Abstract
Several dietary and non-dietary factors and genetic predispositions may play an important role in gastric carcinogenesis. The association between fruit and vegetable with gastric cancer (GC) is still unclear. This study aimed to investigate the association between fruit and vegetable intake and GC risk. This case-control study enrolled 173 patients with GC and 313 GC-free controls between March 2015 and August 2018. Dietary data was collected separately from each patient using an interview-based food frequency questionnaire. The results showed a significant (P ≤ .05) inverse association between grape and melon consumption and GC development. In contrast, the consumption of 100% orange juice, orange, pear, and strawberry increased significantly (P ≤ .05) the risk of GC. However, mixed vegetables, vegetables fried with olive oil, and onion consumption showed a significant (P ≤ .05) protective effect against GC. No association was detected for the other examined fruits and vegetables (dates, watermelon, peach, green beans, peas, carrot, and stuffed vegetables). Although many fruits and vegetables may reduce the risk of GC, others may increase it. The method of vegetable and fruit preparation may affect the nutritive value. More studies are warranted to confirm our findings.
“The present study findings demonstrated that orange consumption in the third tertile might have a protective effect against gastric cancer.”
Introduction
Gastric cancer (GC) is one of the most rapidly lethal malignancies. It has been the fifth most common cancer worldwide, with 1,089,103 new cases in 2020. According to Bosman et al. GC is the second-highest death rate among malignancies accounting for 769,000 deaths in 2020 1 . Despite a decrease in occurrence in some parts of the world, GC continues to be a major clinical problem since most cases are detected late, with limited prediction and scarce treatment options. 2 The prevalence of GC all over the world varies greatly from country to country and area to region. For example, in Japan, it is projected to be 12 times greater than in India and Iran, and seven times more common than in Iraq. 2 In 2017, the Jordanian Ministry of Health reported that gastric cancer has ranked ninth among the most diagnosed cancers with 3.2% in males and 1.8% in females of the total cancer cases in Jordan. 3 GC is a disease in which malignant (cancer) cells form in the lining of the stomach. The mucosa layer that produces the gastric acids in the stomach is the area where most cancers begin. 2
GC is a complex disease with several causes. 4 Many risk factors may affect the prevalence of GC including gender, age, sex, body weight, body fat, smoking, alcohol consumption, medication, physical activity, family history, and genetics. 4 Additionally, some medical conditions may contribute to an increased risk in the development of GC like H. pylori infection. 5
Dietary pattern is a modifiable risk factor that could be improved to reduce the likelihood of developing GC. Various studies observed an association between one’s high intake of processed meat, fat, sugar, salty, fried food, and dairy products and risk of GC.6,7 In addition, low consumption of fruit and vegetable may increase the risk of GC.6,7 Fruits and vegetables are considered one of the well-studied dietary protective factors; they are rich in anticarcinogenic components such as fiber, vitamins, carotenoids, and phytochemicals. 7 These nutrients and bioactive chemicals have antioxidant and antiproliferative properties and the ability to modify steroid hormone concentrations and metabolism. Additionally, fruits and vegetables enhance the immune system and DNA synthesis and methylation. 8 Fruit and non-starchy vegetable consumption are related to a lower risk of GC. 9 Flavonoids have a link with a 20% reduction in the probability of getting GC in women. 10 Since fruits and vegetables contain high levels of antioxidants, they may have increased the ability to protect against the development of GC. 9
An increased prevalence of GC has been reported in the Middle East with a high mortality rate worldwide due to delayed diagnosis. Additionally, poor quality of the diet overall with specifically low consumption of fruits and vegetables has been linked to the increased risk of GC. Thus, the aim of our study is to investigate the association between fruit and vegetable consumption and GC risk in Jordan that witnessed a remarkable nutrition transition towards the Western dietary pattern.
Materials and Methods
Participants’ Enrollment
A case-control study included 173 GC patients and 313 population-based controls was conducted between March 2015 and August 2018. All cases were confirmed by an upper endoscopy with biopsy followed by pathological and radiological confirmation to evaluate for respectability, while controls were included based on thorough medical history, physical examination, and noninvasive screening methods. The endoscopic biopsy followed by pathologic confirmation was within the past 6 months of the enrollment in the study. Data regarding demographics, smoking, medical history, physical activity, and nutrient intake was collected using reliable interview-based questionnaires. Information on fruit and vegetable intake was collected from the participants using a reliable and valid Arabic food frequency questionnaire (FFQ). The study protocol was approved by the Institutional Review Board Ethics Committee of the King Hussein Cancer Center (IRB No. 15 KHCC 03, Amman, Jordan), King Abdullah University Hospital, Jordan University Hospital, and Al-Bashir Hospital. Prior to participation in this study, all participants were asked to give a signed written informed consent. The study was conducted based on the Declaration of Helsinki.
Inclusion criteria included participants aged 18 years or older at enrollment, able to communicate verbally, and absence of any chronic diseases that require dietary modifications, such as kidney disease, liver disease, and celiac disease. For the GC cases, patients must have been diagnosed within the last six months. Exclusion criteria included individuals who were critically ill, hospitalized, or unable to communicate verbally. Informed consent was obtained from all participants before enrollment.
Anthropometric measurements
Participants’ current and pre-diagnosis body weight and height were measured using standardized techniques and calibrated tools by a trained dietician. 11 Body mass index (BMI) was computed as the ratio of weight (kg) to height squared (m), 11 and classified according to World Health Organization guidelines. 12 However, the Pre-diagnosis body weight before diagnosis was self-reported from the cases and controls.
Physical Activity Assessment
The validated physical activity recall (PAR) questionnaire that was originally developed by Sallis et al. was used in this study to evaluate the level of physical activity for each participant. 13 Participants were asked to recall the usual time spent practicing physical activity over a period of a week. The total metabolic equivalent task (MET) minutes per week was obtained by summing the METs. 13
Dietary Assessment
The assessment of dietary intake was achieved using a validated Arabic Food Frequency Questionnaire (FFQ). 14 The FFQ addressed the diet of the past 12 months which would reflect the seasonal variations of fruits and vegetables. Seventeen types of vegetables and 14 main fruits were selected in this study. Fruits and vegetables were compiled according to cultural influences (e.g., tomatoes and sweet peppers were categorized as vegetables, not fruits). Participants were asked to estimate the average frequency of consumption of a standardized serving of each food item consumed during the previous year. The frequency of consumption was classified into nine different categories (≤1 per month, 2-3 times per month, 1-2 times per week, 3-4 times per week, 5-6 times per week, 1 per day, 2-3 times per day, 4-5 times per day, or 6 times per day). To assist participants in precisely estimating the consumed portion size, food models and standard measuring tools were used. Knowing the frequency of consumption and the specified serving size for each food item, the average daily amount of each food item consumed by each participant was then calculated.
Statistical Analyses
IBM SPSS Statistics for Windows version 28.0 (IBM, Armonk, New York) was used to conduct all statistical analyses. The normality of continuous variables was assessed by the Shapiro test. The Chi-square test was used to detect the differences among categorical variables while the t-test will be used for continuous ones. Non-normally distributed variables (vegetables and fruit intake) were presented as middle (first-third) tertiles. Mann–Whitney U test was used to find differences in intakes of vegetables and fruits between GC cases and controls. Multinomial logistic regression was performed to calculate odds ratios (ORs) and confidence intervals (CIs). Before performing any statistical analysis, several factors were identified as potential confounders including age, sex, pre-diagnosis BMI, physical activity level, total energy intake, education level, smoking, marital status, and family history of participants. The level of significance was set at P-value ≤.05.
Results
Based on the number of servings of fruits and vegetables consumed on a daily basis, Tables 1 and 2 show the characteristics of the GC cases and the controls, respectively. There was a significant difference in the pre-diagnosis BMI for the case group who consumed different servings of vegetables with a P-value = .024 (Table 1). Moreover, significant differences were detected in the family history of GC (P-value = .003) and the number of daily consumed snacks (P-value = .008) in GC patients who consumed vegetables. Regarding fruit consumption among the GC cases, the results demonstrated significant differences in tobacco use (P-value = .035), educational level (P- value = .013), and daily water intake (P-value = .008). On the other hand, significant differences were detected in current weight (P-value = .002), usual body weight (P-value = .006), tobacco use, and BMI (P- value = .003) among the controls who consumed vegetables in different servings numbers. In addition, the results showed a significant difference in marital status, number of meals eaten outside the home and daily water intake (P value <.05). Furthermore, skipped meals and the number of snacks consumed daily were significantly different among the control group who consumed different servings of fruits with P-value = .042 and P-value = .001, respectively. Sociodemographic Characteristics of the Case Group Based on Fruit and Vegetable Intake.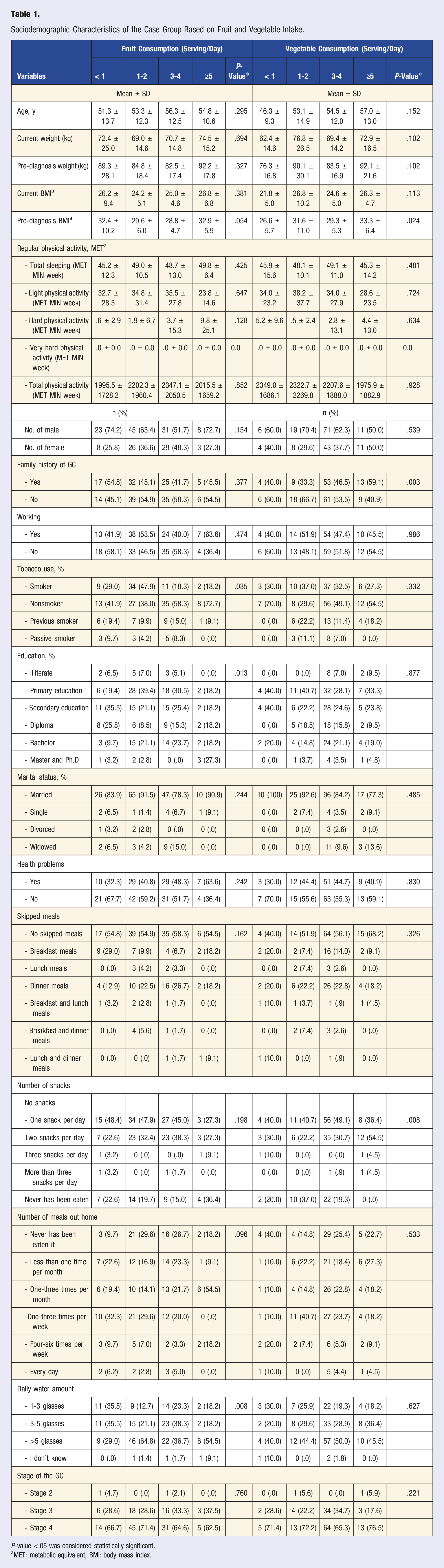
P-value <.05 was considered statistically significant.
aMET: metabolic equivalent, BMI: body mass index.
Sociodemographic Characteristics of the Control Group Based on Fruit and Vegetable Intake.
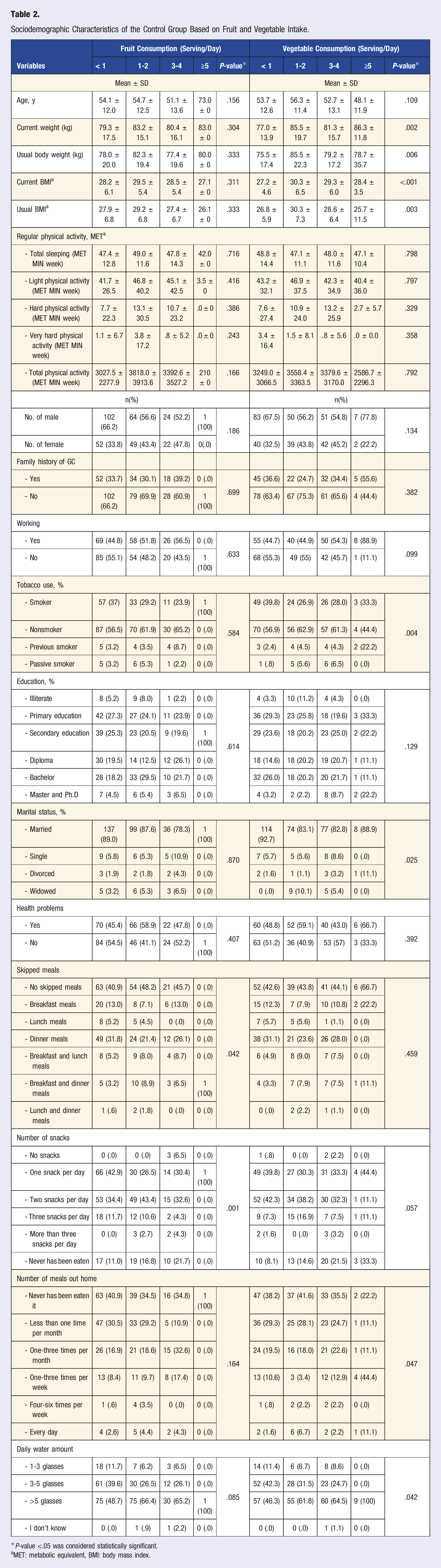
*P-value <.05 was considered statistically significant.
aMET: metabolic equivalent, BMI: body mass index.
Fruit and Vegetable Consumption Among the GC Cancers and Controls.
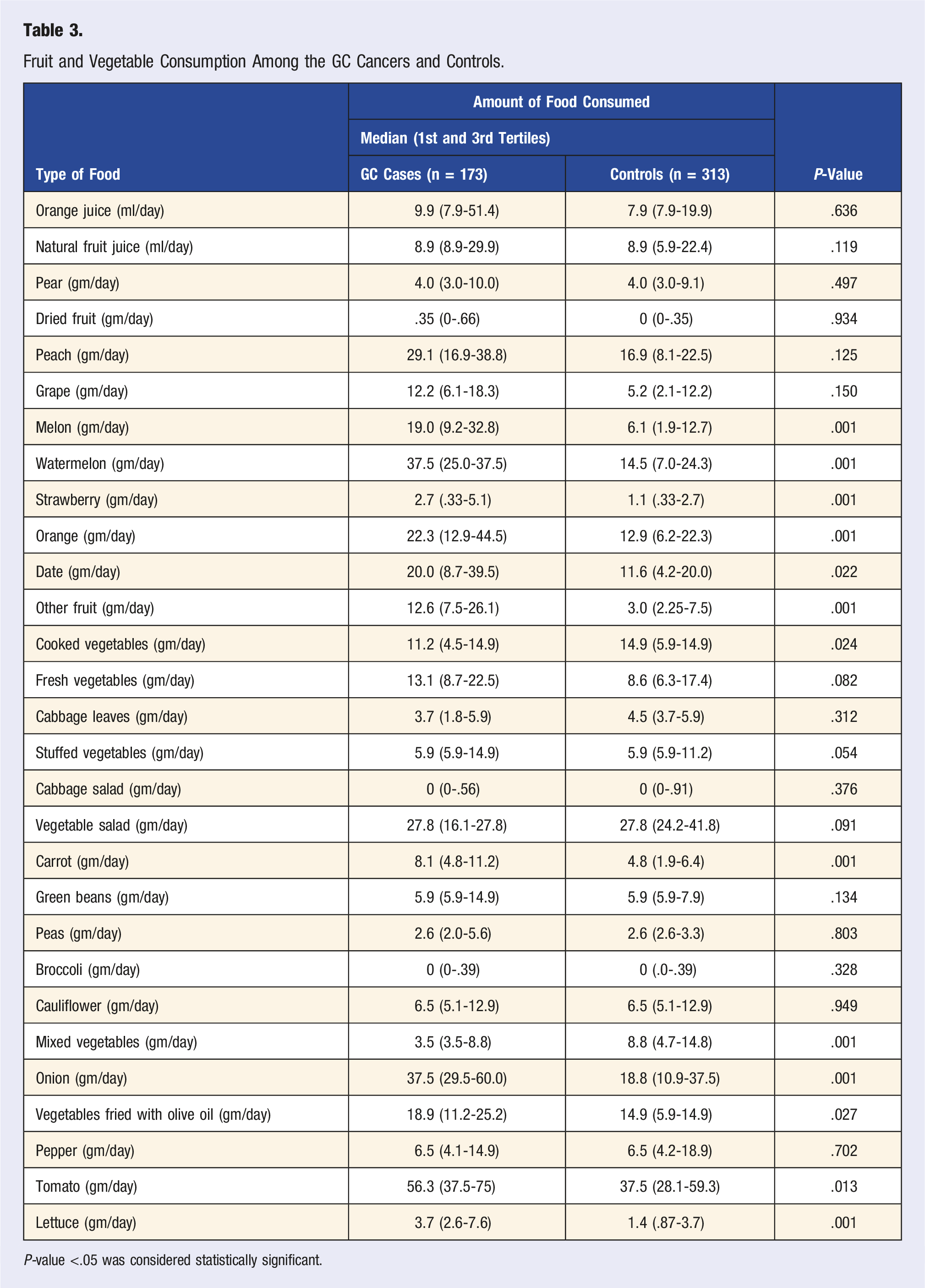
P-value <.05 was considered statistically significant.
Association Between Fruit Intake and GC Risk Among GC Cases and Controls.
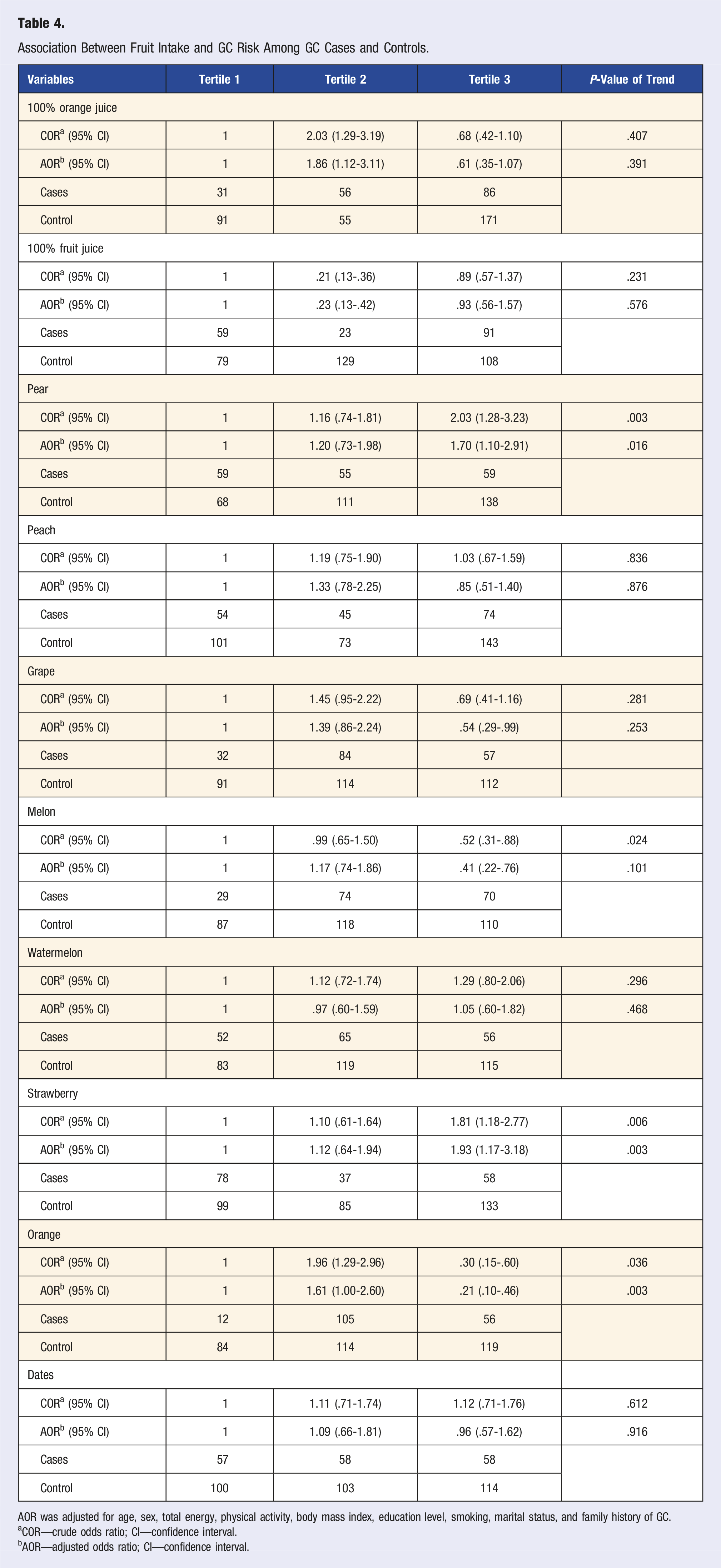
AOR was adjusted for age, sex, total energy, physical activity, body mass index, education level, smoking, marital status, and family history of GC.
aCOR—crude odds ratio; CI—confidence interval.
bAOR—adjusted odds ratio; CI—confidence interval.
Association Between Vegetable Intake and GC Risk Among GC Cases and Controls.
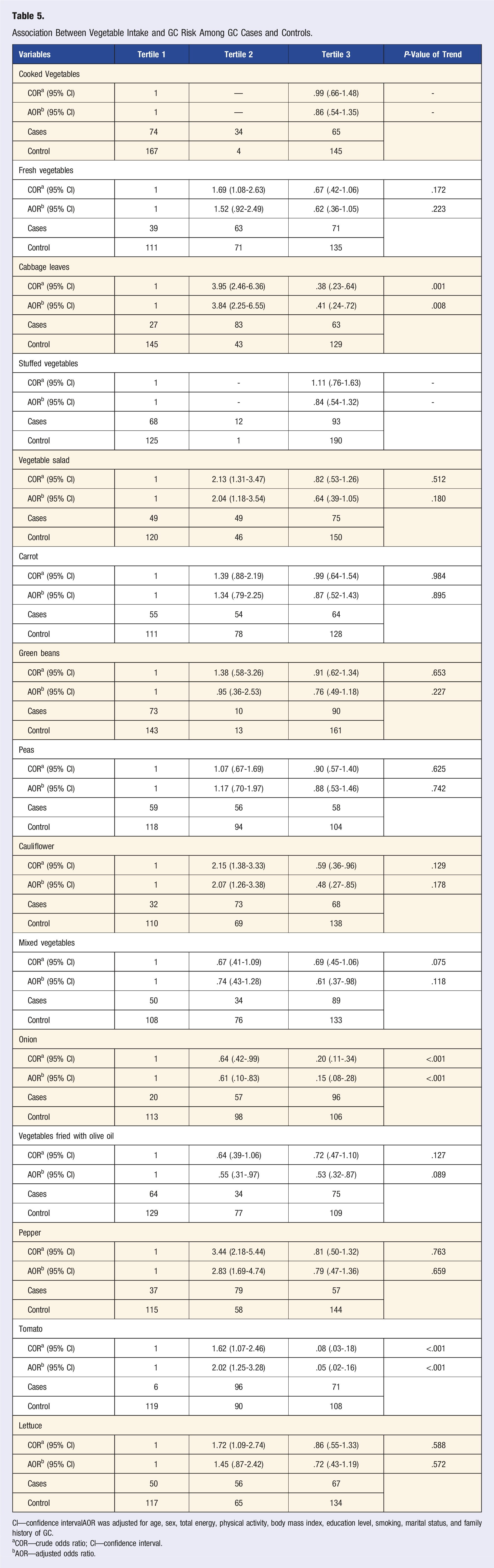
CI—confidence intervalAOR was adjusted for age, sex, total energy, physical activity, body mass index, education level, smoking, marital status, and family history of GC.
aCOR—crude odds ratio; CI—confidence interval.
bAOR—adjusted odds ratio.
Furthermore, the risk of GC significantly increased to about 2 odds (AOR = 1.70 (1.10-2.91) and COR = 2.03 (1.28-3.23), AOR = 1.93 (1.17-3.18) and COR = 1.81 (1.18-2.77), respectively) in the third tertile of pear and strawberry consumption. In contrast, the results showed an inverse association between grape and melon consumption and GC development (AOR = .54 (.29-.99)), AOR = .41 (.22-.76) and COR = .52 (.31-.88), respectively) in the third tertile.
The risk significantly increased in the second tertile of orange consumption but this relationship was not seen with the orange consumption after adjusting for confounding factors (AOR = 1.61 (1.00-2.60) and COR = 1.96 (1.29-2.96)).
Table 5 showed the ORs of cases and controls according to the number of vegetables consumed daily. The results indicated that the consumption of cooked and fresh vegetables, vegetable salad, and pepper lettuce in the second tertile might be a risk factor for GC. However, mixed vegetable consumption in the third tertile was protective against GC (AOR = .61 (.37-.98). In addition, similar results were found in the second and third tertiles of onion consumption (AOR = .61 (.38-.10) and AOR = .15 (.08-.28), respectively). The association between cabbage leaves, cauliflower and tomato consumption and the risk of GC significantly increased in the second tertile. However, the above risk decreased in the third tertile. The results showed that the consumption of vegetables fried with olive oil in the second and third tertile have a protective effect against cancer. No association was detected for all other examined fruits and vegetables (dates, watermelon, peach, green beans, peas, carrot, and stuffed vegetables).
Discussion
In the current study, we examined the association between the study participants' consumption of common fruits and vegetables and the risk of developing GC. The findings of the study show that cases who consumed more vegetable servings per day were more likely to have higher pre-diagnosis BMIs than those who consumed less. Similarly, Table 2 shows a significant difference between controls’ usual weight and current weight in vegetable consumption; showing that controls with higher weight tend to consume more vegetables. Controversially, a study showed that obese people had significantly lower fruit and vegetable intakes compared to people with normal weight (OR .78; 95% CI .63-.97). 15 There was a significant difference between the fruit and vegetable intake of the cases and controls when comparing their use of tobacco, showing that the cases with tobacco use consumed less fruit while the controls with tobacco use consumed fewer servings of vegetables daily. Nonsmokers tend to consume more fruit and vegetables compared to smokers. A similar association was reported by Dehghan et al. (2011), where nonsmokers were found to consume more fruits and vegetables comparing to occasional and daily smokers. 15 Another study showed that women who are nonsmokers or previous smokers were consuming more fruits and vegetables. 16 Additionally, when comparing the number of snacks in the cases and controls, there was a significant difference in the intake of fruit and vegetables. A smaller number of snacks in a day was associated with a higher intake of fruit in controls and vegetables in cases. A study conducted by Lazzeri et al. (2013) found a strong correlation between irregular snack consumption and frequent fruit consumption (OR = 1.20 (1.02-1.42)). 17 Furthermore, our study showed a significant relationship between daily water amount and vegetable servings intake in the control group while the significant difference was observed in the fruit consumption in the case group. These findings demonstrate that daily water intake was higher with more vegetable intake in controls and more fruit intake in cases. Similar findings were documented by Goodman et al, where participants who had 1 cup or less of fruits or vegetables daily tended to drink fewer than 4 cups of water on average, compared to those who drank 4.5 cups or more (AOR, 3.0).18 In addition, it was found that in case group who had just completed primary and secondary education consumed fruit less than those with higher education did. A study found that fruit and vegetables consumption was around 1.5 times higher for those with post-secondary education compared to those with secondary and lower levels of education (OR for all fruit and vegetables was 1.86; 95% CI 1.44-2.39). 15 Another study showed that women with a higher level of education consumed higher amounts of fruits and vegetables. 16 Cases with a family history of GC tended to consume vegetable more than those who have no history of GC. The marital status and the number of meals eaten outside affected the servings of vegetables consumed daily in the control group. Dehghan et al. (2011) showed that single/never-married people consumed slightly more fruits compared to the married/common law and widow/separated/divorced groups. 15 Another study showed that married women tend to consume more fruit and vegetables than others. 16 A significant difference was found between controls who skipped meals and their daily intake of fruit; controls who do not skip meals consumed more fruit than others did.
Several studies reported that fruit and vegetable consumption was associated with a significant reduction in gastric cancer risk.18-20 It has been postulated that the anticarcinogenic effects of fruits and vegetables may be attributed to the antioxidant effects of their vitamin content, particularly vitamin c and ß-carotene.19,20 Due to the presence of free radicals, oxidative stress occurs and leads to cell damage and DNA changes. It plays a causal role in the development of cancer and other health conditions. 21 Antioxidants present in fruits neutralize reactive oxygen free radicals (ROS) that cause DNA damage.22,23 A study conducted by Poorolajal et al. (2020) showed that fruits have a protective effect against GC. 24 Based on 13 studies, the overall OR for fruit consumption 3 times/week compared to fruit consumption <3 times/week was .48 (95% CI, .37 to .63). The overall effect measurement showed that fruit consumption significantly reduced the risk of gastric cancer by 48% (P = .001). 24
The present study findings demonstrated that orange consumption in the third tertile might have a protective effect against gastric cancer. In fact, orange fruit contains hesperidin and naringenin, two major flavanone compounds found in oranges and tangerines involved in the growth, migration of the human stomach, preventing intrusions, and inhibiting the proliferation of cancer cells. The effect on the proliferation of gastric cancer was dose and time dependent.25-27 Hesperetin and hesperidin's anti-inflammatory properties have been studied in several cell culture research reviewed by Chanet et al. (2012). 28 Hesperetin inhibited lipopolysaccharide-stimulated macrophages' ability to produce TNF- in a concentration-dependent manner;29,30 IL-6 production was unaffected. 30 In many cell culture and animal feeding investigations, naringenin's anti-inflammatory properties have been investigated. 28 Naringenin might inhibit lipopolysaccharide-stimulated macrophages' production of TNF-, IL-1, IL-6, and prostaglandin E2, as well as the expression of cyclooxygenase-2 and inducible nitric oxide synthase.31,32 Moreover, a study found that citrus fruit peels are rich in polyethoxylated flavones, Tageretin, and nobiletin. 33 Tageretin inhibits NF-kB activity, lowers oxidative stress and pro-inflammatory cytokines. 34 Nobiletin is a polyethoxylated flavonoid found in fruits and vegetables. Its pharmacological effects include those that promote apoptosis, reduce inflammation, fight tumors, fight against free radicals, and fight against diabetes. 35 On contrary, the present study shows that the consumption of 100% orange juice in the second tertile increased the risk of GC, while this result was not shown in the third tertile. The amount of orange juice consumed daily by GC cases was higher compared to controls (Table 3). A wide range of potential health benefits has been ascribed to citrus fruits and juices, including anticancer, antimicrobial, antioxidant, and anti-inflammatory, 36 closely linked to their flavonoid content. 37
Our study showed that strawberry consumption in the third tertile might increase the risk of GC. Our data have shown that cases consume a significantly higher amount of strawberries than controls. Strawberries are rich in anthocyanin and non-anthocyanin phenolic compounds that are known for their antioxidant properties.38,39 However, strawberries are often commercialized and marketed as processed products. The effect of fruit drying on the antioxidative potential was presented and studied by Mendez-Lagunas et al, who revealed a 74.7% loss of antioxidant activity in the processed fruit, with thermal treatment at 50 Co. 40 Another study investigated 128 pesticides, 18 phthalates, and three heavy metals. The authors found 51 pesticide residues in strawberry samples, with levels of at least one of the 51 pesticides exceeding the detection limit in 97.91% of the samples. Furthermore, pesticide residue levels in 2.39% of samples exceeded the Chinese maximum residue limit. Most samples contained multiple pesticide residues. 41 The findings indicate that a variety of pollutants could contaminate strawberries. 42 Moreover, it has been found that exposure to the pesticide methyl bromide raises the risk of developing stomach cancer (Barry et al., 2012). 43 It has been found that there is a link between pesticide exposure and diffuse-type gastric cancer based on their analysis of the association between stomach cancer histological types and exposure to pesticides. 44
On the other hand, grape and melon consumption in the third tertile decreased the risk of GC and an inverse association was shown with GC development. In a study conducted in Korea, daily grape juice consumption results in reduced oxidative DNA damage as measured in peripheral lymphocytes and increased plasma antioxidant capacity in healthy Korean participants. The findings of these studies strongly suggest that grapes and grape-based products are the sources of many potential anticancer and cancer chemopreventive agents. 45 The phenolic components, antioxidants, and antiproliferative properties of melon residue extracts were studied. 46 The findings indicate that melon residue extracts had a significant biological activity against the development of human tumor cells and high antioxidant activity in in vitro experiment. 46 Another study found that the phytochemical composition and functional characteristics of melon peel (Maazouncultivar) might contain high levels of polyphenols and flavonoids (332 and 95.46 mg/100 g extract, respectively), both are potent antioxidants. According to the data, their primary phenolic classes include hydroxybenzoic acids and flavones. The primary phenolic ingredient in the melon peel is 3-hydroxybenzoic acid, which makes up 33.45 mg/100 g, followed by apigenin-7-glycoside (29.34 mg/100 g). 47
Regarding vegetable consumption, the study findings reveal the presence of association between the intake of commonly consumed vegetables and the risk of GC among study participants. Based on the AOR, our results indicated that the consumption of cooked and fresh vegetables, vegetable salad, and pepper in the second might be a risk factor for GC. This can be explained by the fact that the amount consumed from these vegetables in the second is below the level that can prevent GC. Therefore, a protective association was observed in the third tertile with the consumption of these vegetables, which includes cooked and fresh vegetables, vegetable salad, and peppers. Consistent with findings from other studies, our research indicates that a significant reduction in risk is associated with higher consumption of cooked and fresh vegetables, vegetable salad, and peppers. Studies have suggested that almost all vegetables exhibit protective effects against gastric cancer, attributed to the presence of carotenoids, vitamins C and E, selenium, dietary fiber, dithiolthiones, glucosinolates and indoles, isothiocyanates, flavonoids, phenols, protease inhibitors, plant sterols, allium compounds, and limonene.48,49 However, a recent meta-analysis found that GC risk increased by 1.51 times with a high intake of chilli or capsaicin. 50 Furthermore, a study conducted by cancer research found that capsaicin could act as an adjunct cancer promoter by activating epidermal growth factor receptor (EGFR) to increase GC development. 51 Additionally, our study found that mixed vegetable consumption in the third tertile had a protective effect against GC (AOR = .61) with a significant lower intake in the cases group compared to the control (shown in Table 3). A case-control study conducted by Kim et al. (2021), illustrated that pattern with low vegetable loading was linked to a greater risk of developing GC compared to the pattern with high vegetable. 52 Moreover, Poorolajal et al. (2020) study indicated that eating vegetables significantly lowers the chance of developing stomach cancer. 53 Vegetables are anticarcinogenic due to the antioxidant effects of their vitamin content, particularly vitamin C and beta-carotene. 53
The association between cabbage leaves, cauliflower, and tomato consumption with the likelihood risk of GC reduced in the third tertile. Cruciferous vegetables contain vitamin C, folate, dietary fiber, phytoestrogens, selenium, and phytochemicals. It is a rich source of vitamins and minerals that may prevent cancer. 54 Wu et al 55 (2013) demonstrated that the presence of anticancer components (such as glucosinolates, the precursors of isothiocyanates, and indole‐3‐carbinol) in cauliflower are linked to a reduced risk of gastric cancer. 55 Moreover, tomatoes have been considered a food with potential anticancer properties due to their antioxidant properties, especially the carotenoid. 56 Tomato can inhibit cancer cell proliferation during the G0–G1 phase of the cell cycle, 57 neutralizes reactive oxygen species, protect DNA from oxidation, 58 decrease cell proliferation and induce apoptosis, 59 change cell-cell communication, 60 and enhance immunity against cancer. 61
Furthermore, onion consumption in the second and third tertiles was found protective against GC (AOR = .61 (.38-.10) and AOR = .15 (.08-.28)). Eating onions increase serum pepsinogen I (PG-I) levels and the PG-I/II ratio that act as preventive factors for stomach cancer. 62 Furthermore, flavonoids and polyphenols in onions may act as antioxidants to reduce the risk of GC. 63 Another study that was conducted by Ghosh et al (2021) demonstrated that kaempferol and anthocyanin pigments found in onions protect against GC. 64 Onions have antibacterial properties that contribute to anticarcinogenic effects in the stomach. 64 Inhibiting the growth of bacteria in the stomach lowers the conversion of nitrate to nitrite, decreasing the likelihood of carcinogens like nitrogen-nitroso compounds forming internally, and H pylori infection becoming less common. 64 Additionally, our study found that sautéed or fried vegetables with olive oil have protective effect against GC in the second and third tertiles (AOR = .55 (.31-.97) and AOR = .53 (.32-.87)). The result might be attributed to the fact that olive oil is used for frying in Jordanian cuisine. In fact, recent studies have found that an increase in the consumption of virgin olive oil may protect against cancer due its rich content in polyphenols. 65 Polyphenols have an antioxidant activity, anticancer, and anti- Inflammatory properties. 65 It has been proved that the natural polyphenols found in olive oil may protect against oxidative damage and modulate the immune system. 65 Moreover, a recent systematic review and meta-analysis showed that the risk for gastrointestinal cancer was 23% lower for those who had the highest intake of olive oil. 66 The type of fat used for frying might affect the relationship between fried food and the risk of cancer. A study conducted by Galeone et al. (2007) found that frying with olive oil, but not other types of oils, had a protective effect against colon cancer risk. 67 The association between frying food in olive oil and gastric cancer is not sufficiently studied.
Study Strengths and Limitations
This case-control study has several strengths including the adjustment of statistical analyses for many substantial confounders is believed to strengthen our findings by reducing the effects of these variables on GC risk. Compliance with the questionnaire was high with an eminent response rate of >95%. On the other hand, it is worth mentioning the limitations of this research including some questionnaire-inherited limitations that cannot be excluded; nevertheless, these factors were attenuated by precautionary measures. The accuracy of dietary recall tools is affected by possible recall bias and estimation errors; however, their effects were lessened using a validated FFQ along with food models and measuring tools to enhance the accuracy of portion size estimation. The used FFQ includes cultural foods, commonly used portion sizes for each food item, and accounted for seasonal variations for a more precise estimation of participants’ dietary history. Moreover, recall bias may have influenced the dietary data due to knowledge of the disease status as well as due to the possibility that cases might have changed diet due to their disease or corresponding symptoms; yet, the inclusion of recently diagnosed patients could have reduced such bias. In addition, the interviewers were not blinded for the diagnosis of the participants (i.e., cases and control); nevertheless, all interviewers were well-trained and treated the participants professionally and identically, regardless of their case and control status.
The findings of this study revealed that consumption of 100% orange juice, orange, pear, and strawberry may increase significantly the risk of GC. In contrast, the results showed an inverse association between grape and melon consumption and GC development. Regarding the vegetable consumption, the consumption of cooked and fresh vegetables, vegetable salad, pepper, lettuce cabbage leaves, cauliflower, and tomato might be a risk factor of GC. However, mixed vegetables, vegetables fried with olive oil, and onion consumption showed a protective effect against GC. No association was detected for all other investigated fruits and vegetables. Further research is required to confirm our findings and elucidate the underlying mechanisms of these associations.
Footnotes
Acknowledgments
The authors would like to express their thanks to the Deanship of Scientific Research at The Hashemite University for funding the research projects. We would like to thank Dr Narmeen Al-Awwad for her support in the administrative work at The Hashemite University.
Author Contributions
RT and TA were responsible for the study conception and design and responsible for the development of the methodology. SA, TA, AH, and YR were responsible for the acquisition of data. RT, MA, NA, and RA were responsible for the analysis and interpretation of data. RT, MA, NA, RA, and SA were responsible for drafting the manuscript. All authors were responsible for critically revising the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Deanship of Scientific Research at The Hashemite University.
