Abstract
A Lifestyle Medicine approach to compliment cancer care is less commonly researched or implemented for women with gynecologic cancers as compared to better funded malignancies such as breast, prostate, and colorectal. Yet, several gynecologic malignancies are linked to obesity, estrogen/metabolic signaling pathways, and altered tumor microenvironment which could benefit greatly from a lifestyle medicine program. Lifestyle medicine, an evidenced-based branch of science, has expanded to the prevention and treatment of disorders caused by lifestyle factors (including cancer). Modifiable lifestyle factors such as obesity, lack of physical activity/nutrient density, microbial dysbiosis, sleep disturbance, and chronic stressors contribute greatly to cancer morbidity and mortality worldwide. This overarching area of research is evolving with some subtopics in their infancy requiring further investigation. Modern tools have allowed for better understanding of mechanisms by which adiposity and inactivity affect tumor promoting signaling pathways as well as the local tumor environment. Through the evolving use of these sophisticated techniques, novel prognostic biomarkers have emerged to explore efficacy of pharmacologic and lifestyle interventions in cancer. This state-of-the-art review article appraises recent evidence for a lifestyle medicine approach, beyond diet and exercise, to optimize survivorship and quality of life for patients with gynecologic cancers and introduces the 8-week web-based comprehensive HEAL-GYN program.
Keywords
“Modifiable lifestyle factors such as obesity, lack of physical activity, sleep disruption, and chronic stress, affect the risk of cancer, survivorship, and QoL after cancer diagnosis.”
Introduction
Cancer is the second leading cause of death in the United States and marginalized subgroups are disproportionately affected. Furthermore, obesity-related dysfunction is not only linked to cancer pathogenesis but also to treatment resistance through various mechanisms. 1 In fact, obesity has surpassed tobacco consumption as a lifestyle-related factor of overall mortality. Now, there is increasing evidence supporting the benefits of lifestyle interventions such as physical activity in preventing many chronic diseases and cancer as well as reducing all-cause mortality (with the strongest evidence for breast and colon cancers). 2
Recently, the both the American Cancer Society (ACS) nutrition and physical activity guideline for cancer survivors (2022 version) 3 and the American Society of Clinical Oncology (ASCO) guidelines, 4 were published to address evidence-based lifestyle interventions that could reduce risk of recurrence and mortality while increasing disease-free survival for the 16.9 million people currently living with or from cancer.3,4 These guidelines are consistent with the data and recommendations published by other authoritative bodies including the World Cancer Research Fund/American Institute for Cancer Research (WCRF/AICR); the American College of Sports Medicine (ACSM); and the Academy of Nutrition and Dietetics (AND).3,4 These guidelines are also consistent with research and recommendations from the American College of Lifestyle Medicine (ACLM). Specifically, being physically active, consuming foods that reflect a healthy dietary pattern, and avoiding obesity during and after the completion of cancer treatment improves long-term survival.3,4
The ACLM continues to define lifestyle medicine for the country and the world. The challenge for ACLM is to standardize the evolving definition of lifestyle medicine, all while acknowledging its current definition as “the use of evidence-based lifestyle therapeutic approaches, such as a predominately whole food, plant-based diet, physical activity, sleep, stress management, tobacco cessation, and other non-drug modalities, to prevent, treat, and, oftentimes, reverse the lifestyle-related chronic disease that are all too prevalent.” 5 Lifestyle medicine operates to complement care and to work synergistically with the current public health initiatives globally. Lifestyle medicine leverages the provider-patient relationship to put the person at the center of their own health and health care. 6
Gynecologic cancer refers to any cancer that originates in the reproductive tract of phenotypic human females with an estimated 115,130 new cases in the United States.
7
Cancers of the endometrium and ovaries have a strong obesity/hormonal etiology and physical activity has been postulated as a potential modifiable risk factor for prevention of these cancers because it can influence weight, energy balance, circulating hormone levels, and insulin-mediated pathways that are thought to be important mediators underlying the associations (Figure 1).
8
Few studies have evaluated the association of physical activity with cervical cancer (as well as rare vulvar/vaginal cancer) because the main etiologic factor is infection with certain types of human papillomavirus (HPV), although hormonal and immune co-factors are also thought to play a role. Interplay of underlying factors linking gynecologic cancer and lifestyle choices.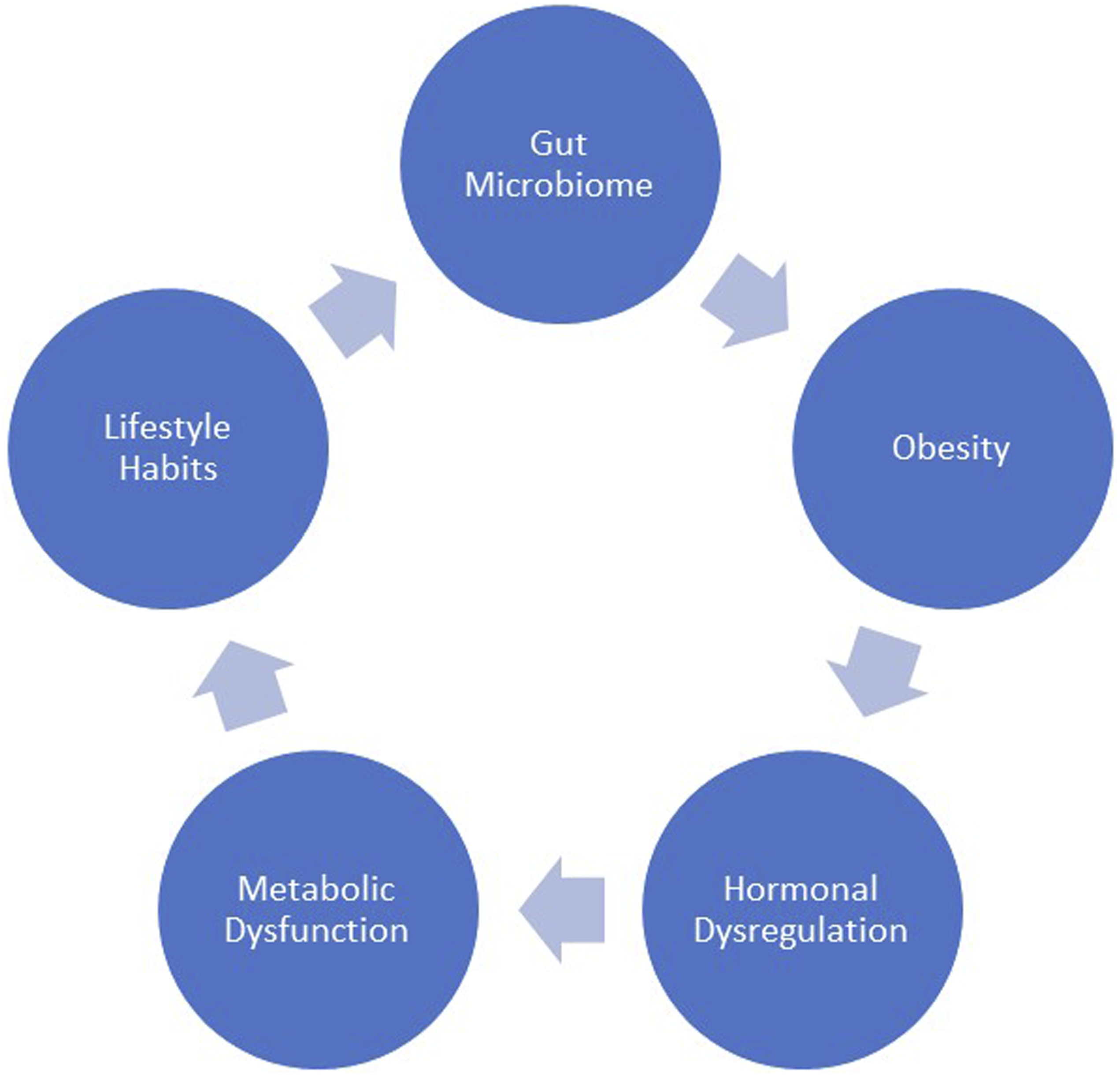
It has been postulated that risk factors that predispose to gynecologic cancer, such as obesity, hypoestrogenism, nulliparity, and menopause may be linked to the pathways by which the microbiome alters gynecologic cancer risk and prognosis. Additionally, there appears to be a convincing bidirectional relationship between diet, physical activity, inflammation, altered metabolism, stress, the gut microbiome, and gynecologic cancers.
9
The microbiome is now known to play an intricate role in carcinogenesis and response to cancer treatment.
10
In fact, the newly revised hallmark of cancer now includes polymorphic microbes, and all of these hallmarks are applicable to gynecologic cancers (Figure 2). This state-of-the-art review discusses emerging data highlighting the role of lifestyle interventions in the risk of developing and survival from gynecologic cancers with special focus on endometrial and epithelial ovarian cancers. Revised hallmarks of cancer are applicable to gynecologic cancers.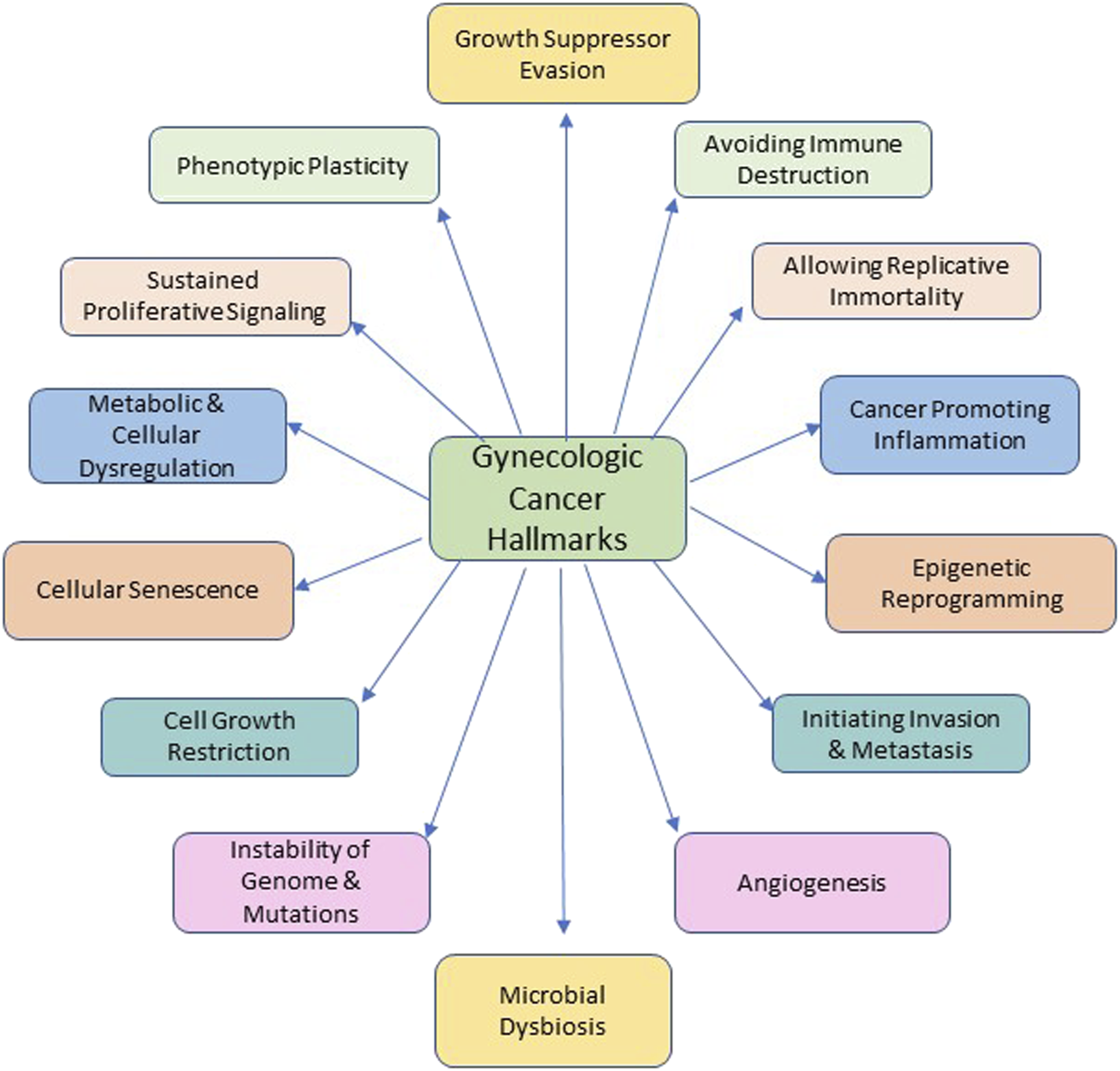
Diet and Obesity
Obese women have 2.4–4.5 times the risk of being diagnosed with endometrial cancer compared with normal-weight women.11,12 The relative risk (RR) of mortality is 6.25 times higher in morbidly obese survivors compared to normal-weight survivors. 13 Obesity may also be a risk factor for ovarian cancer, although its effect is much less than for endometrial cancer. In obese women, observational studies reveal a 6.25-fold increase in the relative risk of death from uterine cancer. The epidemiologic evidence to date suggests that physical activity probably protects against endometrial cancer with a risk reduction of about 20–30%. 14
Studies have shown that weight loss, especially significant weight reduction, reduces endometrial cancer risk.15,16 The sole randomized controlled trial (RCT) on diet, the Women’s Health Initiative trial, found a reduction in epithelial ovarian cancer (EOC) risk using a low-fat diet with an hazards ratio (HR) of .60 (95% confidence interval [CI] .38-.96) at 8-years post-intervention, suggesting that long-term adherence to a low-fat diet was needed for benefit. 17 Cohort studies, including the Nurses’ Health Study, have found no significant association between fat intake and EOC incidence.18,19 The Nurses’ Health Study also found no association with 2 healthy eating indices or a Mediterranean diet.20,21 Vegetable intake may have a small impact on EOC risk: 2 meta-analyses of cohort and case-control studies reported 11–16% risk reduction with daily vegetable consumption (95% CI 0.81-.99 and .75-.94, respectively).22,23 Other meta-analyses have found no significant association with dairy, 24 fish, 25 fiber, or dietary and antioxidant nutrients. 19 These studies highlight that although diet and weight control may play a role in gyn cancer risk reduction, these factors alone may be insufficient to greatly impact risk and/or survival.
Physical Activity
Physical activity is associated with decreased risk of gynecologic cancers. Exercise has demonstrated a 20-30% risk reduction of endometrial cancer in women who report moderate-intensity or high-intensity exercise compared to non-exercisers.26,27 The Nurses’ Health Study, a large prospective cohort study started in the year 1976, demonstrated a temporal effect of exercise on endometrial cancer risk: women who reported recent high-intensity exercise had a 35% reduced risk of endometrial cancer compared to those who did not perform any vigorous activity (RR = .65, 95% CI 0.47-.88). 18 Cannioto and colleagues 28 conducted a pooled analysis of 12 studies from the ovarian cancer association consortium to investigate the association between pre-diagnostic physical inactivity and mortality and found on multivariate analysis that inactive women had significantly higher mortality risks. These authors reported on the association between physical inactivity and risk of EOC (all histotypes). 29 Benefits of physical activity and/or exercise are further discussed in the subsequent section on gynecologic cancer survivorship. In their review, Rubenstein et al. 1 beautifully highlight the effects of obesity on insulin, adipokine, estrogen signaling pathways and the tumor microenvironment, but also how diet and exercise alter the pro-tumorigenic effects of these pathways. 1
Sleep and Stress
Women with cancer often suffer from both physical and psychological disturbance. These include fatigue, pain, and sleep disturbances, loneliness, lack of connection, stress, anxiety, alteration in cognitive functioning, depression, and fear of recurrence. These symptoms reduce both quality-of-life (QoL) and function.30-32 Circadian disruption and sleep deprivation have been characterized as carcinogenic factors and contributor of decreased survival, and reduced QoL in cancer patients. 33 Sleep is more commonly disrupted in women compared to men and further disrupted by surgical menopause, chemotherapy, and the cancer itself. Furthermore, sleep dysfunction is more prevalent among gynecologic cancer survivors (40–55%) as compared with all cancer survivors (30–55%). 34 Several tools exist to assess sleep disturbance such as the General Sleep Disturbance Scale (GSDS). 35 Lack of sleep is also linked to inadequate pain management, depression, and overall QoL scores among women with cancer. Zhao and colleagues 36 provide supporting evidence and strategies for sleep optimization in women with gynecologic cancers. The ACLM website (www.lifestylemedicine.org) provides patient education material to address sleep disruptors and proposes evidence-based strategies to improve sleep hygiene.
Chronic stress, also known as chronic unpredictable mild stress (CUMS), has been shown to boost tumorigenesis and impact cancer outcomes through a variety of mechanisms most of which are linked to elevations in stress hormones. 37 For example, we have learned through mostly mouse model studies that CUMS results in changes to the body’s immune function and inflammatory response, leading to long-term inflammatory response and the decline of the body’s immune surveillance capabilities are implicated in cancer promoting pathways. 37 Elevated glucocorticoid levels initiate a cascade leading to the inhibition of p53 affecting proper DNA repair, and other pathways, which ultimately allow for tumor formation and disturb response to therapy. 37 These animal models/translational research findings are hypothesis generating and an opportunity for further clinical research. Strategies to mitigate or manage stress as proposed by the ACLM have been shown to play a role in stress reduction as well as improve symptoms of anxiety and depression. 38 Bird et al. 39 also summarized recent published work revealing the benefits of stress management for cancer patients. These authors recommend exercise referrals, social prescriptions, and social movements as a strategy to mitigate the “three P’s (people, purpose, place) thereby achieving resilience in the face of CUMS which in turn decreases chronic inflammation and promotes wellbeing of the patient with cancer.” The association between sleep and stress to cancer, and particularly gynecologic cancer outcomes, remains in its infancy. Clearly, this patient population could benefit from sleep optimization and stress management, at the very least, to positively affect QoL. Hence, more research in this field is needed to strengthen our understanding of the association linking stress and or sleep to gynecologic cancer outcomes as well as its underlying mechanism(s).
Studies are underway to explore underlying pathways by which behavior modification and/or lifestyle choices affects cancer biology. The next few sections of the article discuss the potential biological mechanisms of various lifestyle factors. The gut microbiome, adiposity, and physical activity are among the mechanisms described which may affect tumor-modulating pathways.
Gut Microbiome Interplay
The composition of the microbiome appears to be individually unique. It is mostly established in early life and plays a very important role in lifelong health.40-43 In our recent publication, we described further the interplay between the gut microbiome, disease pathogenesis, and response to gynecologic cancer therapeutics. 9 Obesity interplays with both the gut microbiome and estrogen metabolism; and is a prime driver of carcinogenesis in endometrial cancer. 44 Obesity is also one of the strongest determinants of gut microbiome and uterine microbiome composition. A recent study found that microbiota profiles of endometrial tumors and stool in lean mice were distinctly different from that of obese mice.45,46 Overall, it can be concluded that the obesity estrogen metabolism pathways simultaneously modulate both the gut and uterine microbiome.47,48 Dietary alterations have potential applications in gynecologic cancers, where obesity and an inflammatory diet are associated with worse disease-specific outcomes.49-51 Behavioral changes such as exercise are another modifier that can significantly affect the composition of the microbiota and gut inflammation.52,53 This finding has strong implications in gynecologic cancer prevention and control as both risk factors are amenable to modification though lifestyle and dietary interventions.
Molecular Pathways and the Tumor Microenvironment
Metabolomics is a promising molecular tool to identify novel etiologic pathways leading to cancer.
Dashti et al. 54 evaluated the mediating effects of and anti-inflammatory adipocytokine, adiponectin, as well as other inflammatory biomarkers; C-peptide (a hyperinsulinemia biomarker); and free estradiol and estrone-estrogen biomarkers in the adiposity-endometrial cancer link in postmenopausal women. The authors 54 found that reduced adiponectin and increased inflammatory biomarkers, C-peptide, and estrogens mediated approximately 70% of increased odds of endometrial cancer in women with obesity vs normal weight. 54 In another study, His and colleagues 55 evaluated the link between serum metabolite concentrations, lifestyle, adiposity, and breast cancer. Blood concentrations of acetylcarnitine, a metabolite, and breast cancer risk have been reported as well as lower risk associated with higher blood concentrations of seven other metabolites. These metabolites are either negatively or positively linked to adiposity, total and saturated fat intakes, and alcohol consumption. They are also associated with scores reflecting adherence to a healthy lifestyle. These associations may indicate possible mechanisms underlying interplay between lifestyle and anthropometric factors, and risk of breast cancer.
Additionally, women who inherit the germline BRCA1 or BRCA2 gene mutations face a high lifetime risk (penetrance) of developing breast/ovarian cancer and this penetrance appears to be affected by serum levels of adiponectin. 56 A recent meta-analysis confirmed an inverse relation between adiponectin and cancer risk while high leptin was associated with a significantly higher risk. 57 Bruno and colleagues 58 evaluated the influence of lifestyle characteristics on the lifetime penetrance of developing BRCA-related cancer and found that higher fat mass and dysmetabolism were significantly associated with BRCA-related cancers and greater effect for BRCA2-positive cancers. These authors are currently investigating strategies such as the Mediterranean diet to reduce potential modulators of BRCA penetrance. 59
Crown-like structures (CLS), a histologic marker of local inflammation, are composed of necrotic adipocytes encircled by macrophages and are associated with obesity. CLSs have also been found to be related to formation, progression, and prognosis of many caner types. The CLS within the breast are associated with local increased aromatase activity and elevation of pro-inflammatory mediators, thereby potentially affecting the local tumor environment. 60 The tumor microenvironment is a conceptual framework encompassing how local immune cells promote or inhibit cancer formation and development via sophisticated signaling pathways. Liang et al. 61 demonstrated that the presence of omental CLSs is associated with poor prognosis in some histologic types of advanced-stage ovarian cancer.
Most recently, Emery et al. 62 in their exhaustive review, highlight how physical activity appears to modulate the cancer immunoediting process. Using a cancer immunogram as a basis for this evaluation, the authors explored the effects of physical activity on serum T-cell status, T-cell infiltration of tissues, presence of immune checkpoints associated with T-cell exhaustion and anergy, presence of inflammatory inhibitors of T-cells, and lastly presence of metabolic inhibitors of T-cells. Newer investigational tools have allowed for better understanding of the underlying mechanisms by which adiposity affects tumor promoting signaling pathways as well as the local tumor environment and how lifestyle interventions can modulate these processes. The use of these sophisticated analyses will help determine appropriate markers and predictors of pharmacologic as well as lifestyle interventions for cancer patients.
Lifestyle Medicine for Gynecologic Cancer Survivorship
Gynecologic cancers and their treatments are associated with functional status decline that has impactful consequences on physical function, QoL, and care continuum (Figure 3). Many patients present with or experience clinical deconditioning and various symptoms due to surgery, chemotherapy, radiation therapy, and/or biologic therapy.
63
Often, gynecologic cancer survivors are plagued by adverse physical (e.g., fatigue, pain, and sleep disturbances), social (e.g., loneliness, lack of connection), and psychological (e.g., stress, anxiety, alteration in cognitive functioning, depression, and fear of recurrence) symptoms that may remain and/or occur after the treatment completion, thereby reducing both QoL and functionality.30-32 These patients also often experience issues with sexuality and spiritual domains of QoL.
64
A recent 1-year longitudinal study demonstrated that half of women with gynecologic cancers experience significant fatigue and depression after surgery and chemotherapy with no spontaneous regression of symptoms.
65
Pain has also been reported by up to 33% of cancer survivors after curative therapy.
63
The prevalence of sleep disturbances is reported to be twice as high among cancer survivors in comparison with the general population.66,67 It is estimated that up to 70% of cancer survivors experience 6 or more co-occurring symptoms.
68
Although 46% of cancer survivors report depressive symptoms as a concern, only 34% attempt to seek help and among these, 25% report difficulty obtaining care.69,70 Cancer survivors also desire healthy lifestyle programs, since they often engage in sedentary lifestyle, unhealthy diet, smoking, and alcohol use.
71
In addition to symptom burden, at least one third of survivors experience physical deconditioning and exercise intolerance,
72
further highlighting the need for a multidisciplinary approach to cancer survivor wellness. Common side effects of gynecologic cancer therapy.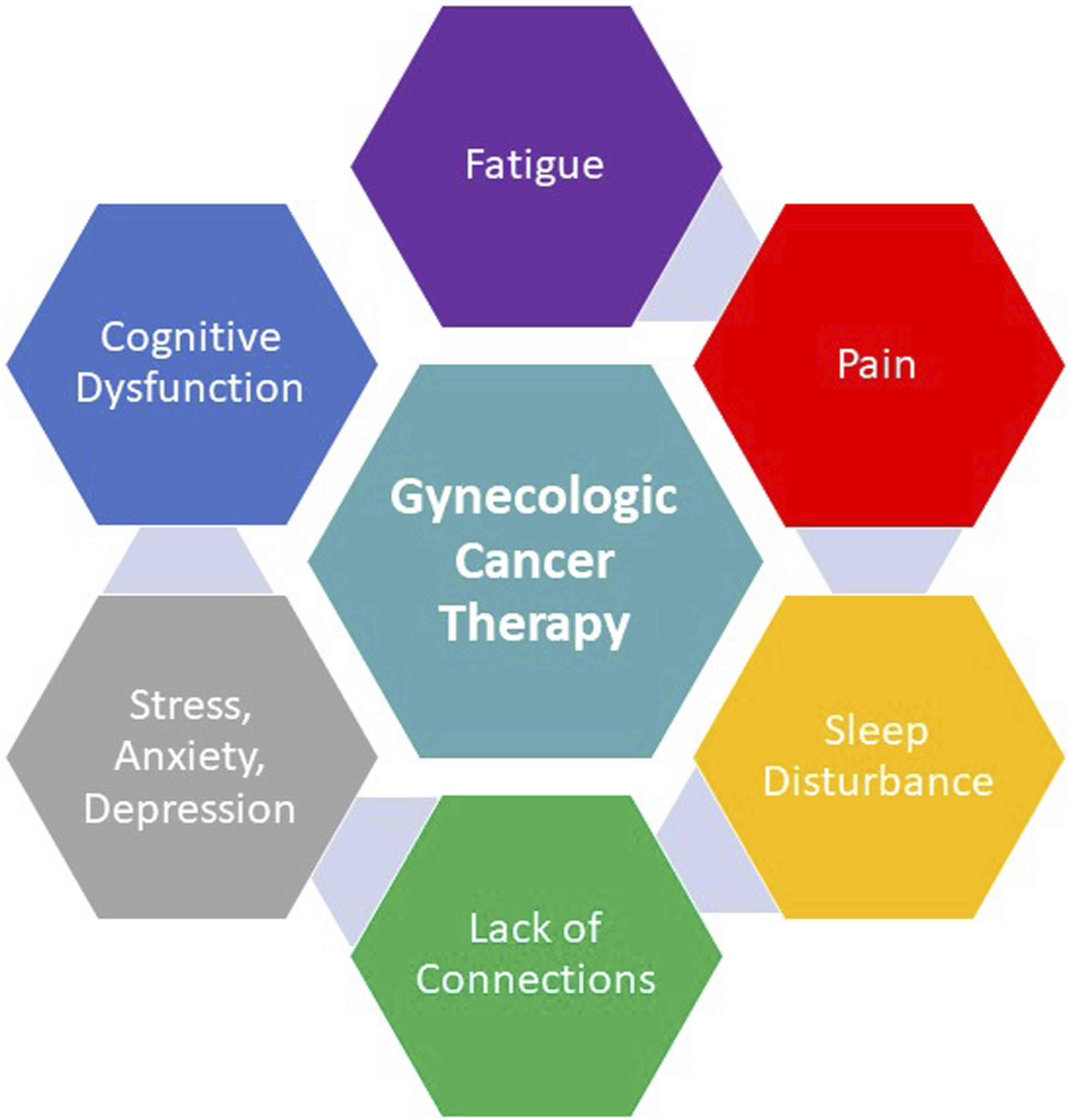
Nutrition and physical activity recommendations recently established by the ACS 3 and the ASCO 4 guidelines for the primary prevention of cancer are broadly relevant to survivors undergoing and immediately after cancer treatment. 4 There is sufficient evidence that exercise during cancer treatment is beneficial in managing several aspects of QoL during cancer including anxiety, depression, physical function, fatigue, and lymphedema. 3 Endometrial cancer survivors frequently report wanting to eat better, be healthier, and lose weight.73,74 Yet, only 50% of women with history of endometrial hyperplasia or cancer survivors are aware that obesity contributed to their cancer risk. 75 Interestingly, only 10% of members of the Society for Gynecologic Oncology (SGO) reported having any training in weight loss counseling. 76
Internationally endorsed physical activity guidelines recommend that cancer survivors should participate in 150-min/week of physical activity and strength-training and suggest incorporating these into standard of care. 3 Jones et al. 77 highlight the evidence behind these recommendations as well as their own longitudinal cohort study through which they found more than 50% of ovarian cancer survivors did not meet physical activity guidelines post diagnosis. Yet, two cross-sectional studies found that exercise has been associated with improved QoL and mental health and reduced fatigue in EOC survivors.78,79 Obese endometrial cancer survivors also stand to gain from positive effects of exercise, as surveys indicate that they have lower QoL than normal-weight survivors. 80 Nonetheless, most endometrial cancer survivors are not able to meet the physical activity guidelines. Only 1% of early-stage endometrial cancer survivors are able to meet all the ACS guidelines, and only 12% are able to meet the physical activity guidelines, which has been associated with worse QoL and fatigue. 81
Several recent studies evaluated the benefit of lifestyle interventions. The survivors of uterine cancer empowered by exercise and healthy diet (SUCCEED) randomized controlled trial (RCT) showed significant weight loss, increased physical activity, greater fruit and vegetable consumption, and improved QoL in endometrial cancer survivors after an intensive 6-month group and individual lifestyle intervention.82,83 The Steps to Health study found similar results with a home-based exercise intervention, with significant improvement in physical activity, heart rate, and systolic blood pressure in obese and non-obese participants. 84 Low-cost intervention with a mobile health application Loseit! also showed short-term weight loss in endometrial cancer survivors. 85 The revving-up exercise for sustained weight loss by altering neurological reward and drive (REWARD) RCT is currently recruiting obese endometrial cancer survivors to study different types of exercise and weight loss. 86 Yet, another study evaluated an 8-week diet and exercise RCT pilot behavioral program for endometrial cancer patients and found that at 24-weeks, global QoL improved in intervention. 87 However, not all exercise studies have been successful. Rossi et al. 73 recruited ethnically diverse, low-income women with a history of endometrial cancer. While 86% of the women surveyed expressed some interest in joining a free exercise program, only 5% came to the scheduled exercise classes. The authors emphasize the fact that care needs to be taken to determine barriers to exercise in vulnerable low-income and/or minority populations. 73
Hausmann et al. 88 reported combined aerobic and resistance training improves physical capacity in women treated with gynecologic malignancies with beneficial effects lasting even 1-year later. Recently, Fleming et al. 89 also demonstrated that many gynecologic cancer survivors can improve their physical activity levels. In addition, Hansen et al. 90 retrospectively evaluated a large dataset of women whose healthy lifestyle index (HLI) scores were calculated before and after primary treatment. These authors found that there was trend towards pre-treatment HLI score, but a stronger association with post-treatment HLI suggesting that an intervention during the treatment or after would have the greatest impact on survival.
Based on these collective findings, it appears that a comprehensive behavioral intervention integrating physical activity, education on optimal micronutrient-dense nutrition, management of stress, anxiety, and support into standard care could lead to gains in QoL throughout the gynecologic cancer care continuum. 89 These findings altogether underscore the importance of developing cumulative, scalable, and effective transdiagnostic interventions to ameliorate general health symptoms and improve QoL in survivors of gynecologic cancer.
HEAL-GYN (a Lifestyle Medicine-Based Gynecologic Cancer Survivorship Program)
Lifestyle medicine strategies have been explored outside of gynecologic oncologic subspeciality. Orman et al. 91 introduced this concept for breast cancer patients highlighting the role that lifestyle plays in primary breast cancer prevention, breast cancer treatment, and tertiary breast cancer prevention. Current data regarding the benefits of a predominantly plant-based diet in combination with physical activity and maintenance of a healthy body weight are reviewed. Some of the authors went to further define the use of a lifestyle medicine approach to compliment breast cancer care via a 12-week program including all six-pillars of lifestyle medicine, as well as positive psychology, gratitude, and goal-setting work. PAVING the Path to Wellness Program for Breast Cancer Survivors empowers breast cancer survivors with knowledge regarding evidence-based lifestyle recommendations and helps them achieve an improved sense of wellbeing following treatment. 92
Schedule and Details of the McKenzie-Whole Life Integration Curriculum™ of the HEAL-GYN Program Based on the ACLM Adult Starter Kit.
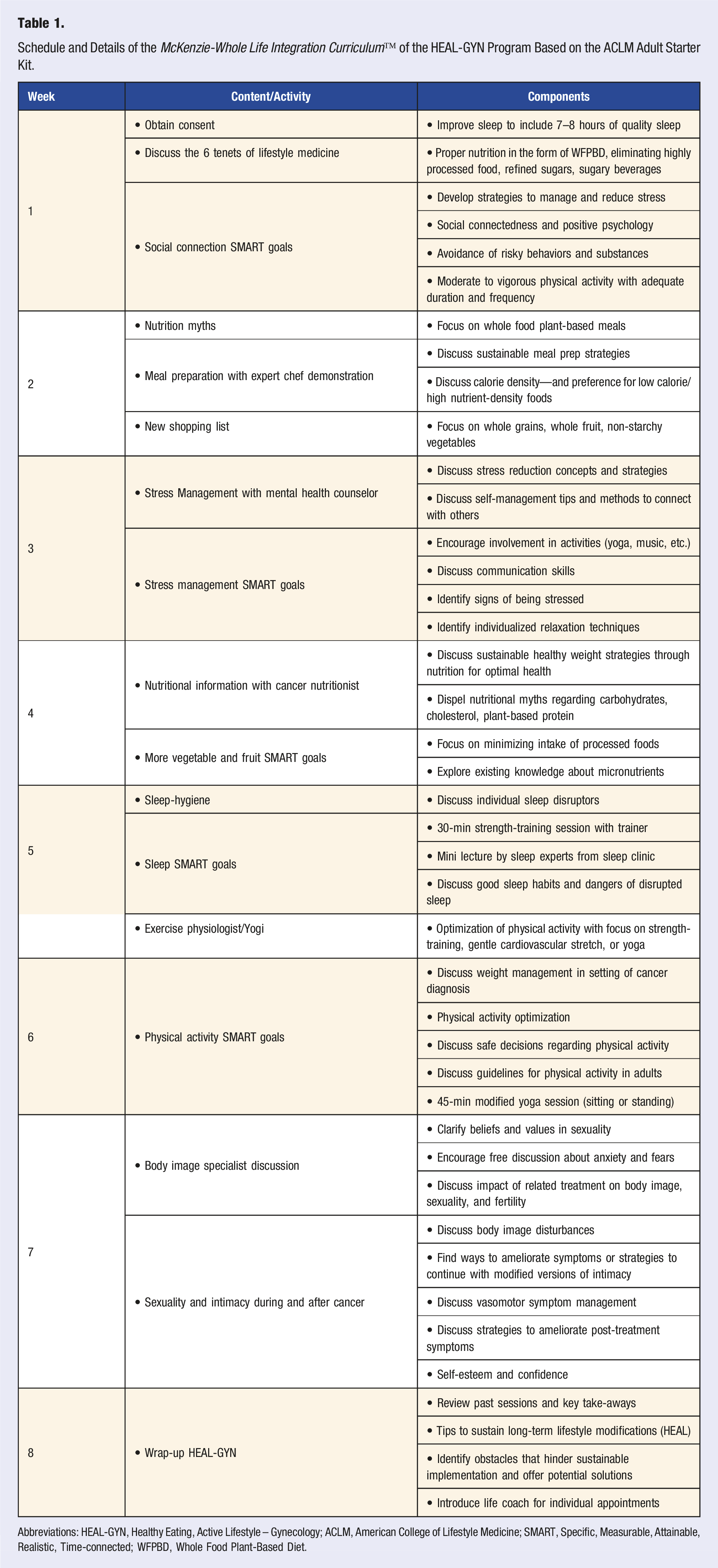
Abbreviations: HEAL-GYN, Healthy Eating, Active Lifestyle – Gynecology; ACLM, American College of Lifestyle Medicine; SMART, Specific, Measurable, Attainable, Realistic, Time-connected; WFPBD, Whole Food Plant-Based Diet.
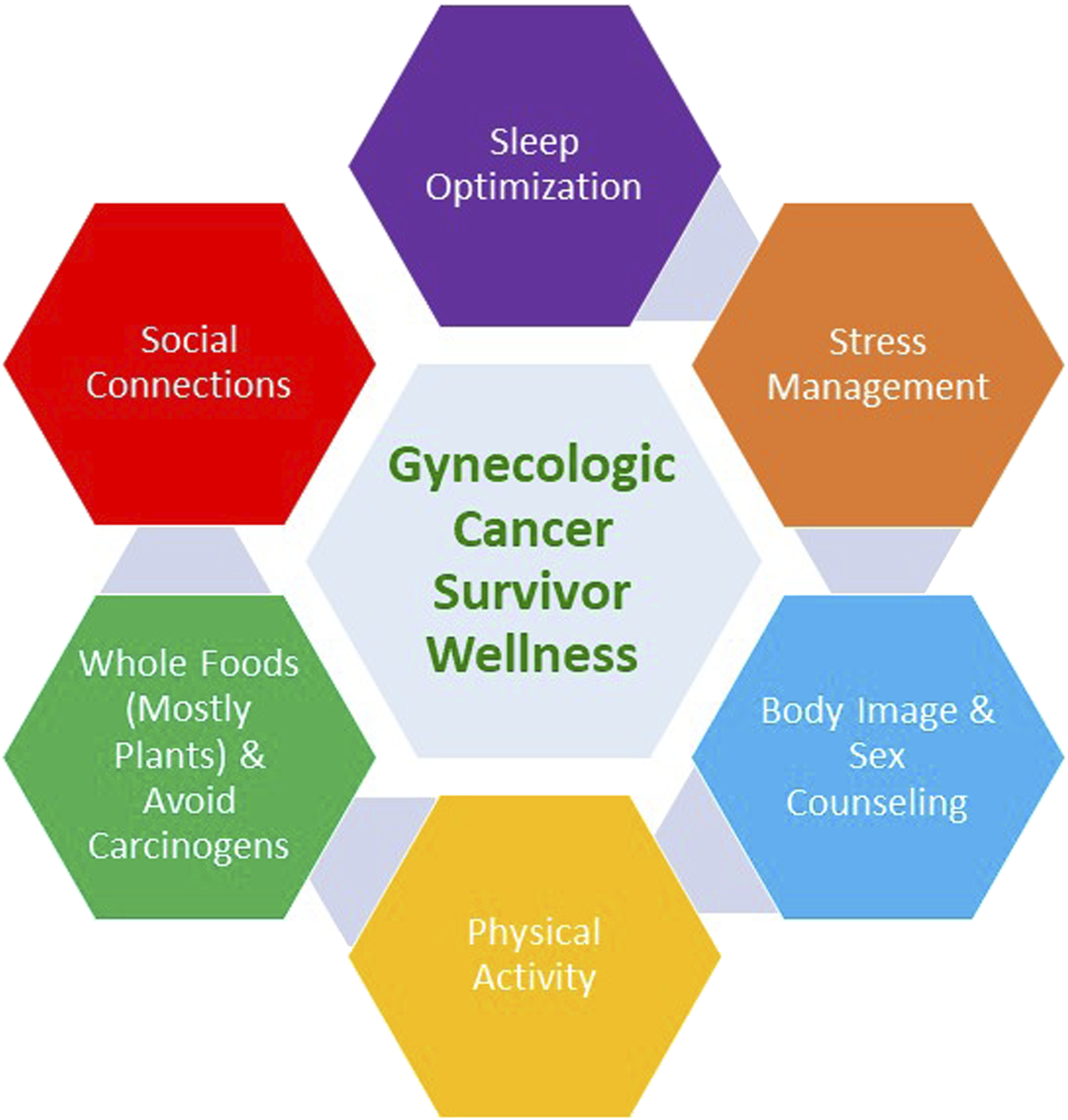
Whole life integration approach to gynecologic cancer survivor wellness.
The 8-week HEAL-GYN program is actively collecting ongoing prospective data on metabolic, functional, and anthropometric parameters to inform a randomized controlled trial. Particularly important, is whether a short comprehensive program such as ours is sufficient to impact short-term QoL and affect oncologic outcomes. Other studies aim to explore extended interventions. For example, results of the ongoing GOG-225 (LIVES) randomized trial of a 24-month intervention involving physical activity and diet for women with ovarian cancer will inform whether lifestyle interventions might improve progression-free survival after oncologic treatment. 93 Also, the DUET study will evaluate a 6-month web-based weight loss intervention among a heterogenous group of cancer survivors and their friends/family. 94 Yet, another group at Yale Cancer Center and University of Miami recently received funding for “Trial of Exercise and Lifestyle (TEAL) in women with ovarian cancer” (https://www.drjimmirios.com/yale-cancer-center-and-school-of-public-health-receive-grant-to-study-lifestyle-intervention-in-women-with-ovarian-cancer/). Clearly, there is a growing interest in lifestyle interventions to mitigate cancer treatment toxicity to further boost QoL and survival outcomes.
Conclusions and Future Perspectives
Modifiable lifestyle factors such as obesity, lack of physical activity, sleep disruption, and chronic stress, affect the risk of cancer, survivorship, and QoL after cancer diagnosis. Obesity interplays with estrogen-metabolic signaling pathways and is a prime driver of carcinogenesis in many endometrial and some ovarian cancers. Obesity is also one of the strongest determinants of gut microbiome and uterine microbiome composition. Small interventional trials show promise in increasing physical activity and weight maintenance for endometrial cancer and ovarian cancer survivors, although the impact on long-term health, including cancer recurrence and overall mortality is unknown. While each of these subtopics are in varying degrees of evolution, we have noted to the growing evidence in theses respective areas of research as well as the need for ongoing investigation. A lifestyle medicine approach to optimizing the health of gynecologic patients has the potential to confer additional benefits with patient reported outcomes and perhaps decreasing resistance to cancer treatment. The HEAL-GYN cancer survivor program is one proposed example of a lifestyle medicine strategy using ACLM-based education. 74 The ongoing 8-week HEAL-GYN longitudinal cohort study will not only provide preliminary data for a future randomized clinical trial but will also inform whether a short comprehensive web-based program could impact short-term QoL and affect oncologic outcomes.
Footnotes
Authors’ Contributions
Both authors have diligently contributed to the development and preparation of this state-of-the-art review article, including literature search, concept organization, data interpretation, artworks, and writings.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
