain, and supplementation may have a role in optimizing sleep and improving pain.37,40
Phytonutrients
Phytonutrients are physiologically active compounds found in plants that play a dietary role in human health. 41 The role of phytonutrients with antioxidant properties, including plant polyphenols known as flavonoids, has generated interest. 42 Plant polyphenols are naturally occurring chemicals that give plants their purple, orange, red, and blue colors, and high levels are found in many plants consumed as food, such as fruits and vegetables. 43 However, consumption of these nutrients in the US is low, especially with the shift from freshly prepared foods to processed foods. 20 This is consistent with the low intake of vegetables, fruits, and whole grains, with most dietary polyphenols coming from coffee (39.6%). 44
Several phytonutrients are especially notable concerning musculoskeletal pain conditions. Resveratrol, for example, is a phenolic micronutrient that has garnered attention for having a therapeutic effect on discogenic LBP.45-47 Resveratrol acts by reducing mRNA levels of inflammatory cytokines and disc matrix-degrading enzymes. 47 Also, the polyphenol oleocanthal in olive oil has been found to have anti-inflammatory properties that can help relieve pain. Oleocanthal works synergistically as part of the Mediterranean diet and also independently in the arachidonic acid pathway by inhibiting the enzymatic activity of cyclooxygenase.48-51 As a general guideline, patients with LBP can benefit from increasing their intake of fruits and vegetables, consuming a variety of brightly colored fruits and vegetables, and incorporating oils such as olive oil, which may help reduce inflammation and potentially impact pain. Turmeric, or curcumin, is a polyphenol with anti-inflammatory and antioxidant properties. Curcumin has demonstrated potential in various pain conditions, including joint pain from osteoarthritis, 52 postoperative pain, and neuropathic pain. 53 However, more research is needed to determine the specific benefits of individual phytonutrients for LBP.
Omega-3 Fatty Acids
Omega-3 fatty acids (O3FA) have a well-established role in the treatment of rheumatoid arthritis, with a modulating effect on the inflammatory response and prostaglandin metabolism,54,55 and may provide a protective effect in intervertebral disc degeneration. 56 Further study is needed to delineate the clinical impact of supplementation vs the dietary pattern approach. Moreover, the role of fish oil supplementation vs algae or other plant-based sources warrants attention as the combination of docosahexaenoic acid (DHA) and eicosapentaenoic acid (EPA) subtypes may be relevant, and an understanding of the optimal ratio may inform recommendations. 57 A study examining the association between LBP and plasma levels of O3FA found a reduced risk of LBP in individuals with genetically elevated plasma O3FA levels, 58 implicating a role for increasing consumption of O3FA-rich foods and possibly for supplementation.
The ratio of omega-6 fatty acids (O6FA) to O3FA has been implicated as a factor in pain, as an excess of O6FA relative to O3FA can amplify nociception and increase inflammation. 59 O6FA are pervasive in the SAD due to their presence in a variety of seed oils (including soybean, corn, cottonseed, and safflower) and processed foods, and in general, have pro-inflammatory effects. 60
In contrast, O3FAs have anti-inflammatory and antinociceptive properties. In a cross-sectional study of over 500 adults with one of two chronic pain conditions, including LBP, a higher level of circulating O6FA relative to O3FA was associated with a greater likelihood of experiencing LBP. 61 Patients can be advised to consume foods naturally rich in O3FA, such as cold water oily fish (e.g., salmon), flaxseed, and walnuts, and limit their relative intake of O3FA; in some cases, O3FA supplementation may be appropriate.
B Vitamins
Vitamin B12 may reduce pain by inhibiting cyclooxygenase enzymes and promoting neural regeneration, and may have synergistic effects with other analgesics, including non-steroidal anti-inflammatory drugs and opioids. Clinical trials support the efficacy of treatment for LBP; however, larger clinical trials are still needed. 62 In a randomized, placebo-controlled, double-blinded study of 60 adults with chronic axial or radicular LBP lasting between 6 months and 5 years, intramuscular vitamin B12 in the form of cyanocobalamin (1000 micrograms) administered daily over 2 weeks resulted in decreased pain intensity and disability scores by 87% and 82%, respectively. However, the placebo group also demonstrated pain and disability reduction to a lesser degree. 63 In another small clinical trial, 60 adults who had a diagnosis of nonspecific LBP for at least 6 months were given 500 micrograms of vitamin B12 in the form of methylcobalamin three times weekly for 2 weeks. This resulted in a 31% reduction in pain intensity and a 27% reduction in disability. 64 It should be noted that vitamin B12 is found in higher levels in animal-based foods such as meat, milk, eggs, fish, and shellfish, and that intrinsic factor-mediated gastrointestinal absorption of vitamin B12 is often decreased in older adults. 65 This suggests that fortified foods and supplementation should be considered in those following plant-based and/or vegan diets, and in some older adults. 66
Other B vitamins may also play a modulating role in LBP. The multiple mechanisms of action of combined thiamine (vitamin B1), pyridoxine (vitamin B6), and cyanocobalamin include antioxidant and anti-inflammatory effects, antinociceptive effects, potentiation of opioid analgesic effects, modulation of neurotransmitter pathways, activation of adenosine receptors, and GABA-ergic and serotonergic effects. 67 In a systematic review and meta-analysis investigating the combined effect of a non-steroidal anti-inflammatory drug (diclofenac) with thiamine (vitamin B1), pyridoxine (vitamin B6), and cyanocobalamin on LBP compared with diclofenac monotherapy, possible analgesic superiority was observed with combination therapy over monotherapy, suggesting a role in combination therapy, although evidence was not sufficient to support routine supplementation with B vitamins and NSAID therapy. 67
Gastrointestinal Microbiome
The gastrointestinal microbiome has a fundamental immune system role in inflammation. Alterations can fuel systemic inflammation, thereby affecting pain perception through the gut-brain axis.
68
A connection may exist between the degenerative process of intervertebral discs and microbiome dysbiosis. The relationship is not well understood, and there are multiple potential mechanisms by which the microbiome may contribute to disc degeneration, which could potentially lead to LBP. These include the translocation of gut bacteria into the discs, the regulation of immunity, and the diffusion of nutrients and metabolites from the gut into discs.
69
The intestinal microbiome may play a role in LBP in individuals with obesity due to increased low-grade inflammation. A study comparing the gut microbiome of 36 overweight or obese individuals with and without LBP found that those with LBP had a higher abundance of
Intestinal inflammation may be involved in the development of chronic pain, and people with food hypersensitivities and intolerances are susceptible. Identifying foods that trigger this process may provide an opportunity to modify diets for pain reduction. The relationship between chronic inflammation and pain related to gastrointestinal immune and neurologic systems can be measured via immunoglobulin G4 (IgG4), which represents antibodies from gastrointestinal exposure to foods, particularly eggs, dairy, cereals, and dried fruit. In a study of 40 adults with chronic pain, including 17 participants with LBP, a 4-week elimination diet resulted in reduced pain and improved quality of life, with pain intensity scores decreasing by more than 50%. 70 Nutritional evaluation and intervention, with a focus on optimizing gut microbiome health, can be considered for patients with LBP in primary care or specialty settings.
Advanced Glycation End Products
Advanced glycation end products (AGEs) are heterogeneous compounds derived from physiologic processes, including the reduction of sugars, amino groups, and free amines. They can form irreversibly and accumulate in tissues, leading to degenerative changes and tissue damage. 71 AGEs are produced in cooking methods such as baking, frying, and grilling. AGEs are implicated in several disease processes, including diabetes, cardiovascular disease, certain cancers, and neurodegenerative diseases, and have been linked to intradiscal inflammation. 71 In a prospective cohort study of more than 600 patients that examined the relationship between AGEs and clinical symptoms in people with LBP, quantified AGEs were found to correlate positively with higher visual analog scale ratings in LBP, lower extremity pain, and presence of numbness in patients under 50 years of age. 72 As the accumulation of AGEs in the intervertebral discs can lead to degeneration of the annulus, endplate, and nucleus pulposus, dietary interventions aimed at reducing exogenous exposure to AGEs are worthwhile areas for investigation. Investigating the effect of lowering AGE dietary load upon discogenic LBP can help inform nutritional recommendations to reduce the risk of chronic LBP.
Discussion
Nutrition, as it pertains to LBP as a distinct entity from chronic pain related to other painful conditions (including inflammatory arthropathy, osteoarthritis, headaches, neuropathic pain, and cancer pain), would benefit from further investigation. Although studying nutritional interventions for specific causes of LBP, such as discogenic LBP, can help elucidate underlying biological mechanisms, the challenge remains that most LBP cases are nonspecific. Therefore, having broad nutritional guidelines for nonspecific LBP that can be personally tailored to the individual on an as-needed basis would be a worthwhile approach, aligning with the mission of patient-centered care. Moving from health care for LBP that has historically been costly and invasive to a value-based care model, prevention and education efforts that reduce LBP-related morbidity have become a focus.
Although nutrition is an essential aspect of individual and population health, it also plays a role in pain, obesity, and other pain-related comorbidities. Many health care professionals have traditionally had limited exposure to nutrition in their training. Yet, the health care workforce is becoming increasingly informed. An increasing number of health care professionals are gaining training in integrative medicine and lifestyle medicine. Many are participating in nutrition-focused professional societies and continuing medical education, while also developing and implementing competencies for the health care workforce. 73
The American College of Physicians’ 2017 non-invasive treatment clinical practice guidelines for LBP include extensive pharmacologic and non-pharmacologic treatments; however, 74 nutritional recommendations were not included. Similarly, the World Health Organization clinical guidelines on non-surgical management of LBP do not include nutritional recommendations. 75 Interdisciplinary rehabilitation, exercise, mind–body therapies, and other treatment modalities can be augmented with nutrition education. This highlights the need for further research to inform clinical practice.
When evaluating the impact of nutrition on chronic, multifactorial conditions, such as LBP, confounders and effect modifiers must be considered in the study design and analysis. Still, dietary patterns over long periods may influence the development and progression of chronic pain. 28 Consuming a nutrient-dense diet that includes fruits, vegetables, whole grains, and legumes, while reducing highly processed foods and saturated fats, can help reduce the risk of chronic musculoskeletal pain. 76 It may be helpful to assess diet in a study prospectively and have a long follow-up period to identify the development and progression of LBP into a recurrent and/or chronic condition, allowing for statistical analysis.
As an exposure variable, diet is a complex concept. In the case of macronutrient studies, substitution patterns are relevant, as the percentage of protein, fat, or carbohydrates may fluctuate while overall energy intake remains constant. As nutrition is a universal exposure throughout everyone’s lifetime, most people will be exposed to variables of interest at some point, with confounding factors related to sociodemographic variables and health behaviors. Individual dietary components may have minor effects; therefore, studying nutritional patterns may be a more feasible approach. In studies on nutrition and LBP, examining significant exposure variability alongside small variability in potential confounders is of interest. Furthermore, the relevant exposure period may not be known and may vary depending on the nutrient or dietary pattern of interest, as well as the target population. Additionally, confounding factors can occur if nutrients with a positive effect on LBP are deficient in the diet.
Despite the challenges, continued exploration of best practices in advising patients and educating health care professionals on nutrition is essential for achieving comprehensive pain management.
The Social Determinants of Health (SDoH) concerning LBP are of interest, 77 as the conditions in which people are born, grow, live, work, and play can have a profound impact on the prevalence of non-communicable diseases and health outcomes. 78 A systematic review of the relationship between SDoH and chronic LBP found that the strongest links were related to educational and socioeconomic status. Additionally, previous research has shown correlations between LBP frequency, intensity, and disability and SDoH factors such as poverty, low income, low social class, low education, and social support deprivation. 78 Nutrition-related health outcomes tend to follow similar patterns, as obesity and poor dietary habits are often linked to food insecurity, low levels of education, and socioeconomic disadvantages.77,79 These findings highlight the need to promote and mobilize public health strategies and systems/institutional-level interventions to address these health disparities, as they fall beyond the scope of purely individual-level interventions. Thus, achieving health equity should be the goal, which Braverman et al. describe as both the ethical responsibility and the justice‐based strategy to reduce these disparities by addressing the underlying social and structural injustices in a systematic way. 80 Further research may clarify how neighborhood design, structural racism, the availability of nutrient-rich food, or the presence of food deserts contribute to the prevalence and persistence of LBP. This knowledge can offer a more detailed understanding of how to support efforts to promote positive dietary changes.
Other related topics within the broad scope of public health that warrant exploration and consideration include global food systems, sustainability, and the impact of population growth on planetary health. The impact of food production on the environment has a worldwide ecological effect, which can ultimately influence food availability and the nutrient content of food, thereby affecting the health of populations with nutritional, cultural, and socioeconomic implications. Notably, the Mediterranean Dietary Pyramid has been redesigned to reflect current nutrition and health guidelines, while incorporating a third dimension that emphasizes environmental aspects. This update highlights reducing consumption of red meat and dairy while increasing intake of locally grown plant-based foods and legumes. 81 Although this topic falls outside the scope of this review, attention to sustainability in dietary recommendations aligns with the United Nations Sustainable Development Goals, including ending hunger, promoting food security, improving nutrition, and developing sustainable agricultural practices. 82
Conclusion
In summary, general dietary recommendations that can be recommended as having benefits for LBP include the following: • Achieve and maintain a healthy body weight over time to reduce chronic inflammation. • Adhere to an anti-inflammatory dietary pattern for primary and secondary prevention of comorbidities such as cardiovascular disease and diabetes. • Consider vitamin D optimization in at-risk populations. • Promote dietary macronutrients, including antioxidants such as plant polyphenols, through adequate consumption of fruits and vegetables, as well as olive oil. • Consume foods that are rich in O3FA and limit the relative consumption of dietary O6FA. • Optimizing intestinal microbiome health may require individualized recommendations.
Nutrition may significantly impact chronic LBP, and general strategies for reducing systemic inflammation can be part of a comprehensive, whole-health approach for the overall population. More personalized nutrition plans can be adjusted based on medical comorbidities. Since most people seeking medical care for LBP do not have a specific pathoanatomic diagnosis, having broadly applicable nutritional guidelines may be useful. Patients with a precise pain diagnosis or associated systemic disease might benefit from customized dietary therapy. Given the evidence presented in this review, which highlights the impact of plant-based dietary patterns on chronic diseases and pain sensitivity, further research elucidating nutritional patterns that may help patients with chronic LBP presents itself as a valuable next step. Therefore, a prospective, staged research effort is in the works focused on evaluating whole-food plant-predominant diets in chronic LBP. Studies like this will inform scalable, nutrition-focused interventions in spine health.
Adopting a more comprehensive approach that considers a person’s overall health rather than focusing solely on traditional treatments like medication, surgery, and interventions is essential. The whole-person health perspective views health and illness as a two-way continuum influenced by lifestyle choices, including diet, stress, sleep, and physical activity. More broadly, addressing SDoH and identifying health inequities in the food system present an opportunity to improve nutritional health equity through policy, potentially leading to better population health outcomes. Factors such as education level, income, living environment, and structural racism should inform health recommendations aimed at promoting health equity. In the future, integrating nutrition into LBP care may prove to be an effective approach.
Footnotes
Acknowledgments
Sarah Cantrell, MLIS, Duke University Medical Center Library & Archives, Duke University School of Medicine
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
