Abstract
Physical activity and nutrition are important in a healthy lifestyle with potential benefits to immunity often overlooked. Infection of the upper respiratory tract, and the associated symptoms, are the most frequent presentations to general practitioners and may have significant economic and social impact. In this review, we consider the role of physical activity and nutrition in improving immunity. Evidence suggests that regular moderate activity is particularly beneficial for immune enhancement and reducing the risk of infection. We also discuss some nutritional strategies. Unfortunately, the evidence for many is weak. Avoiding nutritional deficiencies seems the most pragmatic recommendation. This can be achieved with a balanced diet. Including a variety of fruits and vegetables may help ensure adequate intake of essential nutrients with little risk of excess intake of any single nutrient. Supplementation with individual nutrients is generally not recommended. Multinutrients may be beneficial for those with a preexisting deficiency but not if normal dietary intake is sufficient. Further benefit may be gained from some supplements including probiotics, bovine colostrum, and some plant-derived products (Echinacea, black elderberry, and some polyphenols) but only in specific situations/contexts. Individuals should consider their personal needs, use caution, and avoid the indiscriminate use of supplements.
‘Immune function changes throughout the lifespan with distinct differences evident at particular phases.’
Physical activity and nutrition are important in a healthy lifestyle. Indeed, an insufficient amount of physical activity has been classified by the World Health Organization as a leading risk factor for global mortality from noncommunicable diseases.1,2 As such, engaging in an appropriate amount of regular physical activity is associated with reductions in all-cause mortality. 3 Likewise, a poor diet is associated with negative health consequences. While many of the lifestyle (eg, physical activity and diet)-related benefits are well documented, such as improvements in fitness, health, well-being, and perceived quality of life, 4 the potential benefits to immunity and infection susceptibility are of significant value yet are often overlooked. Infection of the upper respiratory tract (URT) and the associated symptoms, in the general population, are the most frequent presentations to general practitioners.5-8 This has a significant economic impact for many reasons including absence from work and health care costs. There is also a significant human and social impact, which may include increased morbidity; reduced feelings of well-being, health, and quality of life; and reduced social interaction. 7 In this review, we consider the role of physical activity and nutrition in improving immune function and URT infection (URTI) risk. The overall focus is on improving immunity and infection risk. We hope that this review is particularly useful for clinicians and practitioners not already well-versed in this area, but also with useful contemporary and practical considerations for those who are.
The Immune System
Immunity is the ability to defend against infection and disease. The immune system is composed of a variety of cells, tissues, and molecules. The immune system is generally classified into 2 main “arms”—innate (nonspecific) and adaptive (specific and acquired)—but all components work synergistically and function in a highly orchestrated manner. 9 The innate arm (eg, involving phagocytes, Natural Killer cells) can respond very quickly but lacks specificity (ie, the same response to all challenges), whereas the adaptive response (eg, involving T and B lymphocytes) has both antigen specificity and “memory” properties, but takes much longer to develop on initial exposure to an infectious agent, although the speed and magnitude of this response is increased with subsequent exposures.9-11
Immune function changes throughout the lifespan with distinct differences evident at particular phases. At birth, neither the innate nor adaptive immune systems are fully developed. Throughout the younger years, the immune system is challenged by a large number of infectious agents where the naivety of immune mechanisms leads to an increased incidence of infection in infancy compared to adulthood. 12 Each response to a pathogenic challenge results in a further modulation of the adaptive immune system. 13 Once adulthood is reached, the adaptive and innate immune systems are fully developed and generally considered to be robust. 14 A decline in cellular and humoral immune function has been noted in older adults. 15 This decline in physiological and immune function can be attributed to many factors including declining nutritional habits, lifestyle choices, and immunosenescence. 16 Immunosenescence is characterized by impaired function of immune cell populations and an “overcrowding” of the so-called immune space whereby the population of leukocyte subsets essential in responses to novel pathogens (naïve T lymphocytes) have been gradually replaced by expanded clones (effector and effector-memory T lymphocytes) that have a late-stage differentiation phenotype and limited antigenic specificity.13,15,17-19 An increased inflammatory status has also been associated with the ageing process (inflamm-aging), which may give rise to other health issues in the older population (cardiovascular disease, type 2 diabetes). 20 Immunosenescence is a primary factor contributing to increased infection susceptibility and decreased response to vaccinations in the elderly. Exercise and nutritional habits, however, are important modifiable factors that can have a significant impact on how well the immune system responds to challenge at all stages of life.10,11,21
Physical Activity Interventions
The relationship between physical activity, immunity, and susceptibility to URTI has been well investigated. A great deal of work in this area came after the J-shaped model on exercise and URTI risk was first hypothesized.21,22 This model suggests that a moderately active individual is less likely to contract an URTI compared to a sedentary individual and those engaged in activities with a strenuous workload (eg, endurance athletes) on a regular basis. Observational and experimental studies have further investigated the proposed greater resistance to pathogens with moderately active lifestyles. Animal investigations have demonstrated that brief bouts of moderate physical activity (20-30 minutes running) compared to inactivity prior to or immediately following inoculation with pathogens leads to decreased mortality and morbidity from infection.23,24 Early exercise training studies of older and obese humans also demonstrated that 12 to 15 weeks of moderate exercise (eg, 30-45 minutes at 60% to 75% heart rate reserve, 5 times per week) resulted in lower incidence or duration of URTI compared to sedentary individuals.25-27 These effects have been supported by several longitudinal studies of the wider general population (ages 18-85 years) where maintenance of a moderately active lifestyle leads to lower self-reported28-33 or laboratory confirmed34,35 URTI episodes.
The adaptation of the immune system to physical activity is dependent on the duration, intensity, and type of exertion.
36
One of the main mechanisms responsible for changes in host defense with moderate activity seems to be a greater immunosurveillance associated with moderate activity. Acute bouts of moderate-intensity activity generally induce transient improvements in the immune system.
37
For example, moderate activity (cycling) of 1 hour duration has been shown to increase the capacity of blood neutrophils (phagocytic cells) to respond to both receptor independent and receptor dependent in vitro stimulation.
38
This exercise-induced priming of neutrophil microbiocidal capacity has been suggested to be due to an increased presence of immunostimulating factors in the circulation and a greater proportion of responsive cells. Dhabhar
39
suggests that the immunopotentiation from moderate exercise is due to the bidirectional effect of stress hormones on immunity where subtle elevations are beneficial, whereas significant and sustained elevations (as seen with prolonged and/or intensive exertion) are detrimental to the host. It is these short-lived changes in cell-mediated immunity that occur during and shortly after each acute moderate exercise bout itself that are proposed to contribute to the lower risk of URTI.
40
There are various factors that mediate these relationships, one of which is the fitness status of participants. It is worthy to note that sedentary individuals, or those with low fitness or physical capacity (eg, the “new exerciser” taking up physical activity after a prolonged period of inactivity or a previously sedentary lifestyle, hereafter referred to as
Regular moderate activity in both populations (sedentary and active) is considered to promote an anti-inflammatory environment. This is an important underlying mechanism in the protection against chronic inflammatory conditions (eg, cardiovascular disease, type 2 diabetes, obesity) gained from physical activity in older populations. 11 Despite moderate activity induced decreases in resting concentrations of inflammatory mediators (eg, acute phase reactants, C-reactive protein), 41 evidence of an influence of regular moderate activity on resting leukocyte function remains equivocal however. It is likely that additional mechanisms are also responsible for the benefits of physical activity on overall immunity and infection risk. To further investigate the effect of moderate activity on cell-mediated immunity, animal and human experimental studies have monitored the production of cytokines by T lymphocytes. It is well established that intracellular pathogens trigger a (type 1) cell-mediated immune response resulting in the differentiation of naïve CD4+ and CD8+ T cells into T helper and T cytotoxic type 1 lymphocytes (Th1/Tc1) characterized by a phenotype of interferon (IFN)-γ and interleukin (IL)-2 production. 42 In contrast, extracellular pathogens induce a humoral (type 2) immune response resulting in the differentiation of naïve CD4+ and CD8+ T cells into Th2/Tc2 lymphocytes characterized by a phenotype of IL-4, IL-5, IL-10, and IL-13 production. 42 Type 1 and type 2 phenotypes are mutually inhibitory, whereby upregulation or downregulation of either leads to an imbalance in immune responses. 43 For example, it has been suggested that moderate activity can induce heightened type 1 responses and, given the importance of type 1 responses toward viral infection,44,45 contribute to decreased URTI risk. Baum et al 46 showed that an acute bout of moderate cycling caused increases in IFN-γ production by peripheral blood lymphocytes of young adults where concentrations remained elevated compared to a control group 24 hours postexercise. Longitudinal studies (0.5-4 years) of moderate activity (cycling or walking for 30 minutes each day) have to date only been investigated in the older population where an age-associated decline in type 1 lymphocytes and cytokines have been reversed.47,48 Although a type 1 response is critical for immunosurveillance and early viral clearance, it is important to note that a sustained or excessive shift toward this phenotype has been implicated in tissue damage within lung pathology. 49 Martin et al 50 have hypothesized, albeit from animal models, that moderate activity also potentiates immunoregulatory mechanisms to skew toward a Th2 phenotype and anti-inflammatory environment during responses to infection. Further research is warranted to determine the role of type 1/type 2 balance and cell-derived cytokines toward infection risk.
Longitudinal interventions (~16 weeks) also suggest that aerobic activities of a moderate intensity mediate the resting levels of other immune parameters in the circulatory and mucosal (saliva) compartments. This may include increasing (“restoring”) plasma immunoglobulin (Ig) levels or preventing further age-related decline in salivary concentrations (salivary secretory immunoglobulin A [SIgA]) in the elderly or increasing levels (eg, SIgA secretion rate) in the younger adult population.51-55
SIgA of the mucosal immune system expressed as a saliva secretion rate has previously been found to be a strong predictor of URTI incidence (risk) during exposure to intense, prolonged exertion. 56 As a reduction in incidence of URTI symptoms during 12 weeks of moderate activity has been associated with an increase in SIgA, 52 it is likely that this parameter plays an important role in the differential dose–response relationship of physical activity and infection risk. The underlying mechanisms of these changes are unclear, but evidence from animal studies suggests that moderate activity may mediate an upregulation of SIgA production by stimulating the expression of cytokines involved in the synthesis of the Ig. 57
Based on the available evidence, regular moderate (eg, 3-5 times per week, 30-45 minutes of moderate intensity) aerobic activity should be promoted as a preventive strategy against URTI due to short-term acute changes after each bout and/or a summation effect over a longer period, with the exact temporal pattern being dependent on the investigated immune parameter and nature of the exercise. Inconsistencies in study design (eg, lack of resting control group) make it difficult to identify the changes in immune measures that represent a clinically significant change and hence contribute to a reduction in rates of URTI. To what extent moderate exercise can exert an effect on immune function likely depends on the specific population. The addition of in vivo measures of immune function, which are considered more meaningful than in vitro measures, will increase understanding of the multicellular, whole system response to (antigenic or pathogenic) challenge.58,59 Further evaluation of both the acute and chronic response to moderate exercise will aid in maximizing its potential in a public health context.
Resistance exercise has been studied less comprehensively in the context of its effects on immunity. The majority of research in this area has focused on the postexercise changes in leukocyte counts. 60 Following an acute bout of resistance exercise, Natural Killer cells, and phagocytes (monocytes and neutrophils) increase in the circulation whereby the number of some of these subsets (monocytes, neutrophils) remain elevated for up to 2 hours postexercise. 61 The duration and magnitude of the postexercise leukocytosis is diminished in the older population as a consequence of age-related decline in leukocytes. 60 Differentiation of monocytes into macrophages and their infiltration into muscle tissue along with neutrophils is essential for the regeneration and repair of muscle tissue after resistance exercise–induced damage.62,63 This leukocyte redistribution has been shown to be dependent on individual components of the resistance exercise (eg, rest length, training load).64,65 The extent of this damage depends largely on the eccentric component of the exercise bout. 66 The infiltration of immune cells and release of chemokines and other inflammatory mediators associated with this muscle damage creates a localized pro-inflammatory environment. 67 Although focus of investigations with resistance exercise has not been on changes in infection risk per se, this heightened inflammatory environment has been proposed to be an effective adjuvant that may enhance responses to vaccination, 68 as discussed in the vaccination section below.
A convincing level of evidence has been developed in recent years showing that protocols of 3 to 7 repeated maximal “all out” cycling efforts (eg, using the 30-second Wingate protocol) interspersed with short recovery intervals (typically 3-5 minutes) initiate physiological adaptations for both human health and performance. 69 However, immunoendocrine responses to this new phenomenon remain underexplored. 70 Investigations, to date, have only been completed in active adults but these studies have reported decrements in salivary SIgA, with similar protocols (eg, 30-second all out efforts71,72 or 60-second all out efforts 73 ) with total exercise time as low as ~10 minutes. Despite such effects on mucosal immunity not being replicated in a more recent study with active males, decreases in neutrophil functional capacity (degranulation and oxidative burst) were observed following such activity. 70 However, a more “traditional” interval training session (20 efforts at 100% VO2max with 2-minute recovery intervals at 30%, similar to the type of session that is common for many endurance athletes) was previously shown to have little effect on salivary SIgA. The majority of these studies have also not measured URTI incidence and/or have lacked power to determine change in incidence with interval-type exertion. Further research is warranted to confirm the longer term implications on markers of immunity and clinically relevant endpoints from this type of activity across the wider population. Despite the many general health benefits that can be gained from this type of training, given the short-term immune disturbances reported in many studies (or no effect in others) in active populations, the current evidence does not favor low-volume high-intensity physical activity as an intervention to improve immunity (see Table 1).
Overview of Evidence for Likely Benefit From Physical Activity Interventions.
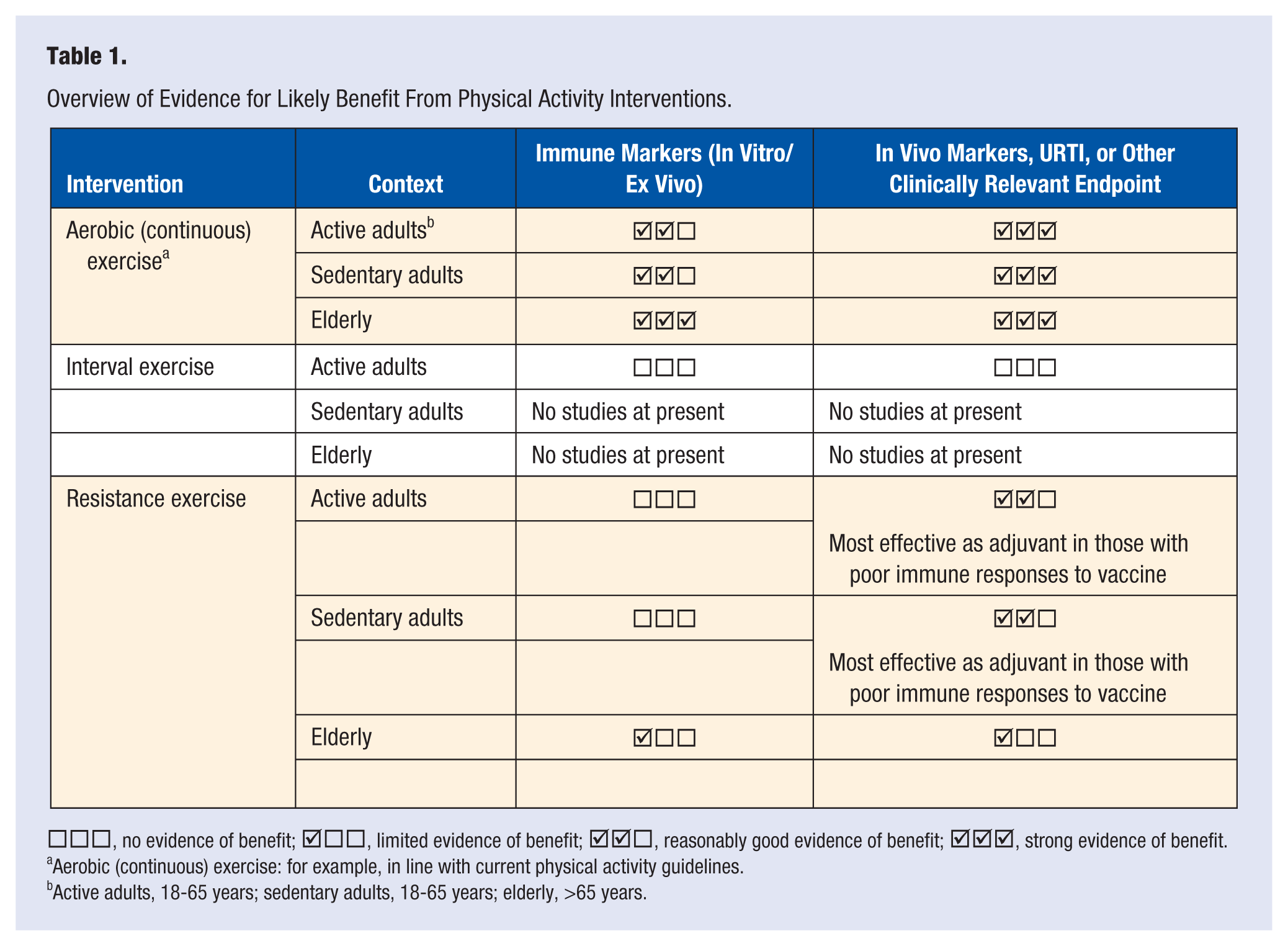
□□□, no evidence of benefit;  □□, limited evidence of benefit;
□□, limited evidence of benefit; 
 □, reasonably good evidence of benefit;
□, reasonably good evidence of benefit; 

 , strong evidence of benefit.
, strong evidence of benefit.
Aerobic (continuous) exercise: for example, in line with current physical activity guidelines.
Active adults, 18-65 years; sedentary adults, 18-65 years; elderly, >65 years.
Ageing and Immunosenescence
As discussed previously, ageing is associated with immunosenescence. It is possible that regular exercise can contribute to reducing or delaying the onset of immunosenescence. 15 Simpson 13 has suggested that exercise can increase the output of naïve T lymphocytes from the thymus and/or eliminate exhausted/senescent T lymphocytes, which can help reverse the “overcrowding” of the so-called immune space, but this requires further research. Many studies are cross-sectional,25,47,74-78 so it may be that exercise is more beneficial in preventing or limiting the decrease in the first place, which would suggest that the earlier in life the uptake of physical activity the better. Regardless, even if direct effects on immunosenescence are limited, there appears to be a number of other immune benefits that can be gained from previously sedentary individuals increasing their habitual physical activity levels, including reducing the risk of URTI or improving the response to vaccination (discussed further below).
Vaccination Studies
Older adults are less responsive to vaccination, producing lower hemagglutination-inhibition titers compared to their younger counterparts. This has been attributed to lifestyle factors and immunosenescence, particularly decreases in T lymphocyte function and the ratio of naïve–mature T lymphocytes. 79 As such, many studies have investigated the use of adjuncts (both endogenous and exogenous) to promote vaccine efficacy in these populations.
Long-term physical activity in
The effects of acute exercise on vaccine response have also been investigated. Exercise interventions include resistance exercise,68,88,89 cycling (incremental test followed by 25 minutes at 55% predicted maximum workload),90,91 treadmill exercise (40 minutes at 55% to 65% maximal heart rate), 92 and walking (45 minutes at or above 55% maximal heart rate). 93 The evidence from these studies is less compelling than that of the above-mentioned long-term exercise and physical activity studies. However, a greater proportion of these acute interventions have been undertaken in young adults where there is generally less scope for improvement than their older counterparts due to the lack of age-related decline in immunity. Indeed, there is a growing body of evidence advocating the application of eccentric resistance exercise pre-vaccination in immunocompromised populations.68,89 This may be due to the local inflammatory response (with some suggestions that IL-6 is particularly important), activation of the innate immune system, and leukocyte trafficking responses that arise as a result of exercise, potentiating the immune response to the vaccine, but the exact mechanisms, including age-related differences, are still under investigation.68,90,92,94-96
Nutritional Interventions
Nutrition has both direct and indirect effects on immune responses to challenges from foreign agents. Overnutrition, exemplified by obesity, 97 is an area of particular concern. The number of obese individuals worldwide has doubled over the past 2 decades, and prevalence is expected to continue to rise over the coming years. 2 Although the relationships of obesity with other health outcomes (eg, heart disease, diabetes) are well documented, an area that has received limited attention is the influence of obesity on host defense and infection risk. Nevertheless, excess nutrient intake has been suggested to induce immune dysregulation, whereby obesity is associated with a state of chronic low-grade inflammation and a greater risk of developing infections, particularly those of the respiratory tract, 98 compared to nonobese healthy populations. 99 Hence, increased physical activity may be beneficial in obese individuals due to improvements in weight and body composition and/or due to the anti-inflammatory effects of exercise, which may occur independent of weight loss.100,101
Undernutrition/malnutrition is characterized by an insufficient intake of total energy and macronutrients and/or deficiencies in specific micronutrients. 102 Malnutrition is one of the leading causes of immunodeficiency in the developing world. 103 Although malnutrition is rare in developed countries, deficiencies or insufficiencies of certain nutrients may occur simply as a result of poor dietary choices. It would seem, therefore, that one of the most pragmatic “interventions” (and possibly the simplest strategy in some respects) to benefit immunity and reduce infection risk is to consume a healthy and balanced diet. That is, avoiding the overconsumption of any nutrient (or total energy intake and excessive weight gain) but providing sufficient energy and micronutrients to avoid any deficiencies. Although deficiency is more likely in some groups (eg, those with eating disorders, smokers, the elderly), 104 it could be argued that a balanced diet, and the avoidance of deficiency, should be the “normal” baseline for most individuals, and so any nutritional strategies that may further enhance immunity are of particular interest.
A comprehensive evaluation of all areas of nutrition is beyond the scope of this review, and the focus is on potential benefit from additional intake (ie, beyond a normal, healthy, and balanced diet). However, a noteworthy point is that a substantial proportion of the existing research in this area has used isolated (in vitro or ex vivo) markers of immune function (eg, immune cell functions from peripheral blood samples). While these are valuable to help identify potential mechanisms, it is important to bear in mind that such measures may lack clinical relevance (biological relevance). Hence, actual infection incidence (and the associated morbidity) and/or measures of in vivo immune function are considered more “clinically relevant” as they provide information on the ability of the whole integrated system to mount a coordinated “whole body” response and defend against infection.58,59 For this reason, we have focused on a few nutritional interventions for which evidence is available on such high-value outcomes or clinically relevant endpoints. We also consider the effects of nutrition on immunity in exercising individuals, with specific consideration of the implications for
Carbohydrate
Of the many nutritional interventions that have been investigated as potential countermeasures to exercise-induced immunodepression, supplementation with carbohydrate is often cited as the most efficacious and well-supported.9,95,105 There is little doubt that carbohydrate availability has a significant impact on exercise-induced immune changes. Indeed, individuals who consume less than the recommended amount of carbohydrate are at greater risk of immune disruption during exercise (extensively reviewed elsewhere9,95). However, it could be argued that consuming sufficient carbohydrate in the diet falls within the remit of a balanced diet, and the avoidance of deficiency recommendation mentioned previously. Here we aim to consider interventions for which there is evidence of further benefit when dietary intake is already sufficient or not deficient. To this end, it is well established that the ingestion of carbohydrate (eg, 30-60 g/h) acutely before and during immunodepressive exercise effectively blunts the stress response (eg, increased cortisol, cytokine, and leukocyte redistribution responses) and perturbation in a host of immunological measures (eg, stimulated neutrophil degranulation and oxidative burst functions, lymphocyte proliferation and functions, and Natural Killer cell function).9,95,105,106 Despite the vast amount of research showing a beneficial effect of carbohydrate on a wide array of immune markers there is currently a lack of evidence showing that this actually translates to a reduced risk of URTI, 106 meaning the clinical relevance remains to be determined fully. Furthermore, the majority (of the small number) of studies that have assessed the high value (URTI risk) marker,58,59 salivary SIgA, have not found a benefit from carbohydrate supplementation. 106 On the other hand, animal models have demonstrated that increasing carbohydrate content of the diet reduces susceptibility to, and morbidity from, respiratory virus challenge (herpes simplex virus 1 [HSV-1]). 107 Bishop et al 108 have also recently shown that carbohydrate ingestion blunts the postexercise decrease in the in vitro migration of human T lymphocytes toward Human Rhinovirus–infected airway epithelial cells. Given the vast amount of research showing beneficial effects of carbohydrate (albeit predominantly in vitro/ex vivo measures), it would be surprising if this does not translate to a benefit for clinically relevant endpoints and/or URTI incidence. However, at present such evidence is not available. Hence, more research is still required, on the effectiveness of carbohydrate supplementation, with clinically relevant outcomes (something we are currently working on in our laboratory).
Micronutrients
A deficiency of certain micronutrients (eg, iron, zinc, vitamin A) can have negative effects on immune function. This has led some to believe that additional intakes of these substances will further augment immunity. Immune function is usually restored to “normal” once deficiency is corrected with additional intake above the normal (individual-specific) requirement, for most micronutrients, providing no additional benefits. The effect of micronutrient deficiencies on immunity has been reviewed extensively elsewhere103,104 and will not be discussed further here since the focus is on gaining additional benefit above what may be considered the “normal” baseline (balanced diet). However, there is some evidence that additional intake of some micronutrients (ie, more than current RDAs) has beneficial effects on the immune system and infection susceptibility in some situations (eg, individuals exposed to extremes of physiological stress or exposed to environmental extremes, although this could simply reflect a higher than “normal” requirement in these individuals, or under these circumstances), which will be considered below.
Antioxidants
Although, as a general rule for micronutrients, intakes in excess of normal requirements appear to offer little additional protection and, in some cases, might actually be detrimental, beneficial effects have been observed for some antioxidants (such as vitamins C and E). For example, high doses of antioxidant vitamins (ie, in the region of 10 × RDA) have been shown to reduce infection incidence in ultra-endurance athletes. This was originally thought to be mediated indirectly by the effects of antioxidants on minimizing stress hormone and cytokine responses
109
but this has more recently been called into question.110-112 While supplementing with high doses of antioxidants is capable of blunting the cortisol and IL-6 responses to a stressor,
113
recent research has demonstrated that, in well-nourished healthy individuals, this has little effect on most aspects of immunity and URTI incidence114,115 (although small effects of vitamin C on URTI duration have sometimes been observed
116
). So while there may be some benefit for individuals exposed to extreme physical and environmental stressors,
116
this is unlikely to be of significant benefit for the average individual undertaking regular physical activity to meet general health guidelines. Such nutritional practices may appear attractive for
Vitamin D
There has been a great deal of recent interest in the role of vitamin D in immunity and infection risk. Vitamin D is a secosteroid hormone that interacts with more than 200 genes via nuclear receptors, which are present in numerous tissues of the body. It therefore has been implicated to have a wide range of actions and physiological functions beyond the “classical” role in bone health. There is now considerable evidence that suboptimal vitamin D status is associated with compromised immunity and increased illness risk.121-123 Indeed, a large percentage of the population in many developed countries, including athletic populations, are at risk of vitamin D insufficiency and deficiency.124,125 Vitamin D can be obtained from dietary sources, but it is primarily synthesized endogenously when the skin is exposed to solar ultraviolet B radiation. However, this synthesis is often minimal or negligible during winter months in some locations (eg, from October to April at the latitude of 52°N and from November to February at the latitude of 42°N), due to the lower solar zenith angle, meaning deficiency is more likely. 126 It is worthy of note, however, that while some studies have observed associations between vitamin D status and URTI risk, 127 this is not a universal finding. Some of the discrepancies may be related to baseline status, so it may be, as is the case for many micronutrients, that benefit is only likely in those who are deficient to begin with. Once deficiency is corrected there is generally no further benefit to be gained from additional intake. Hence, vitamin D supplementation may be unnecessary, and unlikely to be of benefit unless an individual has poor vitamin D status (which should be confirmed before recommending supplementation). It is also noteworthy that all studies to date have been cross-sectional or observational in nature rather than intervention studies (ie, causality has not been determined). It is of course possible, therefore, that low vitamin D status is in fact a marker of compromised immune defense rather than the cause. Hence, further research is required, specifically supplementation trials, 128 in this area.
Looking Beyond Single Nutrients (Mixed Nutrients From Supplemental or Food Sources)
Deficiency in a single nutrient is rare and combinations of nutrients (eg, multivitamins and mineral supplements) may be particularly effective in those at risk of dietary deficiency. 129 An advantage to this approach is it may be considered lower risk (in terms of potential over intake of specific nutrients) than single nutrient supplements, which are often available in much higher doses (and high doses of single nutrients may interfere with the absorption of others). Robson et al 112 observed an enhancement of neutrophil functional capacity in a group of 12 healthy young (mean age = 30 years) male and female athletes supplemented with a multivitamin and mineral supplement daily for 3 weeks. However, findings such as this may simply reflect the correction of mild deficiency or be indicative of the increased requirement in athletic/training individuals, meaning that the existing diet was suboptimal and do not necessarily prove that “additional” intakes are beneficial. Furthermore, a compelling argument can be made for obtaining nutrients from food rather than supplemental sources. For example, this may provide a natural mixture of nutrients meeting the aim of a multinutrient supplement. It may also provide nutrients that work together in synergy and have better bioavailability than isolated single nutrients or synthetic sources. In support of this, Nieman et al 130 observed a lower URTI incidence in individuals with a high intake of fruit (3 or more portions per day) compared to those who consumed 2 or less portions per day in their normal diet. It would seem, therefore, that a potentially beneficial strategy is to eat more fruit and vegetables as part of a healthy and balanced diet.
Immune-Enhancing Foods and Supplements
The “immune” or “cold and flu” section of many health food shops are often littered with a vast array of supplements and products with claims that they enhance immunity. Unfortunately, the evidence for many of these is scant, if existent at all. Here, we have discussed some for which there is at least a reasonable body of evidence for general benefit (and in situations of additional physical strain) but this is not, however, intended to be an exhaustive list.
Dairy-Derived Supplements or Functional Foods
Bovine colostrum
Bovine colostrum (“early milk”) is the initial milk produced by cows, usually obtained within the first 48 hours postpartum. Like “normal” milk, it contains a rich source of nutrition, both in terms of macronutrients and micronutrients, but it is also abundant in bioactive components including immune, growth, and antimicrobial factors.131,132 Bovine colostrum has received considerable attention within
Probiotics
Probiotics are defined as live microorganisms that, when ingested in adequate quantities, reach the intestine in sufficient numbers (alive) to exert a health benefit to the host.151,152 Before further discussion of probiotics, it is important to first note that many different strains of bacteria exist and may be in commercial products claiming to be probiotics. However, only certain strains are efficacious, and only if consumed in sufficient quantity will they have a beneficial effect on immunity and infection risk (discussion of the various strains and quantities needed is beyond the scope of this review, and although we refer to some specific strains when discussing studies below, this is by no means an exhaustive list of potentially efficacious strains). Supplementation with appropriate probiotics is able to modulate the intestinal microbial flora and improve microbial balance. As the gut microbial flora is able to interact with intestinal epithelial cells and a number of specific immune cells, this can exert beneficial effects on the skin and upper respiratory tract, modulating mucosal and systemic immunity and influencing intestinal permeability and barrier functions when subjected to stressors.153-155 It may also contribute to improving nutritional and microbial balance within the intestinal tract
155
and hence improve immune function. Probiotics are most commonly consumed via fermented foods such as fermented milk or dairy products (eg, yogurt). There is good evidence for beneficial effects of probiotics supplementation in individuals with compromised immune status and thus increased infection risk, for example, reducing gastrointestinal infection risk (from some enteric pathogens) and antibiotic-associated diarrhea and improving outcome in at-risk populations (eg, intensive care patients).
155
There are, however, few well-conducted studies in otherwise healthy individuals. In one study with healthy adults, De Vrese et al
152
investigated the effect of daily probiotics supplementation (5 × 107 colony forming units [CFU], including
In a recent study, Cox et al
158
compared self-reported URT illness symptoms in 20 male athletes during the winter months, taking either a daily probiotics supplement (1.3 × 1010 CFU
Plant-Derived and Herbal Supplements
Echinacea
Surprisingly, given the mainstream and consumer popularity of
Polyphenols and Other Plant-Derived Substances
The consumption of other plant-derived compounds (eg, polyphenols) is now becoming more popular. As well as being potent antioxidants, many of these substances are suggested to have immune-modulatory properties (separate from, and perhaps independent of, their antioxidant properties). Many also possess direct antimicrobial and antiviral properties in vitro. However, many of the benefits have been shown in animal studies and/or in vitro studies, usually with extremely high doses. There is limited research on the efficacy of oral supplementation in humans. The bioflavonoid quercetin has received a considerable amount of attention in the sport and exercise immunology field. This is a type of polyphenol found naturally in many foods including onions, some fruits and berries, green leafy vegetables, and teas. Typical dietary intake is less than 50 mg per day but intakes of up to 300 mg are possible when the diet is abundant in such foods. Nieman et al
164
showed daily quercetin supplementation (1000 mg per day) for 3 weeks before and 2 weeks after a 3-day period of intensified training reduced URTI incidence by a remarkable amount (5% incidence in quercetin vs 45% in placebo) in the following 2 weeks. However, the mechanisms responsible for these effects are unclear as there were no differences from placebo in a comprehensive battery of immune function markers in response to the training (although when consumed with other polyphenols bioavailability, and immune functions, are seen to be improved). Quercetin does have potent antioxidant properties, but unlike some of the other antioxidants mentioned above, it does not seem to be associated with a blunting of training responses and physiological adaptations. On the contrary, some evidence has suggested small, but potentially worthwhile, beneficial effects in enhancing and potentiating the training response in previously untrained individuals.
165
Taken together, this could suggest some benefit for
It has been suggested that regular consumption of foods high in polyphenols confer immune and URTI risk reduction benefits. Zakay-Rones et al
166
conducted a double-blind placebo controlled study with a commercial product containing black elderberry (
Nutritional Interventions in Ageing and Immunosenescence
Although age-related immune modulation is controlled by genetic factors, it is amenable to influence through lifestyle choices such as nutrition.
16
The suboptimal status of macronutrients and micronutrients often found in the older population is suggested to exacerbate immunosenescence.172-174 The majority of nutritional interventions to date have focused on use of dietary lipids (n-3, n-6 polyunsaturated fatty acids) and micronutrients such as vitamin E, carotenoids, and zinc, all of which are essential in mounting immune responses.174-176 However, evidence for the effects of these nutritional supplementation strategies on immunosenescence is equivocal.
177
The inconsistent effects in the literature with micronutrient supplementation are most likely due to variance in dosage between studies and/or the initial nutritional status of individuals.
173
Although some studies have shown single and/or multivitamin or mineral supplementation can reverse age-related declines in immune parameters, the risks associated with oversupplementation must be recognized whereby an approach to “right dose” rather than “megadose” is required.
178
As highlighted for the younger adult population, it is unlikely that micronutrient supplementation beyond the level required to correct for dietary insufficiency will provide any further benefit. Furthermore, the potential benefits must also be weighed against potential detrimental effects. For example, the older population is considered to be more sensitive to the immunological effects of n-3 polyunsaturated fatty acids, which are advantageous in ameliorating inflammation, but the downregulation of innate and adaptive immunity may increase susceptibility to infection in this group.
171
For these reasons, the best recommendation may be to ensure the diet is adequate in order to avoid deficiencies whereby a whole diet approach is likely to be feasible and a more efficacious strategy. The research of other (advanced) nutritional supplements (eg, probiotics) within the older population is continually developing. The use of certain strains of probiotics (eg,
Overview of Evidence for Likely Benefit From Nutritional Interventions a .
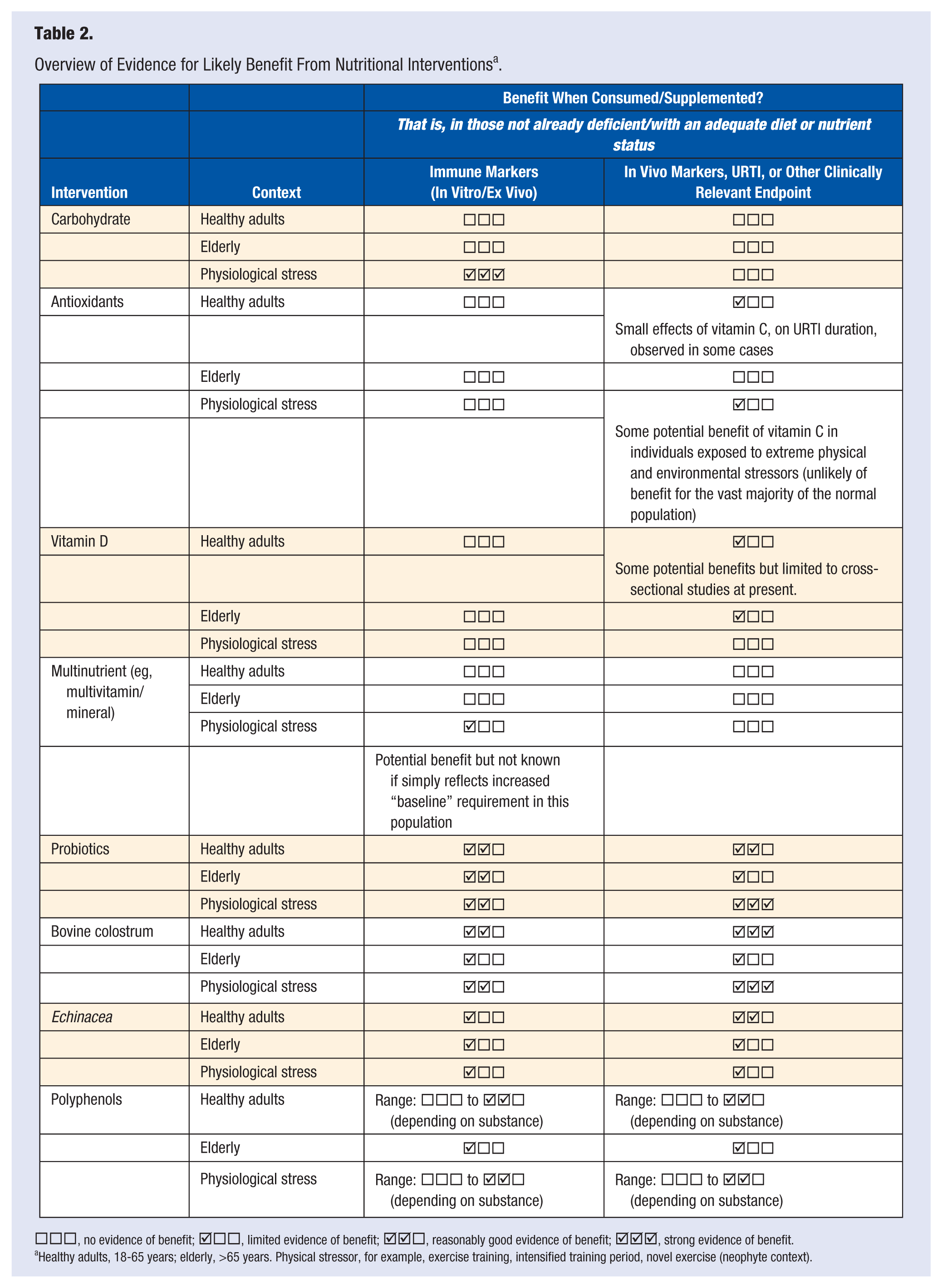
□□□, no evidence of benefit;  □□, limited evidence of benefit;
□□, limited evidence of benefit; 
 □, reasonably good evidence of benefit;
□, reasonably good evidence of benefit; 

 , strong evidence of benefit.
, strong evidence of benefit.
Healthy adults, 18-65 years; elderly, >65 years. Physical stressor, for example, exercise training, intensified training period, novel exercise (neophyte context).
Summary
There are many lifestyle factors that can influence immunity and the risk of infection. Exercise and nutritional habits are important modifiable factors that can have a significant impact on how well the immune system responds to challenge and hence the risk of infection. In this review, we have highlighted 4 main key points (take home messages) on interventions to improve immunity and reduce infection risk as follows:
In summary, both exercise and nutrition can influence immunity and resistance to infection across a wide spectrum of the population. As a result of immunosenescence, the older population (eg, aged over 65 years) are one group for whom interventions to improve immunity are particularly worthwhile. Both physical activity and good nutrition may help minimize the negative consequences of this age-related immune dysfunction although this evidence remains preliminary at present (apart from improved vaccine efficacy, which seems most promising in these “immune compromised” populations).
Footnotes
Acknowledgements
GD was PhD supervisor/advisor to AWJ (from 2010 to 2014), whose PhD was supported by a Knowledge Economy Skills Scholarship, which was a collaborative project with an external partner, The Golden Dairy Ltd (supplier of bovine colostrum for Neovite/Colostrum UK Ltd). This project was part-funded by the European Social Fund through the European Union’s Convergence Programme (West Wales and the Valleys) and administered by the Welsh Government. Some of the work discussed in this review (Jones et al 138 ) in the dairy section came from work undertaken by AWJ during his PhD. GD has also received research funding from Neovite/Colostrum UK Ltd for some of the work mentioned in the same section (Davison and Diment 139 ). GD has also received research funding, from the Nestec Research Centre, Switzerland, for some of the work mentioned in the polyphenols section (Davison et al 170 ). All authors declare no conflict of interest.
