Abstract
Diabetes is a costly disease affecting 387 million individuals globally and 28 million in the United States. Its precursor, prediabetes, affects 316 and 86 million individuals globally and in the United States, respectively. People living with elevated blood glucose levels are at high risk for all-cause mortality and numerous cardiometabolic ailments. Fortunately, diabetes can be prevented or delayed by maintaining a healthy lifestyle and a healthy body weight. In this review, we summarize the literature around lifestyle diabetes prevention programs and provide recommendations for introducing prevention strategies in clinical practice. Overall, evidence supports the efficacy and effectiveness of lifestyle diabetes prevention interventions across clinical and community settings, delivery formats (eg, individual-, group-, or technology-based), and implementers (eg, clinicians, community members). Evidence-based diabetes prevention strategies that can be implemented in clinical practice include brief behavior change counseling, group-based education, community referrals, and health information technologies. These strategies represent opportunities where practitioners, communities, and health care systems can work together to provide individuals with education, support and opportunities to maintain healthy, diabetes-free lifestyles.
‘Obesity and physical inactivity lead to insulin resistance by increasing the nonphysiological deposition of fat in visceral, hepatic, and muscle tissues and by intracellular sequestration of glucose transporter-4 (GLUT-4) in unexercised muscle.’
Overview
Type 2 diabetes mellitus is a costly disease, affecting individuals, health care systems, economies, and whole societies worldwide. Diabetes and its precursor, prediabetes, affect 8% and 7% of the world’s population, respectively.1,2 Type 2 diabetes mellitus (hereafter called diabetes) makes up 95% of all diabetes cases. 2 Overwhelming evidence shows that lifestyle intervention programs promoting healthy diets, physical activity, and modest body weight reductions can prevent or delay the onset of diabetes among high-risk populations, such as those with impaired glucose tolerance (IGT).3-5 In this review, we briefly define diabetes and prediabetes and discuss their worldwide burden, describe the role physical activity and obesity play in the pathophysiology of diabetes, summarize the evidence around the effectiveness of lifestyle interventions for preventing diabetes, and provide evidence-based recommendations for health care practitioners looking to promote healthy lifestyles among their patients at risk of diabetes.
Defining Glucose Intolerance
Hyperglycemia is a major contributor to cardiovascular mortality and morbidity worldwide, 6 and it manifests in the form of prediabetes or diabetes. Prediabetes is a state of hyperglycemia where glucose levels are higher than normal but lower than diabetes thresholds and includes both impaired fasting glucose (IFG) and IGT. IFG is defined as fasting plasma glucose (FPG) levels of 100 to 125 mg/dL, whereas IGT is diagnosed when plasma glucose levels after an oral glucose tolerance test (OGTT) reach 140 to 199 mg/dL. 7 Elevated blood glucose levels represent defects in insulin secretion, insulin action, or both, where IFG is a reflection of impaired β-cell function and IGT of insulin resistance. Glycated hemoglobin (HbA1c) reflects chronic hyperglycemia, and values between 5.7% and 6.4% are also clinically used to define prediabetes.
Hyperglycemia is a strong risk factor for diabetes, and many adults with prediabetes will develop diabetes within the next 10 years, unless lifestyle changes are made. 8 People with IFG/IGT progress to diabetes when they reach FPG ≥126 mg/dL, plasma glucose levels ≥200 mg/dL after an OGTT, or an HbA1c value ≥6.5 (see Figure 1). 7 In some cases (eg, patients with hyperglycemia symptoms), a random plasma glucose ≥200 mg/dL can also be used to detect diabetes. Overall, HbA1c is regarded as a more convenient and more stable method than IFG and OGTT, although it is more expensive and not available in some settings. 9

Prediabetes and Diabetes FBG, OGTT, and HbA1c Cutoff Points According to ADA 2015. 7
The Diabetes Burden
Diabetes is an independent risk factor for premature illness and mortality, mainly because of cardiovascular disease (CVD)10,11 but can also lead to nephropathy, retinopathy, and neuropathy, 12 representing a major global public health burden. The International Diabetes Federation (IDF) reports that in 2014, diabetes prevalence among adults between 20 and 79 years old reached 8.3%, with 77% of the global cases living in low- and middle-income countries. 2 The diabetes prevalence is higher in urban than in rural populations, whereas the prevalence seems to be similar among men and women. 2 Overall, China, India, and the United States have the largest numbers of diabetes cases, with 92, 62, and 24 million people affected, respectively.13-15
Because diabetes can be asymptomatic and remain undetected for long periods of time, around 46% of global diabetes cases are undiagnosed, 16 suggesting that affected persons are not receiving the appropriate treatment to prevent complications. Diabetes has become an important cause of disability and mortality globally, accounting for 46 823 disability-adjusted life years (per 1000 population) in 2010 17 and 4.9 million global deaths in 2014. 2 Given global population growth and ageing trends, 18 the world prevalence of diabetes is expected to escalate and reach 55% by 2035. 1
In the United States, 9.3% of adults older than 20 years were affected by diabetes in 2012, and approximately 28% of diabetes cases were undiagnosed. 19 Together, undiagnosed and diagnosed diabetes affect 12.3% of American adults, with men showing a higher prevalence than women (13.6% vs 11.2%, respectively) and adults >65 years showing the highest prevalence (25.6%). Diabetes, particularly affects minorities, with American Indians and Alaska Natives having the highest prevalence (16%), followed by non-Hispanic blacks (13%), Hispanics (13%), and Asian Americans (9%). Conversely, the prevalence of diabetes among non-Hispanic whites is the lowest in the country (7.6%). 19
The financial burden diabetes poses to individuals and societies is also alarming. The global health expenditure on diabetes reached $612 billion in 2014. 2 The United States has the highest diabetes expenditure in the world, accounting for about 53% of the global diabetes expenditure. 20 In 2012, diabetes costs reached $245 billion in the United States, with $176 billion spent in direct medical costs and $69 billion spent in reduced productivity. 21 The costs of undiagnosed diabetes alone have been estimated at $33 billion, 22 suggesting that direct and indirect diabetes costs may be underestimated. Overall, Americans with diagnosed diabetes have 2.3 times higher medical expenditures than their healthy counterparts, creating a financial burden that affects individuals and societies through higher insurance premiums and taxes, reduced earnings, and reduced standard of living (Table 1). 21
Summary of the Burden of Prediabetes and Diabetes, the Evidence on Lifestyle Prevention and Recommendations.
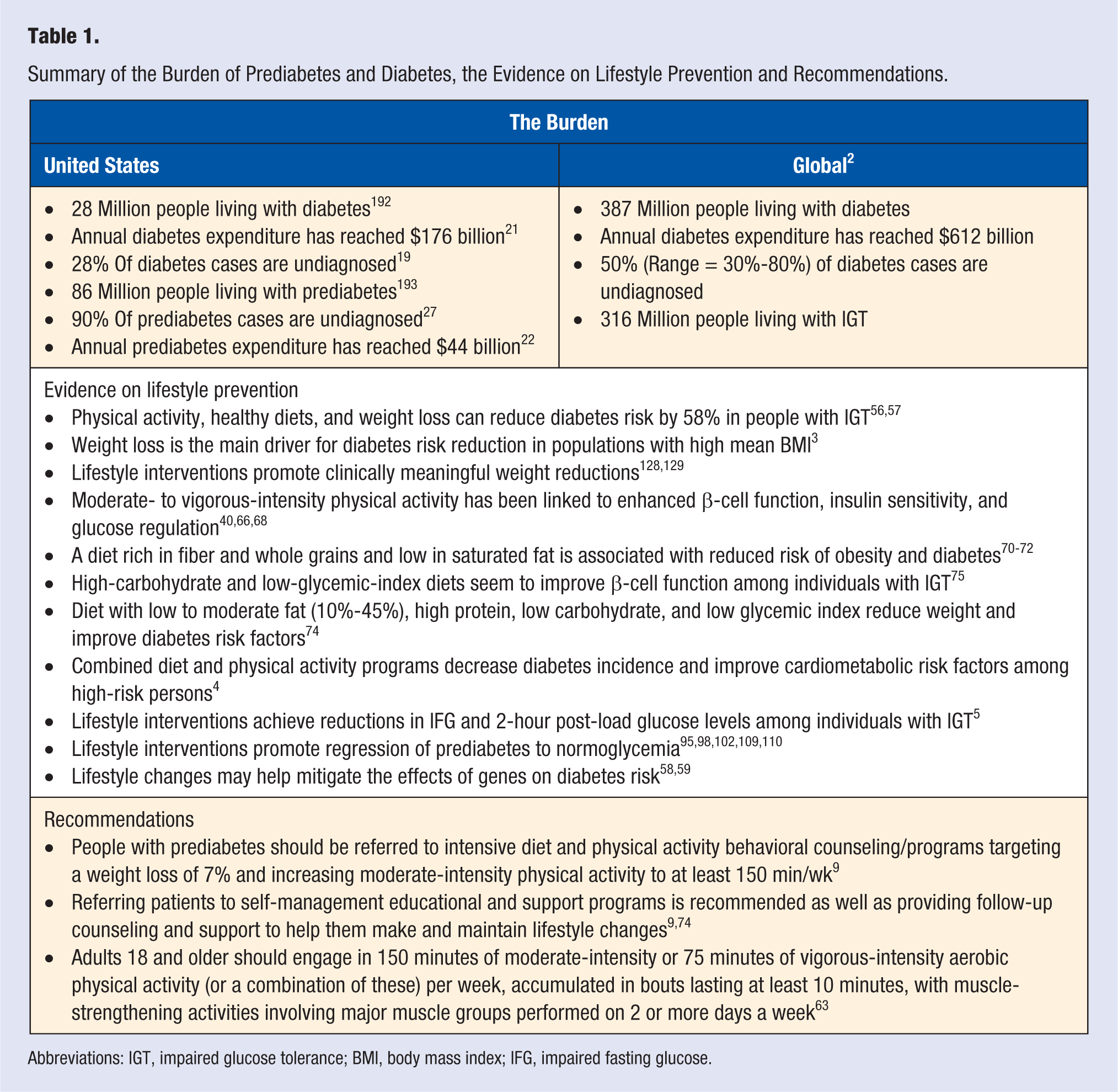
Abbreviations: IGT, impaired glucose tolerance; BMI, body mass index; IFG, impaired fasting glucose.
The Prediabetes Burden
Prediabetes is associated with all-cause mortality and has been shown to increase CVD risk by almost 2-fold.23-25 The IDF reports that the global prevalence of prediabetes reached 7% in 2013 and is expected to rise to 8% by 2035. 2 About 70% of the prediabetes cases are found in low- and middle-income countries and among adults younger than 50 years. 2 The prevalence of prediabetes is higher in Africa and European Regions and lower in the South-East Asia Region. 2 Slightly higher levels of prediabetes have been observed in rural than in urban areas, and IGT seems to be more common among women than men.13,26 Differences in prevalence between ethnic groups have also been observed, even between groups from the same country, as observed in India and China.13,14
In the United States, 37% of adults older than 20 years were affected by prediabetes in 2012, 19 and it is estimated that approximately 90% of prediabetes cases are undiagnosed (Table 1). 27 These estimates are similar for non-Hispanic whites (35%), non-Hispanic blacks (39%), and Hispanics (38%), 19 although recent studies have reported differences in prediabetes prevalence and susceptibility between ethnic groups. For instance, African Americans have shown a higher prevalence of prediabetes than their white counterparts, 28 whereas Asian Indians seem to be particularly susceptible to developing prediabetes. 29 The economic burden of prediabetes in the United States reached $44 billion in direct health care costs in 2012. 22 Prediabetes represents a substantial economic burden in its own right, 30 but given that IFG and IGT are likely to progress to diabetes,23,31 it also represents a potential future increase in new diabetes cases and associated costs.
Energy Balance and Diabetes Development
Energy balance plays an important role in the development of diabetes. The key elements in the energy balance equation are energy intake and energy expenditure, the former directly linked to diet and the latter to physical activity. High energy intake levels have been shown to increase diabetes risk by 11% to 26%, whereas adequate levels of physical activity reduce risk by 8% to 30%. 32 A positive energy balance, where energy intake is higher than energy expenditure, leads to overweight or obesity, further increasing diabetes risk.32,33
Obesity and physical inactivity lead to insulin resistance by increasing the nonphysiological deposition of fat in visceral, hepatic, and muscle tissues 34 and by intracellular sequestration of glucose transporter-4 (GLUT-4) in unexercised muscle. 35 The pathological deposition of fat is a major contributor to insulin resistance. Visceral, particularly intrahepatic fat, is associated with insulin resistance 36 and with lipid accumulation in muscle cells. 37 The excess adipose tissue interferes with glucose transport signaling, mainly by sending toxic messages in the form of free fatty acids, cytokines, and oxidative stress, which impair insulin’s ability to regulate glucose production by the liver and glucose uptake by the muscle.38,39
In contrast, physical activity reduces insulin resistance directly by promoting free fatty acid oxidation and reducing lipotoxicity in skeletal muscle and liver 40 and indirectly by reducing visceral fat. 34 Furthermore, exercise seems to improve serum levels of adiponectin, 41 a hormone that promotes insulin sensitivity 42 and is reduced in the presence of obesity. 43 Physical activity represents a physiological stressor that triggers changes in glucose transport and disposal to satisfy energy demands. The muscle is the main site for insulin-stimulated glucose disposal and, together with the adipose tissue, stores around 90% of GLUT-4, the main insulin-responsive glucose transporter. 35 In the absence of insulin or other stimuli (eg, physical activity), GLUT-4 remains sequestered intracellularly, impairing its ability to transport glucose into the muscle cell. Physical activity leads to GLUT-4 translocation from the intracellular storage vesicles to the plasma membrane, where it can transport glucose into the cell in a noninsulated mediated pathway. 35 Exercise increases glucose uptake by the working muscle 7 to 20 times above the basal rate, with improvements in insulin sensitivity lasting up to 3 days. 44 Among diabetes patients, exercise can increase nonoxidative glucose disposal, which in turn can improve whole-body glucose utilization. 45
β-Cell dysfunction and mass loss result in impaired insulin secretion. Loss in β-cell mass through apoptosis of β-cells has been tightly correlated with diabetes. 46 Studies have found reduced β-cell mass, ranging from 40% among prediabetes patients to 60% among diabetes patients. 47 Reductions in β-cell mass contribute to β-cell dysfunction, where the insulin secretion burden of the remaining cells increases, leading to chronic β-cell stress and ultimately impairing functional insulin secretion. 48 Although the mechanisms are not fully understood, evidence suggests that a variable combination of β-cell mass loss and β-cell dysfunction are involved in the pathogenesis of diabetes. 48 The influence of lifestyle changes on β-cell mass and function has not been fully understood, but preliminary animal and human studies suggest that physical activity improves β-cell function by upregulating insulin signaling pathways and β-cell mass by stimulating proliferation and preventing apoptosis. 49
Diabetes develops progressively as a result of the complex interaction between insulin resistance and β-cell dysfunction. Insulin resistance triggers a compensatory response, where the β-cells increase insulin secretion to maintain glucose homeostasis. If an adequate compensatory insulin secretion response is given, hyperglycemia can be offset, and blood glucose levels can regress to normal. Conversely, if the β-cells fail to cover the insulin demand triggered by insulin resistance, hyperglycemia will remain and potentially progress to IGT and ultimately to diabetes. 50 This pathway results in reductions in β-cell mass, further impairing the body’s ability to sustain normal glucose levels. 51 Figure 2 depicts the diabetes development process.
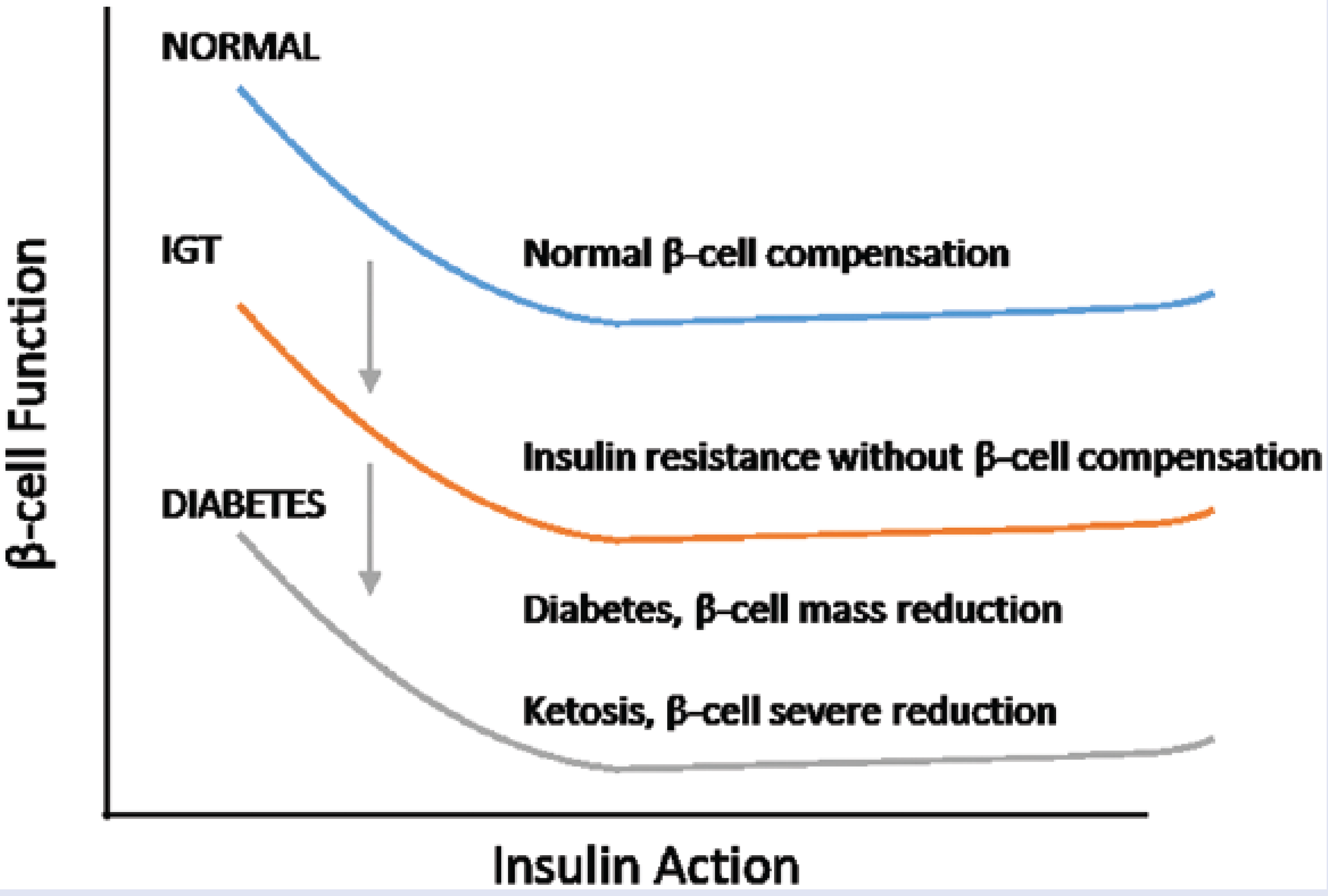
Diabetes development process (adapted from Stumvoll et al 50 ).
In sum, the degree of insulin resistance and the extent of β-cell dysfunction influence the development of glucose intolerance and progression to diabetes. Given the complex interaction among the physiological factors discussed above, people with insulin resistance will not necessarily develop glucose intolerance, nor will all people with prediabetes necessarily progress to diabetes. However, it is important to note that physical inactivity increases diabetes risk by 20%, 52 and each additional kilogram of weight gained translates into a 4.5% increase in diabetes risk. 53 Considering the high obesity and physical inactivity rates and poor diets among US adults, promoting lifestyle changes among at-risk adults before they develop the disease is imperative.
A Lifestyle Prescription for Diabetes Prevention
Overwhelming evidence shows that lifestyle changes—namely, improvements in physical activity and diet, leading to weight loss—reduce diabetes risk significantly.54,55 Randomized controlled trials have shown that lifestyle interventions focused on physical activity, healthy diets, and weight loss can reduce diabetes risk by 58% in people with IGT.56,57 Evidence also shows that lifestyle changes may help mitigate the effects of genes on diabetes risk. For instance, in the US Diabetes Prevention Program (DPP), the association between susceptible genotypes and progression to diabetes was attenuated in individuals receiving a lifestyle intervention. 58 Similarly, a study among Swedish adults showed that the genetic predisposition to diabetes may be offset by physically active lifestyles. 59
Weight loss is the main driver of diabetes prevention in populations with high mean BMI, such as Americans. 3 In the US DPP for instance, weight loss was the strongest predictor of reduced diabetes incidence; a 5-kg weight loss explained an incidence reduction of 58%. 60 More specifically, for every kilogram participants lost, a 16% reduction in diabetes risk was observed. 60 Similarly, only participants who lost 8-17% of their weight in the Finish Diabetes Prevention Study (DPS) achieved significant improvements in insulin sensitivity. 61 Changes in fat distribution have also been linked to improved insulin sensitivity, particularly changes in visceral fat mass and liver fat content. 62 Based on this, a weight loss of 7% of total body weight has been recommended for the primary prevention of diabetes.
Because obesity is in part an outcome of positive energy balance, targeting physical activity and diet in weight loss–driven diabetes prevention efforts is essential. Physical activity involves any bodily movement produced by skeletal muscles that requires energy expenditure and includes leisure time physical activity, transportation (eg, walking or cycling), occupational (ie, work), household chores, play, games, and sports or planned exercise (see Table 2). 63 A range of physical activities and intensities are associated with 20% to 30% diabetes risk reduction, especially among high-risk individuals. 64 For instance, ≥2.5 h/wk of moderate-intensity brisk walking is associated with a diabetes risk reduction of 27%, independent of BMI. 65 Furthermore, moderate- to vigorous-intensity physical activity has been linked to enhanced β-cell function and glucose regulation, independent of obesity. 66 Structured exercise training has been found to reduce HbA1c by 67% among diabetes patients 67 and to improve β-cell function and insulin sensitivity among high-risk patients.40,68 These effects are comparable or superior to those achieved with common antidiabetic drugs.68,69 Overall, international guidelines recommend that adults 18 years and older engage in 150 minutes of moderate-intensity or 75 minutes of vigorous-intensity aerobic physical activity (or a combination of these) per week, accumulated in bouts lasting at least 10 minutes with muscle-strengthening activities involving major muscle groups performed on 2 or more days a week. 63
Physical Activity Definitions According to the World Health Organization 2010. 63
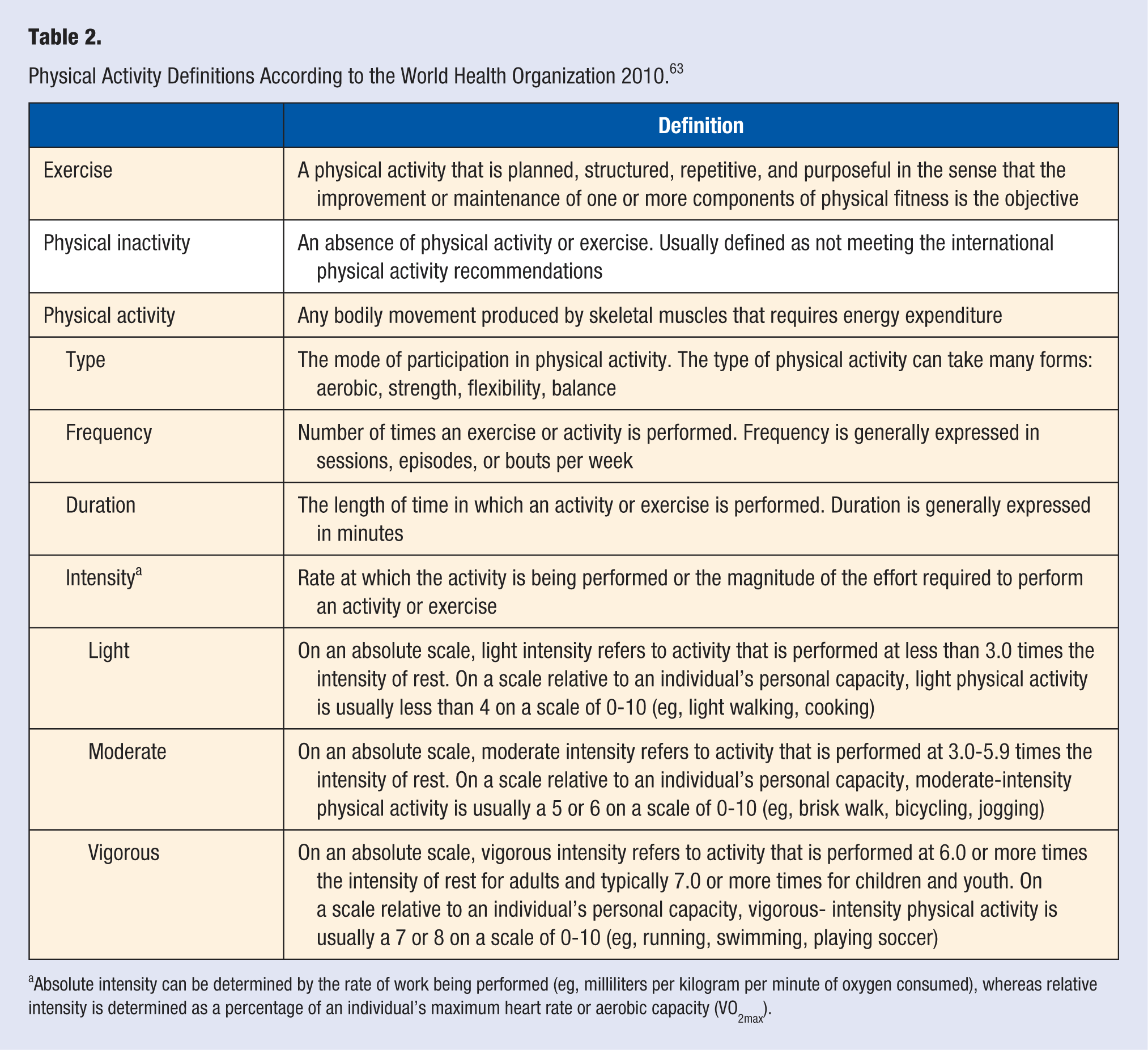
Absolute intensity can be determined by the rate of work being performed (eg, milliliters per kilogram per minute of oxygen consumed), whereas relative intensity is determined as a percentage of an individual’s maximum heart rate or aerobic capacity (VO2max).
Caloric intake and diet quality are important drivers of obesity and diabetes. Overall, a diet rich in fiber and whole grains and low in saturated fat is associated with reduced risk of obesity and diabetes.70,71 Prospective studies have consistently shown that consumption of cereal fiber or mixtures of whole grains and bran reduce diabetes risk by 18% to 40%. 70 Furthermore, with a 2 serving/d increment in whole-grain intake, diabetes risk can be reduced up to 21%. 72 It has also been shown that a saturated fat intake less than 7% of the total energy intake is associated with reduced diabetes risk. 71 Because people who drink more than 1 sugar-sweetened beverage per day have a 26% increase in diabetes risk compared with those who drink less than 1 per month, 73 reducing sugar-sweetened beverage consumption may also decrease risk. Regarding dietary strategies, low to moderate fat (10%-45%), high protein, low carbohydrate, and low glycemic index diets have been shown to effectively reduce weight and improve diabetes risk factors. 74 High-carbohydrate and low glycemic index diets seem to improve β-cell function among those with IGT. 75 The ADA recommends that high-risk individuals are encouraged to increase their fiber intake and consume whole grain foods. 9 Behavioral counseling and ongoing support should be used to help patients, 74 whereas individualized nutrition counseling should be provided by a registered dietitian (see Table 1).
The extent to which lifestyle changes can prevent or delay the onset of diabetes is associated with the degree of β-cell dysfunction and hyperglycemic nature. For example, in persons with mild β-cell dysfunction, who exhibit IGT as a result of peripheral insulin resistance, lifestyle changes can improve insulin sensitivity and ultimately restore normoglycemia. Conversely, in patients with moderate β-cell dysfunction, who present with isolated IFG, lifestyle changes may not restore normoglycemia but may help regulate glucose levels. 76 In other words, lifestyle changes may have a small effect on the progression to diabetes in individuals with isolated IFG concentrations but they are highly beneficial among those with IGT. 76
Other lifestyle behaviors and mental health factors such as smoking, sedentary behavior (time spent seating), sleeping patterns, and stress have also been linked to diabetes risk, independent of sociodemographic factors, physical activity, and obesity.77-85 Meta-analyses have shown that smoking significantly increases diabetes risk, with active smokers showing a 44% increased risk. 78 A diabetes risk 1 to 2 times higher has been observed among men who watch television for more than 20 hours per week, compared to those who do it for less than 2 hours per week. 79 Similarly, a 3.4% increase in diabetes risk for each additional hour spent watching television was observed among DPP participants. 80 Sleeping disturbances have also been linked to abnormal glucose metabolism, where trouble sleeping, sleep apnea, and sleep loss have been shown to significantly increase diabetes risk.81-83 Likewise, psychosocial stress predicts diabetes incidence in men 84 and abnormal glucose metabolism among women. 85 Because these behaviors are out of the scope of the present review, we invite interested readers to review the articles cited in this paragraph and other articles available elsewhere.
Efficacy of Lifestyle Diabetes Prevention
Five highly controlled, large-scale studies assessing the efficacy of lifestyle DPPs have been implemented across the globe.57,86-91 In the US DPP, adults with IGT were randomly assigned to receive a placebo, metformin (a glucose-lowering drug), or a lifestyle program consisting of 24 individual counseling sessions on diet and physical activity. After 2.8 years, diabetes risk was reduced by 58% in the lifestyle group and by 31% in the metformin group. Lifestyle participants also achieved a significant weight loss, which was higher than that of controls (5.6 vs 0.1 kg, respectively). 86 At the end of the original DPP, participants in all groups were offered a group-based lifestyle program, and 15 years after original randomization, diabetes incidence was reduced by 27% among lifestyle participants and 18% among metformin participants. 92
In Finland, the DPS showed that patients receiving a 4-year intervention comprising individualized dietary counseling and free access to gym-based supervised exercise sessions achieved a diabetes risk reduction of 58%. 93 Likewise, intervention patients showed greater weight reductions, better glycemic and lipid levels, and healthier diets than patients receiving usual care. 87 After 13 years of randomization, diabetes incidence was reduced by 43% among intervention patients while improvements in body weight, FBG and 2-hour blood glucose, and dietary habits were maintained. 94
Three studies in Asia, the Da Qing IGT and Diabetes Study, the Indian DPP (IDPP) and the Japanese DPP also showed the benefits of lifestyle intervention. The Da Qing IGT and Diabetes Study implemented in China showed that compared with control participants, those receiving individualized dietary prescription combined with physical activity counseling over 6 years had a 51% lower diabetes incidence at the end of the intervention and a 43% lower incidence over a 20-year follow-up period.88,89 In the IDPP, participants randomized to receive monthly in-person advice on physical activity and diet to prevent diabetes achieved a 28.5% relative risk reduction compared with the control group. This risk reduction was similar to that achieved among participants assigned to a metformin or a lifestyle plus metformin group (28% and 26%, respectively). 90 Finally, the Japanese DPP showed that participants receiving frequent individual instructions and lifestyle support from medical staff over 3 years achieved a diabetes risk reduction of 44%. 91
Not only can lifestyle programs significantly reduce diabetes incidence among people with prediabetes, but they can also improve the CVD risk profile. Numerous randomized controlled trials have shown that lifestyle interventions can reduce FPG (from −3 to −19 mm/dL),93,95-102 2-hour post-load plasma glucose (from −6 to −29 mm/dL),93,96,98-101,103,104 systolic blood pressure (from −1.6 to −11 mm Hg),96,98,103,105,106 total cholesterol (from −3 to −15 mg/dL),96,98,99,105-107 triglycerides (from −2 to −15 mm/dL),96,98,101,105 HbA1c (from −0.09% to −19%),100,102,103,106 and body weight (from −2 to −10 kg).95,96,98,108,109 Furthermore, a meta-analytic synthesis showed that lifestyle interventions among individuals with IGT achieve a standardized mean reduction of −0.27 (95% CI = −0.38 to −0.15) on FPG and −0.56 (95% CI = −1.01 to −0.10) on 2-hour post-load glucose levels, with effects varying between intervention strategies (eg, diet vs physical activity). 5 Finally, lifestyle interventions have also been shown to promote regression to normoglycemia (Table 1).95,98,102,109,110
Effectiveness of Lifestyle Diabetes Prevention
The efficacy-driven interventions discussed above are resource intensive, directed at homogeneous populations, implemented in highly controlled settings, and often delivered by health care professionals, thereby limiting their real-world applicability and impact. Thus, researchers have focused on investigating low-cost strategies that can be implemented in real-world practice, utilizing group-based formats and lay community members or technology, and implemented in a variety of community settings where heterogeneous populations can be reached. For instance, the 16-week original DPP curriculum has been adapted for group-based delivery and has been implemented in YMCAs, churches, primary care clinics, and other community settings, by trained staff, physicians, lay community members, and technology-assisted devices (eg, Internet, text messages).111-123 The achieved effects include weight reductions ranging from −1.9 to −8.7 kg112,113,115,116,118-122,124 and improvements in fasting glucose levels ranging from −2.1 to −9 mm/dL.113,115,119,121,125-127
A recent meta-analysis of 26 intervention studies aimed at translating the DPP in the United States found that lifestyle interventions achieved a pooled weight reduction of 4%, regardless of whether the intervention was delivered by health care professionals or lay community educators. 128 Another meta-analysis summarizing the evidence on the effectiveness of 22 translational DPPs from 11 countries showed that lifestyle interventions achieved a mean weight loss of 2.3 kg (95% CI = −2.9 to −1.7 kg), although effects varied widely across studies. 129 Factors associated with heterogeneity in weight change effects include intervention dose (eg, number of sessions delivered, adherence to guidelines), the delivery agent (eg, health care provider, community member), and study design and follow-up length.128,129
In light of this, the Community Preventive Services Task Force, the ADA, the IDF, and the World Health Organization recommend combined diet and physical activity counseling and education promotion programs for people at increased risk of diabetes across a range of intervention intensities, settings, and implementers.9,130-132
Lifestyle Diabetes Prevention in Clinical Practice
Lifestyle diabetes prevention in clinical practice is effective and feasible, leading to the adaptation of large prevention trials for the clinical setting.113,120,126,133-135 A meta-analytic review showed that dietary and physical activity counseling for diabetes prevention in routine clinical practice promotes weight and waist circumference reductions in high-risk patients. 54 Implementing lifestyle counseling in clinical practice has been shown to be feasible and cost-effective3,54,136-139 and acceptable among practitioners and patients. 140 This has led to the conclusion that lifestyle-related health care provider services should be widely implemented and potentially reimbursed. 141
Based on this, a number of clinical recommendations have been released. The US Preventive Services Task Force 2014 recommendations support offering or referring adults at risk of CVD to physical activity and nutrition counseling, a strategy that under the Affordable Care Act must be included in new health plans.142,143 The American Heart Association recommends using behavior change techniques such as goal setting and self-monitoring for promoting lifestyle change, 144 and the ADA promotes referring high-risk patients to programs that target weight loss and increased physical activity. 9 Despite this, the adoption and implementation of lifestyle diabetes prevention strategies in regular clinical practice remains a challenge.
The US Department of Health and Human Services reports that in 2010, 32.4% of adults and 56% of diabetes patients who had seen a health professional in the past year were advised to engage in physical activity. 145 The barriers to lifestyle counseling practitioners face—mainly lack of time, training, and organizational resources 146 —as well as the disconnection between clinical practice and community health programs 147 contribute to these low rates. Furthermore, clinic-level factors such as organizational characteristics and capacity/resources may hinder the adoption of diabetes preventive practices. Recognizing this, strategies to integrate lifestyle diabetes prevention in clinical practice have been identified. Collaborating with community-based lifestyle programs, establishing patient referrals to such programs, creating multidisciplinary teams to provide patient education and follow-up, training physicians on effective brief counseling, and using health information technologies to promote behavior change represent the most promising strategies.141,148
From Evidence to Practice
The IDF has proposed a simple 3-step plan for the prevention of diabetes in high-risk individuals that includes (1) identification of those who may be at increased risk, (2) risk evaluation, and (3) intervention to prevent diabetes. 149 Following these steps, we now outline the evidence-based diabetes prevention strategies that health care practitioners can implement in their practice (see Table 3).
Steps Outlined by IDF 149 for the Prevention of Diabetes in High-Risk Individuals.

Abbreviations: IDF, International Diabetes Federation; BMI, body mass index; HbA1c, hemoglobin A1c; FBG, fasting blood glucose; OGTT, oral glucose tolerance test; CVD, cardiovascular disease.
Identification
The IDF recommends that high-risk individuals should be identified through opportunistic screening by physicians, nurses, and pharmacists and through self-screening. 149 Simple, noninvasive diabetes risk questionnaires exist150-152 and are useful tools for identifying patients at high risk. According to ADA 2015 criteria, patients of any age with overweight or obesity (BMI ≥25 kg/m2 or ≥23 kg/m2 in Asian Americans) and who have one or more additional risk factors (ie, family history of diabetes, history of gestational diabetes, high risk race/ethnicity [African American, Latino, Native American, Asian American, Pacific Islander]) are at increased risk. 9 The opportunistic screening of high-risk people could help lower undiagnosed diabetes rates.
Measurement
According to ADA, high-risk patients of any age (see criteria above) and low-risk patients ≥45 years of age should be tested for glucose intolerance. HbA1c, FPG, or OGTT are appropriate methods for identifying prediabetes as well as diabetes cases. If tests are normal, testing should be repeated at least once every 3 years. In people with prediabetes, testing should be repeated yearly. 9 Although this is still under debate, the US Preventive Services Task Force’s 2015 recommendations and others support the testing of glucose intolerance among individuals who are at increased risk.30,153,154 Among patients with prediabetes, screening for and treatment of modifiable CVD risk factors is recommended, 9 and preexisting CVD should be treated. 149
Lifestyle Intervention
The objective of an intervention should be helping patients lose body weight (7% is recommended, but patient’s ethnicity should be considered), increase moderate-intensity physical activity levels to at least 150 min/wk, and increase fiber intake. 9 In terms of intervention strategies, individual or group (or a combination of both) dietary and physical activity counseling from health care professionals, group educational sessions, and tailoring of diet or physical activity plans can be used.4,54 For promoting lifestyle behavior change, helping patients set lifestyle goals, providing information on the health consequences of current behaviors, and using follow-up prompts and plans have been shown to be effective behavior change techniques.144,155 Similarly, empathy, nonjudgmental interactions, and specific personalized recommendations have been identified as essential components of weight loss counseling. 156 Regarding “dose,” a minimum of 4 to 6 months of weekly weight loss education sessions and consistent follow-up for 1 year are necessary to achieve a clinically meaningful weight loss. 157 High-intensity counseling strategies (>360 minutes of total patient time) show greater effects on lifestyle and health outcomes than medium-intensity counseling (31-360 minutes). 158 Overall, DPPs have been shown to be effective across a range of counseling intensities, settings, and implementers, 4 and evidence-based strategies to help integrate lifestyle promotion in clinical practice are available.
Clinician-Based Strategies
Health care professionals interested in providing individual lifestyle counseling to their patients can use existing evidence-based tools such as the 5-As (Assess, Advice, Assist, Agree, and Arrange) model and Brief Action Planning. The 5-As model is a 5-step tool designed to assist health care providers in their lifestyle counseling efforts. The model can help health care professionals improve their lifestyle and weight loss counseling, which can in turn improve patient behavior.159,160 Using diet as an example, the practitioner first assesses patient’s dietary behavior, then advices the patient to increase fiber intake, agrees on a collaboratively set goal (eg, increasing dietary fiber 20-30 g/d), follows with assisting the patient on identifying barriers to their goal and strategies to overcome these, and finally arranges follow-up visits and support (see Table 4).
Steps Outlined in the 5-As Model for Lifestyle Counseling With Corresponding Tools and Behavior Change Strategies.
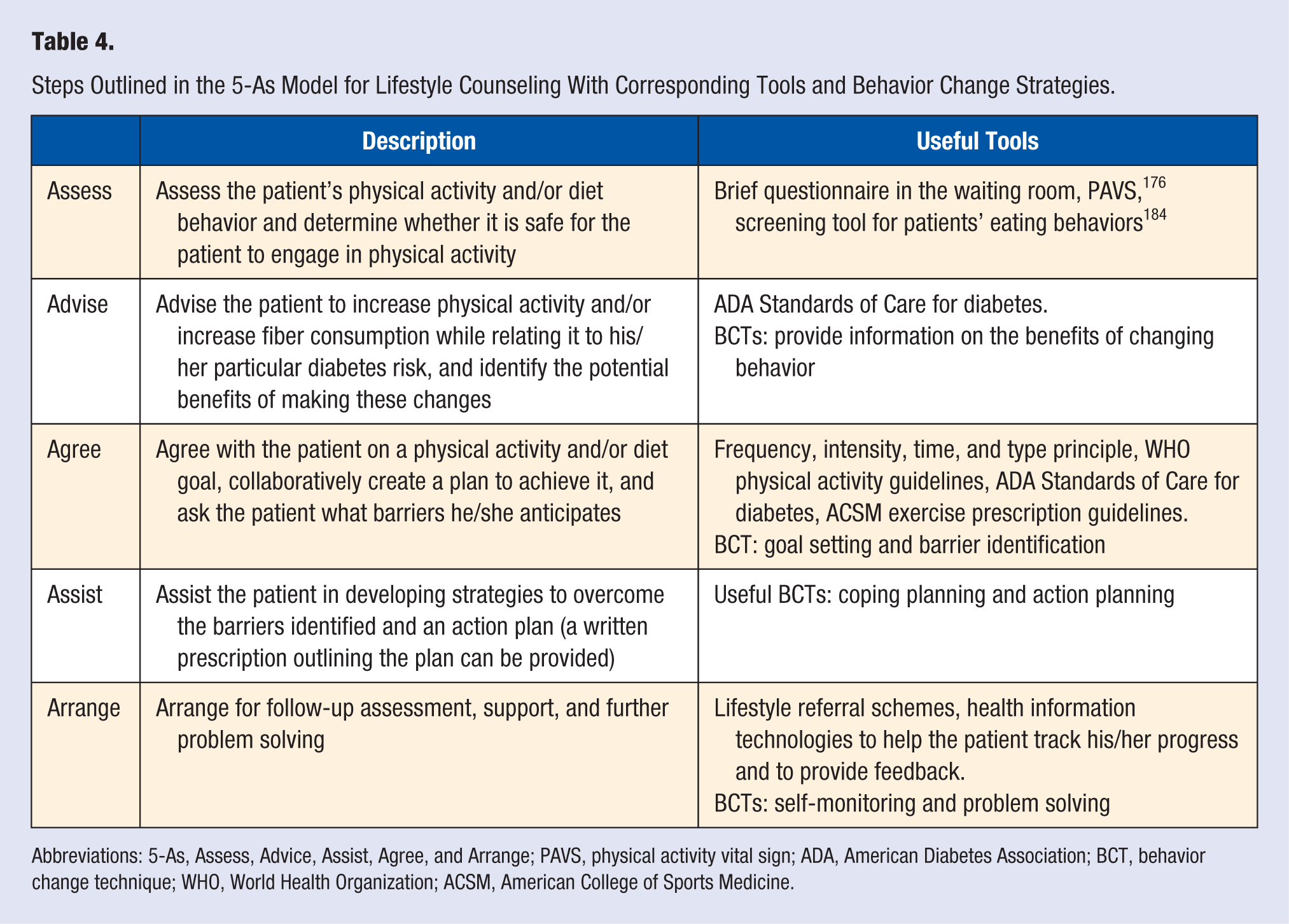
Abbreviations: 5-As, Assess, Advice, Assist, Agree, and Arrange; PAVS, physical activity vital sign; ADA, American Diabetes Association; BCT, behavior change technique; WHO, World Health Organization; ACSM, American College of Sports Medicine.
To provide more in-depth behavior change counseling, practitioners can turn to brief action planning. 161 This is a highly structured, self-management support technique that includes 3 questions and 5 skills focused on helping patients set goals and form action plans (ie, when, where, and how to enact a goal-directed behavior) to change their behavior. With training, using interactive Web-based tools and guidelines, brief action planning can be learned by health care practitioners and used in clinical settings. 161 However, brief action planning takes about 20 minutes to complete, whereas the 5-As model can take 3 to 5 minutes, which challenges the successful incorporation of individual lifestyle counseling in busy clinical settings.148,156 Recognizing that many health care professionals may not have time to provide individual counseling, other strategies such as group-based, lifestyle referrals and health information technology have been proposed.
Group and Referral Strategies
The implementation of group-based DPPs in the clinical setting has been successful in reducing diabetes risk while avoiding placing time burdens on health care practitioners. Examples include DPPs consisting of 1 to 6 educational group sessions on physical activity and healthful eating delivered by health care proressionals.162,163 Furthermore, the DPP has been adapted for implementation in clinical settings,120,133 and course curriculum, program materials, and training are available to support implementation.164,165 If implementing group-based lifestyle programs is not possible, collaborating with existing community-based programs and resources is a feasible strategy for supporting lifestyle change in clinical settings. 148
Lifestyle referral strategies in primary care are cost-effective, 166 whereas practical collaborative approaches among health care practitioners (eg, physicians, nurses, dietitians) show promising weight outcomes. 156 A great example is the physical activity referral scheme, which has been widely tested and shown to promote small, short-term improvements in patients’ physical activity. 167 Moreover, lifestyle referral schemes have the potential to support patient behavior change and maintenance while also promoting the creation of clinical-community linkages. 168 In line with this, the ADA recommends referring high-risk patients to weight loss and physical activity programs as well as to educational programs for lifestyle change support. 9 Because referring patients to lifestyle programs and resources may not be enough to promote behavior change, 169 the collaborative effort between clinicians, organizations, and community settings is warranted to provide patients with healthy eating and physical activity opportunities in all settings.
Health Information Technology
The use of health information technology to facilitate behavior change has been identified as a promising strategy for promoting lifestyle change in the clinical context. 148 Electronic health records, online tools and websites, electronic physical activity monitors, and mobile technologies (eg, smart phones and applications) are among technologies that have been developed to assist patients in their self-management efforts.170,171 Health information technology used in primary care represents a feasible, cost-effective, and acceptable strategy to deliver or augment standardized interventions, improve patient self-management and access to resources, and provide flexibility of care services.172-174
A major example of such technologies is the electronic health record, which has been strongly supported by the US federal government to improve diabetes prevention and quality of care. 175 The usefulness of electronic health records for identification of obesity and the delivery of interventions has also been emphasized. 156 Through electronic health records, the physical activity vital sign, a 2-question tool to screen for physical inactivity, has been implemented in the United States, achieving favorable changes in physician practice and patient metabolic outcomes. 176
Health information technologies facilitate the delivery of effective interventions. Physical activity electronic monitors for instance have been used to implement behavior change strategies such as self-monitoring, feedback, and environmental change 177 and for physical activity counseling in clinical and community settings. 178 Likewise, telephone-based strategies have been used to deliver dietary and physical activity counseling, which have been shown to have a wide patient reach 162 and promote behavior change. 179 Other strategies include DVD and Internet program delivery, both of which have shown promising impact on weight loss.120,133 Although current evidence supporting the use of health information technologies for diabetes prevention is lacking, such an approach has the potential to facilitate the delivery of targeted interventions to people with prediabetes, accelerate the diffusion of new evidence, and generate automatic referrals to lifestyle intervention programs. 180
A Case Study
One example of a comprehensive lifestyle intervention that combines the elements described above is Exercise is Medicine, a program aimed at making physical activity part of the standard medical care in the US and global health care systems. 147 Using a multisector, multilevel approach, Exercise is Medicine is focused on integrating physical activity assessment within health care systems, creating clinical-community linkages through physical activity referrals, and advancing health information technologies to support patient behavior change. 147 Pilot studies are promising and support the feasibility and effectiveness of integrating physical activity assessment, counseling, and referrals in clinical practice.181,182 Comprehensive tools, customized physical activity prescriptions for several diseases, and resources to help health care practitioners and systems integrate physical activity promotion in their practice are available on the Exercise is Medicine website. 183
The Exercise is Medicine model can be used to guide lifestyle diabetes prevention efforts in clinical practice. Comprising behavior assessment, counseling, and referral steps, the clinical component of the model outlines strategies that can help patients improve their diabetes-related behaviors. 147 First, the patient’s physical activity and dietary behaviors can be assessed using available tools such as the physical activity vital sign 176 and brief dietary and obesity-related behaviors questionnaires184-186 or simply by asking patients about their lifestyle behaviors. Second, brief lifestyle counseling can be provided, where health care practitioners can turn to the evidence-based tools previously described (ie, 5-As model, brief action planning) or implement behavior change techniques such as informing patients on the risks of their current behaviors, helping them set behavioral goals, and providing support and follow-up.144,155,187 Here, health care practitioners can offer the patient a verbal or written prescription that summarizes the patient’s goals or that can be based on weight loss, physical activity, and dietary recommendations for diabetes prevention.9,63 Finally, health care practitioners can refer patients to self-management resources (eg, websites, electronic physical activity monitors, diet and exercise tracking apps),177,178,188,189 individual counseling with physical activity and nutrition experts,139,190 or existing group-based DPPs and resources in the community.112,120,191 The community and health information technology components of the model can be consulted elsewhere. 147
Conclusion
Affecting individuals, health care systems, economies, and whole societies worldwide, diabetes has become a major public health threat. Lifestyle intervention programs promoting healthy diets, physical activity, and modest body weight reductions can prevent or delay the onset of diabetes among high-risk populations. Evidence supports the efficacy and effectiveness of such interventions across clinical and community settings, delivery formats, and implementers. At-risk individuals need education, access to ongoing support, and an adequate environment to engage in health-enhancing behaviors. Health care practitioners and systems can contribute by providing individual counseling, establishing referral systems and linkages with community programs and resources, or by introducing health information technologies to screen for unhealthy behaviors or deliver interventions. These strategies represent opportunities where practitioners, communities, and health care systems can work together to provide individuals with education, support, and opportunities to maintain healthy, diabetes-free lifestyles.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
